Experience in Multiple Sclerosis Patients with COVID-19 and Disease-Modifying Therapies: A Review of 873 Published Cases
Abstract
1. Introduction
2. Materials & Methods
3. Results
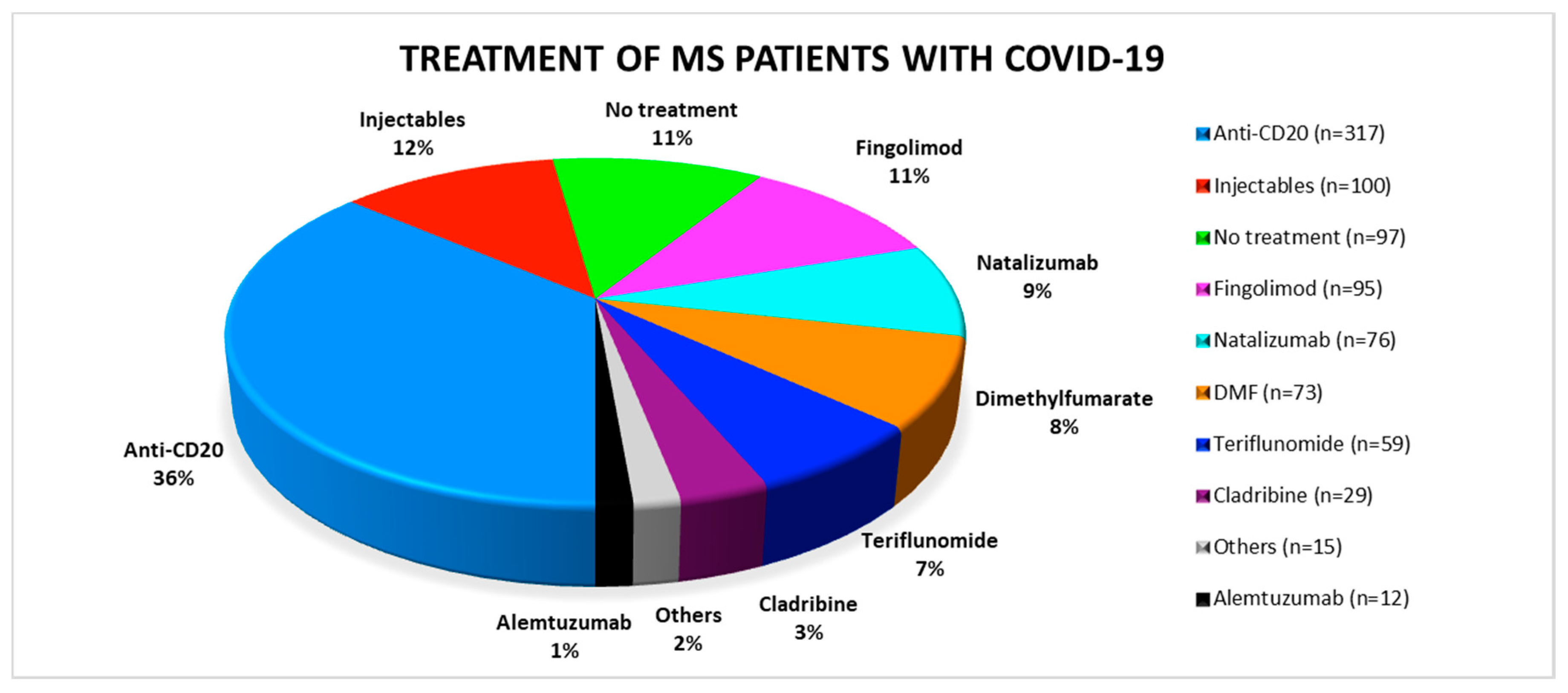
3.1. Distribution of DMTs among Published SARS-CoV-2 Positive MS Patients
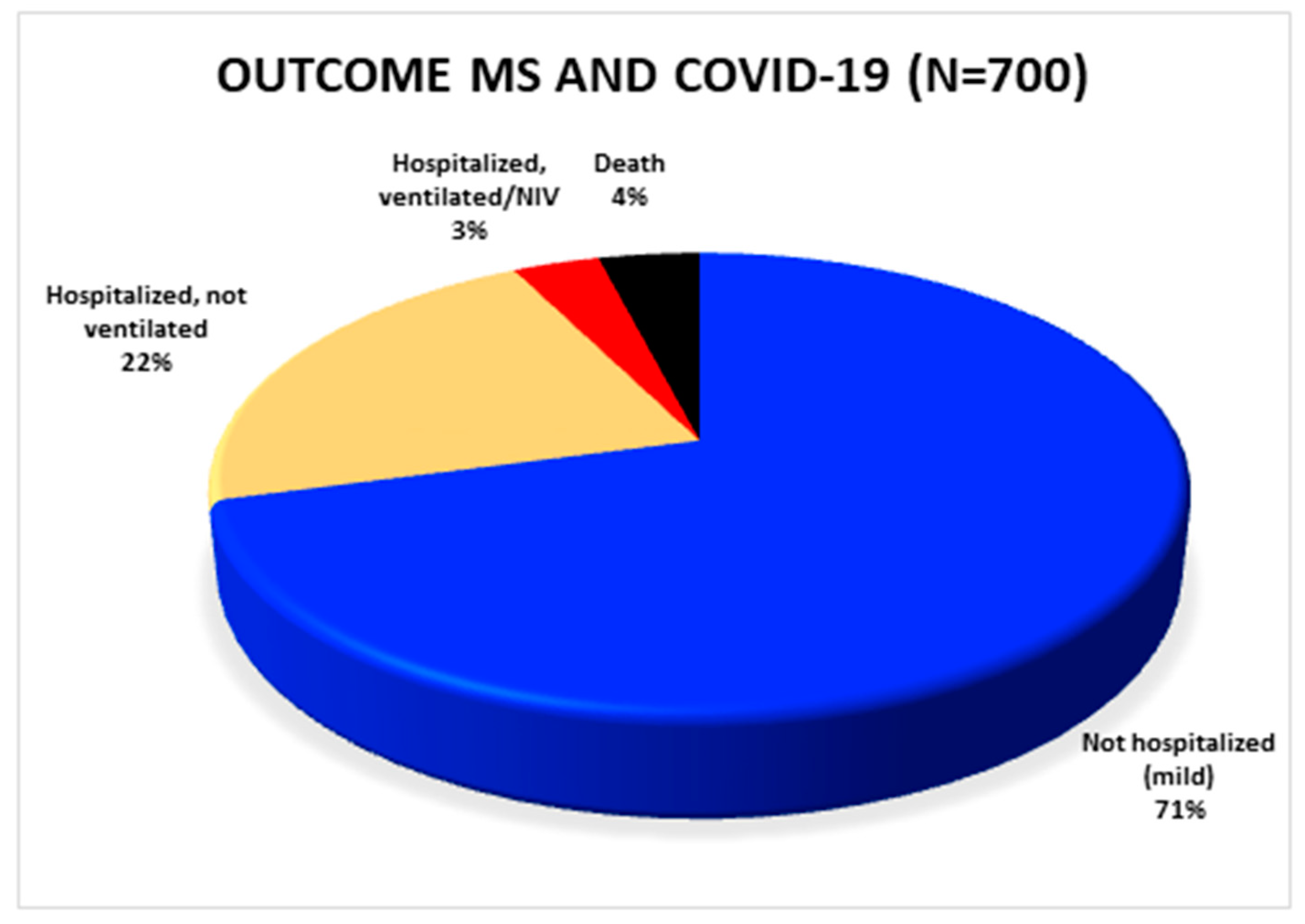
3.2. Outcome of SARS-CoV-2 Positive MS Patients
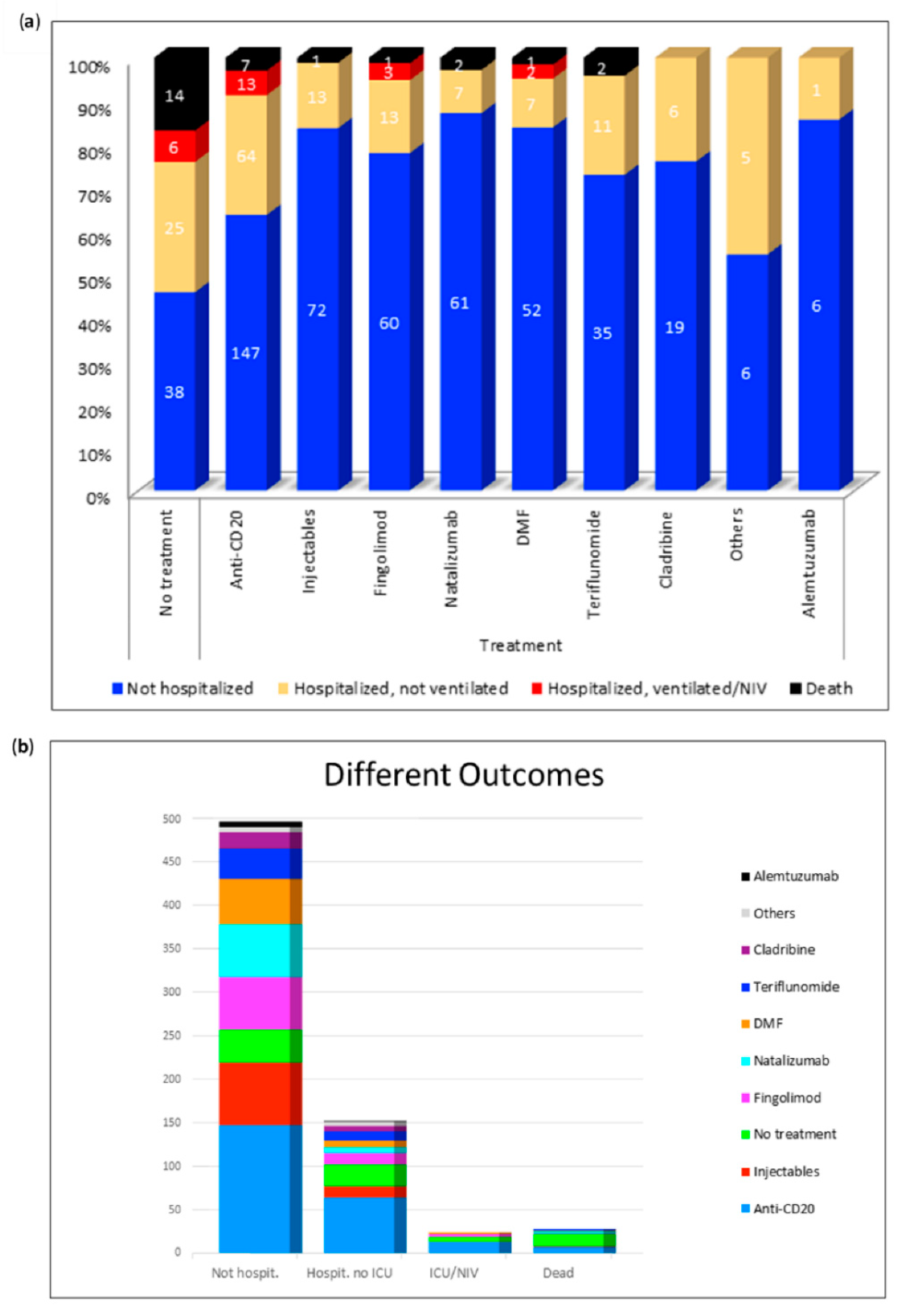
3.3. Outcome of SARS-CoV-2 Positive MS Patients in Association with the Respective DMTs
3.4. Characteristics of Deceased Patients
3.5. Analysis of Detailed Case Reports
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- Force, A.D.T.; Ranieri, V.M.; Rubenfeld, G.D.; Thompson, B.T.; Ferguson, N.D.; Caldwell, E.; Fan, E.; Camporota, L.; Slutsky, A.S. Acute respiratory distress syndrome: The Berlin Definition. JAMA 2012, 307, 2526–2533. [Google Scholar] [CrossRef]
- Chu, D.K.W.; Pan, Y.; Cheng, S.M.S.; Hui, K.P.Y.; Krishnan, P.; Liu, Y.; Ng, D.Y.M.; Wan, C.K.C.; Yang, P.; Wang, Q.; et al. Molecular diagnosis of a novel coronavirus (2019-nCoV) causing an outbreak of pneumonia. Clin. Chem. 2020, 66, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Ashour, H.M.; Elkhatib, W.F.; Rahman, M.M.; Elshabrawy, H.A. Insights into the Recent 2019 Novel coronavirus (SARS-CoV-2) in light of past human coronavirus outbreaks. Pathogens 2020, 9, 186. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.B.; June, C.H. Cytokine release syndrome in severe COVID-19. Science 2020, 368, 473–474. [Google Scholar] [CrossRef]
- Zhang, C.; Wu, Z.; Li, J.W.; Zhao, H.; Wang, G.Q. Cytokine release syndrome in severe COVID-19: Interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int. J. Antimicrob. Agents 2020, 55, 105954. [Google Scholar] [CrossRef]
- Grifoni, A.; Weiskopf, D.; Ramirez, S.I.; Mateus, J.; Dan, J.M.; Moderbacher, C.R.; Rawlings, S.A.; Sutherland, A.; Premkumar, L.; Jadi, R.S.; et al. Targets of T Cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell 2020, 181, 1489–1501.e15. [Google Scholar] [CrossRef]
- Park, M.D. Macrophages: A Trojan horse in COVID-19? Nat. Rev. Immun. 2020, 20, 351. [Google Scholar] [CrossRef]
- Yuki, K.; Fujiogi, M.; Koutsogiannaki, S. COVID-19 pathophysiology: A review. Clin. Immun. 2020, 215, 108427. [Google Scholar] [CrossRef]
- Goumenou, M.; Sarigiannis, D.; Tsatsakis, A.; Anesti, O.; Docea, A.O.; Petrakis, D.; Tsoukalas, D.; Kostoff, R.; Rakitskii, V.; Spandidos, D.A.; et al. COVID19 in Northern Italy: An integrative overview of factors possibly influencing the sharp increase of the outbreak (Review). Mol. Med. Rep. 2020, 22, 20–32. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Available online: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/groups-at-higher-risk.html (accessed on 18 October 2020).
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef]
- Mantero, V.; Abate, L.; Balgera, R.; Basilico, P.; Salmaggi, A.; Cordano, C. Assessing the susceptibility to acute respiratory illness COVID-19-related in a cohort of multiple sclerosis patients. Mult. Scler. Relat. Disord. 2020, 46, 102453. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Jung, S.; Kim, A.; Park, J.E. MERS transmission and risk factors: A systematic review. BMC Public Health 2018, 18, 574. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.S.; Zheng, J.P.; Mok, Y.W.; Li, Y.M.; Liu, Y.N.; Chu, C.M.; Ip, M.S. SARS: Prognosis, outcome and sequelae. Respirology 2003, 8, S36–S40. [Google Scholar] [CrossRef] [PubMed]
- Mohn, N.; Pul, R.; Kleinschnitz, C.; Pruss, H.; Witte, T.; Stangel, M.; Skripuletz, T. Implications of COVID-19 outbreak on immune therapies in multiple sclerosis patients-lessons learned from SARS and MERS. Front. Immunol. 2020, 11, 1059. [Google Scholar] [CrossRef]
- Owji, H.; Negahdaripour, M.; Hajighahramani, N. Immunotherapeutic approaches to curtail COVID-19. Int. Immunopharmacol. 2020, 88, 106924. [Google Scholar] [CrossRef]
- Louapre, C.; Collongues, N.; Stankoff, B.; Giannesini, C.; Papeix, C.; Bensa, C.; Deschamps, R.; Creange, A.; Wahab, A.; Pelletier, J.; et al. Clinical characteristics and outcomes in patients with coronavirus disease 2019 and Multiple Sclerosis. JAMA Neurol. 2020, 77, 1079–1088. [Google Scholar] [CrossRef]
- Parrotta, E.; Kister, I.; Charvet, L.; Sammarco, C.; Saha, V.; Charlson, R.E.; Howard, J.; Gutman, J.M.; Gottesman, M.; Abou-Fayssal, N.; et al. COVID-19 outcomes in MS: Observational study of early experience from NYU Multiple Sclerosis Comprehensive Care Center. Neurol. R Neuroimmunol. Neuroinflamm. 2020, 7. [Google Scholar] [CrossRef]
- Aguirre, C.; Meca-Lallana, V.; Barrios-Blandino, A.; Del Rio, B.; Vivancos, J. Covid-19 in a patient with multiple sclerosis treated with natalizumab: May the blockade of integrins have a protective role? Mult. Scler. Relat. Disord. 2020, 44, 102250. [Google Scholar] [CrossRef]
- Arca, K.N.; Starling, A.J. Treatment-refractory headache in the setting of COVID-19 pneumonia: Migraine or meningoencephalitis? Case Report. SN Compr. Clin. Med. 2020, 1–4. [Google Scholar] [CrossRef]
- Barzegar, M.; Mirmosayyeb, O.; Nehzat, N.; Sarrafi, R.; Khorvash, F.; Maghzi, A.H.; Shaygannejad, V. COVID-19 infection in a patient with multiple sclerosis treated with fingolimod. Neurol. R Neuroimmunol. Neuroinflamm. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Carandini, T.; Pietroboni, A.M.; Sacchi, L.; De Riz, M.A.; Pozzato, M.; Arighi, A.; Fumagalli, G.G.; Martinelli Boneschi, F.; Galimberti, D.; Scarpini, E. Alemtuzumab in multiple sclerosis during the COVID-19 pandemic: A mild uncomplicated infection despite intense immunosuppression. Mult. Scler. 2020, 26, 1268–1269. [Google Scholar] [CrossRef] [PubMed]
- Celius, E.G. Normal antibody response after COVID-19 during treatment with cladribine. Mult. Scler. Relat. Disord. 2020, 46, 102476. [Google Scholar] [CrossRef] [PubMed]
- Chiarini, M.; Paghera, S.; Moratto, D.; Rossi, N.; Giacomelli, M.; Badolato, R.; Capra, R.; Imberti, L. Immunologic characterization of a immunosuppressed multiple sclerosis patient that recovered from SARS-CoV-2 infection. J. Neuroimmunol. 2020, 345, 577282. [Google Scholar] [CrossRef]
- Ciardi, M.R.; Zingaropoli, M.A.; Pasculli, P.; Perri, V.; Tartaglia, M.; Valeri, S.; Russo, G.; Conte, A.; Mastroianni, C.M. The peripheral blood immune cell profile in a teriflunomide-treated multiple sclerosis patient with COVID-19 pneumonia. J. Neuroimmunol. 2020, 346, 577323. [Google Scholar] [CrossRef]
- Conte, W.L. Attenuation of antibody response to SARS-CoV-2 in a patient on ocrelizumab with hypogammaglobulinemia. Mult. Scler. Relat. Disord. 2020, 44, 102315. [Google Scholar] [CrossRef]
- De Angelis, M.; Petracca, M.; Lanzillo, R.; Brescia Morra, V.; Moccia, M. Mild or no COVID-19 symptoms in cladribine-treated multiple sclerosis: Two cases and implications for clinical practice. Mult. Scler. Relat. Disord. 2020, 45, 102452. [Google Scholar] [CrossRef]
- Dersch, R.; Wehrum, T.; Fahndrich, S.; Engelhardt, M.; Rauer, S.; Berger, B. COVID-19 pneumonia in a multiple sclerosis patient with severe lymphopenia due to recent cladribine treatment. Mult. Scler. 2020, 26, 1264–1266. [Google Scholar] [CrossRef]
- Devogelaere, J.; D’Hooghe, M.B.; Vanderhauwaert, F.; D’Haeseleer, M. Coronavirus disease 2019: Favorable outcome in an immunosuppressed patient with multiple sclerosis. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2020, 41, 1981–1983. [Google Scholar] [CrossRef]
- Fernandez-Diaz, E.; Gracia-Gil, J.; Garcia-Garcia, J.G.; Palao, M.; Romero-Sanchez, C.M.; Segura, T. COVID-19 and multiple sclerosis: A description of two cases on alemtuzumab. Mult. Scler. Relat. Disord. 2020, 45, 102402. [Google Scholar] [CrossRef]
- Fiorella, C.; Lorna, G. COVID-19 in a multiple sclerosis (MS) patient treated with alemtuzumab: Insight to the immune response after COVID. Mult. Scler. Relat. Disord. 2020, 46, 102447. [Google Scholar] [CrossRef]
- Foerch, C.; Friedauer, L.; Bauer, B.; Wolf, T.; Adam, E.H. Severe COVID-19 infection in a patient with multiple sclerosis treated with fingolimod. Mult. Scler. Relat. Disord. 2020, 42, 102180. [Google Scholar] [CrossRef] [PubMed]
- Gemcioglu, E.; Davutoglu, M.; Ozdemir, E.E.; Erden, A. Are type 1 interferons treatment in Multiple Sclerosis as a potential therapy against COVID-19? Mult. Scler. Relat. Disord. 2020, 42, 102196. [Google Scholar] [CrossRef] [PubMed]
- Ghajarzadeh, M.; Mirmosayyeb, O.; Barzegar, M.; Nehzat, N.; Vaheb, S.; Shaygannejad, V.; Maghzi, A.H. Favorable outcome after COVID-19 infection in a multiple sclerosis patient initiated on ocrelizumab during the pandemic. Mult. Scler. Relat. Disord. 2020, 43, 102222. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Mayordomo, V.; Montero-Escribano, P.; Matias-Guiu, J.A.; Gonzalez-Garcia, N.; Porta-Etessam, J.; Matias-Guiu, J. Clinical exacerbation of SARS-CoV2 infection after fingolimod withdrawal. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Guevara, C.; Villa, E.; Cifuentes, M.; Naves, R.; Grazia, J. Mild COVID-19 infection in a patient with multiple sclerosis and severe depletion of T-lymphocyte subsets due to alemtuzumab. Mult. Scler. Relat. Disord. 2020, 44, 102314. [Google Scholar] [CrossRef] [PubMed]
- Iannetta, M.; Cesta, N.; Stingone, C.; Malagnino, V.; Teti, E.; Vitale, P.; De Simone, G.; Rossi, B.; Ansaldo, L.; Compagno, M.; et al. Mild clinical manifestations of SARS-CoV-2 related pneumonia in two patients with multiple sclerosis under treatment with ocrelizumab. Mult. Scler. Relat. Disord. 2020, 45, 102442. [Google Scholar] [CrossRef]
- Lucchini, M.; Bianco, A.; Del Giacomo, P.; De Fino, C.; Nociti, V.; Mirabella, M. Is serological response to SARS-CoV-2 preserved in MS patients on ocrelizumab treatment? A case report. Mult. Scler. Relat. Disord. 2020, 44, 102323. [Google Scholar] [CrossRef]
- Maghzi, A.H.; Houtchens, M.K.; Preziosa, P.; Ionete, C.; Beretich, B.D.; Stankiewicz, J.M.; Tauhid, S.; Cabot, A.; Berriosmorales, I.; Schwartz, T.H.W.; et al. COVID-19 in teriflunomide-treated patients with multiple sclerosis. J. Neurol. 2020, 267, 2790–2796. [Google Scholar] [CrossRef]
- Bollo, L.; Guerra, T.; Bavaro, D.F.; Monno, L.; Saracino, A.; Angarano, G.; Paolicelli, D.; Trojano, M.; Iaffaldano, P. Seroconversion and indolent course of COVID-19 in patients with multiple sclerosis treated with fingolimod and teriflunomide. J. Neurol. Sci. 2020, 416, 117011. [Google Scholar] [CrossRef]
- Borriello, G.; Ianniello, A. COVID-19 occurring during Natalizumab treatment: A case report in a patient with extended interval dosing approach. Mult. Scler. Relat. Disord. 2020, 41, 102165. [Google Scholar] [CrossRef] [PubMed]
- Mallucci, G.; Zito, A.; Fabbro, B.D.; Bergamaschi, R. Asymptomatic SARS-CoV-2 infection in two patients with multiple sclerosis treated with fingolimod. Mult. Scler. Relat. Disord. 2020, 45, 102414. [Google Scholar] [CrossRef] [PubMed]
- Meca-Lallana, V.; Aguirre, C.; Beatrizdel, R.; Cardenoso, L.; Alarcon, T.; Vivancos, J. COVID-19 in 7 multiple sclerosis patients in treatment with ANTI-CD20 therapies. Mult. Scler. Relat. Disord. 2020, 44, 102306. [Google Scholar] [CrossRef] [PubMed]
- Mohn, N.; Saker, F.; Bonda, V.; Respondek, G.; Bachmann, M.; Stoll, M.; Wattjes, M.P.; Stangel, M.; Skripuletz, T. Mild COVID-19 symptoms despite treatment with teriflunomide and high-dose methylprednisolone due to multiple sclerosis relapse. J. Neurol. 2020, 267, 2803–2805. [Google Scholar] [CrossRef] [PubMed]
- Novi, G.; Mikulska, M.; Briano, F.; Toscanini, F.; Tazza, F.; Uccelli, A.; Inglese, M. COVID-19 in a MS patient treated with ocrelizumab: Does immunosuppression have a protective role? Mult. Scler. Relat. Disord. 2020, 42, 102120. [Google Scholar] [CrossRef] [PubMed]
- Rimmer, K.; Farber, R.; Thakur, K.; Braverman, G.; Podolsky, D.; Sutherland, L.; Migliore, C.; Ryu, Y.K.; Levin, S.; De Jager, P.L.; et al. Fatal COVID-19 in an MS patient on natalizumab: A case report. Mult. Scler. J. Exp. Transl. Clin. 2020, 6, 2055217320942931. [Google Scholar] [CrossRef] [PubMed]
- Suwanwongse, K.; Shabarek, N. Benign course of COVID-19 in a multiple sclerosis patient treated with Ocrelizumab. Mult. Scler. Relat. Disord. 2020, 42, 102201. [Google Scholar] [CrossRef]
- Thornton, J.R.; Harel, A. Negative SARS-CoV-2 antibody testing following COVID-19 infection in Two MS patients treated with ocrelizumab. Mult. Scler. Relat. Disord. 2020, 44, 102341. [Google Scholar] [CrossRef]
- Valencia-Sanchez, C.; Wingerchuk, D.M. A fine balance: Immunosuppression and immunotherapy in a patient with multiple sclerosis and COVID-19. Mult. Scler. Relat. Disord. 2020, 42, 102182. [Google Scholar] [CrossRef]
- Woo, M.S.; Steins, D.; Haussler, V.; Kohsar, M.; Haag, F.; Elias-Hamp, B.; Heesen, C.; Lutgehetmann, M.; Schulze Zur Wiesch, J.; Friese, M.A. Control of SARS-CoV-2 infection in rituximab-treated neuroimmunological patients. J. Neurol. 2020, 1–3. [Google Scholar] [CrossRef]
- Wurm, H.; Attfield, K.; Iversen, A.K.; Gold, R.; Fugger, L.; Haghikia, A. Recovery from COVID-19 in a B-cell-depleted multiple sclerosis patient. Mult. Scler. 2020, 26, 1261–1264. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.D.; Brink, J.; Brown, T.R.; Lucassen, E.B.; Smoot, K.; Wundes, A.; Repovic, P. COVID-19 in MS: Initial observations from the Pacific Northwest. Neurol. R Neuroimmunol. Neuroinflamm. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Matias-Guiu, J.; Montero-Escribano, P.; Pytel, V.; Porta-Etessam, J.; Matias-Guiu, J.A. Potential COVID-19 infection in patients with severe multiple sclerosis treated with alemtuzumab. Mult. Scler. Relat. Disord. 2020, 44, 102297. [Google Scholar] [CrossRef] [PubMed]
- Mantero, V.; Baroncini, D.; Balgera, R.; Guaschino, C.; Basilico, P.; Annovazzi, P.; Zaffaroni, M.; Salmaggi, A.; Cordano, C. Mild COVID-19 infection in a group of teriflunomide-treated patients with multiple sclerosis. J. Neurol. 2020, 1–2. [Google Scholar] [CrossRef]
- Mantero, V.; Abate, L.; Basilico, P.; Balgera, R.; Salmaggi, A.; Nourbakhsh, B.; Cordano, C. COVID-19 in dimethyl fumarate-treated patients with multiple sclerosis. J. Neurol. 2020, 1–3. [Google Scholar] [CrossRef]
- Sahraian, M.A.; Azimi, A.; Navardi, S.; Rezaeimanesh, N.; Naser Moghadasi, A. Evaluation of COVID-19 infection in patients with Neuromyelitis optica spectrum disorder (NMOSD): A report from Iran. Mult. Scler. Relat. Disord. 2020, 44, 102245. [Google Scholar] [CrossRef]
- Ciampi, E.; Uribe-San-Martin, R.; Carcamo, C. COVID-19 pandemic: The experience of a multiple sclerosis centre in Chile. Mult. Scler. Relat. Disord. 2020, 42, 102204. [Google Scholar] [CrossRef]
- Loonstra, F.C.; Hoitsma, E.; van Kempen, Z.L.; Killestein, J.; Mostert, J.P. COVID-19 in multiple sclerosis: The Dutch experience. Mult. Scler. 2020, 26, 1256–1260. [Google Scholar] [CrossRef]
- Barzegar, M.; Mirmosayyeb, O.; Ghajarzadeh, M.; Nehzat, N.; Vaheb, S.; Shaygannejad, V.; Vosoughi, R. Characteristics of COVID-19 disease in multiple sclerosis patients. Mult. Scler. Relat. Disord. 2020, 45, 102276. [Google Scholar] [CrossRef]
- Crescenzo, F.; Marastoni, D.; Bovo, C.; Calabrese, M. Frequency and severity of COVID-19 in multiple sclerosis: A short single-site report from northern Italy. Mult. Scler. Relat. Disord. 2020, 44, 102372. [Google Scholar] [CrossRef]
- Hughes, R.; Pedotti, R.; Koendgen, H. COVID-19 in persons with multiple sclerosis treated with ocrelizumab—A pharmacovigilance case series. Mult. Scler. Relat. Disord. 2020, 42, 102192. [Google Scholar] [CrossRef] [PubMed]
- Safavi, F.; Nourbakhsh, B.; Azimi, A.R. B-cell depleting therapies may affect susceptibility to acute respiratory illness among patients with multiple sclerosis during the early COVID-19 epidemic in Iran. Mult. Scler. Relat. Disorders 2020, 43, 102195. [Google Scholar] [CrossRef] [PubMed]
- Rejdak, K.; Grieb, P. Adamantanes might be protective from COVID-19 in patients with neurological diseases: Multiple sclerosis, parkinsonism and cognitive impairment. Mult. Scler. Relat. Disord. 2020, 42, 102163. [Google Scholar] [CrossRef] [PubMed]
- Nesbitt, C.; Rath, L.; Yeh, W.Z.; Zhong, M.; Wesselingh, R.; Monif, M.; Richards, J.; Minh, V.B.; Jokubaitis, V.G.; Skibina, O.; et al. MSCOVID19: Using social media to achieve rapid dissemination of health information. Mult. Scler. Relat. Disord. 2020, 45, 102338. [Google Scholar] [CrossRef] [PubMed]
- Ciampi, E.; Uribe-San-Martin, R.; Soler, B.; Fernandez, R.; Garcia, P.; Navarrete-Asenjo, C.; Tirapegui, J.M.; Torres, R.; Polanco, J.; Suarez, F.; et al. COVID-19 in MS and NMOSD: A multicentric online national survey in Chile. Mult. Scler. Relat. Disord. 2020, 45, 102392. [Google Scholar] [CrossRef]
- Montero-Escribano, P.; Matias-Guiu, J.; Gomez-Iglesias, P.; Porta-Etessam, J.; Pytel, V.; Matias-Guiu, J.A. Anti-CD20 and COVID-19 in multiple sclerosis and related disorders: A case series of 60 patients from Madrid, Spain. Mult. Scler. Relat. Disord. 2020, 42, 102185. [Google Scholar] [CrossRef]
- Olivares Gazca, J.C.; Gomez Almaguer, D.; Gale, R.P.; Ruiz Arguelles, G.J. Melange interessante: COVID-19, autologous transplants and multiple sclerosis. Hematology 2020, 25, 320. [Google Scholar] [CrossRef]
- Maillart, E.; Papeix, C.; Lubetzki, C.; Roux, T.; Pourcher, V.; Louapre, C. Beyond COVID-19: DO MS/NMO-SD patients treated with anti-CD20 therapies develop SARS-CoV2 antibodies? Mult. Scler. Relat. Disord. 2020, 46, 102482. [Google Scholar] [CrossRef]
- Jack, D.; Nolting, A.; Galazka, A. Favorable outcomes after COVID-19 infection in multiple sclerosis patients treated with cladribine tablets. Mult. Scler. Relat. Disord. 2020, 46, 102469. [Google Scholar] [CrossRef]
- Louapre, C.; Maillart, E.; Roux, T.; Pourcher, V.; Bussone, G.; Lubetzki, C.; Papeix, C. Patients with MS treated with immunosuppressive agents: Across the COVID-19 spectrum. Rev. Neurol. 2020, 176, 523–525. [Google Scholar] [CrossRef]
- Talotta, R.; Robertson, E. Autoimmunity as the comet tail of COVID-19 pandemic. World J. Clin. Cases 2020, 8, 3621–3644. [Google Scholar] [CrossRef] [PubMed]
- Galeotti, C.; Bayry, J. Autoimmune and inflammatory diseases following COVID-19. Nat. Rev. Rheumatol. 2020, 16, 413–414. [Google Scholar] [CrossRef] [PubMed]
- Cardone, M.; Yano, M.; Rosenberg, A.S.; Puig, M. Lessons learned to date on COVID-19 Hyperinflammatory Syndrome: Considerations for interventions to mitigate SARS-CoV-2 viral infection and detrimental hyperinflammation. Front. Immunol. 2020, 11, 1131. [Google Scholar] [CrossRef]
- Hu, G.; Christman, J.W. Editorial: Alveolar macrophages in lung inflammation and resolution. Front. Immunol. 2019, 10, 2275. [Google Scholar] [CrossRef]
- Joshi, N.; Walter, J.M.; Misharin, A.V. Alveolar macrophages. Cell Immunol. 2018, 330, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xie, J.; Zhao, L.; Fei, X.; Zhang, H.; Tan, Y.; Nie, X.; Zhou, L.; Liu, Z.; Ren, Y.; et al. Alveolar macrophage dysfunction and cytokine storm in the pathogenesis of two severe COVID-19 patients. EBioMedicine 2020, 57, 102833. [Google Scholar] [CrossRef]
- Kloc, M.; Ghobrial, R.M. The multiple sclerosis (MS) drugs as a potential treatment of ARDS in COVID-19 patients. Mult. Scler. Relat. Disord. 2020, 45, 102437. [Google Scholar] [CrossRef]
- Li, X.; Xu, S.; Yu, M.; Wang, K.; Tao, Y.; Zhou, Y.; Shi, J.; Zhou, M.; Wu, B.; Yang, Z.; et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J. Allergy Clin. Immunol. 2020, 146, 110–118. [Google Scholar] [CrossRef]
- Suleyman, G.; Fadel, R.A.; Malette, K.M.; Hammond, C.; Abdulla, H.; Entz, A.; Demertzis, Z.; Hanna, Z.; Failla, A.; Dagher, C.; et al. Clinical characteristics and morbidity associated with coronavirus disease 2019 in a series of patients in metropolitan Detroit. JAMA Netw. Open. 2020, 3, e2012270. [Google Scholar] [CrossRef]
- Yu, C.; Lei, Q.; Li, W.; Wang, X.; Li, W.; Liu, W. Epidemiological and clinical characteristics of 1663 hospitalized patients infected with COVID-19 in Wuhan, China: A single-center experience. J. Infect. Public Health 2020, 13, 1202–1209. [Google Scholar] [CrossRef]
- Petrakis, D.; Margina, D.; Tsarouhas, K.; Tekos, F.; Stan, M.; Nikitovic, D.; Kouretas, D.; Spandidos, D.A.; Tsatsakis, A. Obesity a risk factor for increased COVID19 prevalence, severity and lethality (Review). Mol. Med. Rep. 2020, 22, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Scribano, M.L. Why Do Immunosuppressed patients with inflammatory bowel disease not seem to be at a higher risk of COVID-19? Dig. Dis. Sci. 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pablos, J.L.; Galindo, M.; Carmona, L.; Lledo, A.; Retuerto, M.; Blanco, R.; Gonzalez-Gay, M.A.; Martinez-Lopez, D.; Castrejon, I.; Alvaro-Gracia, J.M.; et al. Clinical outcomes of hospitalised patients with COVID-19 and chronic inflammatory and autoimmune rheumatic diseases: A multicentric matched cohort study. Ann. Rheum. Dis. 2020, 79, 1544–1549. [Google Scholar] [CrossRef] [PubMed]
- Freites Nunez, D.D.; Leon, L.; Mucientes, A.; Rodriguez-Rodriguez, L.; Font Urgelles, J.; Madrid Garcia, A.; Colomer, J.I.; Jover, J.A.; Fernandez-Gutierrez, B.; Abasolo, L. Risk factors for hospital admissions related to COVID-19 in patients with autoimmune inflammatory rheumatic diseases. Ann. Rheum. Dis. 2020, 79, 1393–1399. [Google Scholar] [CrossRef] [PubMed]
| Pat. ID | Sex (m/f) | Age | Disease Duration (years) | Disease Phase | EDSS | DMT | Comorbidities |
|---|---|---|---|---|---|---|---|
| 01 | N/A | 6th decade | 25 | SPMS | 7.5 | None | None |
| 02 | N/A | 8th decade | 51 | SPMS | 8 | None | Chronic myelomonocytic leukemia |
| 03 | N/A | 6th decade | 28 | SPMS | 8.5 | None | Ischemic heart disease, COPD |
| 04 | N/A | 8th decade | 22 | PPMS | 8.5 | None | None |
| 05 | N/A | 6th decade | 48 | SPMS | 9 | None | Colorectal cancer |
| 06 | N/A | 7th decade | 35 | SPMS | 9 | None | Arterial hypertension |
| 07 | N/A | 4th decade | 28 | SPMS | 9.5 | None | None |
| 08 | N/A | N/A | N/A | N/A | 4 | None | Obesity |
| 09 | N/A | N/A | N/A | N/A | 7 | None | Asthma |
| 10 | f | 50 | 13 | RRMS | N/A | None | Obesity, hypertension, hypothyroidism |
| 11 | f | 65 | 31 | SPMS | N/A | None | Intrathecal baclofen pump |
| 12 | N/A | 5th decade | 23 | RRMS | 1.5 | None | Obesity |
| 13 | N/A | 7th decade | 47 | RRMS | 5 | None | None |
| 14 | m | 74 | N/A | SPMS | 8.5 | None | Coronary artery disease, hypertension, diabetes mellitus, COPD, cardiomyopathy |
| 15 | N/A | 5th decade | 22 | PPMS | 7 | Rituximab | None |
| 16 | N/A | N/A | N/A | N/A | N/A | Rituximab | Sjögren’s syndrome, hypothyroidism |
| 17 | N/A | N/A | N/A | N/A | N/A | Rituximab | Obesity |
| 18 | f | 43 | 18 | SPMS | 6.5 | Rituximab | Hypothyroidism |
| 19 | m | 42 | 18 | RRMS | N/A | Rituximab | Hodgkin lymphoma, anticoagulation for VTE, intrathecal baclofen pump |
| 20 | m | 66 | 33 | SPMS | N/A | Ocrelizumab | History of testicular and prostate cancer, intrathecal baclofen pump |
| 21 | N/A | N/A | N/A | N/A | 6 | Ocrelizumab | COPD |
| 22 | f | 60 | 19 | RRMS | N/A | Natalizumab | Obesity, coronary artery disease, hypertension |
| 23 | f | 51 | 14 | RRMS | 6.5 | Natalizumab | Obesity, hypertension, recurrent urinary tract infections |
| 24 | f | 55 | N/A | SPMS | 7.5 | Teriflunomide | Myotonic dystrophy |
| 25 | N/A | 3th decade | 5 | RRMS | 3 | Teriflunomide | Obesity |
| 26 | N/A | N/A | N/A | N/A | N/A | Fingolimod | Severe cognitive impairment |
| 27 | N/A | 5th decade | 5 | RRMS | 3 | Dimethyl-fumarate | Obesity, grade 2 lymphopenia, schizophrenia |
| 28 | m | 71 | 30 | SPMS | N/A | Glatiramer-acetate | Obesity, anticoagulation for VTE |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Möhn, N.; Konen, F.F.; Pul, R.; Kleinschnitz, C.; Prüss, H.; Witte, T.; Stangel, M.; Skripuletz, T. Experience in Multiple Sclerosis Patients with COVID-19 and Disease-Modifying Therapies: A Review of 873 Published Cases. J. Clin. Med. 2020, 9, 4067. https://doi.org/10.3390/jcm9124067
Möhn N, Konen FF, Pul R, Kleinschnitz C, Prüss H, Witte T, Stangel M, Skripuletz T. Experience in Multiple Sclerosis Patients with COVID-19 and Disease-Modifying Therapies: A Review of 873 Published Cases. Journal of Clinical Medicine. 2020; 9(12):4067. https://doi.org/10.3390/jcm9124067
Chicago/Turabian StyleMöhn, Nora, Franz F. Konen, Refik Pul, Christoph Kleinschnitz, Harald Prüss, Torsten Witte, Martin Stangel, and Thomas Skripuletz. 2020. "Experience in Multiple Sclerosis Patients with COVID-19 and Disease-Modifying Therapies: A Review of 873 Published Cases" Journal of Clinical Medicine 9, no. 12: 4067. https://doi.org/10.3390/jcm9124067
APA StyleMöhn, N., Konen, F. F., Pul, R., Kleinschnitz, C., Prüss, H., Witte, T., Stangel, M., & Skripuletz, T. (2020). Experience in Multiple Sclerosis Patients with COVID-19 and Disease-Modifying Therapies: A Review of 873 Published Cases. Journal of Clinical Medicine, 9(12), 4067. https://doi.org/10.3390/jcm9124067








