Application of Adult and Pluripotent Stem Cells in Interstitial Cystitis/Bladder Pain Syndrome Therapy: Methods and Perspectives
Abstract
1. Introduction
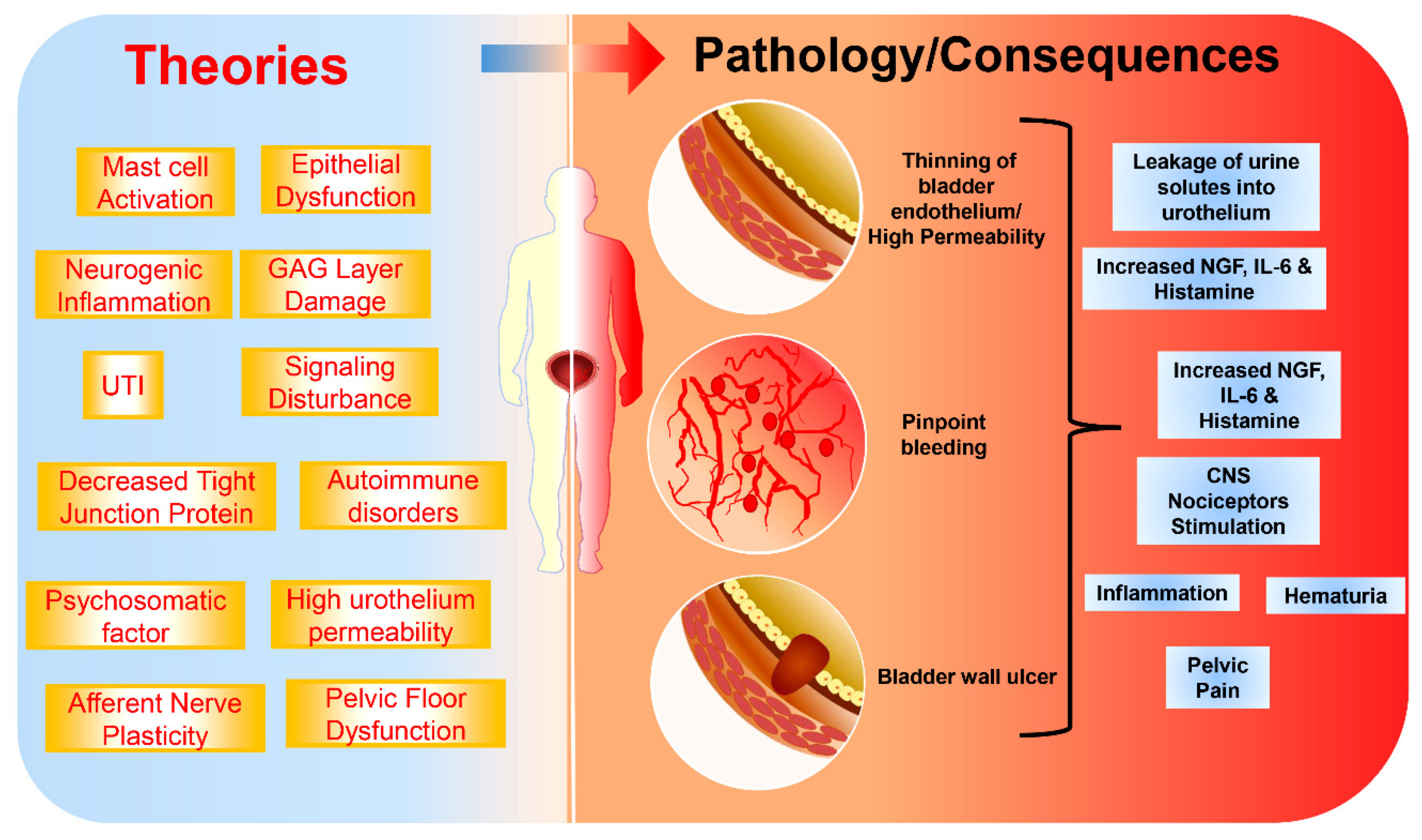
2. Etiology and Pathophysiology of IC/BPS
3. IC/BPS Diagnosis
4. IC/BPS Therapeutic Approaches
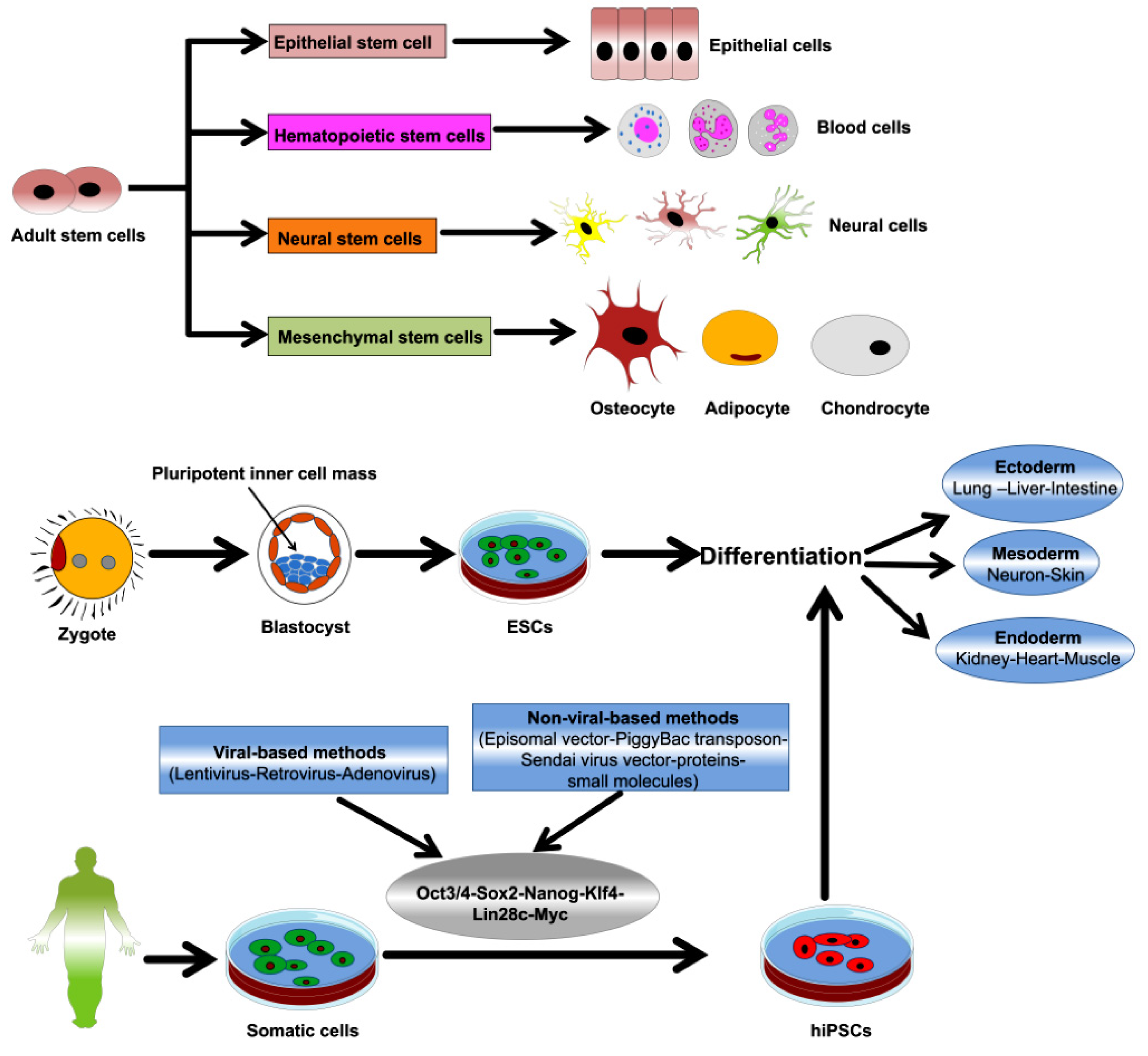
5. Overview of ASCs and PSCs
6. ACSs and IC/BPS Therapy
6.1. Differentiation into Bladder Cells
6.2. Direct Transplantation
6.3. Bladder Augmentation
7. PSCs in Bladder Regeneration and IC/BPS Therapy
7.1. Differentiation into Bladder Cells
7.2. Direct Transplantation
8. Conclusions and Future Prospects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chancellor, M.B.; Yoshimura, N. Treatment of interstitial cystitis. Urology 2004, 63, 85–92. [Google Scholar] [CrossRef]
- Nickel, J.C.; Tripp, D.A.; Pontari, M.; Moldwin, R.; Mayer, R.; Carr, L.K.; Doggweiler, R.; Yang, C.C.; Mishra, N.; Nordling, J. Psychosocial phenotyping in women with interstitial cystitis/painful bladder syndrome: A case control study. J. Urol. 2010, 183, 167–172. [Google Scholar] [CrossRef]
- Simon, L.J.; Landis, J.R.; Erickson, D.R.; Nyberg, L.M.; Group, I.S. The Interstitial Cystitis Data Base Study: Concepts and preliminary baseline descriptive statistics. Urology 1997, 49, 64–75. [Google Scholar] [CrossRef]
- Koziol, J.A.; Clark, D.C.; Gittes, R.F.; Tan, E.M. The natural history of interstitial cystitis: A survey of 374 patients. J. Urol. 1993, 149, 465–469. [Google Scholar] [CrossRef]
- Persu, C.; Cauni, V.; Gutue, S.; Blaj, I.; Jinga, V.; Geavlete, P. From interstitial cystitis to chronic pelvic pain. J. Med. Life 2010, 3, 167. [Google Scholar]
- Ogawa, T.; Ishizuka, O.; Ueda, T.; Tyagi, P.; Chancellor, M.B.; Yoshimura, N. Current and emerging drugs for interstitial cystitis/bladder pain syndrome (IC/BPS). Expert Opin. Emerg. Drugs 2015, 20, 555–570. [Google Scholar] [CrossRef]
- Bosch, P.C.; Bosch, D.C. Treating interstitial cystitis/bladder pain syndrome as a chronic disease. Rev. Urol. 2014, 16, 83–87. [Google Scholar]
- Robinson, R.; Montejano, L.; Ruiz, K.; Lenhart, G. 317 The economic burden of interstitial cystitis and painful bladder syndrome. J. Urol. 2011, 185, e129. [Google Scholar] [CrossRef]
- Anger, J.T.; Zabihi, N.; Clemens, J.Q.; Payne, C.K.; Saigal, C.S.; Rodriguez, L.V. Treatment choice, duration, and cost in patients with interstitial cystitis and painful bladder syndrome. Int. Urogynecol. J. 2011, 22, 395–400. [Google Scholar] [CrossRef]
- Hu, P.; Meyers, S.; Liang, F.-X.; Deng, F.-M.; Kachar, B.; Zeidel, M.L.; Sun, T.-T. Role of membrane proteins in permeability barrier function: Uroplakin ablation elevates urothelial permeability. Am. J. Physiol. Ren. Physiol. 2002, 283, F1200–F1207. [Google Scholar] [CrossRef]
- Višnjar, T.; Kocbek, P.; Kreft, M.E. Hyperplasia as a mechanism for rapid resealing urothelial injuries and maintaining high transepithelial resistance. Histochem. Cell Biol. 2012, 137, 177–186. [Google Scholar] [CrossRef]
- Sun, T.-T.; Zhao, H.; Provet, J.; Aebi, U.; Wu, X.-R. Formation of asymmetric unit membrane during urothelial differentiation. Mol. Biol. Rep. 1996, 23, 3–11. [Google Scholar] [CrossRef]
- Wu, X.-R.; Kong, X.-P.; Pellicer, A.; Kreibich, G.; Sun, T.-T. Uroplakins in urothelial biology, function, and disease. Kidney Int. 2009, 75, 1153–1165. [Google Scholar] [CrossRef]
- Osborn, S.L.; Thangappan, R.; Luria, A.; Lee, J.H.; Nolta, J.; Kurzrock, E.A. Induction of Human Embryonic and Induced Pluripotent Stem Cells Into Urothelium. Stem Cells Transl. Med. 2014, 3, 610–619. [Google Scholar] [CrossRef]
- Colopy, S.A.; Bjorling, D.E.; Mulligan, W.A.; Bushman, W. A population of progenitor cells in the basal and intermediate layers of the murine bladder urothelium contributes to urothelial development and regeneration. Dev. Dyn. 2014, 243, 988–998. [Google Scholar] [CrossRef]
- Wang, C.; Ross, W.T.; Mysorekar, I.U. Urothelial generation and regeneration in development, injury, and cancer. Dev. Dyn. 2017, 246, 336–343. [Google Scholar] [CrossRef]
- Ogawa, T.; Ishizuka, O.; Ueda, T.; Tyagi, P.; Chancellor, M.B.; Yoshimura, N. Pharmacological management of interstitial cystitis/bladder pain syndrome and the role cyclosporine and other immunomodulating drugs play. Expert Rev. Clin. Pharmacol. 2018, 11, 495–505. [Google Scholar] [CrossRef]
- Teichman, J.; Moldwin, R. The role of the bladder surface in interstitial cystitis/painful bladder syndrome. Can. J. Urol. 2007, 14, 3599–3607. [Google Scholar]
- Neuhaus, J.; Heinrich, M.; Schlichting, N.; Oberbach, A.; Fitzl, G.; Schwalenberg, T.; Horn, L.; Stolzenburg, J. Structure and function of suburothelial myofibroblasts in the human urinary bladder under normal and pathological conditions. Der Urologe. Ausg. A 2007, 46, 1197–1202. [Google Scholar] [CrossRef]
- Parsons, C.L. The role of a leaky epithelium and potassium in the generation of bladder symptoms in interstitial cystitis/overactive bladder, urethral syndrome, prostatitis and gynaecological chronic pelvic pain. BJU Int. 2011, 107, 370–375. [Google Scholar] [CrossRef]
- Birder, L.A.; Kullmann, F.A. Role of neurogenic inflammation in local communication in the visceral mucosa. In Seminars in Immunopathology; Springer: Berlin/Heidelberg, Germany, 2018; pp. 261–279. [Google Scholar]
- Kim, D.S. Potential Mechanisms Underlying the Increased Excitability of the Bladder Afferent Pathways in Interstitial Cystitis/Bladder Pain Syndrome. Urogenit. Tract Infect. 2018, 13, 26–34. [Google Scholar] [CrossRef]
- Hunner, G.L. A rare type of bladder ulcer in women; report of cases. Boston Med Surg. J. 1915, 172, 660–664. [Google Scholar] [CrossRef]
- Engeler, D.; Baranowski, A.; Borovicka, J.; Cottrell, A.; Dinis-Oliveira, P.; Elneil, S.; Hughes, J.; Messelink, E.; van Ophoven, A.; Reisman, Y. EAU guidelines on chronic pelvic pain. Eur. Urol. 2012, 57, 35–48. [Google Scholar]
- Fall, M.; Logadottir, Y.; Peeker, R. Interstitial cystitis is bladder pain syndrome with H unner’s lesion. Int. J. Urol. 2014, 21, 79–82. [Google Scholar] [CrossRef]
- Logadottir, Y.; Fall, M.; Kåbjörn-Gustafsson, C.; Peeker, R. Clinical characteristics differ considerably between phenotypes of bladder pain syndrome/interstitial cystitis. Scand. J. Urol. Nephrol. 2012, 46, 365–370. [Google Scholar] [CrossRef]
- McDermott, P. Painful bladder syndrome/interstitial cystitis (history, epidemiology, symptoms, diagnosis and treatments). Int. J. Urol. Nurs. 2009, 3, 16–23. [Google Scholar] [CrossRef]
- Suskind, A.M.; Berry, S.H.; Ewing, B.A.; Elliott, M.N.; Suttorp, M.J.; Clemens, J.Q. The prevalence and overlap of interstitial cystitis/bladder pain syndrome and chronic prostatitis/chronic pelvic pain syndrome in men: Results of the RAND Interstitial Cystitis Epidemiology male study. J. Urol. 2013, 189, 141–145. [Google Scholar] [CrossRef]
- Clemens, J.Q.; Clauw, D.J.; Kreder, K.; Krieger, J.N.; Kusek, J.W.; Lai, H.H.; Rodriguez, L.; Williams, D.A.; Hou, X.; Stephens, A.; et al. Comparison of baseline urological symptoms in men and women in the MAPP research cohort. J. Urol. 2015, 193, 1554–1558. [Google Scholar] [CrossRef]
- Clemens, J.Q.; Link, C.L.; Eggers, P.W.; Kusek, J.W.; Nyberg, L.M.; McKinlay, J.B.; Investigators, B.S. Prevalence of painful bladder symptoms and effect on quality of life in black, Hispanic and white men and women. J. Urol. 2007, 177, 1390–1394. [Google Scholar] [CrossRef]
- Berry, S.H.; Elliott, M.N.; Suttorp, M.; Bogart, L.M.; Stoto, M.A.; Eggers, P.; Nyberg, L.; Clemens, J.Q. Prevalence of symptoms of bladder pain syndrome/interstitial cystitis among adult females in the United States. J. Urol. 2011, 186, 540–544. [Google Scholar] [CrossRef]
- Konkle, K.S.; Berry, S.H.; Elliott, M.N.; Hilton, L.; Suttorp, M.J.; Clauw, D.J.; Clemens, J.Q. Comparison of an interstitial cystitis/bladder pain syndrome clinical cohort with symptomatic community women from the RAND Interstitial Cystitis Epidemiology study. J. Urol. 2012, 187, 508–512. [Google Scholar] [CrossRef]
- Hendrix, S. Neuroimmune communication in skin: Far from peripheral. J. Investig. Dermatol. 2008, 128, 260–261. [Google Scholar] [CrossRef]
- Van De Merwe, J.P. Interstitial cystitis and systemic autoimmune diseases. Nat. Clin. Pract. Urol. 2007, 4, 484–491. [Google Scholar] [CrossRef]
- Malykhina, A.P.; Qin, C.; Greenwood-van Meerveld, B.; Foreman, R.; Lupu, F.; Akbarali, H. Hyperexcitability of convergent colon and bladder dorsal root ganglion neurons after colonic inflammation: Mechanism for pelvic organ cross-talk. Neurogastroenterol. Motil. 2006, 18, 936–948. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Shin, D.-M.; Choo, M.-S. Stem Cell Therapy for interstitial cystitis/bladder pain syndrome. Curr. Urol. Rep. 2016, 17, 1. [Google Scholar] [CrossRef] [PubMed]
- Kanter, G.; Komesu, Y.M.; Qaedan, F.; Jeppson, P.C.; Dunivan, G.C.; Cichowski, S.B.; Rogers, R.G. Mindfulness-based stress reduction as a novel treatment for interstitial cystitis/bladder pain syndrome: A randomized controlled trial. Int. Urogynecol. J. 2016, 27, 1705–1711. [Google Scholar] [CrossRef]
- Lutgendorf, S.K.; Kreder, K.J.; Rothrock, N.E.; Ratliff, T.L.; Zimmerman, B. Stress and symptomatology in patients with interstitial cystitis: A laboratory stress model. J. Urol. 2000, 164, 1265–1269. [Google Scholar] [CrossRef]
- Pierce, A.N.; Christianson, J.A. Stress and chronic pelvic pain. In Progress in Molecular Biology and Translational Science; Elsevier: Amsterdam, The Netherlands, 2015; Volume 131, pp. 509–535. [Google Scholar]
- Birder, L.A. Pathophysiology of interstitial cystitis. Int. J. Urol. 2019, 26, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Petrovic, V.; Stankovic, J.; Stefanovic, V. Tissue engineering of the urinary bladder: Current concepts and future perspectives. Sci. World J. 2011, 11, 1479–1488. [Google Scholar] [CrossRef] [PubMed]
- Koh, C.J.; Atala, A. Tissue engineering, stem cells, and cloning: Opportunities for regenerative medicine. J. Am. Soc. Nephrol. 2004, 15, 1113–1125. [Google Scholar] [CrossRef] [PubMed]
- Hautmann, R.E. Urinary diversion: Ileal conduit to neobladder. J. Urol. 2003, 169, 834–842. [Google Scholar] [CrossRef] [PubMed]
- Atala, A.; Bauer, S.B.; Hendren, W.H.; Retik, A.B. The effect of gastric augmentation on bladder function. J. Urol. 1993, 149, 1099–1102. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, S.-R.; Song, Y.S.; Lee, H.J. Stem cell therapy in bladder dysfunction: Where are we? And where do we have to go? Biomed Res. Int. 2013, 2013, 930713. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Kim, H.H.; Han, Y.-M. Generation of bladder urothelium from human pluripotent stem cells under chemically defined serum-and feeder-free system. Int. J. Mol. Sci. 2014, 15, 7139–7157. [Google Scholar] [CrossRef] [PubMed]
- McLennan, M.T. Interstitial cystitis: Epidemiology, pathophysiology, and clinical presentation. Obstet. Gynecol. Clin. 2014, 41, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J. Update on the pathology and diagnosis of interstitial cystitis/bladder pain syndrome: A review. Int. Neurourol. J. 2016, 20, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Friedlander, J.I.; Shorter, B.; Moldwin, R.M. Diet and its role in interstitial cystitis/bladder pain syndrome (IC/BPS) and comorbid conditions. BJU Int. 2012, 109, 1584–1591. [Google Scholar] [CrossRef]
- Yoshimura, N.; Oguchi, T.; Yokoyama, H.; Funahashi, Y.; Yoshikawa, S.; Sugino, Y.; Kawamorita, N.; Kashyap, M.P.; Chancellor, M.B.; Tyagi, P. Bladder afferent hyperexcitability in bladder pain syndrome/interstitial cystitis. Int. J. Urol. 2014, 21, 18–25. [Google Scholar] [CrossRef]
- Warren, J.W.; Brown, V.; Jacobs, S.; Horne, L.; Langenberg, P.; Greenberg, P. Urinary tract infection and inflammation at onset of interstitial cystitis/painful bladder syndrome. Urology 2008, 71, 1085–1090. [Google Scholar] [CrossRef] [PubMed]
- Richter, B.; Roslind, A.; Hesse, U.; Nordling, J.; Johansen, J.S.; Horn, T.; Hansen, A.B. YKL-40 and mast cells are associated with detrusor fibrosis in patients diagnosed with bladder pain syndrome/interstitial cystitis according to the 2008 criteria of the European Society for the Study of Interstitial Cystitis. Histopathology 2010, 57, 371–383. [Google Scholar] [CrossRef] [PubMed]
- Dellis, A.; Papatsoris, A.G. Intravesical treatment of bladder pain syndrome/interstitial cystitis: From the conventional regimens to the novel botulinum toxin injections. Expert Opin. Investig. Drugs 2014, 23, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Hurst, R. Structure, function, and pathology of proteoglycans and glycosaminoglycans in the urinary tract. World J. Urol. 1994, 12, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Parsons, C.L.; Greene, R.A.; Chung, M.; Stanford, E.J.; Singh, G. Abnormal urinary potassium metabolism in patients with interstitial cystitis. J. Urol. 2005, 173, 1182–1185. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Keay, S.K.; Freeman, M.R. Heparin-binding epidermal growth factor-like growth factor functionally antagonizes interstitial cystitis antiproliferative factor via mitogen-activated protein kinase pathway activation. BJU Int. 2009, 103, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, M.; Oberbach, A.; Schlichting, N.; Stolzenburg, J.-U.; Neuhaus, J. Cytokine effects on gap junction communication and connexin expression in human bladder smooth muscle cells and suburothelial myofibroblasts. PLoS ONE 2011, 6, e20792. [Google Scholar] [CrossRef]
- Birder, L.; Wolf-Johnston, A.; Buffington, C.; Roppolo, J.; De Groat, W.; Kanai, A. Altered inducible nitric oxide synthase expression and nitric oxide production in the bladder of cats with feline interstitial cystitis. J. Urol. 2005, 173, 625–629. [Google Scholar] [CrossRef]
- Keay, S.; Warren, J.; Zhang, C.; Tu, L.; Gordon, D.; Whitmore, K. Antiproliferative activity is present in bladder but not renal pelvic urine from interstitial cystitis patients. J. Urol. 1999, 162, 1487–1489. [Google Scholar] [CrossRef]
- Slobodov, G.; Feloney, M.; Gran, C.; Kyker, K.D.; Hurst, R.E.; Culkin, D.J. Abnormal expression of molecular markers for bladder impermeability and differentiation in the urothelium of patients with interstitial cystitis. J. Urol. 2004, 171, 1554–1558. [Google Scholar] [CrossRef]
- Yoshimura, N.; Seki, S.; Chancellor, M.B.; de Groat, W.C.; Ueda, T. Targeting afferent hyperexcitability for therapy of the painful bladder syndrome. Urology 2002, 59, 61–67. [Google Scholar] [CrossRef]
- Lutgendorf, S.; Kreder, K.; Rothrock, N.; Ratliff, T.; Zimmerman, B. A laboratory stress model for examining stress and symptomatology in interstitial cystitis patients. Urology 2001, 57, 122. [Google Scholar] [CrossRef]
- Denk, F.; McMahon, S.B. Chronic pain: Emerging evidence for the involvement of epigenetics. Neuron 2012, 73, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Kempuraj, D.; Sant, G.R. Mast cell involvement in interstitial cystitis: A review of human and experimental evidence. Urology 2001, 57, 47–55. [Google Scholar] [CrossRef]
- Ustinova, E.E.; Fraser, M.O.; Pezzone, M.A. Cross-talk and sensitization of bladder afferent nerves. Neurourol. Urodyn. Off. J. Int. Cont. Soc. 2010, 29, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Shea-Donohue, T.; Stiltz, J.; Zhao, A.; Notari, L. Mast cells. Curr. Gastroenterol. Rep. 2010, 12, 349–357. [Google Scholar] [CrossRef]
- Gamper, M.; Regauer, S.; Welter, J.; Eberhard, J.; Viereck, V. Are mast cells still good biomarkers for bladder pain syndrome/interstitial cystitis? J. Urol. 2015, 193, 1994–2000. [Google Scholar] [CrossRef]
- Sant, G.R.; Kempuraj, D.; Marchand, J.E.; Theoharides, T.C. The mast cell in interstitial cystitis: Role in pathophysiology and pathogenesis. Urology 2007, 69, S34–S40. [Google Scholar] [CrossRef]
- Liu, H.T.; Tyagi, P.; Chancellor, M.B.; Kuo, H.C. Urinary nerve growth factor level is increased in patients with interstitial cystitis/bladder pain syndrome and decreased in responders to treatment. BJU Int. 2009, 104, 1476–1481. [Google Scholar] [CrossRef]
- Heck, B.N. Interstitial cystitis: Enhancing early identification in primary care settings. J. Nurse Pract. 2007, 3, 509–519. [Google Scholar] [CrossRef]
- Panzera, A.K. Interstitial cystitis/painful bladder syndrome. Urol Nurs 2007, 27, 13–19. [Google Scholar]
- Theoharides, T.; Sant, G. A pilot open label study of Cystoprotek® in interstitial cystitis. Int. J. Immunopathol. Pharmacol. 2005, 18, 183–188. [Google Scholar] [CrossRef]
- Park, C.-S.; Bochner, B.S. Potential targeting of siglecs, mast cell inhibitory receptors, in interstitial cystitis. Int. Neurourol. J. 2011, 15, 61. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bosch, P.C. A Randomized, Double-blind, Placebo-controlled Trial of Certolizumab Pegol in Women with Refractory Interstitial Cystitis/Bladder Pain Syndrome. Eur. Urol. 2018, 74, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, F.S.; Gershwin, M.E. Human autoimmune diseases: A comprehensive update. J. Intern. Med. 2015, 278, 369–395. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-K.; Tsai, C.-P.; Liao, T.-L.; Huang, W.-N.; Chen, Y.-H.; Lin, C.-H.; Chen, Y.-M. Overactive bladder and bladder pain syndrome/interstitial cystitis in primary Sjögren’s syndrome patients: A nationwide population-based study. PLoS ONE 2019, 14, e0225455. [Google Scholar] [CrossRef]
- Jhang, J.-F.; Kuo, H.-C. Pathomechanism of Interstitial Cystitis/Bladder Pain Syndrome and Mapping the Heterogeneity of Disease. Int. Neurourol. J. 2016, 20, S95–S104. [Google Scholar] [CrossRef]
- Yamada, T. Significance of complications of allergic diseases in young patients with interstitial cystitis. Int. J. Urol. 2003, 10, S56–S58. [Google Scholar] [CrossRef]
- Shorter, B.; Lesser, M.; Moldwin, R.M.; Kushner, L. Effect of comestibles on symptoms of interstitial cystitis. J. Urol. 2007, 178, 145–152. [Google Scholar] [CrossRef]
- Bassaly, R.; Tidwell, N.; Bertolino, S.; Hoyte, L.; Downes, K.; Hart, S. Myofascial pain and pelvic floor dysfunction in patients with interstitial cystitis. Int. Urogynecol. J. 2011, 22, 413–418. [Google Scholar] [CrossRef]
- Hanno, P.M.; Burks, D.A.; Clemens, J.Q.; Dmochowski, R.R.; Erickson, D.; FitzGerald, M.P.; Forrest, J.B.; Gordon, B.; Gray, M.; Mayer, R.D.; et al. AUA guideline for the diagnosis and treatment of interstitial cystitis/bladder pain syndrome. J. Urol. 2011, 185, 2162–2170. [Google Scholar] [CrossRef]
- Hanno, P.M.; Erickson, D.; Moldwin, R.; Faraday, M.M. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome: AUA guideline amendment. J. Urol. 2015, 193, 1545–1553. [Google Scholar] [CrossRef]
- Van de Merwe, J.P.; Nordling, J.; Bouchelouche, P.; Bouchelouche, K.; Cervigni, M.; Daha, L.K.; Elneil, S.; Fall, M.; Hohlbrugger, G.; Irwin, P. Diagnostic criteria, classification, and nomenclature for painful bladder syndrome/interstitial cystitis: An ESSIC proposal. Eur. Urol. 2008, 53, 60–67. [Google Scholar] [CrossRef]
- Homma, Y.; Ueda, T.; Ito, T.; Takei, M.; Tomoe, H. Japanese guideline for diagnosis and treatment of interstitial cystitis. Int. J. Urol. 2009, 16, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Butrick, C.W.; Howard, F.M.; Sand, P.K. Diagnosis and treatment of interstitial cystitis/painful bladder syndrome: A review. J. Women’s Health 2010, 19, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Moutzouris, D.-A.; Falagas, M.E. Interstitial cystitis: An unsolved enigma. Clin. J. Am. Soc. Nephrol. 2009, 4, 1844–1857. [Google Scholar] [CrossRef]
- Rosenberg, M.; Parsons, C.L.; Page, S. Interstitial cystitis: A primary care perspective. Clevel. Clin. J. Med. 2005, 72, 698. [Google Scholar] [CrossRef] [PubMed]
- van de Merwe, J.P. Diagnosis of Interstitial Cystitis: A Clinical, Endoscopic and Pathologic Approach. In Bladder Pain Syndrome—An Evolution; Springer: Berlin/Heidelberg, Germany, 2018; pp. 57–62. [Google Scholar]
- Parsons, C.L.; Dell, J.; Stanford, E.J.; Bullen, M.; Kahn, B.S.; Willems, J.J. The prevalence of interstitial cystitis in gynecologic patients with pelvic pain, as detected by intravesical potassium sensitivity. Am. J. Obstet. Gynecol. 2002, 187, 1395–1400. [Google Scholar] [CrossRef]
- O’Leary, M.P.; Sant, G.R.; Fowler, F.J., Jr.; Whitmore, K.E.; Spolarich-Kroll, J. The interstitial cystitis symptom index and problem index. Urology 1997, 49, 58–63. [Google Scholar] [CrossRef]
- Kushner, L.; Moldwin, R.M. Efficiency of questionnaires used to screen for interstitial cystitis. J. Urol. 2006, 176, 587–592. [Google Scholar] [CrossRef]
- Wennevik, G.E.; Meijlink, J.M.; Hanno, P.; Nordling, J. The role of glomerulations in bladder pain syndrome: A review. J. Urol. 2016, 195, 19–25. [Google Scholar] [CrossRef]
- Cox, A.; Golda, N.; Nadeau, G.; Nickel, J.C.; Carr, L.; Corcos, J.; Teichman, J. CUA guideline: Diagnosis and treatment of interstitial cystitis/bladder pain syndrome. Can. Urol. Assoc. J. 2016, 10, E136. [Google Scholar] [CrossRef]
- Wyndaele, J.-J.; Van Dyck, J.; Toussaint, N. Cystoscopy and bladder biopsies in patients with bladder pain syndrome carried out following ESSIC guidelines. Scand. J. Urol. Nephrol. 2009, 43, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Belknap, S.; Blalock, E.; Erickson, D. The challenges of interstitial cystitis: Current status and future prospects. Drugs 2015, 75, 2057–2063. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, M.T.; Newman, D.K.; Page, S.A. Interstitial cystitis/painful bladder syndrome: Symptom recognition is key to early identification, treatment. Clevel. Clin. J. Med. 2007, 74, S54. [Google Scholar] [CrossRef] [PubMed]
- Homma, Y.; Ueda, T.; Tomoe, H.; Lin, A.T.; Kuo, H.C.; Lee, M.H.; Lee, J.G.; Kim, D.Y.; Lee, K.S.; Interstitial Cystitis Guideline Committee. Clinical guidelines for interstitial cystitis and hypersensitive bladder syndrome. Int. J. Urol. 2009, 16, 597–615. [Google Scholar] [CrossRef] [PubMed]
- Hanno, P.M.; Burks, D.A.; Clemens, J.Q.; Dmochowski, R.R.; Erickson, D.; FitzGerald, M.P.; Faraday, M. Diagnosis and Treatment of Interstitial Cystitis/Bladder Pain Syndrome. September 2014. Available online: https://www.auanet.org/guidelines/interstitial-cystitis-(ic/bps)-guideline. (accessed on 10 February 2020).
- Funaro, M.G.; King, A.N.; Stern, J.N.H.; Moldwin, R.M.; Bahlani, S. Endoscopic Injection of Low Dose Triamcinolone: A Simple, Minimally Invasive, and Effective Therapy for Interstitial Cystitis With Hunner Lesions. Urology 2018, 118, 25–29. [Google Scholar] [CrossRef]
- Hsu, C.C.; Chuang, Y.C.; Chancellor, M.B. Intravesical drug delivery for dysfunctional bladder. Int. J. Urol. 2013, 20, 552–562. [Google Scholar] [CrossRef]
- Chuang, Y.-C.; Chermansky, C.; Kashyap, M.; Tyagi, P. Investigational drugs for bladder pain syndrome (BPS)/interstitial cystitis (IC). Expert Opin. Investig. Drugs 2016, 25, 521–529. [Google Scholar] [CrossRef]
- Verghese, T.S.; Riordain, R.N.; Champaneria, R.; Latthe, P.M. Complementary therapies for bladder pain syndrome: A systematic review. Int. Urogynecol. J. 2016, 27, 1127–1136. [Google Scholar] [CrossRef]
- Andersen, A.V.; Granlund, P.; Schultz, A.; Talseth, T.; Hedlund, H.; Frich, L. Long-term experience with surgical treatment of selected patients with bladder pain syndrome/interstitial cystitis. Scand. J. Urol. Nephrol. 2012, 46, 284–289. [Google Scholar] [CrossRef]
- Lu, Q.; Jiang, F.; Xu, R.; Zhao, X.-K.; Zhong, Z.-H.; Zhang, L.; Jiang, H.-Y.; Yi, L.; Hou, Y.; Zhu, X. A pilot study on intravesical administration of curcumin for cystitis glandularis. Evid. Based Complement. Altern. Med. 2013, 2013, 269745. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Kempuraj, D.; Vakali, S.; Sant, G.R. Treatment of refractory interstitial cystitis/painful bladder syndrome with CystoProtek--an oral multi-agent natural supplement. Can. J. Urol. 2008, 15, 4410–4414. [Google Scholar] [PubMed]
- Whitmore, K.E. Complementary and alternative therapies as treatment approaches for interstitial cystitis. Rev. Urol. 2002, 4, S28. [Google Scholar] [PubMed]
- O’Hare, P.G.; Hoffmann, A.R.; Allen, P.; Gordon, B.; Salin, L.; Whitmore, K. Interstitial cystitis patients’ use and rating of complementary and alternative medicine therapies. Int. Urogynecol. J. 2013, 24, 977–982. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Sant, G.R. New agents for the medical treatment of interstitial cystitis. Expert Opin. Investig. Drugs 2001, 10, 521–546. [Google Scholar] [CrossRef]
- Han, E.; Nguyen, L.; Sirls, L.; Peters, K. Current best practice management of interstitial cystitis/bladder pain syndrome. Ther. Adv. Urol. 2018, 10, 197–211. [Google Scholar] [CrossRef]
- Cruz, F. The Future of Pharmacologic Treatment for Bladder Pain Syndrome/Interstitial Cystitis: Lessons From a Meta-Analysis. Eur. Urol. 2012, 61, 54–55. [Google Scholar] [CrossRef]
- Ratajczak, M.Z.; Ratajczak, J.; Suszynska, M.; Miller, D.M.; Kucia, M.; Shin, D.-M. A novel view of the adult stem cell compartment from the perspective of a quiescent population of very small embryonic-like stem cells. Circ. Res. 2017, 120, 166–178. [Google Scholar] [CrossRef]
- Jeong, H.; Yim, H.W.; Park, H.J.; Cho, Y.; Hong, H.; Kim, N.J.; Oh, I.H. Mesenchymal Stem Cell Therapy for Ischemic Heart Disease: Systematic Review and Meta-analysis. Int. J. Stem Cells 2018, 11, 1–12. [Google Scholar] [CrossRef]
- Keller, G. Embryonic stem cell differentiation: Emergence of a new era in biology and medicine. Genes Dev. 2005, 19, 1129–1155. [Google Scholar] [CrossRef]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef]
- Xue, Y.; Cai, X.; Wang, L.; Liao, B.; Zhang, H.; Shan, Y.; Chen, Q.; Zhou, T.; Li, X.; Hou, J. Generating a non-integrating human induced pluripotent stem cell bank from urine-derived cells. PLoS ONE 2013, 8, e70573. [Google Scholar] [CrossRef]
- Orlando, G.; Wood, K.J.; De Coppi, P.; Baptista, P.M.; Binder, K.W.; Bitar, K.N.; Breuer, C.; Burnett, L.; Christ, G.; Farney, A. Regenerative medicine as applied to general surgery. Ann. Surg. 2012, 255, 867. [Google Scholar] [CrossRef] [PubMed]
- Okita, K.; Ichisaka, T.; Yamanaka, S. Generation of germline-competent induced pluripotent stem cells. Nature 2007, 448, 313. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, S. A fresh look at iPS cells. Cell 2009, 137, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, K.; Honmou, O.; Suzuki, J.; Houkin, K.; Hamada, H.; Kocsis, J.D. Therapeutic time window of mesenchymal stem cells derived from bone marrow after cerebral ischemia. Brain Res. 2010, 1334, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Otto, W.R.; Wright, N.A. Mesenchymal stem cells: From experiment to clinic. Fibrogenes. Tissue Repair 2011, 4, 20. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.R.; Pollock, K.; Hubel, A.; McKenna, D. Mesenchymal stem or stromal cells: A review of clinical applications and manufacturing practices. Transfusion 2014, 54, 1418–1437. [Google Scholar] [CrossRef]
- Mendicino, M.; Bailey, A.M.; Wonnacott, K.; Puri, R.K.; Bauer, S.R. MSC-based product characterization for clinical trials: An FDA perspective. Cell Stem Cell 2014, 14, 141–145. [Google Scholar] [CrossRef]
- Chan, T.-M.; Chen, J.Y.-R.; Ho, L.-I.; Lin, H.-P.; Hsueh, K.-W.; Liu, D.D.; Chen, Y.-H.; Hsieh, A.-C.; Tsai, N.-M.; Hueng, D.-Y. ADSC therapy in neurodegenerative disorders. Cell Transplant. 2014, 23, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Nekanti, U.; Mohanty, L.; Venugopal, P.; Balasubramanian, S.; Totey, S.; Ta, M. Optimization and scale-up of Wharton’s jelly-derived mesenchymal stem cells for clinical applications. Stem Cell Res. 2010, 5, 244–254. [Google Scholar] [CrossRef]
- De Windt, T.S.; Vonk, L.A.; Saris, D.B. Response to: Mesenchymal stem cells: Time to change the name! Stem Cells Transl. Med. 2017, 6, 1747. [Google Scholar] [CrossRef]
- Caplan, A.I. Mesenchymal stem cells: Time to change the name! Stem Cells Transl. Med. 2017, 6, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Conese, M.; Carbone, A.; Castellani, S.; Di Gioia, S. Paracrine effects and heterogeneity of marrow-derived stem/progenitor cells: Relevance for the treatment of respiratory diseases. Cells Tissues Organs 2013, 197, 445–473. [Google Scholar] [CrossRef] [PubMed]
- De Becker, A.; Van Riet, I. Homing and migration of mesenchymal stromal cells: How to improve the efficacy of cell therapy? World J. Stem Cells 2016, 8, 73. [Google Scholar] [CrossRef] [PubMed]
- Leibacher, J.; Henschler, R. Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells. Stem Cell Res. Ther. 2016, 7, 7. [Google Scholar] [CrossRef]
- Fujii, S.; Miura, Y.; Fujishiro, A.; Shindo, T.; Shimazu, Y.; Hirai, H.; Tahara, H.; Takaori-Kondo, A.; Ichinohe, T.; Maekawa, T. Graft-versus-host disease amelioration by human bone marrow mesenchymal stromal/stem cell-derived extracellular vesicles is associated with peripheral preservation of naive T cell populations. Stem Cells 2018, 36, 434–445. [Google Scholar] [CrossRef]
- Kim, Y.; Jin, H.J.; Heo, J.; Ju, H.; Lee, H.-Y.; Kim, S.; Lee, S.; Lim, J.; Jeong, S.Y.; Kwon, J. Small hypoxia-primed mesenchymal stem cells attenuate graft-versus-host disease. Leukemia 2018, 32, 2672. [Google Scholar] [CrossRef]
- Lim, J.; Kim, Y.; Heo, J.; Kim, K.-H.; Lee, S.; Lee, S.W.; Kim, K.; Kim, I.-G.; Shin, D.-M. Priming with ceramide-1 phosphate promotes the therapeutic effect of mesenchymal stem/stromal cells on pulmonary artery hypertension. Biochem. Biophys. Res. Commun. 2016, 473, 35–41. [Google Scholar] [CrossRef]
- Čamernik, K.; Barlič, A.; Drobnič, M.; Marc, J.; Jeras, M.; Zupan, J. Mesenchymal stem cells in the musculoskeletal system: From animal models to human tissue regeneration? Stem Cell Rev. Rep. 2018, 14, 346–369. [Google Scholar] [CrossRef]
- Markovic, B.S.; Kanjevac, T.; Harrell, C.R.; Gazdic, M.; Fellabaum, C.; Arsenijevic, N.; Volarevic, V. Molecular and cellular mechanisms involved in mesenchymal stem cell-based therapy of inflammatory bowel diseases. Stem Cell Rev. Rep. 2018, 14, 153–165. [Google Scholar] [CrossRef]
- Squillaro, T.; Peluso, G.; Galderisi, U. Clinical trials with mesenchymal stem cells: An update. Cell Transplant. 2016, 25, 829–848. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.; Kahlenberg, S.; Hornsby, P. Therapeutic potential of mesenchymal stem cells for diabetes. J. Mol. Endocrinol. 2017, 59, R109–R120. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Murphy, S.V.; Atala, A. Regenerative medicine in urology. In Seminars in Pediatric Surgery; WB Saunders: Philadelphia, PA, USA, 2014; pp. 106–111. [Google Scholar]
- Zazzeroni, L.; Lanzoni, G.; Pasquinelli, G.; Ricordi, C. Considerations on the harvesting site and donor derivation for mesenchymal stem cells-based strategies for diabetes. Cellr4 Repair Replace. Regener. Reprogr. 2017, 5, e2435. [Google Scholar]
- Hass, R.; Kasper, C.; Böhm, S.; Jacobs, R. Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun. Signal. 2011, 9, 12. [Google Scholar] [CrossRef]
- Heo, J.S.; Choi, Y.; Kim, H.-S.; Kim, H.O. Comparison of molecular profiles of human mesenchymal stem cells derived from bone marrow, umbilical cord blood, placenta and adipose tissue. Int. J. Mol. Med. 2016, 37, 115–125. [Google Scholar] [CrossRef]
- Hsiao, S.T.-F.; Asgari, A.; Lokmic, Z.; Sinclair, R.; Dusting, G.J.; Lim, S.Y.; Dilley, R.J. Comparative analysis of paracrine factor expression in human adult mesenchymal stem cells derived from bone marrow, adipose, and dermal tissue. Stem Cells Dev. 2012, 21, 2189–2203. [Google Scholar] [CrossRef]
- Amable, P.R.; Teixeira, M.V.T.; Carias, R.B.V.; Granjeiro, J.M.; Borojevic, R. Protein synthesis and secretion in human mesenchymal cells derived from bone marrow, adipose tissue and Wharton’s jelly. Stem Cell Res. Ther. 2014, 5, 53. [Google Scholar] [CrossRef]
- Abdal Dayem, A.; Lee, S.B.; Cho, S.-G. The impact of metallic nanoparticles on stem cell proliferation and differentiation. Nanomaterials 2018, 8, 761. [Google Scholar] [CrossRef]
- Tian, H.; Bharadwaj, S.; Liu, Y.; Ma, P.X.; Atala, A.; Zhang, Y. Differentiation of human bone marrow mesenchymal stem cells into bladder cells: Potential for urological tissue engineering. Tissue Eng. Part A 2010, 16, 1769–1779. [Google Scholar] [CrossRef]
- Ning, J.; Li, C.; Li, H.; Chang, J. Bone marrow mesenchymal stem cells differentiate into urothelial cells and the implications for reconstructing urinary bladder mucosa. Cytotechnology 2011, 63, 531. [Google Scholar] [CrossRef]
- Wu, S.; Cheng, Z.; Liu, G.; Zhao, X.; Zhong, L.; Zhu, Y.; Zhu, J. Urothelial differentiation of human umbilical cord-derived mesenchymal stromal cells in vitro. Anal. Cell. Pathol. 2013, 36, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; McNeill, E.; Tian, H.; Soker, S.; Andersson, K.-E.; Yoo, J.J.; Atala, A. Urine derived cells are a potential source for urological tissue reconstruction. J. Urol. 2008, 180, 2226–2233. [Google Scholar] [CrossRef] [PubMed]
- Bharadwaj, S.; Liu, G.; Shi, Y.; Markert, C.; Andersson, K.-E.; Atala, A.; Zhang, Y. Characterization of urine-derived stem cells obtained from upper urinary tract for use in cell-based urological tissue engineering. Tissue Eng. Part A 2011, 17, 2123–2132. [Google Scholar] [CrossRef] [PubMed]
- Qin, D.; Long, T.; Deng, J.; Zhang, Y. Urine-derived stem cells for potential use in bladder repair. Stem Cell Res. Ther. 2014, 5, 69. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Atala, A. Regenerative medicine of the bladder. In Principles of Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1263–1279. [Google Scholar]
- Lopes, F.A.; Rolim, N.; Rodrigues, T.; Canhoto, A. Intestinal adenocarcinoma in an augmented ileocystoplasty. BMJ Case Rep. 2013, 2013, bcr2013009499. [Google Scholar] [CrossRef] [PubMed]
- Malone, M.J.; Izes, J.K.; Hurley, L.J. CARCINOGENESIS: The Fate of Intestinal Segments Used in Urinary Reconstruction. Urol. Clin. N. Am. 1997, 24, 723–728. [Google Scholar] [CrossRef]
- Wan, Q.; Xiong, G.; Liu, G.; Shupe, T.D.; Wei, G.; Zhang, D.; Liang, D.; Lu, X.; Atala, A.; Zhang, Y. Urothelium with barrier function differentiated from human urine-derived stem cells for potential use in urinary tract reconstruction. Stem Cell Res. Ther. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Sun, B.; Dong, X.; Zhao, J.; Yang, Z.; Zhang, Y.; Li, L. Differentiation of human urine-derived stem cells into interstitial cells of Cajal-like cells by exogenous gene modification: A preliminary study. Biochem. Biophys. Res. Commun. 2020, 523, 10–17. [Google Scholar] [CrossRef]
- Drumm, B.T.; Koh, S.D.; Andersson, K.-E.; Ward, S.M. Calcium signalling in Cajal-like interstitial cells of the lower urinary tract. Nat. Rev. Urol. 2014, 11, 555. [Google Scholar] [CrossRef]
- McCloskey, K.D. Interstitial cells of Cajal in the urinary tract. In Urinary Tract; Springer: Berlin/Heidelberg, Germany, 2011; pp. 233–254. [Google Scholar]
- Kang, H.H.; Kang, J.J.; Kang, H.G.; Chung, S.S. Urothelial differentiation of human amniotic fluid stem cells by urothelium specific conditioned medium. Cell Biol. Int. 2014, 38, 531–537. [Google Scholar] [CrossRef]
- Chung, S.S.; Koh, C.J. Bladder cancer cell in co-culture induces human stem cell differentiation to urothelial cells through paracrine FGF10 signaling. In Vitro Cell. Dev. Biol. Anim. 2013, 49, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Bagai, S.; Rubio, E.; Cheng, J.-F.; Sweet, R.; Thomas, R.; Fuchs, E.; Grady, R.; Mitchell, M.; Bassuk, J.A. Fibroblast growth factor-10 is a mitogen for urothelial cells. J. Biol. Chem. 2002, 277, 23828–23837. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Jiang, W.; Alraies, A.; Liu, Q.; Gudla, V.; Oni, J.; Wei, X.; Sloan, A.; Ni, L.; Agarwal, M. Bladder smooth muscle cells differentiation from dental pulp stem cells: Future potential for bladder tissue engineering. Stem Cells Int. 2016, 2016, 6979368. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, D.; Alraies, A.; Liu, Q.; Zhu, B.; Sloan, A.J.; Ni, L.; Song, B. Wnt-GSK3β/β-catenin regulates the differentiation of dental pulp stem cells into bladder smooth muscle cells. Stem Cells Int. 2019, 2019, 8907570. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.G.; Fu, W.J.; Wang, X.X.; Xu, Y.D.; Li, G.; Hong, B.F.; Hu, K.; Cui, F.Z.; Wang, Y.; Zhang, X. Transdifferentiation of human adipose-derived stem cells into urothelial cells: Potential for urinary tract tissue engineering. Cell Tissue Res. 2012, 347, 737–746. [Google Scholar] [CrossRef] [PubMed]
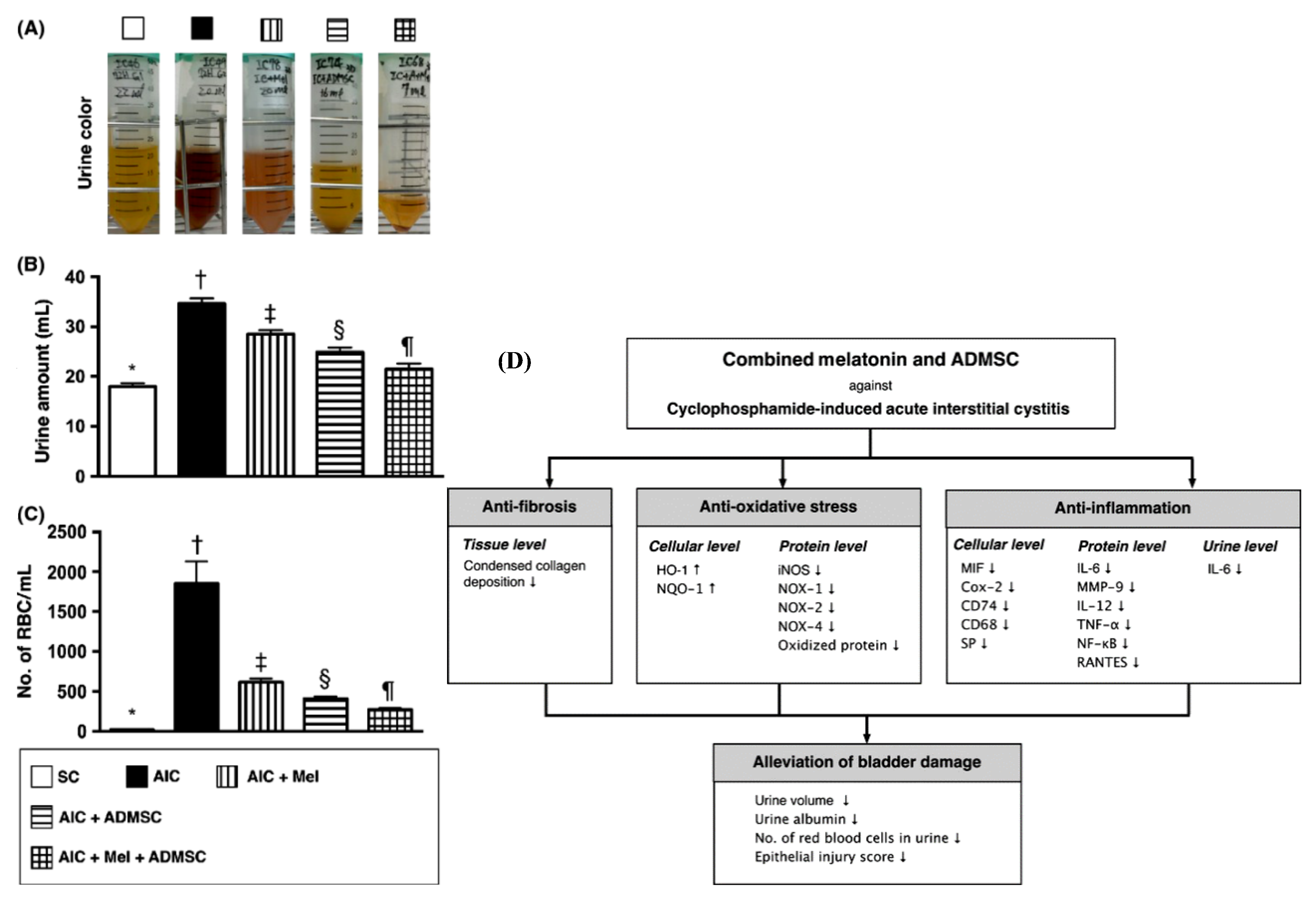
- Chen, Y.T.; Chiang, H.J.; Chen, C.H.; Sung, P.H.; Lee, F.Y.; Tsai, T.H.; Chang, C.L.; Chen, H.H.; Sun, C.K.; Leu, S.; et al. Melatonin treatment further improves adipose-derived mesenchymal stem cell therapy for acute interstitial cystitis in rat. J. Pineal Res. 2014, 57, 248–261. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. Melatonin as a natural ally against oxidative stress: A physicochemical examination. J. Pineal Res. 2011, 51, 1–16. [Google Scholar] [CrossRef]
- Galano, A.; Tan, D.X.; Reiter, R.J. On the free radical scavenging activities of melatonin’s metabolites, AFMK and AMK. J. Pineal Res. 2013, 54, 245–257. [Google Scholar] [CrossRef]
- Mauriz, J.L.; Collado, P.S.; Veneroso, C.; Reiter, R.J.; González-Gallego, J. A review of the molecular aspects of melatonin’s anti-inflammatory actions: Recent insights and new perspectives. J. Pineal Res. 2013, 54, 1–14. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, D.; Wang, X.; Chen, X.; Long, Y.; Chai, W.; Zhou, X.; Rui, X.; Zhang, Q.; Wang, H. Melatonin improved rat cardiac mitochondria and survival rate in septic heart injury. J. Pineal Res. 2013, 55, 1–6. [Google Scholar] [CrossRef]
- Yip, H.K.; Chang, Y.C.; Wallace, C.G.; Chang, L.T.; Tsai, T.H.; Chen, Y.L.; Chang, H.W.; Leu, S.; Zhen, Y.Y.; Tsai, C.Y. Melatonin treatment improves adipose-derived mesenchymal stem cell therapy for acute lung ischemia–reperfusion injury. J. Pineal Res. 2013, 54, 207–221. [Google Scholar] [CrossRef]
- Urata, Y.; Honma, S.; Goto, S.; Todoroki, S.; Iida, T.; Cho, S.; Honma, K.; Kondo, T. Melatonin induces γ-glutamylcysteine synthetase mediated by activator protein-1 in human vascular endothelial cells. Free Radic. Biol. Med. 1999, 27, 838–847. [Google Scholar] [CrossRef]
- Winiarska, K.; Drozak, J.; Wegrzynowicz, M.; Fraczyk, T.; Bryla, J. Diabetes-induced changes in glucose synthesis, intracellular glutathione status and hydroxyl free radical generation in rabbit kidney-cortex tubules. Mol. Cell. Biochem. 2004, 261, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Mayo, J.C.; Tan, D.-X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef]
- Song, M.; Lim, J.; Yu, H.Y.; Park, J.; Chun, J.Y.; Jeong, J.; Heo, J.; Kang, H.; Kim, Y.; Cho, Y.M.; et al. Mesenchymal Stem Cell Therapy Alleviates Interstitial Cystitis by Activating Wnt Signaling Pathway. Stem Cells Dev. 2015, 24, 1648–1657. [Google Scholar] [CrossRef] [PubMed]
- Hirose, Y.; Yamamoto, T.; Nakashima, M.; Funahashi, Y.; Matsukawa, Y.; Yamaguchi, M.; Kawabata, S.; Gotoh, M. Injection of Dental Pulp Stem Cells Promotes Healing of Damaged Bladder Tissue in a Rat Model of Chemically Induced Cystitis. Cell Transplant. 2016, 25, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Furuta, A.; Yamamoto, T.; Igarashi, T.; Suzuki, Y.; Egawa, S.; Yoshimura, N. Bladder wall injection of mesenchymal stem cells ameliorates bladder inflammation, overactivity, and nociception in a chemically induced interstitial cystitis-like rat model. Int Urogynecol J. 2018, 29, 1615–1622. [Google Scholar] [CrossRef]
- Xiao, Y.; Song, Y.J.; Song, B.; Huang, C.B.; Ling, Q.; Yu, X. TGF-beta/MAPK signaling mediates the effects of bone marrow mesenchymal stem cells on urinary control and interstitial cystitis after urinary bladder transplantation. Am. J. Transl. Res. 2017, 9, 1193–1202. [Google Scholar]
- Li, J.; Luo, H.; Dong, X.; Liu, Q.; Wu, C.; Zhang, T.; Hu, X.; Zhang, Y.; Song, B.; Li, L. Therapeutic effect of urine-derived stem cells for protamine/lipopolysaccharide-induced interstitial cystitis in a rat model. Stem Cell Res. 2017, 8, 107. [Google Scholar] [CrossRef]
- Ryu, C.-M.; Yu, H.Y.; Lee, H.-Y.; Shin, J.-H.; Lee, S.; Ju, H.; Paulson, B.; Lee, S.; Kim, S.; Lim, J. Longitudinal intravital imaging of transplanted mesenchymal stem cells elucidates their functional integration and therapeutic potency in an animal model of interstitial cystitis/bladder pain syndrome. Theranostics 2018, 8, 5610. [Google Scholar] [CrossRef]
- Ryu, C.-M.; Shin, J.H.; Yu, H.Y.; Ju, H.; Kim, S.; Lim, J.; Heo, J.; Lee, S.; Shin, D.-M.; Choo, M.-S. N-acetylcysteine prevents bladder tissue fibrosis in a lipopolysaccharide-induced cystitis rat model. Sci. Rep. 2019, 9, 8134. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.-W.; Chun, S.Y.; Lee, E.H.; Ha, Y.-S.; Lee, J.N.; Song, P.H.; Yoo, E.S.; Kwon, T.G.; Chung, S.K.; Kim, B.S. Verification of mesenchymal stem cell injection therapy for interstitial cystitis in a rat model. PLoS ONE 2019, 14, e0226390. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Liu, B.; Chen, J.; Xu, Y.; Zhan, H.; Yang, F.; Li, W.; Zhou, X. Umbilical cord-derived mesenchymal stem cells alleviated inflammation and inhibited apoptosis in interstitial cystitis via AKT/mTOR signaling pathway. Biochem. Biophys. Res. Commun. 2018, 495, 546–552. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Yu, H.Y.; Heo, J.; Song, M.; Shin, J.H.; Lim, J.; Yoon, S.J.; Kim, Y.; Lee, S.; Kim, S.W.; et al. Mesenchymal stem cells protect against the tissue fibrosis of ketamine-induced cystitis in rat bladder. Sci. Rep. 2016, 6, 30881. [Google Scholar] [CrossRef]
- Chu, P.S.; Kwok, S.; Lam, K.; Chu, T.; Chan, S.W.; Man, C.; Ma, W.; Chui, K.; Yiu, M.; Chan, Y.-C. Street ketamine’-associated bladder dysfunction: A report of ten cases. Hong Kong Med. J. 2007, 13, 311. [Google Scholar] [PubMed]
- Phatak, S.; Foster, H.E., Jr. The management of interstitial cystitis: An update. Nat. Rev. Urol. 2006, 3, 45. [Google Scholar] [CrossRef]
- Persson, J. Wherefore ketamine? Curr. Opin. Anesthesiol. 2010, 23, 455–460. [Google Scholar] [CrossRef]
- Chu, P.S.K.; Ma, W.K.; Wong, S.C.W.; Chu, R.W.H.; Cheng, C.H.; Wong, S.; Tse, J.M.l.; Lau, F.L.; Yiu, M.K.; Man, C.W. The destruction of the lower urinary tract by ketamine abuse: A new syndrome? BJU Int. 2008, 102, 1616–1622. [Google Scholar] [CrossRef]
- Sharma, A.K.; Bury, M.I.; Marks, A.J.; Fuller, N.J.; Meisner, J.W.; Tapaskar, N.; Halliday, L.C.; Matoka, D.J.; Cheng, E.Y. A nonhuman primate model for urinary bladder regeneration using autologous sources of bone marrow-derived mesenchymal stem cells. Stem Cells 2011, 29, 241–250. [Google Scholar] [CrossRef]
- Bury, M.I.; Fuller, N.J.; Wethekam, L.; Sharma, A.K. Bone marrow derived cells facilitate urinary bladder regeneration by attenuating tissue inflammatory responses. Cent. Eur. J. Urol. 2015, 68, 115. [Google Scholar] [CrossRef]
- Snow-Lisy, D.C.; Diaz, E.C.; Bury, M.I.; Fuller, N.J.; Hannick, J.H.; Ahmad, N.; Sharma, A.K. The role of genetically modified mesenchymal stem cells in urinary bladder regeneration. PLoS ONE 2015, 10, e0138643. [Google Scholar] [CrossRef]
- Zhe, Z.; Jun, D.; Yang, Z.; Mingxi, X.; Ke, Z.; Ming, Z.; Zhong, W.; Mujun, L. Bladder acellular matrix grafts seeded with adipose-derived stem cells and incubated intraperitoneally promote the regeneration of bladder smooth muscle and nerve in a rat model of bladder augmentation. Stem Cells Dev. 2016, 25, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Salem, S.A.; HWIE, A.N.M.; Aminuddin, S.; Kong, C.H.C.; Sagap, I.; Singh, R.; Yusof, M.R.; Zainuddin, Z.M.; Idrus, R.H. Human adipose tissue derived stem cells as a source of smooth muscle cells in the regeneration of muscular layer of urinary bladder wall. Malays. J. Med. Sci. MJMS 2013, 20, 80. [Google Scholar] [PubMed]
- Hou, X.; Shi, C.; Chen, W.; Chen, B.; Jia, W.; Guo, Y.; Ma, C.; Ye, G.; Kang, J.; Dai, J. Transplantation of human adipose-derived mesenchymal stem cells on a bladder acellular matrix for bladder regeneration in a canine model. Biomed. Mater. 2016, 11, 031001. [Google Scholar] [CrossRef] [PubMed]
- Drewa, T. Using hair-follicle stem cells for urinary bladder-wall regeneration. Regener. Med. 2008, 3, 939–944. [Google Scholar] [CrossRef] [PubMed]
- Drewa, T.; Joachimiak, R.; Kaznica, A.; Sarafian, V.; Pokrywczynska, M. Hair stem cells for bladder regeneration in rats: Preliminary results. In Transplantation Proceedings; Elsevier: Amsterdam, The Netherlands, 2009; pp. 4345–4351. [Google Scholar]
- Lee, J.N.; Chun, S.Y.; Lee, H.-J.; Jang, Y.-J.; Choi, S.H.; Kim, D.H.; Oh, S.H.; Song, P.H.; Lee, J.H.; Kim, J.K. Human urine-derived stem cells seeded surface modified composite scaffold grafts for bladder reconstruction in a rat model. J. Korean Med Sci. 2015, 30, 1754–1763. [Google Scholar] [CrossRef]
- Bharadwaj, S.; Liu, G.; Shi, Y.; Wu, R.; Yang, B.; He, T.; Fan, Y.; Lu, X.; Zhou, X.; Liu, H. Multipotential differentiation of human urine-derived stem cells: Potential for therapeutic applications in urology. Stem Cells 2013, 31, 1840–1856. [Google Scholar] [CrossRef]
- Suzuki, K.; Koyanagi-Aoi, M.; Uehara, K.; Hinata, N.; Fujisawa, M.; Aoi, T. Directed differentiation of human induced pluripotent stem cells into mature stratified bladder urothelium. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef]
- Boumelhem, B.B.; Fraser, S.T.; Assinder, S.J. Differentiation of Urothelium from Mouse Embryonic Stem Cells in Chemically Defined Conditions. In Progenitor Cells; Springer: Berlin/Heidelberg, Germany, 2019; pp. 103–115. [Google Scholar]
- Osborn, S.L.; Kurzrock, E.A. In Vitro Differentiation and Propagation of Urothelium from Pluripotent Stem Cell Lines. In Urothelial Carcinoma; Springer: Berlin/Heidelberg, Germany, 2018; pp. 137–144. [Google Scholar]
- Inoue, Y.; Kishida, T.; Kotani, S.-I.; Akiyoshi, M.; Taga, H.; Seki, M.; Ukimura, O.; Mazda, O. Direct conversion of fibroblasts into urothelial cells that may be recruited to regenerating mucosa of injured urinary bladder. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Xie, C.-Q.; Huang, H.; Wei, S.; Song, L.-S.; Zhang, J.; Ritchie, R.P.; Chen, L.; Zhang, M.; Chen, Y.E. A comparison of murine smooth muscle cells generated from embryonic versus induced pluripotent stem cells. Stem Cells Dev. 2009, 18, 741–748. [Google Scholar] [CrossRef]
- Mauney, J.R.; Ramachandran, A.; Richard, N.Y.; Daley, G.Q.; Adam, R.M.; Estrada, C.R. All-trans retinoic acid directs urothelial specification of murine embryonic stem cells via GATA4/6 signaling mechanisms. PLoS ONE 2010, 5, e11513. [Google Scholar] [CrossRef] [PubMed]
- Franck, D.; Gil, E.S.; Adam, R.M.; Kaplan, D.L.; Chung, Y.G.; Estrada, C.R., Jr.; Mauney, J.R. Evaluation of Silk Biomaterials in Combination with Extracellular Matrix Coatings for Bladder Tissue Engineering with Primary and Pluripotent Cells. PLoS ONE 2013, 8, e56237. [Google Scholar] [CrossRef] [PubMed]
- Kanai-Azuma, M.; Kanai, Y.; Gad, J.M.; Tajima, Y.; Taya, C.; Kurohmaru, M.; Sanai, Y.; Yonekawa, H.; Yazaki, K.; Tam, P.P. Depletion of definitive gut endoderm in Sox17-null mutant mice. Development 2002, 129, 2367–2379. [Google Scholar] [PubMed]
- Sinner, D.; Rankin, S.; Lee, M.; Zorn, A.M. Sox17 and β-catenin cooperate to regulate the transcription of endodermal genes. Development 2004, 131, 3069–3080. [Google Scholar] [CrossRef] [PubMed]
- Varley, C.L.; Stahlschmidt, J.; Lee, W.-C.; Holder, J.; Diggle, C.; Selby, P.J.; Trejdosiewicz, L.K.; Southgate, J. Role of PPAR γ and EGFR signalling in the urothelial terminal differentiation programme. J. Cell Sci. 2004, 117, 2029–2036. [Google Scholar] [CrossRef] [PubMed]
- Lowell, B.B. An essential regulator of adipogenesis and modulator of fat cell function: PPARγ. Cell 1999, 99, 239–242. [Google Scholar] [CrossRef]
- Oottamasathien, S.; Wang, Y.; Williams, K.; Franco, O.E.; Wills, M.L.; Thomas, J.C.; Saba, K.; Sharif-Afshar, A.-R.; Makari, J.H.; Bhowmick, N.A. Directed differentiation of embryonic stem cells into bladder tissue. Dev. Biol. 2007, 304, 556–566. [Google Scholar] [CrossRef]
- Thomas, J.C.; Oottamasathien, S.; Makari, J.H.; Honea, L.; Sharif-Afshar, A.R.; Wang, Y.; Adams, C.; Wills, M.L.; Bhowmick, N.A.; Adams, M.C.; et al. Temporal-spatial protein expression in bladder tissue derived from embryonic stem cells. J. Urol. 2008, 180, 1784–1789. [Google Scholar] [CrossRef]
- Kinebuchi, Y.; Johkura, K.; Sasaki, K.; Imamura, T.; Mimura, Y.; Nishizawa, O. Direct induction of layered tissues from mouse embryonic stem cells: Potential for differentiation into urinary tract tissue. Cell Tissue Res. 2008, 331, 605–615. [Google Scholar] [CrossRef]
- Abdal Dayem, A.; Lee, S.B.; Kim, K.; Lim, K.M.; Jeon, T.-I.; Seok, J.; Cho, S.-G. Production of Mesenchymal Stem Cells through Stem Cell Reprogramming. Int. J. Mol. Sci. 2019, 20, 1922. [Google Scholar] [CrossRef]
- Kimbrel, E.A.; Kouris, N.A.; Yavanian, G.J.; Chu, J.; Qin, Y.; Chan, A.; Singh, R.P.; McCurdy, D.; Gordon, L.; Levinson, R.D. Mesenchymal stem cell population derived from human pluripotent stem cells displays potent immunomodulatory and therapeutic properties. Stem Cells Dev. 2014, 23, 1611–1624. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Yan, L.; Wang, X.; Li, E.; Murphy, K.; Vaccaro, K.; Li, Y.; Xu, R.H. Concise Review: Mesenchymal Stem Cells Derived from Human Pluripotent Cells, an Unlimited and Quality-Controllable Source for Therapeutic Applications. Stem Cells 2019, 37, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.W.; Ryu, C.-M.; Shin, J.-H.; Choi, D.; Kim, A.; Yu, H.Y.; Han, J.-Y.; Lee, H.-Y.; Lim, J.; Kim, Y.H.; et al. The Therapeutic Effect of Human Embryonic Stem Cell-Derived Multipotent Mesenchymal Stem Cells on Chemical-Induced Cystitis in Rats. Int. Neurourol. J. 2018, 22, S34–S45. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Yu, H.Y.; Lim, J.; Ryu, C.M.; Kim, Y.H.; Heo, J.; Han, J.Y.; Lee, S.; Bae, Y.S.; Kim, J.Y.; et al. Improved efficacy and in vivo cellular properties of human embryonic stem cell derivative in a preclinical model of bladder pain syndrome. Sci. Rep. 2017, 7, 8872. [Google Scholar] [CrossRef] [PubMed]
- Bullock, A.D.; Becich, M.J.; Klutke, C.G.; Ratliff, T.L. Experimental Autoimmune Cystitis: A Potential Murhne Model for Ulcerative Interstitial Cystitis. J. Urol. 1992, 148, 1951–1956. [Google Scholar] [CrossRef]
- Lin, Y.H.; Liu, G.; Kavran, M.; Altuntas, C.Z.; Gasbarro, G.; Tuohy, V.K.; Daneshgari, F. Lower urinary tract phenotype of experimental autoimmune cystitis in mouse: A potential animal model for interstitial cystitis. BJU Int. 2008, 102, 1724–1730. [Google Scholar] [CrossRef]
- Jin, X.-W.; Liu, B.-K.; Zhang, X.; Zhao, Z.-H.; Shao, Y. Establishment of a novel autoimmune experimental model of bladder pain syndrome/interstitial cystitis in C57BL/6 mice. Inflammation 2017, 40, 861–870. [Google Scholar] [CrossRef]
- Hauser, P.J.; Buethe, D.A.; Califano, J.; Sofinowski, T.M.; Culkin, D.J.; Hurst, R.E. Restoring barrier function to acid damaged bladder by intravesical chondroitin sulfate. J. Urol. 2009, 182, 2477–2482. [Google Scholar] [CrossRef][Green Version]
- Homan, T.; Tsuzuki, T.; Dogishi, K.; Shirakawa, H.; Oyama, T.; Nakagawa, T.; Kaneko, S. A novel mouse model of chronic inflammatory and overactive bladder by a single intravesical injection of hydrogen peroxide. J. Pharmacol. Sci. 2013, 121, 327–337. [Google Scholar] [CrossRef]
- Gonzalez, R.R.; Fong, T.; Belmar, N.; Saban, M.; Felsen, D.; Te, A. Modulating bladder neuro-inflammation: RDP58, a novel anti-inflammatory peptide, decreases inflammation and nerve growth factor production in experimental cystitis. J. Urol. 2005, 173, 630–634. [Google Scholar] [CrossRef]
- Shin, J.H.; Ryu, C.-M.; Yu, H.Y.; Shin, D.-M.; Choo, M.-S. Current and Future Directions of Stem Cell Therapy for Bladder Dysfunction. Stem Cell Rev. Rep. 2020, 16, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Vasyutin, I.; Zerihun, L.; Ivan, C.; Atala, A. Bladder Organoids and Spheroids: Potential Tools for Normal and Diseased Tissue Modelling. Anticancer Res. 2019, 39, 1105–1118. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.P.; Lapi, E.; Martínez de Villarreal, J.; Álvaro-Espinosa, L.; Fernández-Barral, A.; Barbáchano, A.; Domínguez, O.; Laughney, A.M.; Megías, D.; Muñoz, A.; et al. Urothelial organoids originating from Cd49fhigh mouse stem cells display Notch-dependent differentiation capacity. Nat. Commun. 2019, 10, 4407. [Google Scholar] [CrossRef]
- Fujita, Y.; Kadota, T.; Araya, J.; Ochiya, T.; Kuwano, K. Clinical Application of Mesenchymal Stem Cell-Derived Extracellular Vesicle-Based Therapeutics for Inflammatory Lung Diseases. J. Clin. Med. 2018, 7, 355. [Google Scholar] [CrossRef] [PubMed]
- Mendt, M.; Rezvani, K.; Shpall, E. Mesenchymal stem cell-derived exosomes for clinical use. Bone Marrow Transplant. 2019, 54, 789–792. [Google Scholar] [CrossRef] [PubMed]
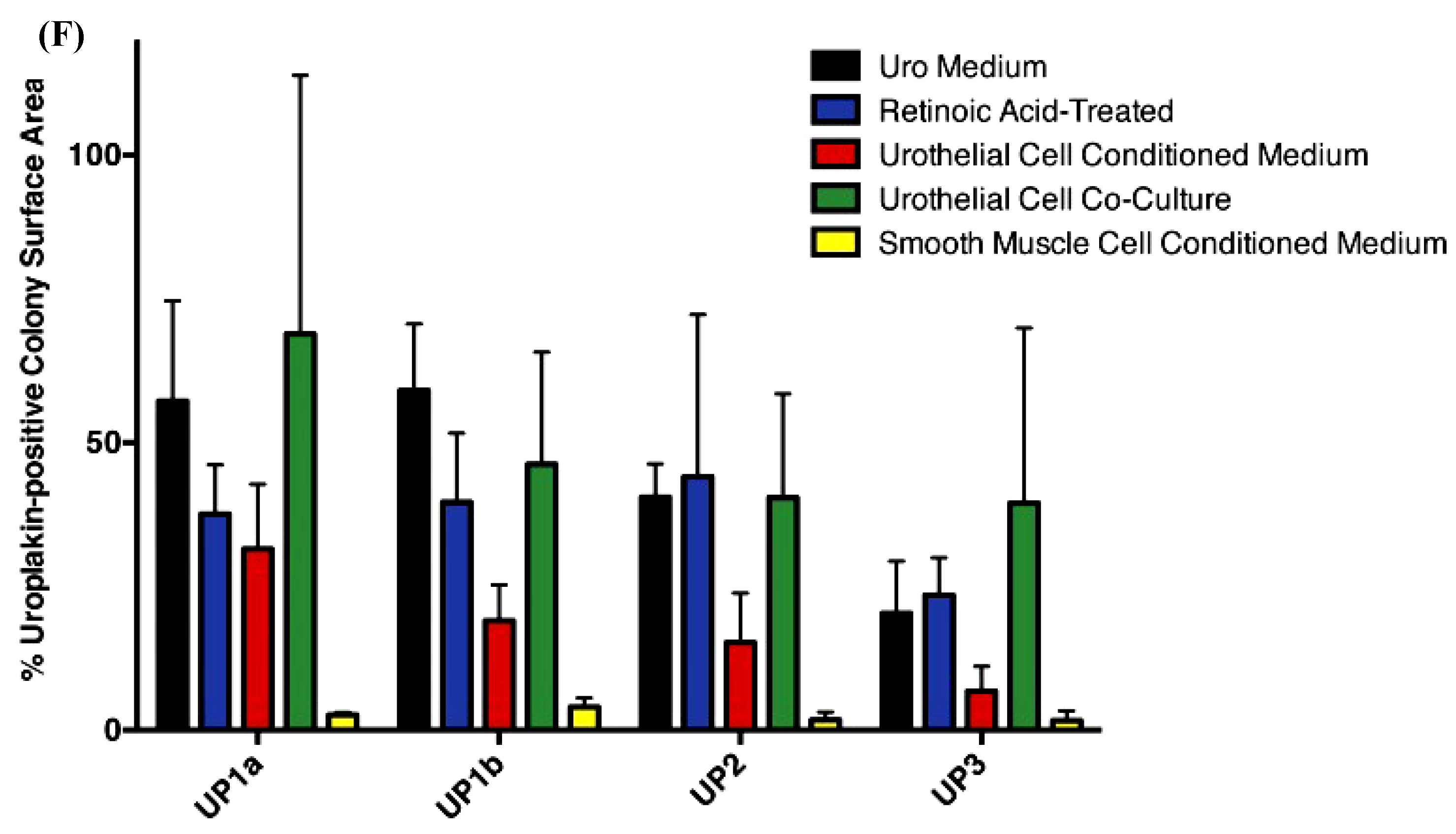
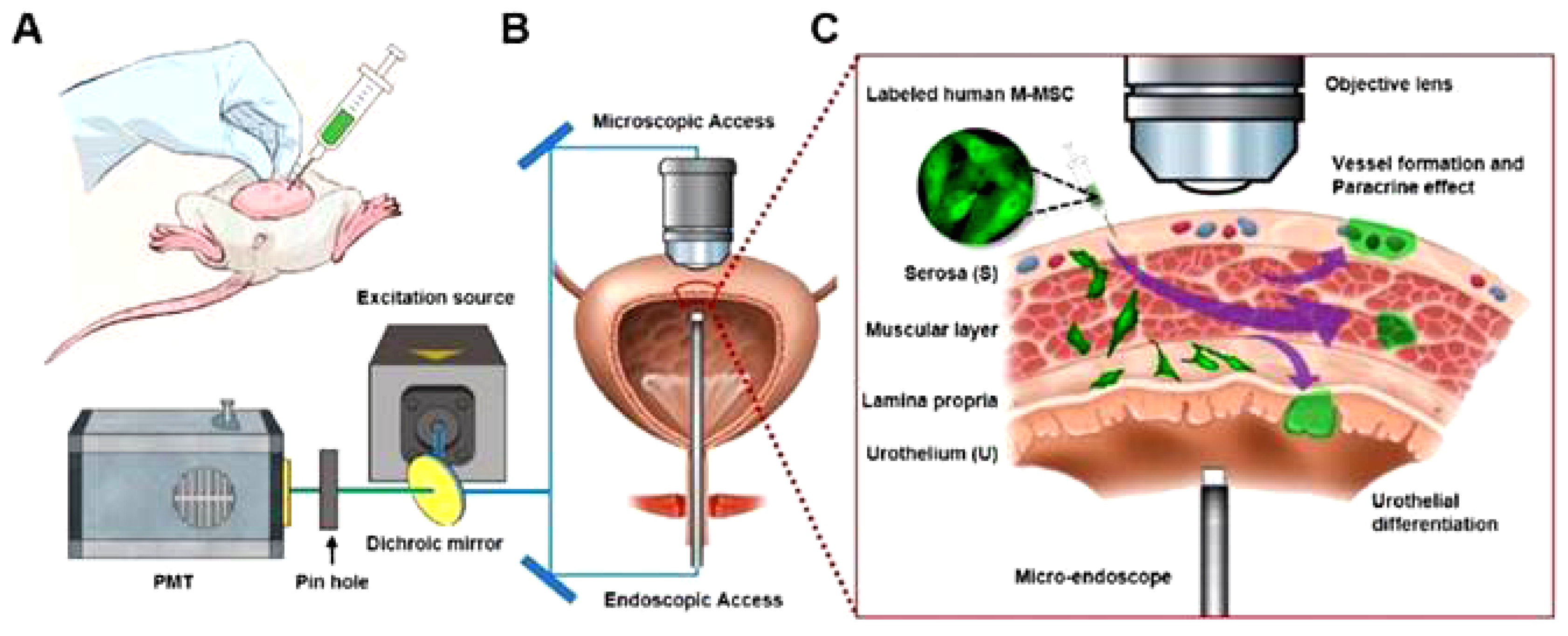
| Stem Cell Type | Cell Line Name | Regeneration Method and In Vitro Verifications | In Vivo Model | References |
|---|---|---|---|---|
| ASCs | BM-MSCs | - Urothelium and SMCs differentiations using urothelial cell-derived CM or co-culture with urothelial cells using Transwell plates. - High expression of UP1a, CK-7, and CK-13 in differentiated cells. - CM showed superior action than co-culture system. - High content of PDGF-BB, TGF-β1, and VEGF in the CM. | n.a. | [144] |
| - Urothelial differentiation through co-culture with neonatal urothelial cells. - High expression of urothelial-related keratin after two weeks of co-culturing. - Detection of microstructural epithelial cell-like features with the electron microscopy. | n.a. | [145] | ||
| - Transplantation was carried out through the injection of BrdU-labeled BMSCs (2 × 105/mL) into the tunica muscularis vesicae urinariae. | - PS-induced IC rat model. - BM-MSCs injection improved the urine excretion kinetics and recovered the pathological lesions. - TGF-β/MAPK signaling pathway-dependent repair mechanism. | [175] | ||
| hUC-MSCs | - Urothelial differentiation by co-culturing with urothelial cell-derived CM containing 20 ng/mL exogenous EGF. - High expression of UPs and CK. | n.a. | [146] | |
| - Single injection of hUC-MSCs into the submucosal layer of the urinary one week after HCl injection. | - HCl-induced IC/BPS female rat model. - Marked improvement of IC/BPS-related symptoms. - WNT signaling is involved in hUC-MSC-mediated IC/BPS recovery in the rat. | [172] | ||
| - i.v. injection of hUC-MSCs (1 × 106 cells) in CYP-induced chronic IC/BPS rat model. - In vitro verification via the co-culture of UC-MSCs with TNF-α-treated SV-HUC-1. This co-culture led to decreases in apoptotic changes (decrease in caspase 3 cleavage) and increases in the phosphorylation of AKT and mTOR. | - CYP-induced chronic IC/BPS rat model. - Significant recovery of the bladder function and the pathological changes in bladder tissue. | [180] | ||
| - Single i.v. of UCB-MSCs at a dose of 1 × 106 cells. | - Ketamine-induced cystitis in rat. - UCB-MSC injection led to a significant improvement in bladder functions and pathological changes. - Upregulation of TGF-β signaling-associated genes and high phosphorylation of Smad2 and Smad3 proteins. | [181] | ||
| hAFSCs | - Urothelial differentiation through co-culturing with bladder cancer-derived CM. - Low expression of pluripotency markers and increase in urothelial markers expression, such as UPII, CK8, and FGF10. - Increase in the expression of MuSK, Tie-1, and EphA4 receptor tyrosine kinases and the downregulation of EphA7 and FGF R1 kinases. | n.a. | [157] | |
| - Urothelial differentiation through co-culturing with the immortalized bladder cancer cell lines. - FGF10-mediated differentiation mechanism. - High mRNA and protein expression levels of UP II, UP III, and CK-8. - MEK inhibitor, U0126, inhibited urothelium differentiation. | n.a. | [158] | ||
| DPSCs | - SMCs differentiation using bladder SMC-derived CM containing TGF-β1. - Upregulation of SMC-associated genes and proteins (α-SMA, desmin, and calponin). - Wnt-GSK3β/β-catenin pathway is involved in differentiation. | n.a. | [160,161] | |
| - Cell injection into the bladder (2.0 × 106 cells in PBS). - DPSC-derived CM contains high level of FGF-2, VEGF, C-C and C-X-C chemokine families. | - HCl-induced cystitis in female F344/NSlc rat model. - Reduction in MPO and proinflammatory cytokines (IL-1β, IL-6, and TNF-α) in the bladder tissue and urine after DSPSC injection. | [173] | ||
| USCs | - Urothelial differentiation induction using urothelial-derived CM. - Upregulation of urothelial markers, UP II and UP1a, tight junction markers, Cingulin, E-cadherin, ZO-1, and ZO-2, and the epithelial markers (AE1/AE3 and CK20). - Differentiated urothelium possess barrier function. | n.a. | [153] | |
| - Interstitial cell differentiation via transfection of USCs with lentiviral vectors encoding SCF, c-kit, AZI2 gene, and HCN4. - Differentiated cells showed high expression of the c-kit after one week. | n.a. | [154] | ||
| - i.v. injection of USCs (1.2 × 106 cells). | - PS/LPS-mediated chronic IC/BPS rat model. - Marked recovery of the bladder functions in USC-injected rat. - Upregulation of the antioxidant and the anti-apoptotic proteins in the bladder tissue. - Downregulation of oxidative stress-related and autophagy-related markers. | [176] | ||
| ADSCs | - Urothelial differentiation induction using urothelial-derived CM. - Polygonal epithelium-like morphology. - Upregulation of UP2 and CK-18 and downregulation of vimentin. - High content of PDGF-BB and VEGF in CM is implicated in differentiation induction. | n.a. | [162] | |
| - The combination of i.v. injection of autologous AD-MSCs (1.2 × 106 cells) and i.p. injection of melatonin. | - CYP-induced acute IC/BPS. - Marked decrease in urine volume, urine albumin, hematuria, and proteinuria. - Decreased tissue fibrosis (low collagen deposition). - Upregulation of the cellular antioxidant markers, HO-1 and NQO. - Downregulation of the inflammatory markers in tissue and urine. | [163] | ||
| - Injection of AD-MSCs (1 × 106 cells in PBS) into the anterior and posterior bladder walls during injection of the second dose of HCl. | - HCl-induced IC/BPS rat model. - Significant decrease of nociceptive behavior, mast cell infiltration, and bladder fibrosis. - Downregulation of TNF-α and TGF-β. | [174] | ||
| PSCs | ESCs | - Urothelium and SMCs differentiation of mESCs via culture on collagen matrices and RA treatment in a dose-dependent manner. - Downregulation of pluripotency markers and significant increase in the mRNA level of UPs. - Upregulation of the transcription factors, GATA4 and GATA6, associated with UP1B and UP2 activation. | n.a. | [201] |
| - Urothelium differentiation of mESCs through the recombination with rEBM. - High expression of UPs. | - Engraftment of the recombinant mESCs and rEBM under a kidney capsule of mouse. - Harvesting of the grafts at various time points. - At day 16: high expression of UPs, androgen receptor expression, and loss of the expression of Foxa2. | [207,208] | ||
| - Differentiation of mESCs into bladder cells. - Seeding of mESC-derived EBs on mono-layered collagen membrane using various KSFM-based differentiation media. - High expression of SMC-related markers (αSMA and hl-calponin, and SM-MHC), UPII, and Pax-2. | - On day 28 after the in vitro culture on collagen and application of several differentiation media, the cultured tissues were transplanted subcutaneously into the back of male nude mice (BALB/c nu/nu). - The transplantation of tissue cultures using KSFM and 3T3-CM resulted in the formation of layered tissue like that of the urinary tract. | [209] | ||
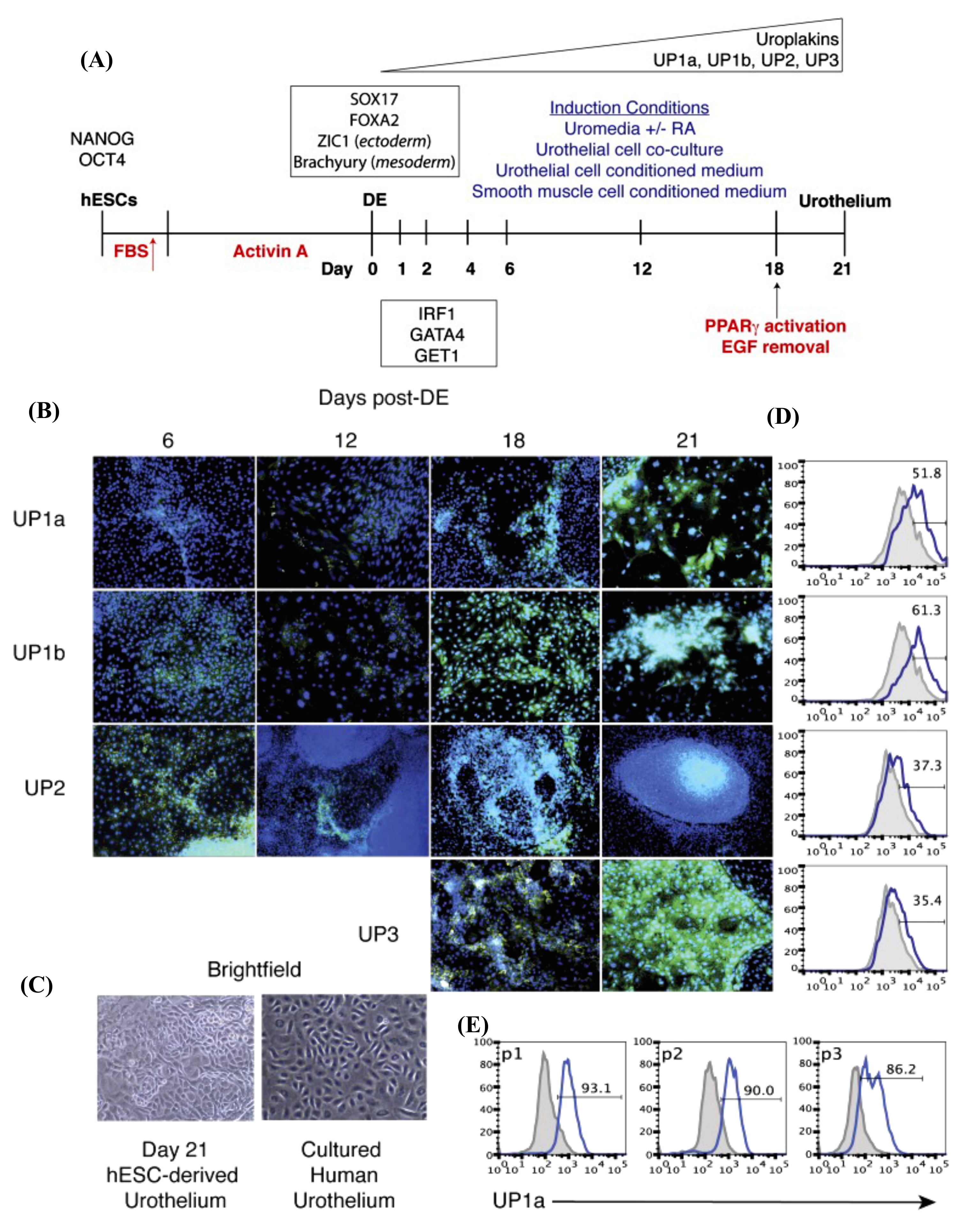
| - hESC-differentiated DE was directed toward urothelium differentiation using keratinocyte basal medium containing RA, EGF, and BPE and 2% FBS. - RA is implicated in GATA signaling activation. - Pure urothelium (up to 90%) was obtained after several passages and selection using specific media. - Co-expression of IRF1, GET1, and GATA4 with UPs expression. | n.a. | [14] | ||
| - Differentiation of hESC-DE into urothelium using KSFM supplemented with RA. - Upregulation of UPIb, UPII, UPIIIa, P63, CK-8/18, CK-7, E-cadherin, and ZO-1. - hESC-derived urothelium possesses a barrier function. | n.a. | [46] | ||
| - Direct transplantation of M-MSCs through the injection of cells in a dose-dependent manner (0.25, 0.5, and 1 × 106 cells) into an anterior bladder wall or bladder dome. | - ketamine-induced IC/BPS rat model. - Significant improvement in bladder function with an effect superior to that of BM-MSCs when injected at a low concentration (1 × 105 cells). | [213] | ||
| - Injection of M-MSCs (1 × 106 cells) into the outer layer of the anterior wall and dome of the bladder. | - HCl-induced IC/BPS rat model. - Significant restoration of bladder function and pathological changes in bladder tissue. - No tumor formation or immune rejection was detected over a long period post-injection (12 months). - Injected cells were differentiated into several cell types and progressively assimilated into a vascular-like structure. - Wnt-mediated and IGF-mediated repair mechanism. | [214] | ||
| - Dose-dependent injection of M-MSCs (0.1, 0.25, 0.5, and 1 × 106 cells) into the outer layer of the bladder (In vivo urothelium differentiation of M-MSCs). | - PS/LPS-induced IC/BPS rat model. - Marked recovery of the bladder function and pathological consequences in the tissue. - Stability: Long-term maintenance of the therapeutic capacity of the transplant. - Differentiation of the transplant into multiple cell types and integration into a perivascular-like structure (up to a month) post-injection). | [177] | ||
| hiPSCs | - Urothelium differentiation of hiPSCs using a high dose of GSK3β inhibitor CHIR99021 during DE differentiation. - Combination of EGFR inhibitor, PD153035 and PPARγ agonist, Troglitazone enhanced terminal urothelium differentiation. - High expression of UPs, CK13, and CK20. - FGF10 and Transwell culture system promoted further stratification. -Barrier function. | n.a. | [196] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdal Dayem, A.; Kim, K.; Lee, S.B.; Kim, A.; Cho, S.-G. Application of Adult and Pluripotent Stem Cells in Interstitial Cystitis/Bladder Pain Syndrome Therapy: Methods and Perspectives. J. Clin. Med. 2020, 9, 766. https://doi.org/10.3390/jcm9030766
Abdal Dayem A, Kim K, Lee SB, Kim A, Cho S-G. Application of Adult and Pluripotent Stem Cells in Interstitial Cystitis/Bladder Pain Syndrome Therapy: Methods and Perspectives. Journal of Clinical Medicine. 2020; 9(3):766. https://doi.org/10.3390/jcm9030766
Chicago/Turabian StyleAbdal Dayem, Ahmed, Kyeongseok Kim, Soo Bin Lee, Aram Kim, and Ssang-Goo Cho. 2020. "Application of Adult and Pluripotent Stem Cells in Interstitial Cystitis/Bladder Pain Syndrome Therapy: Methods and Perspectives" Journal of Clinical Medicine 9, no. 3: 766. https://doi.org/10.3390/jcm9030766
APA StyleAbdal Dayem, A., Kim, K., Lee, S. B., Kim, A., & Cho, S.-G. (2020). Application of Adult and Pluripotent Stem Cells in Interstitial Cystitis/Bladder Pain Syndrome Therapy: Methods and Perspectives. Journal of Clinical Medicine, 9(3), 766. https://doi.org/10.3390/jcm9030766







