Comprehensive Care for Patients with Sarcoidosis
Abstract
1. Introduction
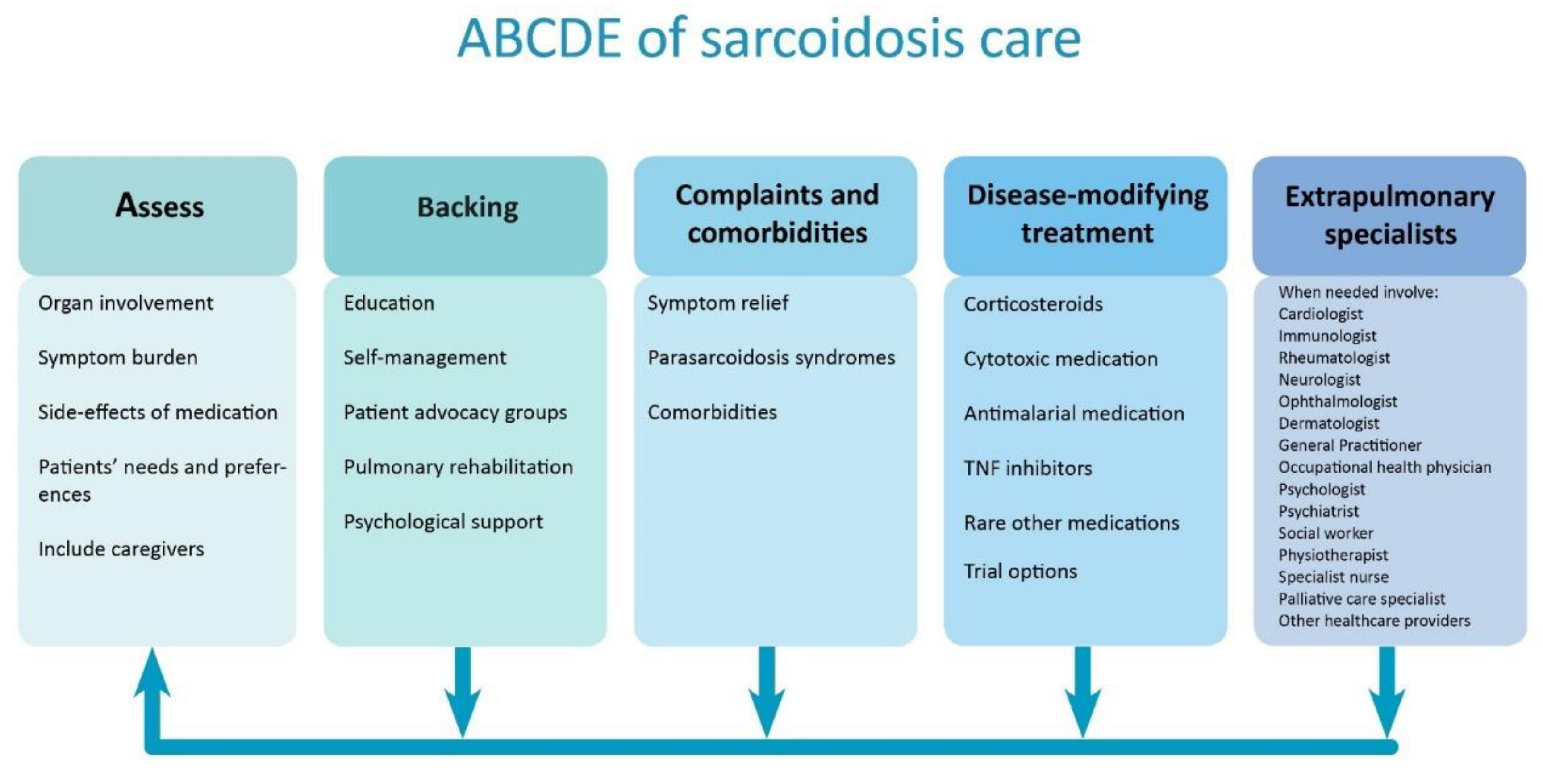
2. ABCDE Model
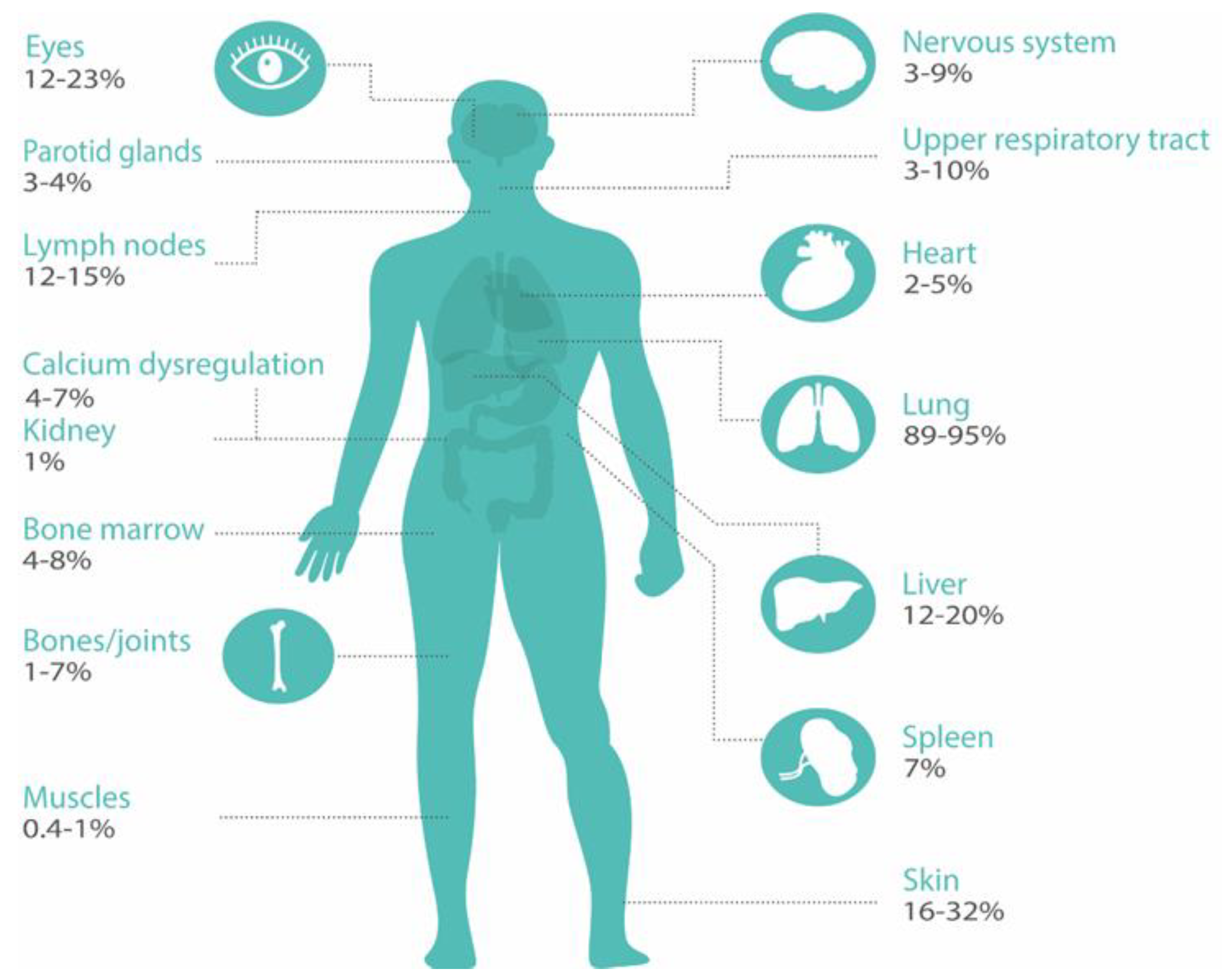
2.1. Assess
2.2. Backing
2.3. Complaints and Comorbidities
2.4. Disease Modifying-Treatment
2.5. Extra Pulmonary Specialists
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Grunewald, J.; Grutters, J.C.; Arkema, E.V.; Saketkoo, L.A.; Moller, D.R.; Muller-Quernheim, J. Sarcoidosis. Nat. Rev. Dis. Primers 2019, 5, 45. [Google Scholar] [CrossRef] [PubMed]
- Valeyre, D.; Prasse, A.; Nunes, H.; Uzunhan, Y.; Brillet, P.Y.; Muller-Quernheim, J. Sarcoidosis. Lancet 2014, 383, 1155–1167. [Google Scholar] [CrossRef]
- Gerke, A.K.; Judson, M.A.; Cozier, Y.C.; Culver, D.A.; Koth, L.L. Disease burden and variability in sarcoidosis. Ann. Am. Thorac. Soc. 2017, 14, S421–S428. [Google Scholar] [CrossRef] [PubMed]
- Harper, L.J.; Gerke, A.K.; Wang, X.F.; Ribeiro Neto, M.L.; Baughman, R.P.; Beyer, K.; Drent, M.; Judson, M.A.; Maier, L.A.; Serchuck, L.; et al. Income and other contributors to poor outcomes in us sarcoidosis patients. Am. J. Respir Crit. Care Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Cox, C.E.; Donohue, J.F.; Brown, C.D.; Kataria, Y.P.; Judson, M.A. Health-related quality of life of persons with sarcoidosis. Chest 2004, 125, 997–1004. [Google Scholar] [CrossRef] [PubMed]
- Baughman, R.P.; Judson, M.A.; Wells, A. The indications for the treatment of sarcoidosis: Wells law. Sarcoidosis Vasc. Diffuse Lung Dis. 2017, 34, 280–282. [Google Scholar]
- Baughman, R.P.; Barriuso, R.; Beyer, K.; Boyd, J.; Hochreiter, J.; Knoet, C.; Martone, F.; Quadder, B.; Richardson, J.; Spitzer, G.; et al. Sarcoidosis: Patient treatment priorities. ERJ Open Res. 2018. [Google Scholar] [CrossRef]
- Judson, M.A.; Baughman, R.P.; Teirstein, A.S.; Terrin, M.L.; Yeager, H., Jr. Defining organ involvement in sarcoidosis: The access proposed instrument. Access research group. A case control etiologic study of sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 1999, 16, 75–86. [Google Scholar]
- Judson, M.A. The clinical features of sarcoidosis: A comprehensive review. Clin. Rev. Allergy Immunol. 2015, 49, 63–78. [Google Scholar] [CrossRef]
- van Manen, M.J.; Geelhoed, J.J.; Tak, N.C.; Wijsenbeek, M.S. Optimizing quality of life in patients with idiopathic pulmonary fibrosis. Ther. Adv. Respir. Dis. 2017, 11, 157–169. [Google Scholar] [CrossRef]
- Kreuter, M.; Bendstrup, E.; Russell, A.M.; Bajwah, S.; Lindell, K.; Adir, Y.; Brown, C.E.; Calligaro, G.; Cassidy, N.; Corte, T.J.; et al. Palliative care in interstitial lung disease: Living well. Lancet Respir. Med. 2017, 5, 968–980. [Google Scholar] [CrossRef]
- Nunes, H.; Jeny, F.; Bouvry, D.; Uzunhan, Y.; Valeyre, D. Indications for treatment of sarcoidosis. Curr. Opin. Pulm. Med. 2019, 25, 505–518. [Google Scholar] [CrossRef] [PubMed]
- Drent, M.; Strookappe, B.; Hoitsma, E.; De Vries, J. Consequences of sarcoidosis. Clin. Chest Med. 2015, 36, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Judson, M.A.; Costabel, U.; Drent, M.; Wells, A.; Maier, L.; Koth, L.; Shigemitsu, H.; Culver, D.A.; Gelfand, J.; Valeyre, D.; et al. The wasog sarcoidosis organ assessment instrument: An update of a previous clinical tool. Sarcoidosis Vasc. Diffuse Lung Dis. 2014, 31, 19–27. [Google Scholar]
- Drent, M.; Marcellis, R.; Lenssen, A.; De Vries, J. Association between physical functions and quality of life in sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 2014, 31, 117–128. [Google Scholar]
- Wijsenbeek, M.S.; Culver, D.A. Treatment of sarcoidosis. Clin. Chest Med. 2015, 36, 751–767. [Google Scholar] [CrossRef]
- Voortman, M.; Hendriks, C.M.R.; Elfferich, M.D.P.; Bonella, F.; Moller, J.; De Vries, J.; Costabel, U.; Drent, M. The burden of sarcoidosis symptoms from a patient perspective. Lung 2019, 197, 155–161. [Google Scholar] [CrossRef]
- Hendriks, C.M.R.; Saketkoo, L.A.; Elfferich, M.D.P.; De Vries, J.; Wijnen, P.; Drent, M. Sarcoidosis and work participation: The need to develop a disease-specific core set for assessment of work ability. Lung 2019, 197, 407–413. [Google Scholar] [CrossRef]
- Waverijn, G.; Spreeuwenberg, P.; Heijmans, M. Leven met een Longziekte in Nederland: Cijfers en Trends over de zorg- en Leefsituatie van Mensen met een Longziekte 2016. Available online: https://www.narcis.nl/publication/RecordID/publicat%3A1003084 (accessed on 30 January 2017).
- Arkema, E.V.; Eklund, A.; Grunewald, J.; Bruze, G. Work ability before and after sarcoidosis diagnosis in sweden. Respir. Med. 2018, 144, S7–S12. [Google Scholar] [CrossRef]
- Voortman, M.; Hendriks, C.M.R.; Lodder, P.; Drent, M.; De Vries, J. Quality of life of couples living with sarcoidosis. Respiration 2019, 98, 373–382. [Google Scholar] [CrossRef]
- Wirnsberger, R.M.; de Vries, J.; Breteler, M.H.; van Heck, G.L.; Wouters, E.F.; Drent, M. Evaluation of quality of life in sarcoidosis patients. Respir. Med. 1998, 92, 750–756. [Google Scholar] [CrossRef]
- De Vries, J.; Drent, M. Quality of life and health status in sarcoidosis: A review of the literature. Clin. Chest Med. 2008, 29, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Michielsen, H.J.; Drent, M.; Peros-Golubicic, T.; De Vries, J. Fatigue is associated with quality of life in sarcoidosis patients. Chest 2006, 130, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Michielsen, H.J.; Peros-Golubicic, T.; Drent, M.; De Vries, J. Relationship between symptoms and quality of life in a sarcoidosis population. Respiration 2007, 74, 401–405. [Google Scholar] [CrossRef]
- Jastrzebski, D.; Ziora, D.; Lubecki, M.; Zieleznik, K.; Maksymiak, M.; Hanzel, J.; Poczatek, A.; Kolczynska, A.; Nguyen Thi, L.; Zebrowska, A.; et al. Fatigue in sarcoidosis and exercise tolerance, dyspnea, and quality of life. Adv. Exp. Med. Biol. 2015, 833, 31–36. [Google Scholar]
- Moor, C.C.; van Manen, M.J.G.; van Hagen, P.M.; Miedema, J.R.; van den Toorn, L.M.; Gur-Demirel, Y.; Berendse, A.P.C.; van Laar, J.A.M.; Wijsenbeek, M.S. Needs, perceptions and education in sarcoidosis: A live interactive survey of patients and partners. Lung 2018, 196, 569–575. [Google Scholar] [CrossRef]
- WHO. Measuring Quality of Life. Available online: https://www.who.int/healthinfo/survey/whoqol-qualityoflife/en/ (accessed on 17 December 2019).
- FDA. Guidance for Industry Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/patient-reported-outcome-measures-use-medical-product-development-support-labeling-claims (accessed on 17 December 2019).
- Thunold, R.F.; Løkke, A.; Cohen, A.L.; Hilberg, O.; Bendstrup, E. Patient reported outcome measures (proms) in sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 2017, 34, 2–17. [Google Scholar]
- Judson, M.A. Quality of life in sarcoidosis. Semin. Respir. Crit. Care Med. 2017, 38, 546–558. [Google Scholar] [CrossRef]
- Judson, M.A.; Mack, M.; Beaumont, J.L.; Watt, R.; Barnathan, E.S.; Victorson, D.E. Validation and important differences for the sarcoidosis assessment tool. A new patient-reported outcome measure. Am. J. Respir. Crit. Care Med. 2015, 191, 786–795. [Google Scholar] [CrossRef]
- De Vries, J.; Michielsen, H.; Van Heck, G.L.; Drent, M. Measuring fatigue in sarcoidosis: The fatigue assessment scale (fas). Br. J. Health Psychol. 2004, 9, 279–291. [Google Scholar] [CrossRef]
- Cox, C.E.; Donohue, J.F.; Brown, C.D.; Kataria, Y.P.; Judson, M.A. The sarcoidosis health questionnaire: A new measure of health-related quality of life. Am. J. Respir. Crit. Care Med. 2003, 168, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, C.; Drent, M.; Elfferich, M.; De Vries, J. The fatigue assessment scale: Quality and availability in sarcoidosis and other diseases. Curr. Opin. Pulm. Med. 2018, 24, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Ishaque, S.; Karnon, J.; Chen, G.; Nair, R.; Salter, A.B. A systematic review of randomised controlled trials evaluating the use of patient-reported outcome measures (proms). Qual. Life Res. 2019, 28, 567–592. [Google Scholar] [CrossRef] [PubMed]
- Basch, E. Patient-reported outcomes: An essential component of oncology drug development and regulatory review. Lancet Oncol. 2018, 19, 595–597. [Google Scholar] [CrossRef]
- Khan, N.A.; Donatelli, C.V.; Tonelli, A.R.; Wiesen, J.; Ribeiro Neto, M.L.; Sahoo, D.; Culver, D.A. Toxicity risk from glucocorticoids in sarcoidosis patients. Respir. Med. 2017, 132, 9–14. [Google Scholar] [CrossRef]
- Judson, M.A.; Chaudhry, H.; Louis, A.; Lee, K.; Yucel, R. The effect of corticosteroids on quality of life in a sarcoidosis clinic: The results of a propensity analysis. Respir. Med. 2015, 109, 526–531. [Google Scholar] [CrossRef]
- Victorson, D.E.; Cella, D.; Grund, H.; Judson, M.A. A conceptual model of health-related quality of life in sarcoidosis. Qual. Life Res. 2014, 23, 89–101. [Google Scholar] [CrossRef]
- Pohle, S.; Baty, F.; Brutsche, M. In-hospital disease burden of sarcoidosis in switzerland from 2002 to 2012. PLoS ONE 2016, 11, e0151940. [Google Scholar] [CrossRef]
- Van Helmondt, S.J.; Polish, L.B.; Judson, M.A.; Grutters, J.C. Patient perspectives in sarcoidosis. Curr. Opin. Pulm. Med. 2019, 25, 478–483. [Google Scholar] [CrossRef]
- Elwyn, G.; Durand, M.A.; Song, J.; Aarts, J.; Barr, P.J.; Berger, Z.; Cochran, N.; Frosch, D.; Galasinski, D.; Gulbrandsen, P.; et al. A three-talk model for shared decision making: Multistage consultation process. BMJ 2017, 359, j4891. [Google Scholar] [CrossRef]
- Karazivan, P.; Dumez, V.; Flora, L.; Pomey, M.P.; Del Grande, C.; Ghadiri, D.P.; Fernandez, N.; Jouet, E.; Las Vergnas, O.; Lebel, P. The patient-as-partner approach in health care: A conceptual framework for a necessary transition. Acad. Med. 2015, 90, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Maier, L.A.; Crouser, E.D.; Martin, W.J.; Eu, J. Executive summary of the nhlbi workshop report: Leveraging current scientific advancements to understand sarcoidosis variability and improve outcomes. Ann. Am. Thorac. Soc. 2017, 14, S415–S420. [Google Scholar] [CrossRef] [PubMed]
- Tavee, J.; Culver, D. Nonorgan manifestations of sarcoidosis. Curr. Opin. Pulm. Med. 2019, 25, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Bonella, F.; Wijsenbeek, M.; Molina-Molina, M.; Duck, A.; Mele, R.; Geissler, K.; Wuyts, W. European idiopathic pulmonary fibrosis patient charter: A missed opportunity. Eur. Respir. J. 2016, 48, 283–284. [Google Scholar] [CrossRef] [PubMed]
- Kingod, N.; Cleal, B.; Wahlberg, A.; Husted, G.R. Online peer-to-peer communities in the daily lives of people with chronic illness: A qualitative systematic review. Qual. Health Res. 2017, 27, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Bodenheimer, T.; Wagner, E.H.; Grumbach, K. Improving primary care for patients with chronic illness. JAMA 2002, 288, 1775–1779. [Google Scholar] [CrossRef]
- Schulman-Green, D.; Jaser, S.S.; Park, C.; Whittemore, R. A metasynthesis of factors affecting self-management of chronic illness. J. Adv. Nurs. 2016, 72, 1469–1489. [Google Scholar] [CrossRef]
- Moor, C.C.; Gur-Demirel, Y.; Wijsenbeek, M.S. Feasibility of a comprehensive home monitoring program for sarcoidosis. J. Pers. Med. 2019. [Google Scholar] [CrossRef]
- Broos, C.E.; Wapenaar, M.; Looman, C.W.N.; van den Toorn, L.M.; Overbeek, M.J.; Grootenboers, M.; Heller, R.; Mostard, R.L.; Poell, L.H.; Hoogsteden, H.C.; et al. Daily home spirometry to detect early steroid treatment effects in newly treated pulmonary sarcoidosis. Eur. Respir. J. 2018, 51, 1702089. [Google Scholar] [CrossRef]
- Bahmer, T.; Watz, H.; Develaska, M.; Waschki, B.; Rabe, K.F.; Magnussen, H.; Kirsten, D.; Kirsten, A.M. Physical activity and fatigue in patients with sarcoidosis. Respiration 2018, 95, 18–26. [Google Scholar] [CrossRef]
- Froidure, S.; Kyheng, M.; Grosbois, J.M.; Lhuissier, F.; Stelianides, S.; Wemeau, L.; Wallaert, B. Daily life physical activity in patients with chronic stage iv sarcoidosis: A multicenter cohort study. Health Sci. Rep. 2019, 2, e109. [Google Scholar] [CrossRef] [PubMed]
- Pilzak, K.; Zebrowska, A.; Sikora, M.; Hall, B.; Lakomy, O.; Kostorz, S.; Ziora, D.; Jastrzebski, D. Physical functioning and symptoms of chronic fatigue in sarcoidosis patients. Adv. Exp. Med. Biol. 2018, 1040, 13–21. [Google Scholar] [PubMed]
- Strookappe, B.; Saketkoo, L.A.; Elfferich, M.; Holland, A.; De Vries, J.; Knevel, T.; Drent, M. Physical activity and training in sarcoidosis: Review and experience-based recommendations. Expert Rev. Respir. Med. 2016, 10, 1057–1068. [Google Scholar] [CrossRef] [PubMed]
- Strookappe, B.; Swigris, J.; De Vries, J.; Elfferich, M.; Knevel, T.; Drent, M. Benefits of physical training in sarcoidosis. Lung 2015, 193, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Marcellis, R.; Van der Veeke, M.; Mesters, I.; Drent, M.; De Bie, R.; De Vries, G.; Lenssen, A. Does physical training reduce fatigue in sarcoidosis? Sarcoidosis Vasc. Diffuse Lung Dis. 2015, 32, 53–62. [Google Scholar]
- Naz, I.; Ozalevli, S.; Ozkan, S.; Sahin, H. Efficacy of a structured exercise program for improving functional capacity and quality of life in patients with stage 3 and 4 sarcoidosis: A randomized controlled trial. J. Cardiopulm. Rehabil. Prev. 2018, 38, 124–130. [Google Scholar] [CrossRef]
- Karadalli, M.N.; Bosnak-Guclu, M.; Camcioglu, B.; Kokturk, N.; Turktas, H. Effects of inspiratory muscle training in subjects with sarcoidosis: A randomized controlled clinical trial. Respir. Care 2016, 61, 483–494. [Google Scholar] [CrossRef]
- Holland, A.E.; Dowman, L.M.; Hill, C.J. Principles of rehabilitation and reactivation: Interstitial lung disease, sarcoidosis and rheumatoid disease with respiratory involvement. Respiration 2015, 89, 89–99. [Google Scholar] [CrossRef]
- Goracci, A.; Fagiolini, A.; Martinucci, M.; Calossi, S.; Rossi, S.; Santomauro, T.; Mazzi, A.; Penza, F.; Fossi, A.; Bargagli, E.; et al. Quality of life, anxiety and depression in sarcoidosis. Gen. Hosp. Psychiatry 2008, 30, 441–445. [Google Scholar] [CrossRef]
- Saketkoo, L.A.; Karpinski, A.; Young, J.; Adell, R.; Walker, M.; Hennebury, T.; Wickremasinghe, M.; Russell, A.M. Feasibility, utility and symptom impact of modified mindfulness training in sarcoidosis. ERJ Open Res. 2018, 4, 00085-2017. [Google Scholar] [CrossRef]
- TIRED trial: A randomized controlled trial of a web-based psychological Intervention to reduce stress and fatigue in patients with sarcoidosis. Available online: https://www.trialregister.nl/trial/7816 (accessed on 20 June 2019).
- Tully, T.; Birring, S.S. Cough in sarcoidosis. Lung 2016, 194, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Sinha, A.; Lee, K.K.; Rafferty, G.F.; Yousaf, N.; Pavord, I.D.; Galloway, J.; Birring, S.S. Predictors of objective cough frequency in pulmonary sarcoidosis. Eur. Respir. J. 2016, 47, 1461–1471. [Google Scholar] [CrossRef] [PubMed]
- Baughman, R.P.; Iannuzzi, M.C.; Lower, E.E.; Moller, D.R.; Balkissoon, R.C.; Winget, D.B.; Judson, M.A. Use of fluticasone in acute symptomatic pulmonary sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 2002, 19, 198–204. [Google Scholar] [PubMed]
- du Bois, R.M.; Greenhalgh, P.M.; Southcott, A.M.; Johnson, N.M.; Harris, T.A. Randomized trial of inhaled fluticasone propionate in chronic stable pulmonary sarcoidosis: A pilot study. Eur. Respir. J. 1999, 13, 1345–1350. [Google Scholar] [CrossRef]
- Milman, N.; Graudal, N.; Grode, G.; Munch, E. No effect of high-dose inhaled steroids in pulmonary sarcoidosis: A double-blind, placebo-controlled study. J. Intern. Med. 1994, 236, 285–290. [Google Scholar] [CrossRef]
- Birring, S.S.; Kavanagh, J.E.; Irwin, R.S.; Keogh, K.A.; Lim, K.G.; Ryu, J.H.; Adams, T.M.; Altman, K.W.; Azoulay, E.; Barker, A.F.; et al. Treatment of interstitial lung disease associated cough: Chest guideline and expert panel report. Chest 2018, 154, 904–917. [Google Scholar] [CrossRef]
- Prasse, A.; Zissel, G.; Lutzen, N.; Schupp, J.; Schmiedlin, R.; Gonzalez-Rey, E.; Rensing-Ehl, A.; Bacher, G.; Cavalli, V.; Bevec, D.; et al. Inhaled vasoactive intestinal peptide exerts immunoregulatory effects in sarcoidosis. Am. J. Respir. Crit. Care Med. 2010, 182, 540–548. [Google Scholar] [CrossRef]
- Lower, E.E.; Harman, S.; Baughman, R.P. Double-blind, randomized trial of dexmethylphenidate hydrochloride for the treatment of sarcoidosis-associated fatigue. Chest 2008, 133, 1189–1195. [Google Scholar] [CrossRef]
- Lower, E.E.; Malhotra, A.; Surdulescu, V.; Baughman, R.P. Armodafinil for sarcoidosis-associated fatigue: A double-blind, placebo-controlled, crossover trial. J. Pain Symptom Manag. 2013, 45, 159–169. [Google Scholar] [CrossRef]
- Elfferich, M.D.; Nelemans, P.J.; Ponds, R.W.; De Vries, J.; Wijnen, P.A.; Drent, M. Everyday cognitive failure in sarcoidosis: The prevalence and the effect of anti-tnf-alpha treatment. Respiration 2010, 80, 212–219. [Google Scholar] [CrossRef]
- Parambil, J.G.; Tavee, J.O.; Zhou, L.; Pearson, K.S.; Culver, D.A. Efficacy of intravenous immunoglobulin for small fiber neuropathy associated with sarcoidosis. Respir. Med. 2011, 105, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Tavee, J.O.; Karwa, K.; Ahmed, Z.; Thompson, N.; Parambil, J.; Culver, D.A. Sarcoidosis-associated small fiber neuropathy in a large cohort: Clinical aspects and response to ivig and anti-tnf alpha treatment. Respir. Med. 2017, 126, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Benn, B.S.; Lehman, Z.; Kidd, S.A.; Miaskowski, C.; Sunwoo, B.Y.; Ho, M.; Sun, S.; Ramstein, J.; Gelfand, J.M.; Koth, L.L. Sleep disturbance and symptom burden in sarcoidosis. Respir. Med. 2018, 144S, S35–S40. [Google Scholar] [CrossRef] [PubMed]
- Fleischer, M.; Hinz, A.; Brahler, E.; Wirtz, H.; Bosse-Henck, A. Factors associated with fatigue in sarcoidosis. Respir. Care 2014, 59, 1086–1094. [Google Scholar] [CrossRef]
- Strookappe, B.; De Vries, J.; Elfferich, M.; Kuijpers, P.; Knevel, T.; Drent, M. Predictors of fatigue in sarcoidosis: The value of exercise testing. Respir. Med. 2016, 116, 49–54. [Google Scholar] [CrossRef]
- van Manen, M.J.G.; Wester, V.L.; van Rossum, E.F.C.; van den Toorn, L.M.; Dorst, K.Y.; de Rijke, Y.B.; Wijsenbeek, M.S. Scalp hair cortisol and testosterone levels in patients with sarcoidosis. PLoS ONE 2019, 14, e0215763. [Google Scholar] [CrossRef]
- Hendriks, C.; Drent, M.; De Kleijn, W.; Elfferich, M.; Wijnen, P.; De Vries, J. Everyday cognitive failure and depressive symptoms predict fatigue in sarcoidosis: A prospective follow-up study. Respir. Med. 2018, 138S, S24–S30. [Google Scholar] [CrossRef]
- Culver, D.A.; Dahan, A.; Bajorunas, D.; Jeziorska, M.; van Velzen, M.; Aarts, L.; Tavee, J.; Tannemaat, M.R.; Dunne, A.N.; Kirk, R.I.; et al. Cibinetide improves corneal nerve fiber abundance in patients with sarcoidosis-associated small nerve fiber loss and neuropathic pain. Invest. Ophthalmol. Vis. Sci. 2017, 58, BIO52–BIO60. [Google Scholar] [CrossRef]
- Ireland, J.; Wilsher, M. Perceptions and beliefs in sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 2010, 27, 36–42. [Google Scholar]
- Chang, B.; Steimel, J.; Moller, D.R.; Baughman, R.P.; Judson, M.A.; Yeager, H., Jr.; Teirstein, A.S.; Rossman, M.D.; Rand, C.S. Depression in sarcoidosis. Am. J. Respir. Crit. Care Med. 2001, 163, 329–334. [Google Scholar] [CrossRef]
- Drent, M.; Wirnsberger, R.M.; Breteler, M.H.; Kock, L.M.; de Vries, J.; Wouters, E.F. Quality of life and depressive symptoms in patients suffering from sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 1998, 15, 59–66. [Google Scholar] [PubMed]
- Martusewicz-Boros, M.M.; Boros, P.W.; Wiatr, E.; Roszkowski-Sliz, K. What comorbidities accompany sarcoidosis? A large cohort (n=1779) patients analysis. Sarcoidosis Vasc. Diffuse Lung Dis. 2015, 32, 115–120. [Google Scholar] [PubMed]
- Brito-Zeron, P.; Acar-Denizli, N.; Siso-Almirall, A.; Bosch, X.; Hernandez, F.; Vilanova, S.; Villalta, M.; Kostov, B.; Paradela, M.; Sanchez, M.; et al. The burden of comorbidity and complexity in sarcoidosis: Impact of associated chronic diseases. Lung 2018, 196, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Nowinski, A.; Puscinska, E.; Goljan, A.; Peradzynska, J.; Bednarek, M.; Korzybski, D.; Kaminski, D.; Stoklosa, A.; Czystowska, M.; Sliwinski, P.; et al. The influence of comorbidities on mortality in sarcoidosis: A observational prospective cohort study. Clin. Respir. J. 2017, 11, 648–656. [Google Scholar] [CrossRef] [PubMed]
- Westney, G.E.; Habib, S.; Quarshie, A. Comorbid illnesses and chest radiographic severity in african-american sarcoidosis patients. Lung 2007, 185, 131–137. [Google Scholar] [CrossRef] [PubMed]
- James, W.E.; Baughman, R. Treatment of sarcoidosis: Grading the evidence. Expert Rev. Clin. Pharmacol. 2018, 11, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Paramothayan, N.S.; Lasserson, T.J.; Jones, P.W. Corticosteroids for pulmonary sarcoidosis. Cochrane Database Syst. Rev. 2005, CD001114. [Google Scholar] [CrossRef]
- Statement on sarcoidosis. Joint statement of the american thoracic society (ats), the european respiratory society (ers) and the world association of sarcoidosis and other granulomatous disorders (wasog) adopted by the ats board of directors and by the ers executive committee, february 1999. Am. J. Respir. Crit. Care Med. 1999, 160, 736–755. [Google Scholar]
- Aryal, S.; Nathan, S.D. Contemporary optimized practice in the management of pulmonary sarcoidosis. Ther. Adv. Respir. Dis. 2019, 13, 1753466619868935. [Google Scholar] [CrossRef]
- Sharma, O.P.; Colp, C.; Williams, M.H., Jr. Course of pulmonary sarcoidosis with and without corticosteriod therapy as determined by pulmonary function studies. Am. J. Med. 1966, 41, 541–551. [Google Scholar] [CrossRef]
- Johns, C.J.; Zachary, J.B.; Ball, W.C., Jr. A ten-year study of corticosteroid treatment of pulmonary sarcoidosis. Johns Hopkins Med. J. 1974, 134, 271–283. [Google Scholar] [PubMed]
- Cremers, J.P.; Drent, M.; Bast, A.; Shigemitsu, H.; Baughman, R.P.; Valeyre, D.; Sweiss, N.J.; Jansen, T.L. Multinational evidence-based world association of sarcoidosis and other granulomatous disorders recommendations for the use of methotrexate in sarcoidosis: Integrating systematic literature research and expert opinion of sarcoidologists worldwide. Curr. Opin Pulm. Med. 2013, 19, 545–561. [Google Scholar] [CrossRef] [PubMed]
- Vorselaars, A.D.M.; Wuyts, W.A.; Vorselaars, V.M.M.; Zanen, P.; Deneer, V.H.M.; Veltkamp, M.; Thomeer, M.; van Moorsel, C.H.M.; Grutters, J.C. Methotrexate vs azathioprine in second-line therapy of sarcoidosis. Chest 2013, 144, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Baughman, R.P.; Winget, D.B.; Lower, E.E. Methotrexate is steroid sparing in acute sarcoidosis: Results of a double blind, randomized trial. Sarcoidosis Vasc. Diffuse Lung Dis. 2000, 17, 60–66. [Google Scholar]
- Adams, J.S.; Diz, M.M.; Sharma, O.P. Effective reduction in the serum 1,25-dihydroxyvitamin d and calcium concentration in sarcoidosis-associated hypercalcemia with short-course chloroquine therapy. Ann. Intern. Med. 1989, 111, 437–438. [Google Scholar] [CrossRef]
- Zic, J.A.; Horowitz, D.H.; Arzubiaga, C.; King, L.E., Jr. Treatment of cutaneous sarcoidosis with chloroquine. Review of the literature. Arch. Dermatol. 1991, 127, 1034–1040. [Google Scholar] [CrossRef]
- Rossman, M.D.; Newman, L.S.; Baughman, R.P.; Teirstein, A.; Weinberger, S.E.; Miller, W., Jr.; Sands, B.E. A double-blinded, randomized, placebo-controlled trial of infliximab in subjects with active pulmonary sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 2006, 23, 201–208. [Google Scholar]
- Judson, M.A.; Baughman, R.P.; Costabel, U.; Flavin, S.; Lo, K.H.; Kavuru, M.S.; Drent, M.; Centocor, T.S.I. Efficacy of infliximab in extrapulmonary sarcoidosis: Results from a randomised trial. Eur. Respir. J. 2008, 31, 1189–1196. [Google Scholar] [CrossRef]
- Vorselaars, A.D.; Crommelin, H.A.; Deneer, V.H.; Meek, B.; Claessen, A.M.; Keijsers, R.G.; van Moorsel, C.H.; Grutters, J.C. Effectiveness of infliximab in refractory fdg pet-positive sarcoidosis. Eur. Respir. J. 2015, 46, 175–185. [Google Scholar] [CrossRef]
- Crommelin, H.A.; van der Burg, L.M.; Vorselaars, A.D.; Drent, M.; van Moorsel, C.H.; Rijkers, G.T.; Deneer, V.H.; Grutters, J.C. Efficacy of adalimumab in sarcoidosis patients who developed intolerance to infliximab. Respir. Med. 2016, 115, 72–77. [Google Scholar] [CrossRef]
- Flaherty, K.R.; Wells, A.U.; Cottin, V.; Devaraj, A.; Walsh, S.L.F.; Inoue, Y.; Richeldi, L.; Kolb, M.; Tetzlaff, K.; Stowasser, S.; et al. Nintedanib in progressive fibrosing interstitial lung diseases. N. Engl. J. Med. 2019, 381, 1718–1727. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.J.; Kallenbach, L.R.; Kreider, M.; Leung, T.H.; Rosenbach, M. Resolution of cutaneous sarcoidosis after janus kinase inhibitor therapy for concomitant polycythemia vera. JAAD Case Rep. 2019, 5, 360–361. [Google Scholar] [CrossRef] [PubMed]
- Drent, M. Sarcoidosis: Benefits of a multidisciplinary approach. Eur. J. Intern. Med. 2003, 14, 217–220. [Google Scholar] [CrossRef]
| Symptom | Pharmacological Treatment | Non-Pharmacological Treatment |
|---|---|---|
| Dyspnea | Regular disease-modifying treatment Treat other causes Supplemental oxygen (in case of hypoxemia) | Physical training, pulmonary rehabilitation [58,59,60] Cognitive behavioral therapy [63] |
| Cough | Regular disease-modifying treatment Treat other causes Inhaled corticosteroids * | Multimodality speech pathology therapy [70] |
| Fatigue | Treat other causes and comorbidities Neurostimulants: armodafinil, (dex)methylphenidate [72,73] TNF inhibitor treatment [74] | Treat reversible causes i.e., obstructive sleep apnea, obesity, depression Physical training or pulmonary rehabilitation [57,58,59,60] Psychosocial counselling [13] Cognitive behavioral therapy [63] |
| Depression and Anxiety | Antidepressants [13] ** Anxiolytics ** | Cognitive behavioral therapy [63] Pulmonary rehabilitation [58] Psychological counselling [46] |
| Small-Fiber Neuropathy | Antidepressants Anticonvulsants Topical anesthetics Opioids [46] Intravenous immunoglobulin [75,76] TNF inhibitor treatment [76] | Mindfulness-based therapy [46] |
| Cognitive Impairment | TNF inhibitor treatment [74] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moor, C.C.; Kahlmann, V.; Culver, D.A.; Wijsenbeek, M.S. Comprehensive Care for Patients with Sarcoidosis. J. Clin. Med. 2020, 9, 390. https://doi.org/10.3390/jcm9020390
Moor CC, Kahlmann V, Culver DA, Wijsenbeek MS. Comprehensive Care for Patients with Sarcoidosis. Journal of Clinical Medicine. 2020; 9(2):390. https://doi.org/10.3390/jcm9020390
Chicago/Turabian StyleMoor, Catharina C., Vivienne Kahlmann, Daniel A. Culver, and Marlies S. Wijsenbeek. 2020. "Comprehensive Care for Patients with Sarcoidosis" Journal of Clinical Medicine 9, no. 2: 390. https://doi.org/10.3390/jcm9020390
APA StyleMoor, C. C., Kahlmann, V., Culver, D. A., & Wijsenbeek, M. S. (2020). Comprehensive Care for Patients with Sarcoidosis. Journal of Clinical Medicine, 9(2), 390. https://doi.org/10.3390/jcm9020390





