Analgesic Efficacy of Melatonin: A Meta-Analysis of Randomized, Double-Blind, Placebo-Controlled Trials
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Searches
2.2. Study Selection and Eligibility
2.3. Assessment of Methodological Quality
2.4. Main and Subgroup Analysis
2.5. Statistical analyses
3. Results
3.1. Identification of Relevant Studies
3.2. General Characteristics of Trials
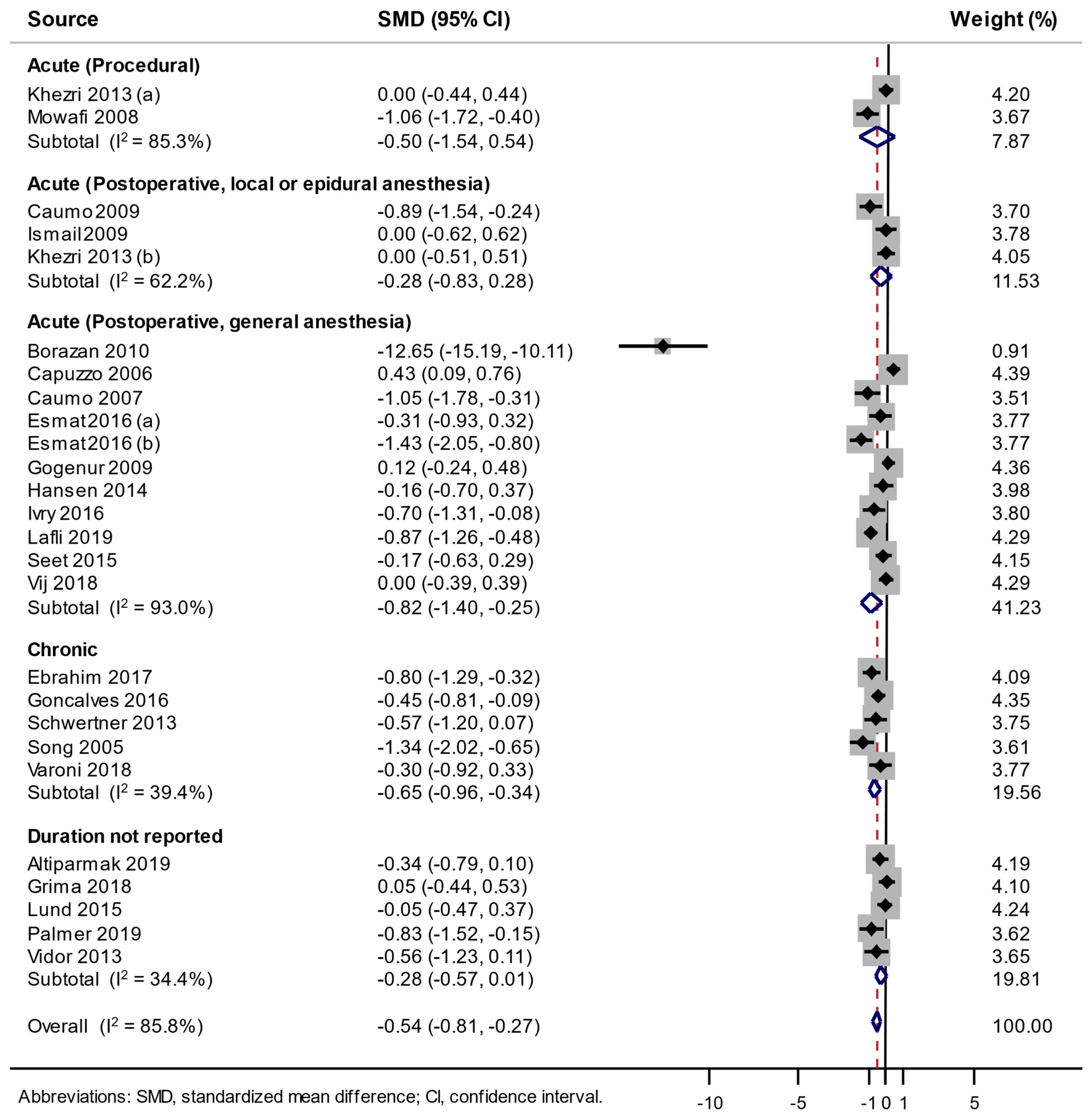
3.3. Association between the Use of Melatonin and Pain Score Changes
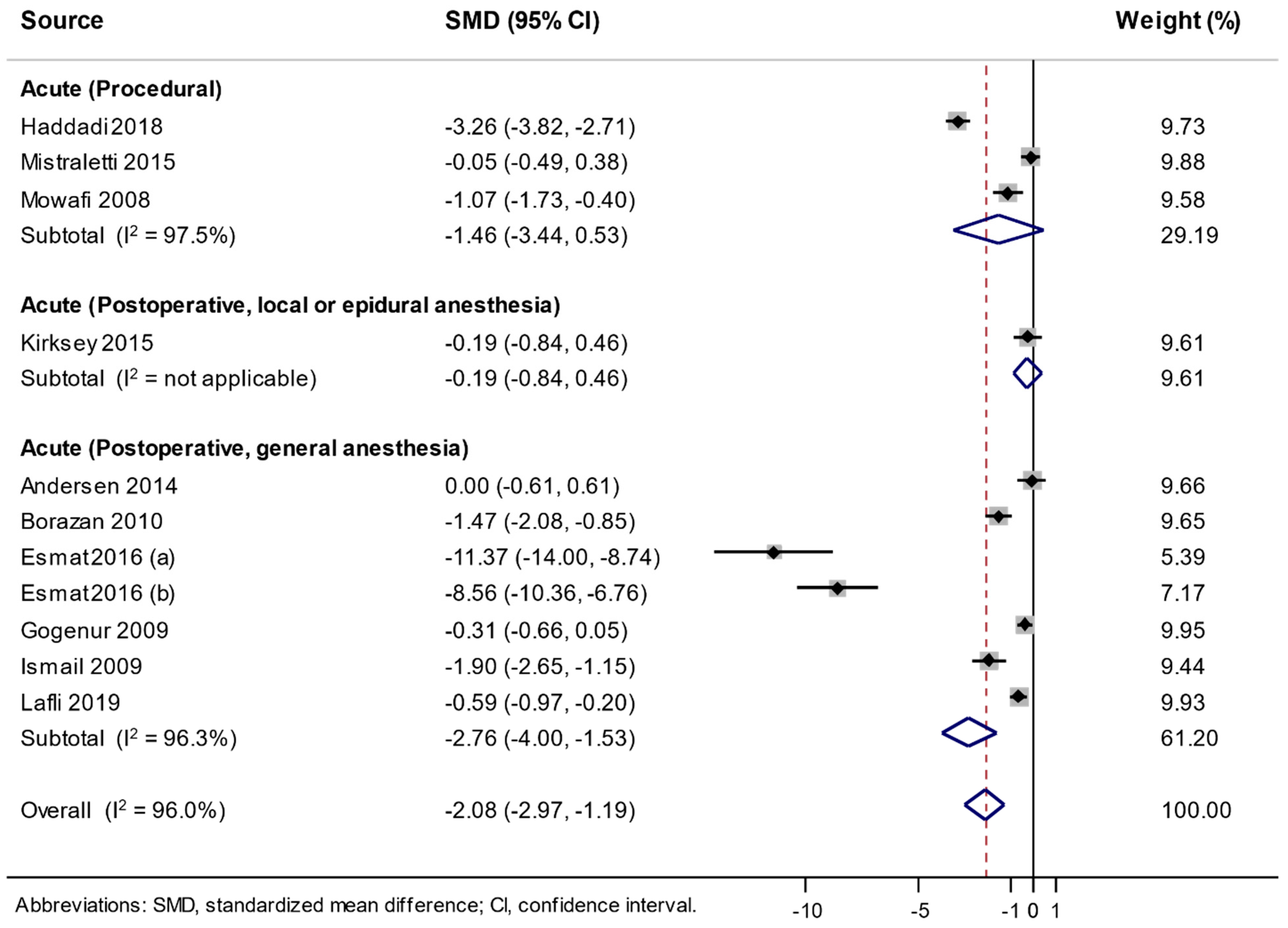
3.4. Association between the Use of Melatonin and Changes in Analgesic Consumption
3.5. Adverse Events
4. Discussion
4.1. Summary of Findings
4.2. Possible Mechanisms for Analgesic Effects of Melatonin
4.3. Comparisons with Previous Studies and Strengths of Our Study
4.4. Possible Reasons for No Analgesic Effect of Melationin in Acute Postoperative or Procedural Pain
4.5. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Brzezinski, A. Melatonin in humans. N. Engl. J. Med. 1997, 336, 186–195. [Google Scholar] [CrossRef]
- Srinivasan, V.; Lauterbach, E.C.; Ho, K.Y.; Acuna-Castroviejo, D.; Zakaria, R.; Brzezinski, A. Melatonin in antinociception: Its therapeutic applications. Curr. Neuropharmacol. 2012, 10, 167–178. [Google Scholar] [CrossRef]
- Ambriz-Tututi, M.; Rocha-Gonzalez, H.I.; Cruz, S.L.; Granados-Soto, V. Melatonin: A hormone that modulates pain. Life Sci. 2009, 84, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, F.; Seet, E.; Venkatraghavan, L.; Abrishami, A.; Chung, F. Efficacy and safety of melatonin as an anxiolytic and analgesic in the perioperative period: A qualitative systematic review of randomized trials. Anesthesiology 2010, 113, 968–976. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.P.; Werner, M.U.; Rosenberg, J.; Gögenur, I. A systematic review of peri-operative melatonin. Anaesthesia 2014, 69, 1163–1171. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Xu, Y.; Duan, Y.; Li, W.; Zhang, L.; Huang, Y.; Zhao, W.; Wang, Y.; Li, J.; Feng, T.; et al. Exogenous melatonin in the treatment of pain: A systematic review and meta-analysis. Oncotarget 2017, 8, 100582–100592. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The prisma statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Higgins, J.P.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration. Available online: https://training.cochrane.org/handbook/archive/v5.1/ (accessed on 19 February 2020).
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Altiparmak, B.; Cil, H.; Celebi, N. Effect of melatonin on the daytime sleepiness side-effect of gabapentin in adults patients with neuropathic pain. Rev. Bras. Anestesiol. 2019, 69, 137–143. [Google Scholar] [CrossRef]
- Andersen, L.P.; Kucukakin, B.; Werner, M.U.; Rosenberg, J.; Gogenur, I. Absence of analgesic effect of intravenous melatonin administration during daytime after laparoscopic cholecystectomy: A randomized trial. J. Clin. Anesth. 2014, 26, 545–550. [Google Scholar] [CrossRef]
- Borazan, H.; Tuncer, S.; Yalcin, N.; Erol, A.; Otelcioglu, S. Effects of preoperative oral melatonin medication on postoperative analgesia, sleep quality, and sedation in patients undergoing elective prostatectomy: A randomized clinical trial. J. Anesth. 2010, 24, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Capuzzo, M.; Zanardi, B.; Schiffino, E.; Buccoliero, C.; Gragnaniello, D.; Bianchi, S.; Alvisi, R. Melatonin does not reduce anxiety more than placebo in the elderly undergoing surgery. Anesth. Analg. 2006, 103, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Caumo, W.; Torres, F.; Moreira, N.L., Jr.; Auzani, J.A.; Monteiro, C.A.; Londero, G.; Ribeiro, D.F.; Hidalgo, M.P. The clinical impact of preoperative melatonin on postoperative outcomes in patients undergoing abdominal hysterectomy. Anesth. Analg. 2007, 105, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Caumo, W.; Levandovski, R.; Hidalgo, M.P. Preoperative anxiolytic effect of melatonin and clonidine on postoperative pain and morphine consumption in patients undergoing abdominal hysterectomy: A double-blind, randomized, placebo-controlled study. J. Pain 2009, 10, 100–108. [Google Scholar] [CrossRef]
- Ebrahimi-Monfared, M.; Sharafkhah, M.; Abdolrazaghnejad, A.; Mohammadbeigi, A.; Faraji, F. Use of melatonin versus valproic acid in prophylaxis of migraine patients: A double-blind randomized clinical trial. Restor. Neurol. Neurosci. 2017, 35, 385–393. [Google Scholar] [CrossRef]
- Esmat, I.M.; Kassim, D.Y. Comparative study between transdermal fentanyl and melatonin patches on postoperative pain relief after lumber laminectomy, a double-blind, placebo-controlled trial. Egypt. J. Anaesth. 2016, 32, 323–332. [Google Scholar] [CrossRef][Green Version]
- Esmat, I.M.; Kassim, D.Y. Comparative study between transdermal nicotine and melatonin patches on postoperative pain relief after laparoscopic cholecystectomy, a double-blind, placebo-controlled trial. Egypt. J. Anaesth. 2016, 32, 299–307. [Google Scholar] [CrossRef]
- Gogenur, I.; Kucukakin, B.; Bisgaard, T.; Kristiansen, V.; Hjortso, N.C.; Skene, D.J.; Rosenberg, J. The effect of melatonin on sleep quality after laparoscopic cholecystectomy: A randomized, placebo-controlled trial. Anesth. Analg. 2009, 108, 1152–1156. [Google Scholar] [CrossRef]
- Gonçalves, A.L.; Ferreira, A.M.; Ribeiro, R.T.; Zukerman, E.; Cipolla-Neto, J.; Peres, M.F.P. Randomised clinical trial comparing melatonin 3 mg, amitriptyline 25 mg and placebo for migraine prevention. J. Neurol. Neurosurg. Psychiatry 2016, 87, 1127–1132. [Google Scholar] [CrossRef]
- Grima, N.A.; Rajaratnam, S.M.W.; Mansfield, D.; Sletten, T.L.; Spitz, G.; Ponsford, J.L. Efficacy of melatonin for sleep disturbance following traumatic brain injury: A randomised controlled trial. BMC Med. 2018, 16, 8. [Google Scholar] [CrossRef] [PubMed]
- Haddadi, S.; Shahrokhirad, R.; Ansar, M.M.; Marzban, S.; Akbari, M.; Parvizi, A. Efficacy of preoperative administration of acetaminophen and melatonin on retrobulbar block associated pain in cataract surgery. Anesth. Pain Med. 2018, 8, e61041. [Google Scholar] [CrossRef]
- Hansen, M.V.; Andersen, L.T.; Madsen, M.T.; Hageman, I.; Rasmussen, L.S.; Bokmand, S.; Rosenberg, J.; Gogenur, I. Effect of melatonin on depressive symptoms and anxiety in patients undergoing breast cancer surgery: A randomized, double-blind, placebo-controlled trial. Breast Cancer Res. Treat. 2014, 145, 683–695. [Google Scholar] [CrossRef]
- Ismail, S.A.; Mowafi, H.A. Melatonin provides anxiolysis, enhances analgesia, decreases intraocular pressure, and promotes better operating conditions during cataract surgery under topical anesthesia. Anesth. Analg. 2009, 108, 1146–1151. [Google Scholar] [CrossRef] [PubMed]
- Ivry, M.; Goitein, D.; Welly, W.; Berkenstadt, H. Melatonin premedication improves quality of recovery following bariatric surgery—A double blind placebo controlled prospective study. Surg. Obes. Relat. Dis. 2017, 13, 502–506. [Google Scholar] [CrossRef] [PubMed]
- Khezri, M.B.; Oladi, M.R.; Atlasbaf, A. Effect of melatonin and gabapentin on anxiety and pain associated with retrobulbar eye block for cataract surgery: A randomized double-blind study. Indian J. Pharmacol. 2013, 45, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Khezri, M.B.; Merate, H. The effects of melatonin on anxiety and pain scores of patients, intraocular pressure, and operating conditions during cataract surgery under topical anesthesia. Indian J. Ophthalmol. 2013, 61, 319–324. [Google Scholar] [CrossRef]
- Kirksey, M.A.; Yoo, D.; Danninger, T.; Stundner, O.; Ma, Y.; Memtsoudis, S.G. Impact of melatonin on sleep and pain after total knee arthroplasty under regional anesthesia with sedation: A double-blind, randomized, placebo-controlled pilot study. J. Arthroplast. 2015, 30, 2370–2375. [Google Scholar] [CrossRef]
- Laflı Tunay, D.; Türkeün Ilgınel, M.; Ünlügenç, H.; Tunay, M.; Karacaer, F.; Biricik, E. Comparison of the effects of preoperative melatonin or vitamin c administration on postoperative analgesia. Bosn. J. Basic Med. Sci. 2019, 20, 117–124. [Google Scholar] [CrossRef]
- Lund Rasmussen, C.; Klee Olsen, M.; Thit Johnsen, A.; Aagaard Petersen, M.; Lindholm, H.; Andersen, L.; Villadsen, B.; Groenvold, M.; Pedersen, L. Effects of melatonin on physical fatigue and other symptoms in patients with advanced cancer receiving palliative care: A double-blind placebo-controlled crossover trial. Cancer 2015, 121, 3727–3736. [Google Scholar] [CrossRef]
- Mistraletti, G.; Umbrello, M.; Sabbatini, G.; Miori, S.; Taverna, M.; Cerri, B.; Mantovani, E.S.; Formenti, P.; Spanu, P.; D’Agostino, A.; et al. Melatonin reduces the need for sedation in icu patients: A randomized controlled trial. Minerva Anestesiol. 2015, 81, 1298–1310. [Google Scholar] [PubMed]
- Mowafi, H.A.; Ismail, S.A. Melatonin improves tourniquet tolerance and enhances postoperative analgesia in patients receiving intravenous regional anesthesia. Anesth. Analg. 2008, 107, 1422–1426. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.C.S.; Souza, A.; Dos Santos, V.S.; Cavalheiro, J.A.C.; Schuh, F.; Zucatto, A.E.; Biazus, J.V.; Da Torres, I.L.S.; Fregni, F.; Caumo, W. The effects of melatonin on the descending pain inhibitory system and neural plasticity markers in breast cancer patients receiving chemotherapy: Randomized, double-blinded, placebo-controlled trial. Front. Pharmacol. 2019, 10, 1382. [Google Scholar] [CrossRef]
- Schwertner, A.; Conceicao Dos Santos, C.C.; Costa, G.D.; Deitos, A.; de Souza, A.; de Souza, I.C.; Torres, I.L.; da Cunha Filho, J.S.; Caumo, W. Efficacy of melatonin in the treatment of endometriosis: A phase ii, randomized, double-blind, placebo-controlled trial. Pain 2013, 154, 874–881. [Google Scholar] [CrossRef] [PubMed]
- Seet, E.; Liaw, C.M.; Tay, S.; Su, C. Melatonin premedication versus placebo in wisdom teeth extraction: A randomised controlled trial. Singapore Med. J. 2015, 56, 666–671. [Google Scholar] [CrossRef] [PubMed]
- Song, G.H.; Leng, P.H.; Gwee, K.A.; Moochhala, S.M.; Ho, K.Y. Melatonin improves abdominal pain in irritable bowel syndrome patients who have sleep disturbances: A randomised, double blind, placebo controlled study. Gut 2005, 54, 1402–1407. [Google Scholar] [CrossRef]
- Varoni, E.M.; Lo Faro, A.F.; Lodi, G.; Carrassi, A.; Iriti, M.; Sardella, A. Melatonin treatment in patients with burning mouth syndrome: A triple-blind, placebo-controlled, crossover randomized clinical trial. J. Oral Facial Pain Headache 2018, 32, 178–188. [Google Scholar] [CrossRef]
- Vidor, L.P.; Torres, I.L.; Custodio de Souza, I.C.; Fregni, F.; Caumo, W. Analgesic and sedative effects of melatonin in temporomandibular disorders: A double-blind, randomized, parallel-group, placebo-controlled study. J. Pain Symptom Manage. 2013, 46, 422–432. [Google Scholar] [CrossRef]
- Vij, V.; Dahiya, D.; Kaman, L.; Behera, A. Efficacy of melatonin on sleep quality after laparoscopic cholecystectomy. Indian J. Pharmacol. 2018, 50, 236–241. [Google Scholar]
- Srinivasan, V.; Pandi-Perumal, S.R.; Spence, D.W.; Moscovitch, A.; Trakht, I.; Brown, G.M.; Cardinali, D.P. Potential use of melatonergic drugs in analgesia: Mechanisms of action. Brain Res. Bull. 2010, 81, 362–371. [Google Scholar] [CrossRef]
- Weaver, D.R.; Rivkees, S.A.; Reppert, S.M. Localization and characterization of melatonin receptors in rodent brain by in vitro autoradiography. J. Neurosci. 1989, 9, 2581–2590. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.M.; Hannah, L.T.; Hastings, M.H.; Maywood, E.S. Melatonin receptors in the rat brain and pituitary. J. Pineal Res. 1995, 19, 173–177. [Google Scholar] [CrossRef]
- Zahn, P.K.; Lansmann, T.; Berger, E.; Speckmann, E.J.; Musshoff, U. Gene expression and functional characterization of melatonin receptors in the spinal cord of the rat: Implications for pain modulation. J. Pineal Res. 2003, 35, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Ayar, A.; Martin, D.J.; Ozcan, M.; Kelestimur, H. Melatonin inhibits high voltage activated calcium currents in cultured rat dorsal root ganglion neurones. Neurosci. Lett. 2001, 313, 73–77. [Google Scholar] [CrossRef]
- Latremoliere, A.; Woolf, C.J. Central sensitization: A generator of pain hypersensitivity by central neural plasticity. J. Pain 2009, 10, 895–926. [Google Scholar] [CrossRef]
- Nelson, C.S.; Marino, J.L.; Allen, C.N. Melatonin receptors activate heteromeric g-protein coupled kir3 channels. Neuroreport 1996, 7, 717–720. [Google Scholar] [CrossRef]
- Vanecek, J. Cellular mechanisms of melatonin action. Physiol. Rev. 1998, 78, 687–721. [Google Scholar] [CrossRef]
- Mantovani, M.; Kaster, M.P.; Pertile, R.; Calixto, J.B.; Rodrigues, A.L.; Santos, A.R. Mechanisms involved in the antinociception caused by melatonin in mice. J. Pineal Res. 2006, 41, 382–389. [Google Scholar] [CrossRef]
- Garcia-Perganeda, A.; Guerrero, J.M.; Rafii-El-Idrissi, M.; Paz Romero, M.; Pozo, D.; Calvo, J.R. Characterization of membrane melatonin receptor in mouse peritoneal macrophages: Inhibition of adenylyl cyclase by a pertussis toxin-sensitive g protein. J. Neuroimmunol. 1999, 95, 85–94. [Google Scholar] [CrossRef]
- Pozo, D.; Reiter, R.J.; Calvo, J.R.; Guerrero, J.M. Inhibition of cerebellar nitric oxide synthase and cyclic gmp production by melatonin via complex formation with calmodulin. J. Cell. Biochem. 1997, 65, 430–442. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Qi, W.; Manchester, L.C.; Karbownik, M.; Calvo, J.R. Pharmacology and physiology of melatonin in the reduction of oxidative stress in vivo. Biol. Signals Recept. 2000, 9, 160–171. [Google Scholar] [CrossRef]
- Stefani, L.C.; Muller, S.; Torres, I.L.; Razzolini, B.; Rozisky, J.R.; Fregni, F.; Markus, R.; Caumo, W. A phase ii, randomized, double-blind, placebo controlled, dose-response trial of the melatonin effect on the pain threshold of healthy subjects. PLoS ONE 2013, 8, e74107. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.P.; Gogenur, I.; Fenger, A.Q.; Petersen, M.C.; Rosenberg, J.; Werner, M.U. Analgesic and antihyperalgesic effects of melatonin in a human inflammatory pain model: A randomized, double-blind, placebo-controlled, three-arm crossover study. Pain 2015, 156, 2286–2294. [Google Scholar] [CrossRef] [PubMed]
- Harpsoe, N.G.; Andersen, L.P.; Gogenur, I.; Rosenberg, J. Clinical pharmacokinetics of melatonin: A systematic review. Eur. J. Clin. Pharmacol. 2015, 71, 901–909. [Google Scholar] [CrossRef] [PubMed]
- Knaggs, R.D. Pain after surgery. Br. J. Pain 2017, 11, 159. [Google Scholar] [CrossRef] [PubMed]
- Woolf, C.J. Central sensitization: Implications for the diagnosis and treatment of pain. Pain 2011, 152, S2–S15. [Google Scholar] [CrossRef]
| Source | Country | Participants (Mean Age, y; Women, %) | Intervention vs. Control | Duration of Medication | Scoring Instrument (Scale); Endpoint | Change in Pain Score (SD) | |
|---|---|---|---|---|---|---|---|
| Intervention Group | Placebo Group | ||||||
| Altiparmak 2019 [11] | Turkey | 80 patients with neuropathic pain (49.3; 48.8) | Melatonin 3 mg PO + gabapentin 900 mg daily vs.placebo + gabapentin 900 mg daily | 30 days | VRS (0–10); secondary | −4.1 (1.3) | −3.7 (1.3) |
| Anderson 2014 [12] | Denmark | 40 patients undergoing laparoscopic cholecystectomy (45; 100) | Melatonin 10 mg IV vs. placebo | At the time of surgical incision | NA | NA | NA |
| Borazan 2010 [13] | Turkey | 52 patients undergoing open prostatectomy (57; 0) | Melatonin 6 mg PO vs. placebo | The night before and 60 min before surgery | VAS (0–100); secondary | 27 (1.0) | 47 (2.0) |
| Capuzzo 2006 [14] | Italy | 150 patients undergoing surgery (73.2, 52) | Melatonin 10 mg PO vs. placebo | 90 min before surgery | VAS (0–10); secondary | 2 (3.0) | 1 (1.5) |
| Caumo 2007 [15] | Brazil | 35 patients undergoing total abdominal hysterectomy (44.8, 100) | Melatonin 5 mg PO vs. placebo | The night before and 60 min before surgery | VAS (0–100); primary | 30 (16.5) | 47 (16.0) |
| Caumo 2009 [16] | Brazil | 63 patients undergoing total abdominal hysterectomy (43.4, 100) | Melatonin 5 mg PO vs. clonidine 100 ug PO vs. placebo | The night before and 60 min before surgery | VAS (0–100); primary | 30 (22.4) | 50 (22.4) |
| Ebrahimi 2017 [17] | Iran | 105 patients with migraine (38.9, 49) | Melatonin 3 mg PO daily vs. Sodium valproate 200 mg vs. placebo | 8 weeks | VAS (0–10); primary | −3.8 (2.8) | −1.3 (3.4) |
| Esmat 2016 [18] | Egypt | 75 patients undergoing lumbar laminectomy (44.6, 48) | Melatonin 7 mg/8 h patch vs. Fentanyl 50 ug/h patch vs. placebo | From 2 h before surgery to 12 h after surgery | VAS (0–10); primary | 3 (0.7) | 4 (0.7) |
| Esmat 2016 [19] | Egypt | 62 patients undergoing laparoscopic cholecystectomy (44.3, 100) | Melatonin 7 mg/8 h patch vs. Nicotine 15 mg/16 h patch vs. placebo | From 2 h before surgery to 12 h after surgery | VAS (0–10); primary | 4 (0.7) | 4.5 (2.2) |
| Gogenur 2009 [20] | Denmark | 136 patients undergoing laparoscopic cholecystectomy (44, 71) | Melatonin 5 mg PO daily vs. placebo | 3 days | VAS (0–100); secondary | 51 (24.5) | 48 (25.0) |
| Gonçalves 2016 [21] | Brazil | 196 patients with migraine (36.9; 44) | Melatonin 3 mg PO daily vs. Amitriptyline 25 mg vs. placebo | 12 weeks | NRS (0–10); secondary | −3.5 (3.95) | −1.8 (3.61) |
| Grima 2018 [22] | Australia | 66 patients with traumatic brain injury (37; 33) | Melatonin 2 mg PO daily vs. placebo | 4 weeks | SF-36 (0–100); secondary | 2.07 (17.64) | 1.27 (17.64) |
| Haddadi 2018 [23] | Iran | 180 patients undergoing retrobulbar eye block for cataract surgery (63.6; 56) | Melatonin 6 mg PO vs. placebo | 60 min before surgery | NA | NA | NA |
| Hansen 2014 [24] | Denmark | 54 patients undergoing breast cancer surgery (51, 100) | Melatonin 6 mg PO daily vs. placebo | From 1 week before surgery to 12 weeks after surgery | VAS (0–100); secondary | 97 (100.7) | 130 (277.8) |
| Ismail 2009 [25] | Saudi Arabia | 40 patients undergoing cataract surgery (72.8, 48) | Melatonin 10 mg PO vs. placebo | 90 min before surgery | VAS (0–100); primary | 30 (14.8) | 30 (11.1) |
| Ivry 2016 [26] | Israel | 60 patients undergoing bariatric surgery (43, 46) | Melatonin 5 mg PO vs. placebo | Night before and 2 h before surgery | QoR-15 (0–10); primary | 3.9 (5.4) | 7.5 (4.9) |
| Khezri 2013 [27] | Iran | 120 patients undergoing retrobulbar eye block for cataract surgery (73; 38) | Melatonin 6 mg PO vs. gabapentin 600 mg PO vs. placebo | 90 min before surgery | VPS (0–10); primary | 4.0 (5.9) | 4.0 (6.3) |
| Khezri 2013 [28] | Iran | 60 patients undergoing cataract surgery (63.5; 60) | Melatonin 3 mg SL vs. placebo | 60 min before surgery | VPS (0–100); secondary | 10.0 (7.4) | 10.0 (7.4) |
| Kirksey 2015 [29] | United States | 37 patients undergoing total knee arthroplasty (70; 73.7) | Melatonin 5 mg PO vs. placebo | 6 days | NA | NA | NA |
| Laflı 2019 [30] | Turkey | 165 patients undergoing major abdominal surgery (47.3; 75) | Melatonin 6 mg PO vs. Vitamin C 2 g PO vs. Placebo | 1 h before surgery | VAS (0–10); primary | 3.04 (1.82) | 4.75 (2.09) |
| Lund 2015 [31] | Denmark | 72 patients with advanced cancer (64; 66) | Melatonin 20 mg PO daily vs. placebo | 15 days | EORTC QLQ-C15-PAL (0–100); secondary | 0.8 (19.3) | 1.9 (22.2) |
| Mistraletti 2015 [32] | Italy | 82 critically ill patients requiring invasive or non-invasive respiratory assistance | Melatonin 6 mg PO daily vs. placebo | From the third intensive care unit (ICU) day to ICU discharge | NA | NA | NA |
| Mowafi 2008 [33] | Saudi Arabia | 40 patients with tourniquet-related pain for hand surgery (44.6; 45) | Melatonin 10 mg PO vs. placebo | 90 min before surgery | VPS (0–100); primary | 30.0 (7.4) | 40.0 (11.1) |
| Palmer 2019 [34] | Brazil | 36 patients with breast cancer receiving adjuvant chemotherapy (54.2; 100) | Melatonin 20 mg PO daily vs. placebo | 10 days | NRS (0–10); primary | −3.25 (1.16) | −1.91 (1.60) |
| Schwertner 2013 [35] | Brazil | 40 patients with endometriosis (36.8; 100) | Melatonin 10 mg PO daily vs. placebo | 8 weeks | VAS (0–10); primary | −3.08 (3.62) | −1.16 (3.13) |
| Seet 2015 [36] | Singapore | 76 patients undergoing all four third molar teeth extraction (22.7; 33) | Melatonin 6 mg PO vs. placebo | 90 min before surgery | VAS (0–100); primary | 11.3 (11.0) | 13.2 (11.0) |
| Song 2005 [37] | Singapore | 42 patients with irritable bowel syndrome (27.2; 60) | Melatonin 3 mg PO daily vs. placebo | 2 weeks | NRS (0–10); primary | −2.35 (1.34) | −0.70 (1.12) |
| Varoni 2018 [38] | Italy | 20 patients with burning mouth syndrome (64.4; 80) | Melatonin 12 mg PO daily | 8 weeks | VAS (0–10); Primary | 0.6 (2.2) | 1.2 (1.8) |
| Vidor 2013 [39] | Brazil | 32 patients with temporomandibular disorders (32.3; NR) | Melatonin 5 mg PO daily vs. placebo | 4 weeks | VAS (0–10); primary | −2.55 (2.96) | −0.91 (2.92) |
| Vij 2018 [40] | India | 100 patients undergoing laparoscopic cholecystectomy (42.8; 74) | Melatonin 5 mg PO daily vs. Placebo | 3 days | VAS (0–100); Secondary | 30.0 (12.5) | 30.0 (15.8) |
| Source | Random Sequence Generation | Allocation Concealment | Blinding of Participants, and Personnel | Blinding of Outcome Assessment | Incomplete Outcome Data | Selective Reporting | Other Bias | No. of Low Risk of Bias |
|---|---|---|---|---|---|---|---|---|
| Altiparmak 2019 [11] | low | unclear | unclear | unclear | low | low | low | 4 |
| Anderson 2014 [12] | low | unclear | unclear | unclear | low | low | low | 4 |
| Borazan 2010 [13] | low | unclear | unclear | low | low | low | low | 5 |
| Capuzzo 2006 [14] | low | low | low | low | low | low | low | 7 |
| Caumo 2007 [15] | low | unclear | unclear | low | low | low | low | 5 |
| Caumo 2009 [16] | low | low | unclear | low | low | low | low | 6 |
| Ebrahimi 2017 [17] | low | low | unclear | low | unclear | low | low | 5 |
| Esmat 2016 [18] | low | low | unclear | low | low | low | low | 6 |
| Esmat 2016 [19] | low | low | unclear | low | low | low | low | 6 |
| Gögenur 2009 [20] | low | low | low | low | low | low | low | 7 |
| Gonçalves 2016 [21] | low | low | low | low | low | low | low | 7 |
| Grima 2018 [22] | unclear | low | low | low | low | low | low | 6 |
| Haddadi 2018 [23] | unclear | unclear | unclear | unclear | low | low | low | 3 |
| Hansen 2014 [24] | low | low | low | low | low | low | low | 7 |
| Ismail 2009 [25] | low | unclear | high | low | low | low | low | 5 |
| Ivry 2016 [26] | low | unclear | unclear | low | low | low | low | 5 |
| Khezri 2013 [27] | low | low | unclear | low | low | low | low | 6 |
| Khezri 2013 [28] | low | unclear | unclear | low | low | low | low | 5 |
| Kirksey 2015 [29] | unclear | low | low | low | low | low | low | 6 |
| Laflı 2019 [30] | low | unclear | unclear | low | low | low | low | 5 |
| Lund 2015 [31] | low | low | low | low | low | low | low | 7 |
| Mistraletti 2015 [32] | low | low | low | low | low | low | low | 7 |
| Mowafi 2008 [33] | low | low | unclear | low | low | low | low | 6 |
| Palmer 2019 [34] | low | low | low | low | low | low | low | 7 |
| Schwertner 2013 [35] | low | low | low | low | low | low | low | 7 |
| Seet 2015 [36] | low | low | low | low | low | low | low | 7 |
| Song 2005 [37] | unclear | low | low | low | low | low | low | 6 |
| Varoni 2018 [38] | low | unclear | low | low | low | low | low | 6 |
| Vidor 2013 [39] | low | low | low | low | low | low | low | 7 |
| Vij 2018 [40] | low | unclear | unclear | unclear | unclear | low | low | 3 |
| Type of Pain | No. of Low Risk of Bias | No. of Trials | Summary SMD (95% CI) | Heterogeneity, I2 (%) |
|---|---|---|---|---|
| Acute pain—postoperative, local or epidural anesthesia | ≥ 6 | 1 [15] | −0.893 (−1.544, −0.241) | NA |
| < 6 | 2 [23,26] | 0.000 (−0.392, 0.392) | 0 | |
| Acute pain—postoperative, general anesthesia | ≥ 6 | 6 [13,17,18,19,22,32] | −0.212 (−0.664, 0.240) | 82.4 |
| < 6 | 5 [12,14,24,27,36] | −2.134 (−3.456, −0.812) | 96.0 | |
| Chronic pain | ≥ 6 | 4 [20,31,33,34] | −0.618 (−1.011, −0.225) | 49.3 |
| < 6 | 1 [16] | −0.803 (−1.291, −0.316) | NA |
| Type of Pain | No. of Low Risk of Bias | No. of Trials | Summary SMD (95% CI) | Heterogeneity, I2 (%) |
|---|---|---|---|---|
| Acute pain—procedural | ≥ 6 | 2 [32,33] | −0.527 (−1.517, 0.464) | 84.0 |
| < 6 | 1 [23] | −3.262 (−3.818, −2.706) | NA | |
| Acute pain—postoperative, general anesthesia | ≥ 6 | 4 [12,18,19,20] | −4.676 (−7.546, −1.806) | 97.9 |
| < 6 | 3 [13,25,30] | −1.271 (−2.087, −0.455) | 83.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, S.N.; Myung, S.-K.; Jho, H.J. Analgesic Efficacy of Melatonin: A Meta-Analysis of Randomized, Double-Blind, Placebo-Controlled Trials. J. Clin. Med. 2020, 9, 1553. https://doi.org/10.3390/jcm9051553
Oh SN, Myung S-K, Jho HJ. Analgesic Efficacy of Melatonin: A Meta-Analysis of Randomized, Double-Blind, Placebo-Controlled Trials. Journal of Clinical Medicine. 2020; 9(5):1553. https://doi.org/10.3390/jcm9051553
Chicago/Turabian StyleOh, Si Nae, Seung-Kwon Myung, and Hyun Jung Jho. 2020. "Analgesic Efficacy of Melatonin: A Meta-Analysis of Randomized, Double-Blind, Placebo-Controlled Trials" Journal of Clinical Medicine 9, no. 5: 1553. https://doi.org/10.3390/jcm9051553
APA StyleOh, S. N., Myung, S.-K., & Jho, H. J. (2020). Analgesic Efficacy of Melatonin: A Meta-Analysis of Randomized, Double-Blind, Placebo-Controlled Trials. Journal of Clinical Medicine, 9(5), 1553. https://doi.org/10.3390/jcm9051553





