Fluid Shifts Induced by Physical Therapy in Lower Limb Lymphedema Patients
Abstract
1. Introduction
2. Experimental Section
2.1. Patients
2.2. Complete Decongestive Therapy Protocol
2.3. Data Collection
2.4. Blood Collection
2.5. Assessment of Volume and Fluid Mobilization
2.6. Assessment of Plasma Volume Changes
2.7. Statistics
3. Results
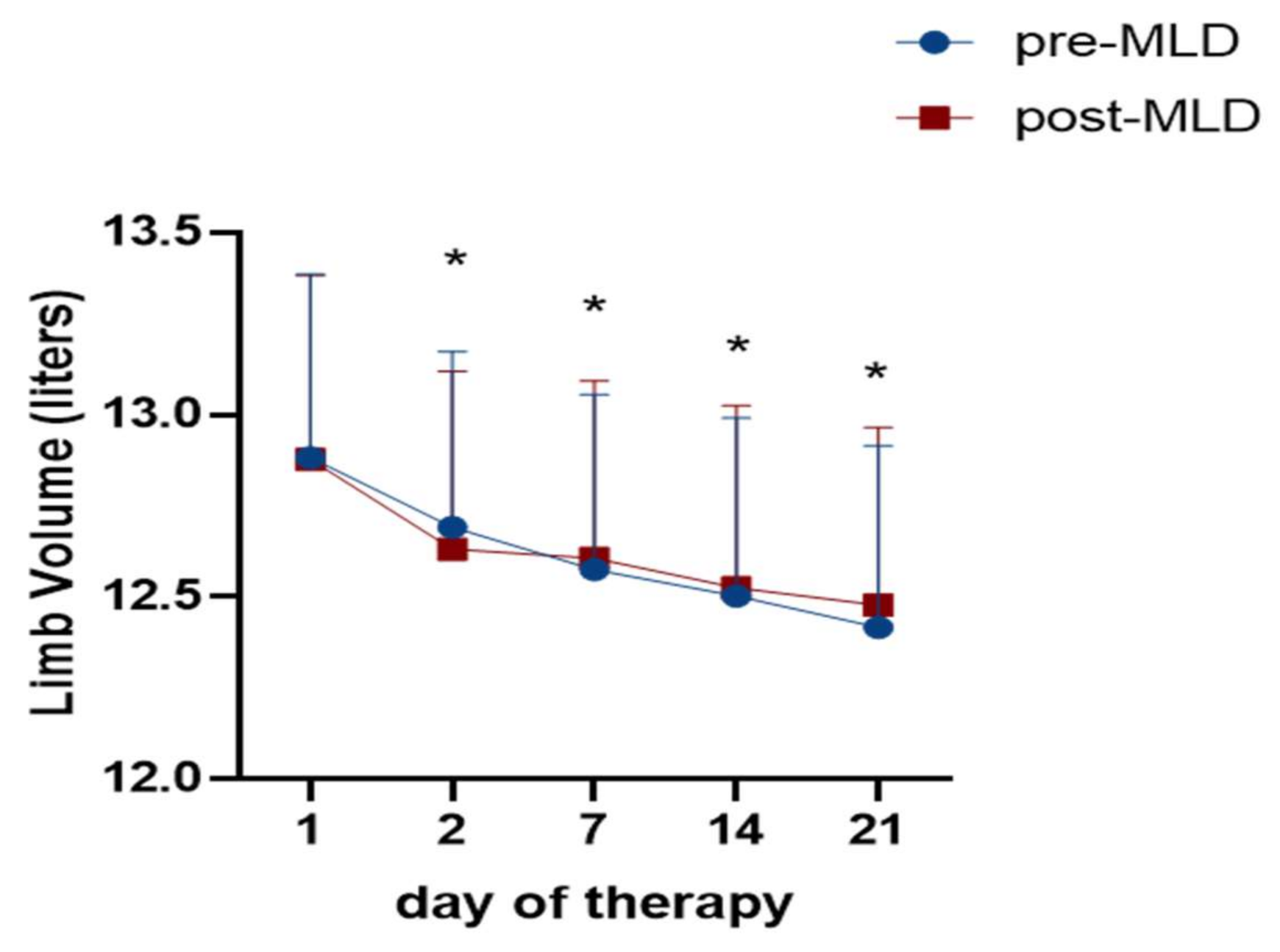
3.1. Reduction of Lower Limb Volume Due to Physical Therapy Assessed via Perometry
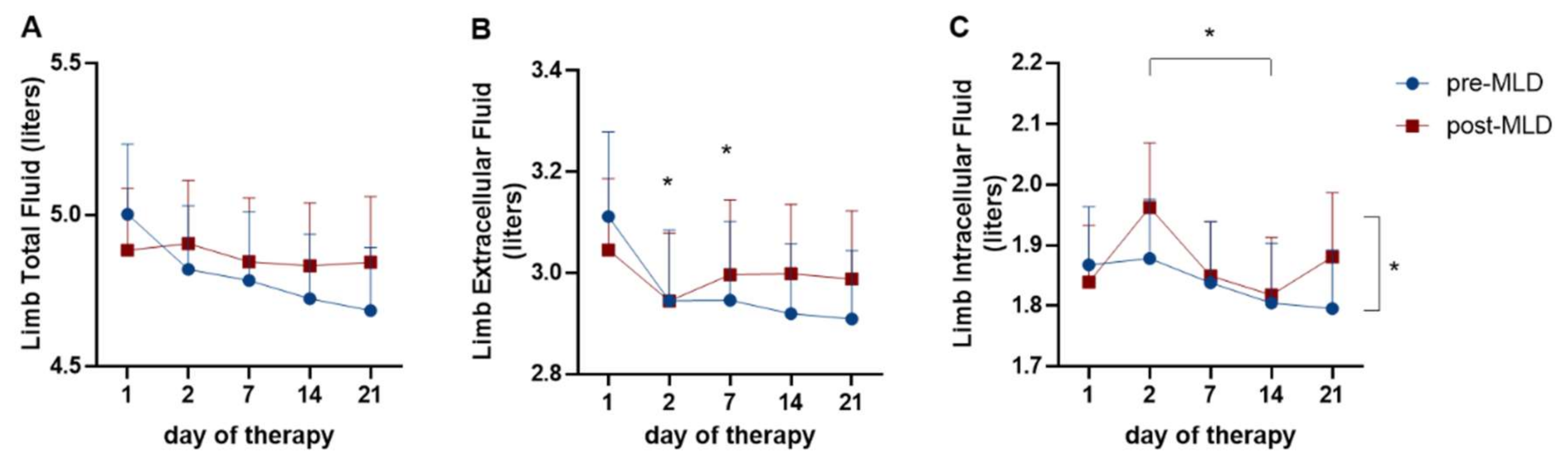
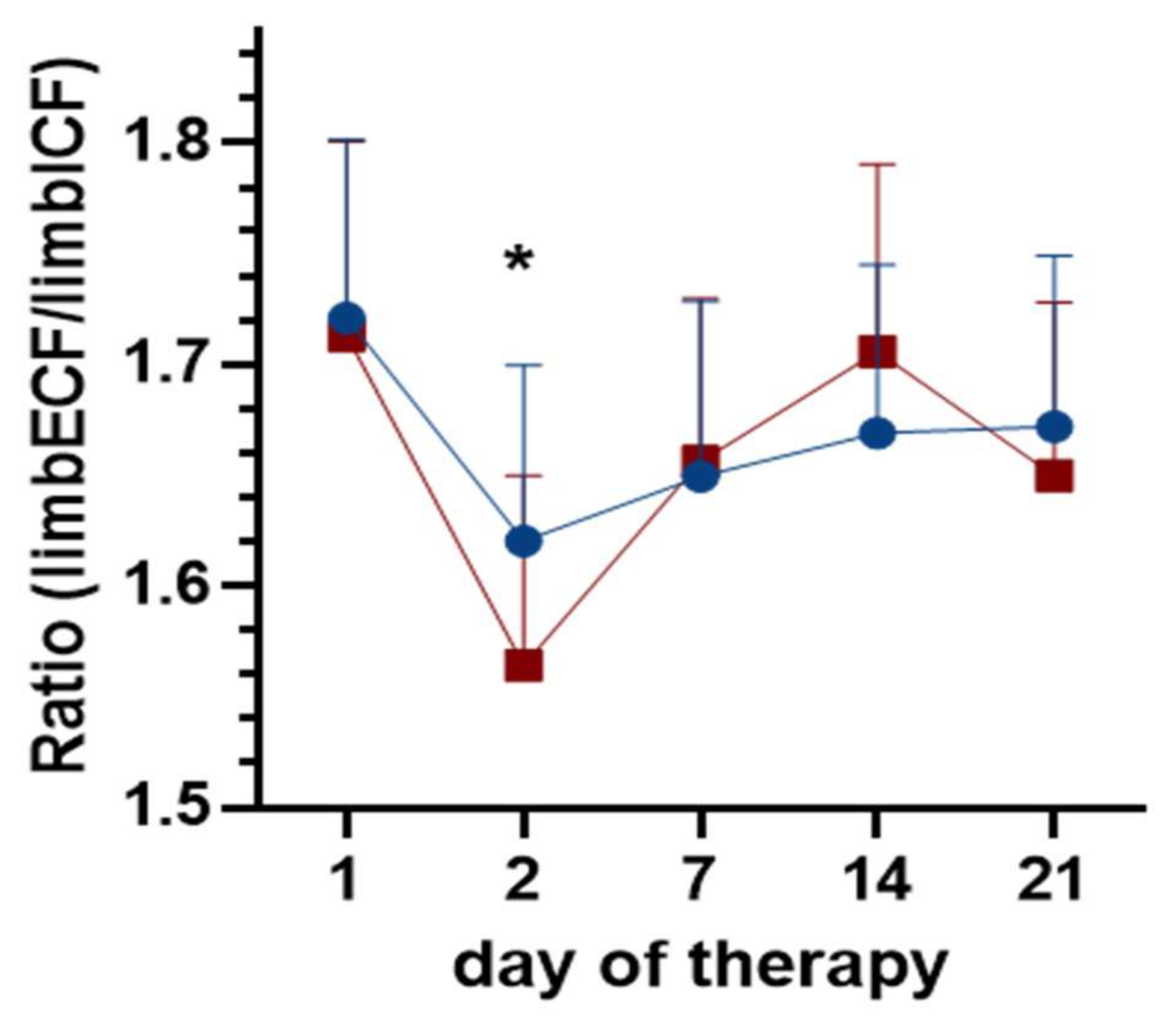
3.2. Changes in Lower Limb Fluid Due to Physical Therapy
3.3. Correlation Between Perometry and BIS
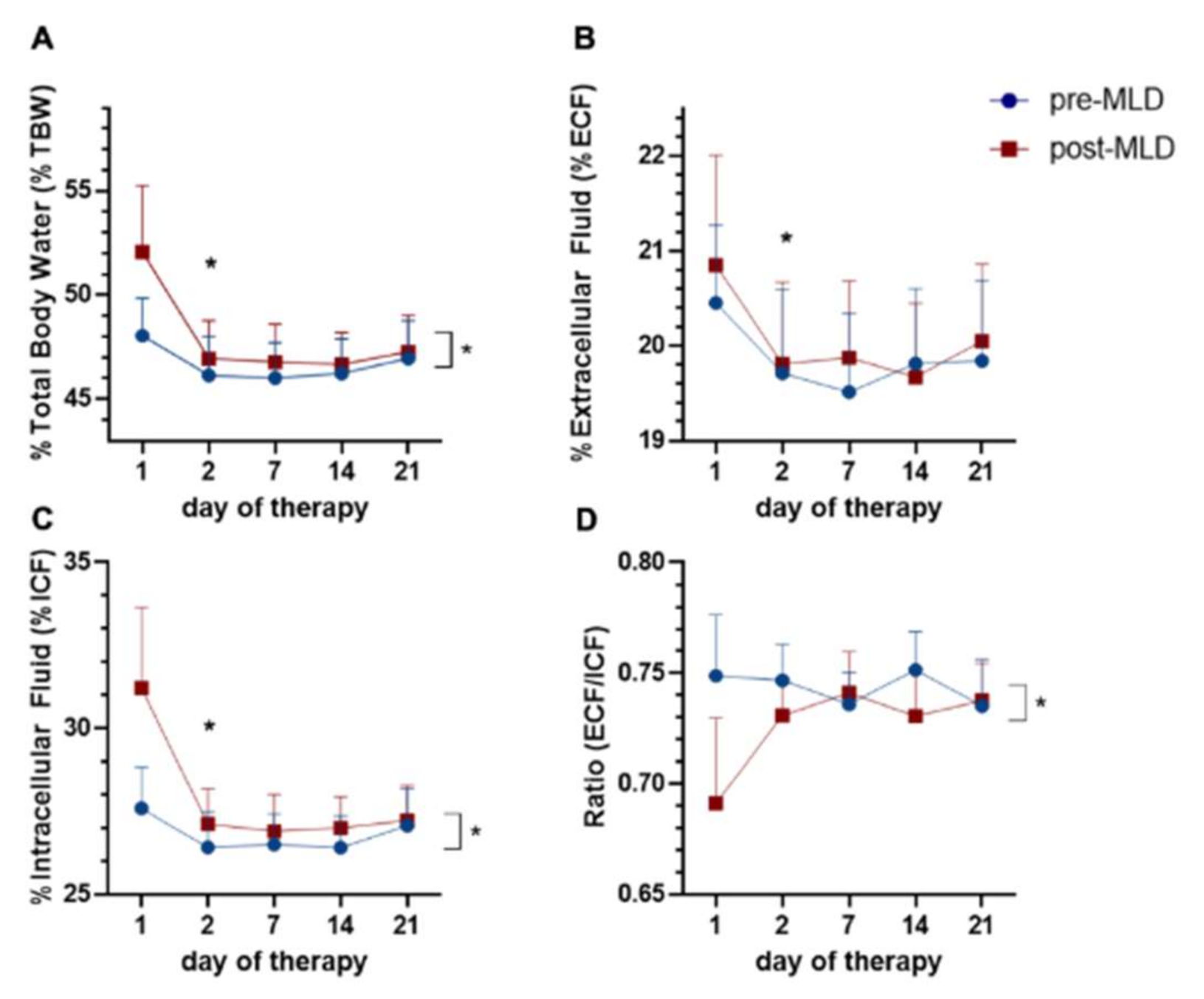
3.4. Whole-Body Fluid Shifts Measured by Bioelectrical Impedance Spectroscopy
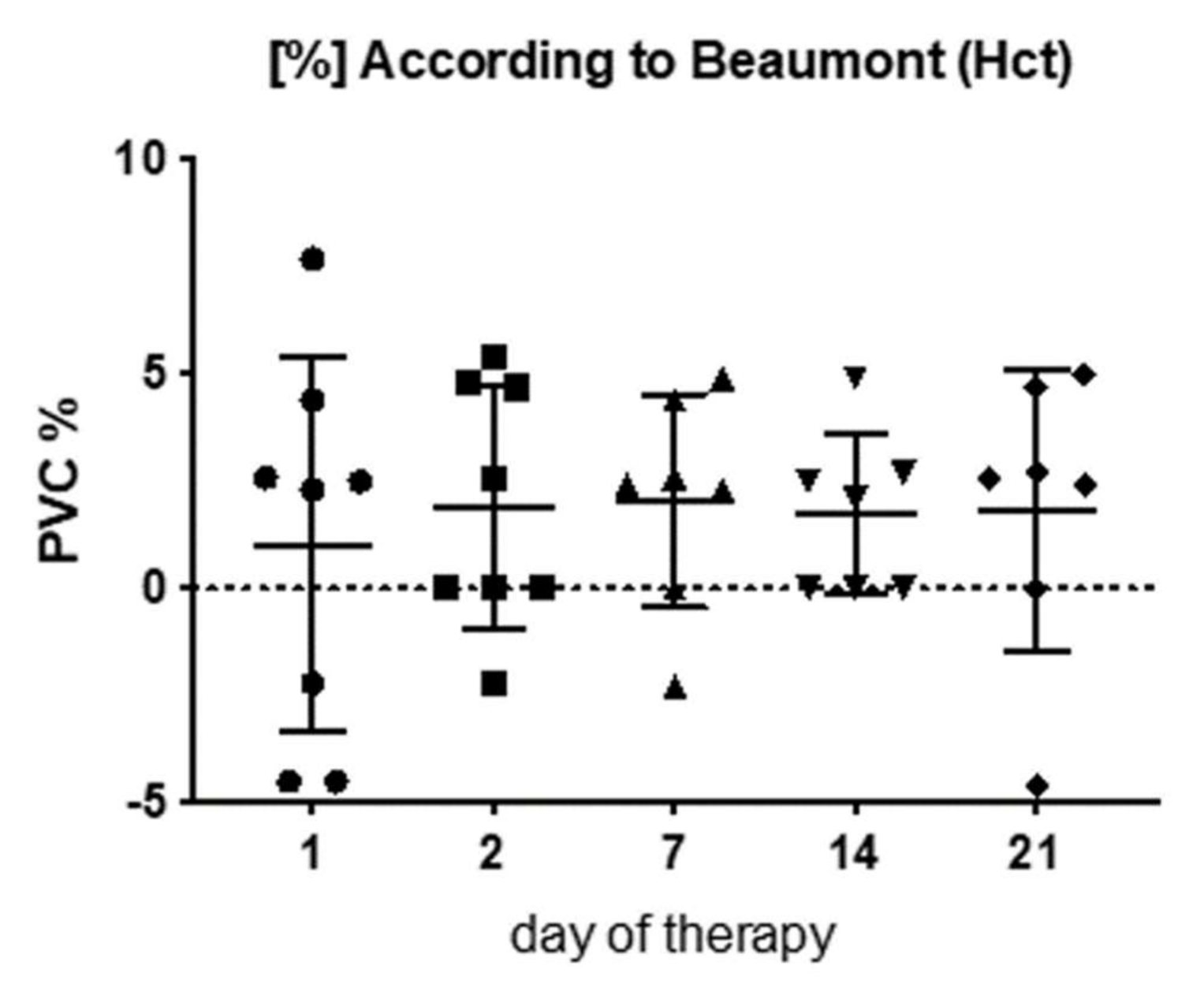
3.5. Plasma Volume Changes (PVC) Due to Physical Therapy
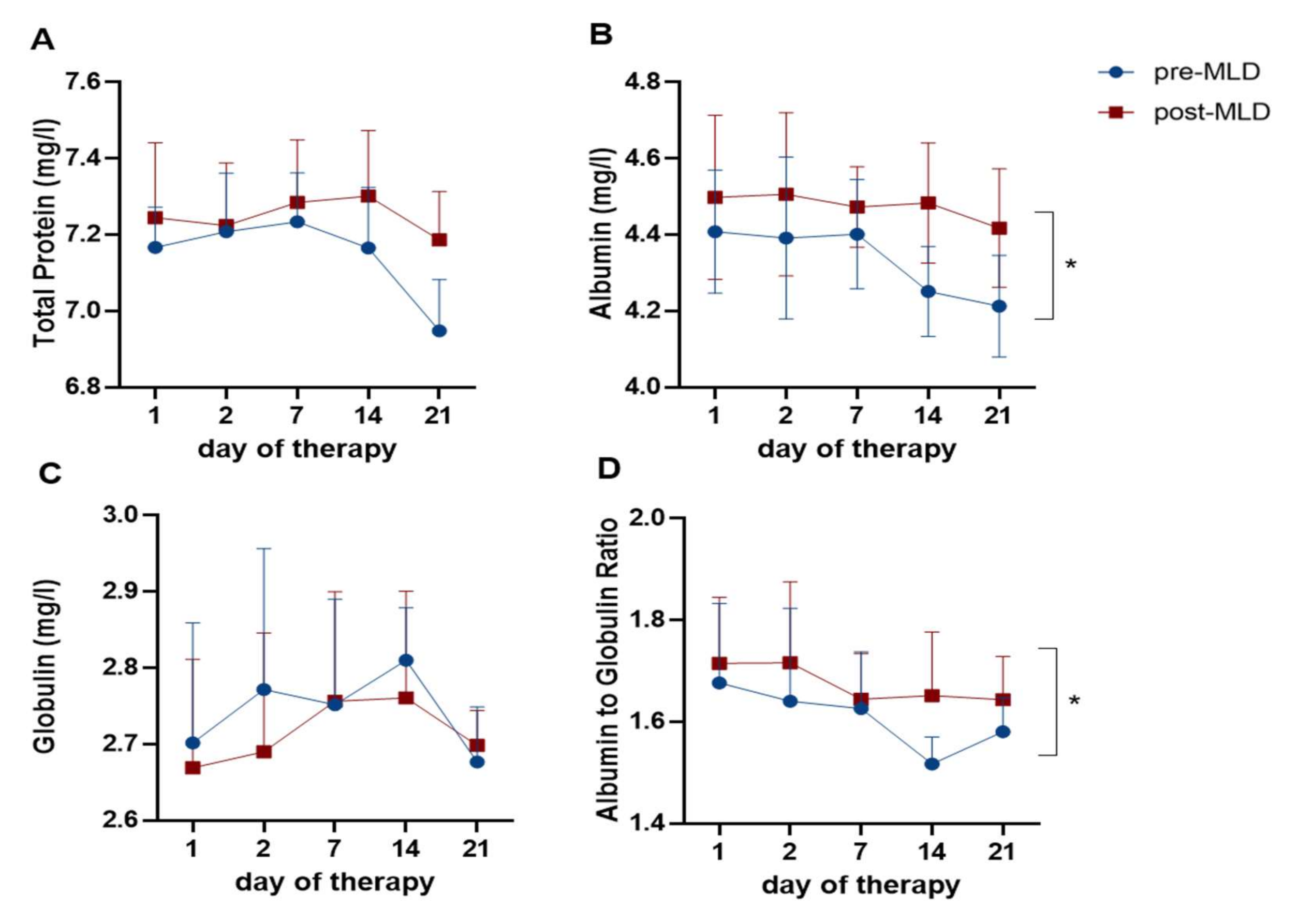
3.6. Plasma Protein Changes Due to Physical Therapy
3.7. Plasma Osmolality and Oncotic Pressure
3.8. Electrolyte Concentrations
4. Discussion
4.1. Reduction of Lower Limb Volume Due to Physical Therapy
4.2. Changes in Lower Limb Fluid Due to Physical Therapy
4.3. Correlation between Perometry and Bioelectrical Impedance Spectroscopy
4.4. Whole-Body Fluid Shifts Due to Lymphedema Therapy
4.5. Plasma Volume and Plasma Component Changes Due to Fluid Shifts During Lymphedema Therapy
4.6. Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Breslin, J.W.; Yang, Y.; Scallan, J.P.; Sweat, R.S.; Adderley, S.P.; Murfee, W.L. Lymphatic Vessel Network Structure and Physiology. Compr. Physiol. 2018, 9, 207–299. [Google Scholar] [CrossRef]
- Adamczyk, L.A.; Gordon, K.; Kholová, I.; Meijer-Jorna, L.B.; Telinius, N.; Gallagher, P.J.; Van Der Wal, A.C.; Baandrup, U. Lymph vessels: The forgotten second circulation in health and disease. Virchows Arch. 2016, 469, 3–17. [Google Scholar] [CrossRef]
- Levick, J.R.; Michel, C.C. Microvascular fluid exchange and the revised Starling principle. Cardiovasc. Res. 2010, 87, 198–210. [Google Scholar] [CrossRef]
- Moore, J.E.; Bertram, C.D. Lymphatic System Flows. Annu. Rev. Fluid Mech. 2018, 50, 459–482. [Google Scholar] [CrossRef] [PubMed]
- Renkin, E.M. Some consequences of capillary permeability to macromolecules: Starling’s hypothesis reconsidered. Am. J. Physiol. Circ. Physiol. 1986, 250, H706–H710. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.; Li, L.; Li, S.; Wu, M.; Ge, N.; Cui, Y.; Lian, Z.; Song, J.; Chengfu, C. Lymphatic system identification, pathophysiology and therapy in the cardiovascular diseases. J. Mol. Cell. Cardiol. 2019, 133, 99–111. [Google Scholar] [CrossRef] [PubMed]
- Kostanoğlu, A.; Ramoğlu, M.; Güneren, E. Results of home-based modified combined decongestive therapy in patients with lower extremity lymphedema. Turk. J. Med Sci. 2019, 49, 610–616. [Google Scholar] [CrossRef]
- Dayan, J.H.; Ly, C.L.; Kataru, R.P.; Mehrara, B.J. Lymphedema: Pathogenesis and Novel Therapies. Annu. Rev. Med. 2018, 69, 263–276. [Google Scholar] [CrossRef]
- Ciudad, P.; Sabbagh, M.D.; Agko, M.; Huang, T.C.; Manrique, O.J.; Roman, L.C.; Reynaga, C.; Delgado, R.; Maruccia, M.; Chen, H.-C. Surgical Management of Lower Extremity Lymphedema: A Comprehensive Review. Indian J. Plast. Surg. 2019, 52, 81–92. [Google Scholar] [CrossRef]
- Apich, G. Konservative Therapie des Lymphödems–Lymphologische Rehabilitationsbehandlung. Wien. Med. Wochenschr. 2013, 163, 169–176. [Google Scholar] [CrossRef]
- Tzani, I.; Tsichlaki, M.; Zerva, E.; Papathanasiou, G.; Dimakakos, E. Physiotherapeutic rehabilitation of lymphedema: State-of-the-Art. Lymphology 2018, 51, 1–12. [Google Scholar] [PubMed]
- Földi, E.; Sauerwald, A.; Hennig, B. Effect of Complex Decpngestive Physiotherapy on Gene Expression for the Inflammatory Response in PEripheral Lymphedema. Lymphology 2000, 33, 151–157. [Google Scholar]
- Bozkurt, M.; Palmer, L.J.; Guo, Y. Effectiveness of Decongestive Lymphatic Therapy in Patients with Lymphedema Resulting from Breast Cancer Treatment Regardless of Previous Lymphedema Treatment. Breast J. 2016, 23, 154–158. [Google Scholar] [CrossRef]
- Lasinski, B.B.; Thrift, K.M.; Squire, D.; Austin, M.K.; Smith, K.M.; Wanchai, A.; Green, J.M.; Stewart, B.R.; Cormier, J.N.; Armer, J.M. A Systematic Review of the Evidence for Complete Decongestive Therapy in the Treatment of Lymphedema from 2004 to 2011. PM&R 2012, 4, 580–601. [Google Scholar] [CrossRef]
- Executive Committee of the International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema: 2020 Consensus Document of the International Society of Lymphology. Lymphology 2020, 53, 3–19. [Google Scholar]
- De Godoy, J.M.P.; Brigidio, P.A.F.; Cunha, S.X.S.; Batigália, F.; De Fatima Guerreiro Godoy, M. Mobilization of fluids in large volumetric reductions during intensive treatment of leg lymphedema. Int. Angiol. 2013, 32, 479–482. [Google Scholar]
- Döller, W. Lymphologie. Wien. Med. Wochenschr. 2013, 163, 153–154. [Google Scholar] [CrossRef][Green Version]
- Hinghofer-Szalkay, H.; Lackner, H.K.; Rössler, A.; Narath, B.; Jantscher, A.; Goswami, N. Hormonal and plasma volume changes after presyncope. Eur. J. Clin. Investig. 2011, 41, 1180–1185. [Google Scholar] [CrossRef]
- Hinghofer-Szalkay, H.G.; Goswami, N.; Rössler, A.; Grasser, E.; Schneditz, D. Reactive hyperemia in the human liver. Am. J. Physiol. Liver Physiol. 2008, 295, G332–G337. [Google Scholar] [CrossRef][Green Version]
- Steele, M.L.; Janda, M.; Vagenas, D.; Ward, L.C.; Cornish, B.H.; Box, R.; Gordon, S.; Matthews, M.; Poppitt, S.D.; Plank, L.D.; et al. A Bioimpedance Spectroscopy-Based Method for Diagnosis of Lower-Limb Lymphedema. Lymphat. Res. Biol. 2020, 18, 101–109. [Google Scholar] [CrossRef]
- Sakuda, H.; Satoh, M.; Sakaguchi, M.; Miyakoshi, Y.; Kataoka, T. Physiological characteristics of the body fluid in lymphedematous patients postbreast cancer surgery, focusing on the intracellular/extracellular fluid ratio of the upper limb. Jpn. J. Nurs. Sci. 2010, 7, 108–118. [Google Scholar] [CrossRef]
- Ward, L.C.; Winall, A.; Isenring, E.; Hills, A.; Czerniec, S.; Dylke, E.; Kilbreath, S. Assessment of Bilateral Limb Lymphedema by Bioelectrical Impedance Spectroscopy. Int. J. Gynecol. Cancer 2011, 21, 409–418. [Google Scholar] [CrossRef]
- Hinghofer-Szalkay, H.G.; Sauseng-Fellegger, G.; Greenleaf, J.E. Plasma volume with alternative tilting: Effect of fluid ingestion. J. Appl. Physiol. 1995, 78, 1369–1373. [Google Scholar] [CrossRef] [PubMed]
- Van Beaumont, W. Evaluation of hemoconcentration from hematocrit measurements. J. Appl. Physiol. 1972, 32, 712–713. [Google Scholar] [CrossRef]
- Dill, D.B.; Costill, D.L. Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J. Appl. Physiol. 1974, 37, 247–248. [Google Scholar] [CrossRef] [PubMed]
- Nadler, S.B.; Hidalgo, J.U.; Bloch, T. Predicition of blood volume in normal humnan adul. Surgery 1962, 51, 224–232. [Google Scholar] [PubMed]
- Cohen, M.D. Complete Decongestive Physical Therapy in a Patient with Secondary Lymphedema Due to Orthopedic Trauma and Surgery of the Lower Extremity. Phys. Ther. 2011, 91, 1618–1626. [Google Scholar] [CrossRef][Green Version]
- Cavezzi, A.; Urso, S.U.; Paccasassi, S.; Mosti, G.; Campana, F.; Colucci, R. Bioimpedance spectroscopy and volumetry in the immediate/short-term monitoring of intensive complex decongestive treatment of lymphedema. Phlebol. J. Venous Dis. 2020, 35, 715–723. [Google Scholar] [CrossRef]
- Weiss, J.M.; Spray, B.J. The effect of complete decongestive therapy on the quality of life of patients with peripheral lymphedema. Lymphology 2002, 35, 46–58. [Google Scholar]
- Bertsch, T. Evaluation of a novel night-time compression garment: A prospective observational study. Br. J. Community Nurs. 2018, 23, 535–541. [Google Scholar] [CrossRef][Green Version]
- Cho, K.H.; Han, E.Y.; Lee, S.A.; Park, H.; Lee, C.; Im, S.H. Feasibility of Bioimpedance Analysis to Assess the Outcome of Complex Decongestive Therapy in Cancer Treatment-Related Lymphedema. Front. Oncol. 2020, 10, 111. [Google Scholar] [CrossRef] [PubMed]
- El Habnouni, C.; Tauveron, V.; Leducq, S.; Gérémia, S.; Allain, P.; Touchard, H.; Benejean, S.-A.; Machet, L.; Maruani, A. Short-Term Effect and Acceptability of Manual Lymphatic Drainage for Paediatric Limb Lymphoedema: A Prospective Study. Acta Derm. Venereol. 2020, 100, adv00125-5. [Google Scholar] [CrossRef] [PubMed]
- Ward, L.C. Assessment of lymphedema by bioelectrical impedance spectroscopy. Jpn. J. Nurs. Sci. 2011, 8, 108. [Google Scholar] [CrossRef] [PubMed]
- De Godoy, J.M.P.; Valente, F.M.; Azoubel, L.M.; Godoy, M.F.G. Evaluation of lymph drainage using bioelectrical impedance of the body. Phlebol. J. Venous Dis. 2011, 26, 298–300. [Google Scholar] [CrossRef]
- Hidding, J.T.; Viehoff, P.B.; Beurskens, C.H.; Van Laarhoven, H.W.; Der Sanden, M.W.N.-V.; Van Der Wees, P.J. Measurement Properties of Instruments for Measuring of Lymphedema: Systematic Review. Phys. Ther. 2016, 96, 1965–1981. [Google Scholar] [CrossRef] [PubMed]
- De Godoy, J.M.P.; De Godoy, H.J.P.; De Godoy, A.C.P.; De Marqui, T.G.; Godoy, M.F.G. Lymphedema and the mobilization of intracellular and extracellular fluids with intensive treatment. Acta Phlebol. 2019, 20. [Google Scholar] [CrossRef]
- Yamamoto, T.; Todo, Y.; Kaneuchi, M.; Handa, Y.; Watanabe, K.; Yamamoto, R. Study of edema reduction patterns during the treatment phase of complex decongestive physiotherapy for extremity lymphedema. Lymphology 2008, 41, 80–86. [Google Scholar]
- Cvirn, G.; Waha, J.E.; Ledinski, G.; Schlagenhauf, A.; Leschnik, B.; Koestenberger, M.; Tafeit, E.; Hinghofer-Szalkay, H.; Goswami, N. Bed rest does not induce hypercoagulability. Eur. J. Clin. Investig. 2014, 45, 63–69. [Google Scholar] [CrossRef]
- Goswami, N. Falls and Fall-Prevention in Older Persons: Geriatrics Meets Spaceflight! Front. Physiol. 2017, 8, 603. [Google Scholar] [CrossRef]
- Goswami, N.; Batzel, J.J.; Clément, G.; Stein, T.P.; Hargens, A.R.; Sharp, M.K.; Blaber, A.P.; Roma, P.G.; Hinghofer-Szalkay, H.G. Maximizing information from space data resources: A case for expanding integration across research disciplines. Eur. J. Appl. Physiol. 2013, 113, 1645–1654. [Google Scholar] [CrossRef]
- Goswami, N.; Kavcic, V.; Marusic, U.; Šimunič, B.; Rössler, A.; Hinghofer-Szalkay, H.; Pišot, R. Effect of computerized cognitive training with virtual spatial navigation task during bed rest immobilization and recovery on vascular function: A pilot study. Clin. Interv. Aging 2015, 10, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Waha, J.E.; Goswami, N.; Schlagenhauf, A.; Leschnik, B.; Koestenberger, M.; Reibnegger, G.; Roller, R.E.; Hinghofer-Szalkay, H.; Cvirn, G. Effects of Exercise and Nutrition on the Coagulation System During Bedrest Immobilization. Medicine 2015, 94, e1555. [Google Scholar] [CrossRef] [PubMed]
- Maher, J.; Refshauge, K.; Ward, L.C.; Paterson, R.; Kilbreath, S.L. Change in extracellular fluid and arm volumes as a consequence of a single session of lymphatic massage followed by rest with or without compression. Support. Care Cancer 2012, 20, 3079–3086. [Google Scholar] [CrossRef] [PubMed]
- Tambour, M.; Holt, M.; Speyer, A.; Christensen, R.; Gram, B. Manual lymphatic drainage adds no further volume reduction to Complete Decongestive Therapy on breast cancer-related lymphoedema: A multicentre, randomised, single-blind trial. Br. J. Cancer 2018, 119, 1215–1222. [Google Scholar] [CrossRef] [PubMed]
- Jeffs, E.; Ream, E.; Taylor, C.; Bick, D. Clinical effectiveness of decongestive treatments on excess arm volume and patient-centered outcomes in women with early breast cancer-related arm lymphedema. JBI Database Syst. Rev. Implement. Rep. 2018, 16, 453–506. [Google Scholar] [CrossRef]
- Huang, T.-W.; Tseng, S.-H.; Lin, C.; Bai, C.-H.; Chen, C.-S.; Hung, C.-S.; Wu, C.-H.; Tam, K.-W. Effects of manual lymphatic drainage on breast cancer-related lymphedema: A systematic review and meta-analysis of randomized controlled trials. World J. Surg. Oncol. 2013, 11, 15. [Google Scholar] [CrossRef]
- Thompson, B.; Gaitatzis, K.; De Jonge, X.J.; Blackwell, R.; Koelmeyer, L.A. Manual lymphatic drainage treatment for lymphedema: A systematic review of the literature. J. Cancer Surviv. 2020, 1–15. [Google Scholar] [CrossRef]
- Jain, M.S.; Danoff, J.V.; Paul, S.M. Correlation between bioelectrical spectroscopy and perometry in assessment of upper extremity swelling. Lymphology 2010, 43, 85–94. [Google Scholar]
- Bundred, N.J.; Stockton, C.; Keeley, V.; Riches, K.; Ashcroft, L.; Evans, A.; Skene, A.; Purushotham, A.; Bramley, M.; Hodgkiss, T.; et al. Comparison of multi-frequency bioimpedance with perometry for the early detection and intervention of lymphoedema after axillary node clearance for breast cancer. Breast Cancer Res. Treat. 2015, 151, 121–129. [Google Scholar] [CrossRef]
- Lopera, C.; Worsley, P.R.; Bader, D.L.; Fenlon, D.F. Investigating the Short-Term Effects of Manual Lymphatic Drainage and Compression Garment Therapies on Lymphatic Function Using Near-Infrared Imaging. Lymphat. Res. Biol. 2017, 15, 235–240. [Google Scholar] [CrossRef]
- de Godoy, J.M.; Santana, K.R.; Godoy Mde, F. Lymphoscintigraphic evaluation of manual lymphatic therapy: The Godoy & Godoy technique. Phlebol. J. Venous Dis. 2013, 30, 39–44. [Google Scholar] [CrossRef]
- Sawka, M.N.; Convertino, V.A.; Eichner, E.R.; Schnieder, S.M.; Young, A.J. Blood volume: Importance and adaptations to exercise training, environmental stresses, and trauma/sickness. Med. Sci. Sports Exerc. 2000, 32, 332–348. [Google Scholar] [CrossRef] [PubMed]
- Fellmann, N. Hormonal and Plasma Volume Alterations Following Endurance Exercise. Sports Med. 1992, 13, 37–49. [Google Scholar] [CrossRef]
- Otaki, Y.; Watanabe, T.; Konta, T.; Watanabe, M.; Asahi, K.; Yamagata, K.; Fujimoto, S.; Tsuruya, K.; Narita, I.; Kasahara, M.; et al. Impact of calculated plasma volume status on all-cause and cardiovascular mortality: 4-year nationwide community-based prospective cohort study. PLoS ONE 2020, 15, e0237601. [Google Scholar] [CrossRef]
- Hinghofer-Szalkay, H.; Moser, M. Fluid and protein shifts after postural changes in humans. Am. J. Physiol. Circ. Physiol. 1986, 250, H68–H75. [Google Scholar] [CrossRef]
- Hayashi, M.; Watanabe-Asaka, T.; Maejima, D.; Nagashio, S.; Kajihara, R.; Amari, K.; Yokoyama, Y.; Kaidoh, M.; Sugano, M.; Honda, T.; et al. Evaluating Lymph Flow Through the Thoracic Duct Using Urine Osmolarity in Human Participants. Lymphat. Res. Biol. 2020, 18, 351–356. [Google Scholar] [CrossRef]
- Dean, S.M.; Valenti, E.; Hock, K.; Leffler, J.; Compston, A.; Abraham, W.T. The clinical characteristics of lower extremity lymphedema in 440 patients. J. Vasc. Surg. Venous Lymphat. Disord. 2020, 8, 851–859. [Google Scholar] [CrossRef]
- Noh, S.; Hwang, J.H.; Yoon, T.H.; Chang, H.J.; Chu, I.H.; Kim, J.H. Limb Differences in the Therapeutic Effects of Complex Decongestive Therapy on Edema, Quality of Life, and Satisfaction in Lymphedema Patients. Ann. Rehabil. Med. 2015, 39, 347–359. [Google Scholar] [CrossRef]
- Keeley, V.; Franks, P.; Quere, I.; Mercier, G.; Michelini, S.; Cestari, M.; Borman, P.; Hughes, A.; Clark, K.; Lisle, J.; et al. LIMPRINT in Specialist Lymphedema Services in United Kingdom, France, Italy, and Turkey. Lymphat. Res. Biol. 2019, 17, 141–146. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brix, B.; Apich, G.; Roessler, A.; Ure, C.; Schmid-Zalaudek, K.; Hinghofer-Szalkay, H.; Goswami, N. Fluid Shifts Induced by Physical Therapy in Lower Limb Lymphedema Patients. J. Clin. Med. 2020, 9, 3678. https://doi.org/10.3390/jcm9113678
Brix B, Apich G, Roessler A, Ure C, Schmid-Zalaudek K, Hinghofer-Szalkay H, Goswami N. Fluid Shifts Induced by Physical Therapy in Lower Limb Lymphedema Patients. Journal of Clinical Medicine. 2020; 9(11):3678. https://doi.org/10.3390/jcm9113678
Chicago/Turabian StyleBrix, Bianca, Gert Apich, Andreas Roessler, Christian Ure, Karin Schmid-Zalaudek, Helmut Hinghofer-Szalkay, and Nandu Goswami. 2020. "Fluid Shifts Induced by Physical Therapy in Lower Limb Lymphedema Patients" Journal of Clinical Medicine 9, no. 11: 3678. https://doi.org/10.3390/jcm9113678
APA StyleBrix, B., Apich, G., Roessler, A., Ure, C., Schmid-Zalaudek, K., Hinghofer-Szalkay, H., & Goswami, N. (2020). Fluid Shifts Induced by Physical Therapy in Lower Limb Lymphedema Patients. Journal of Clinical Medicine, 9(11), 3678. https://doi.org/10.3390/jcm9113678





