Low ALT Levels Associated with Poor Outcomes in 8700 Hospitalized Heart Failure Patients
Abstract
1. Introduction
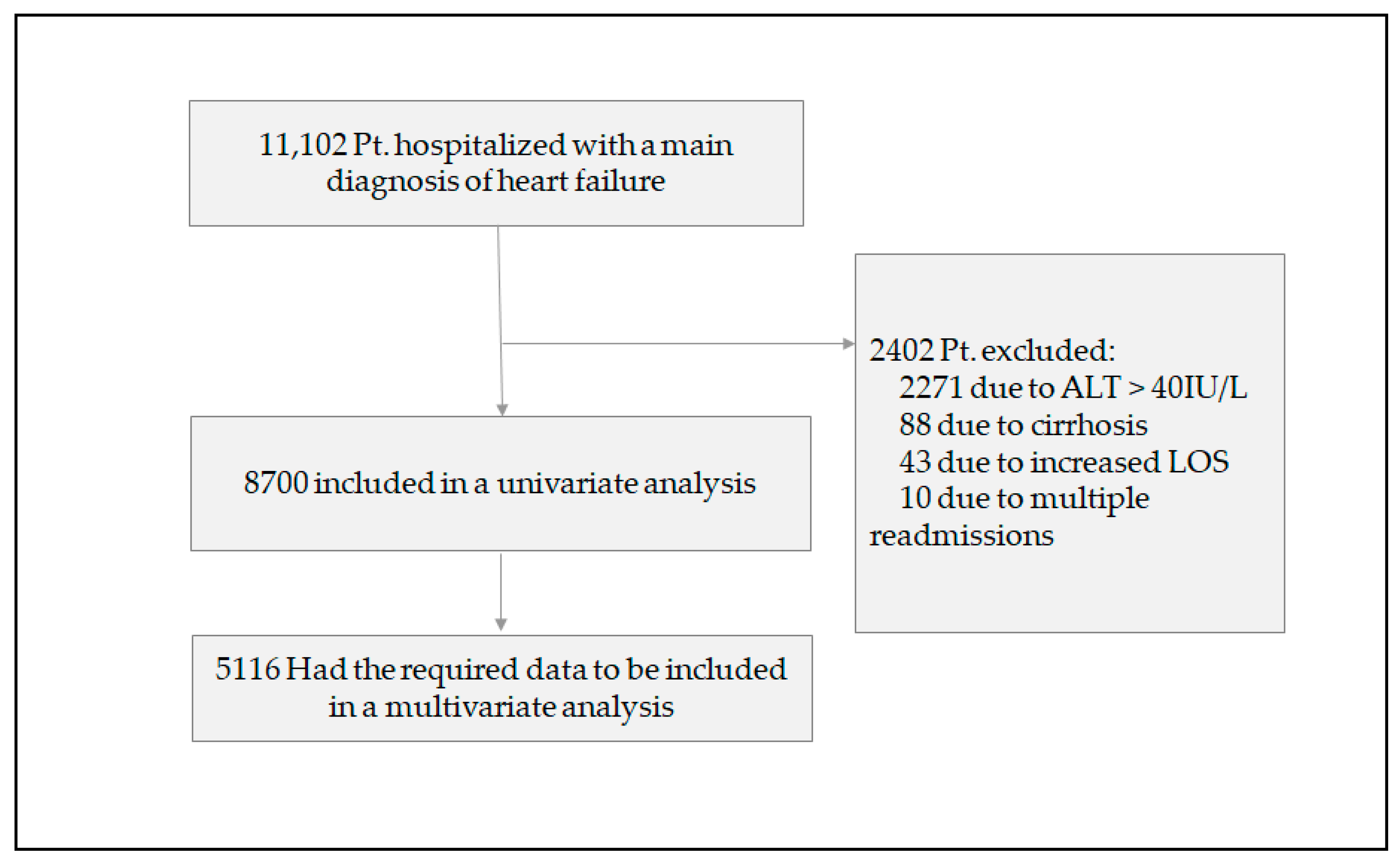
2. Materials and Methods
Statistical Analysis
3. Results
3.1. Patient Characteristics
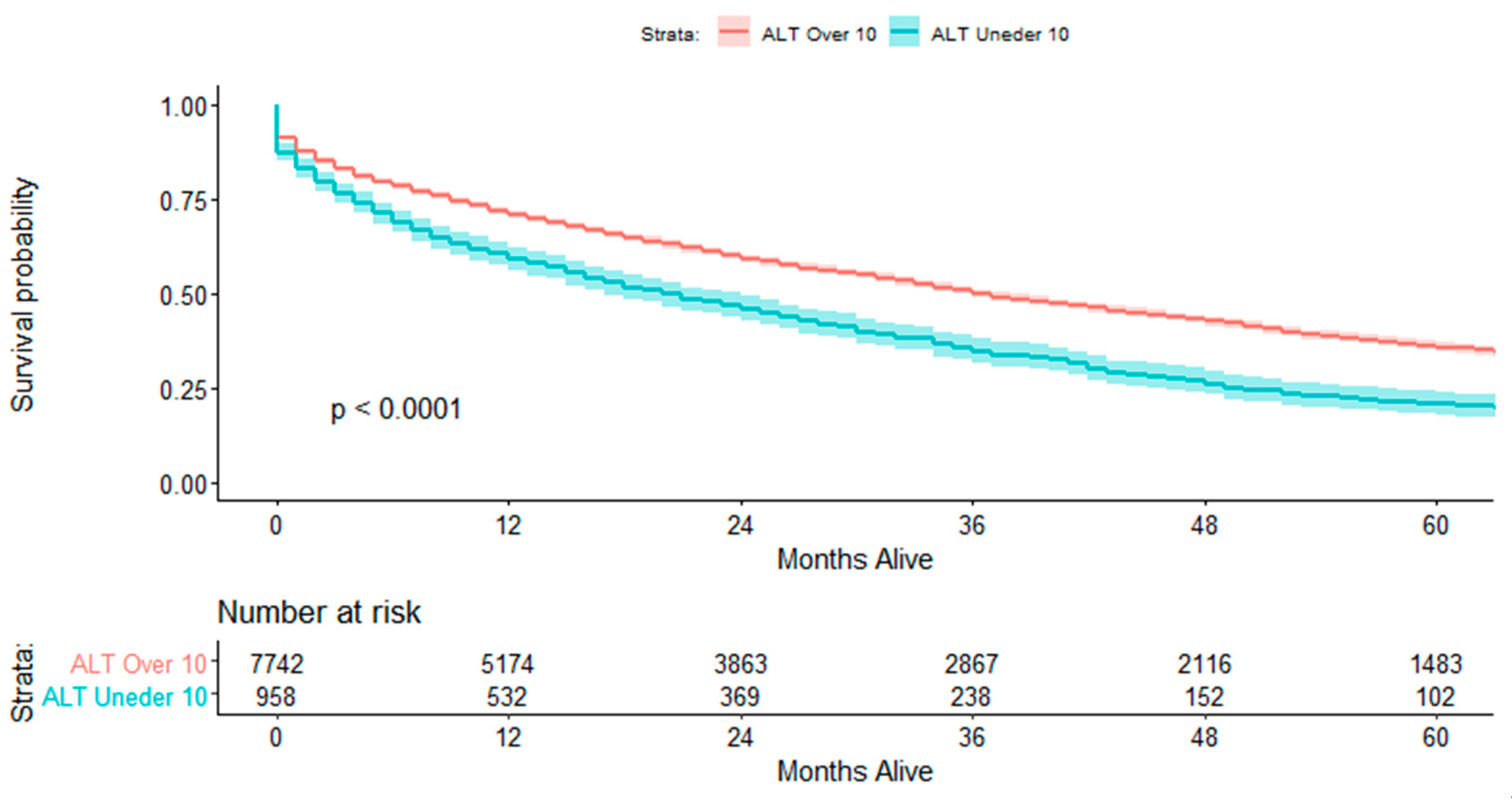
3.2. Short- and Long-Term Outcomes
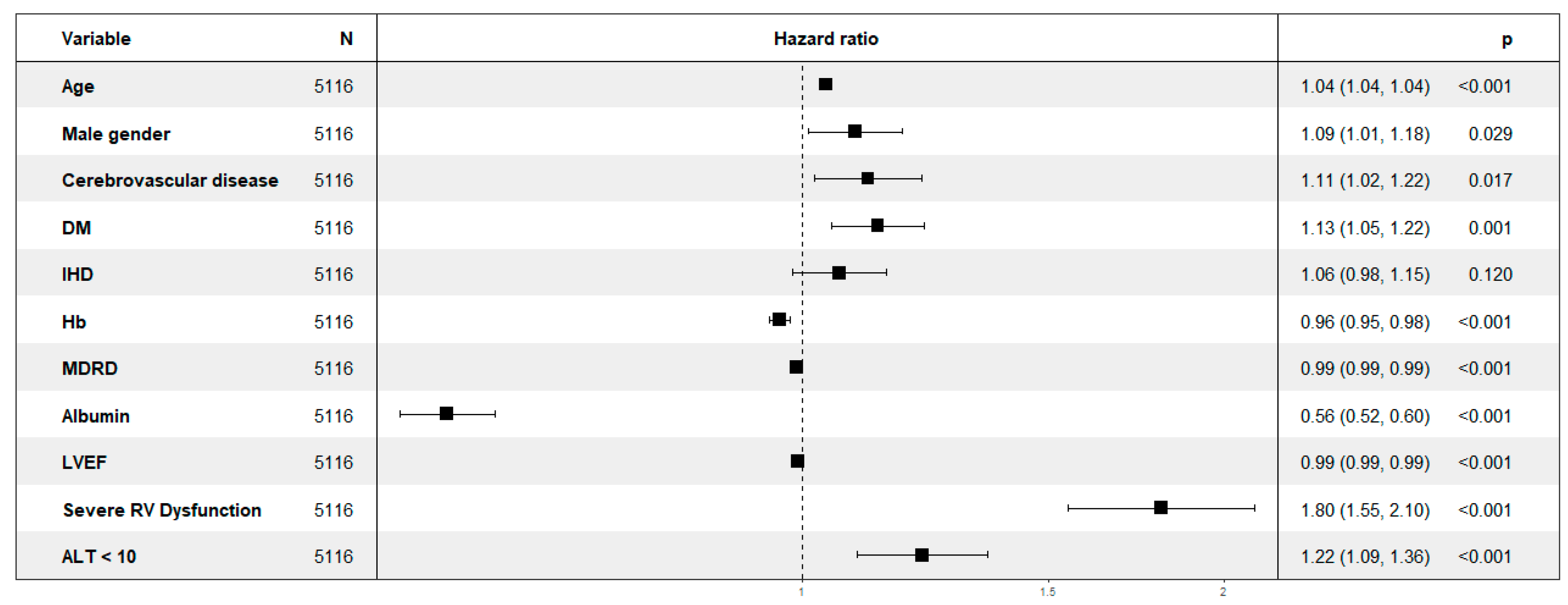
3.3. Multivariate Analysis
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Von Haehling, S.; Arzt, M.; Doehner, W.; Edelmann, F.; Evertz, R.; Ebner, N.; Herrmann-Lingen, C.; Macedo, T.G.; Koziolek, M.; Noutsias, M.; et al. Improving exercise capacity and quality of life using non-invasive heart failure treatments: Evidence from clinical trials. Eur. J. Hear. Fail. 2020. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.; Moreira-Gonçalves, D.; Santos, M.; Leite-Moreira, A.; Oliveira, J. Physical activity and exercise training in heart failure with preserved ejection fraction: Gathering evidence from clinical and pre-clinical studies. Heart Fail. Rev. 2020, 1–14. [Google Scholar] [CrossRef]
- Kurmani, S.; Squire, I. Acute Heart Failure: Definition, Classification and Epidemiology. Curr. Heart Fail. Rep. 2017, 14, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Dharmarajan, K.; Rich, M.W. Epidemiology, Pathophysiology, and Prognosis of Heart Failure in Older Adults. Heart Fail. Clin. 2017, 13, 417–426. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2001, 56, M146–M157. [Google Scholar] [CrossRef]
- Wilkinson, D.; Piasecki, M.; Atherton, P.J. The age-related loss of skeletal muscle mass and function: Measurement and physiology of muscle fibre atrophy and muscle fibre loss in humans. Ageing Res. Rev. 2018, 47, 123–132. [Google Scholar] [CrossRef]
- Allen, S.C. Systemic Inflammation in the Genesis of Frailty and Sarcopenia: An Overview of the Preventative and Therapeutic Role of Exercise and the Potential for Drug Treatments. Geriatrics 2017, 2, 6. [Google Scholar] [CrossRef]
- Dodson, J.A.; Chaudhry, S.I. Geriatric Conditions in Heart Failure. Curr. Cardiovasc. Risk Rep. 2012, 6, 404–410. [Google Scholar] [CrossRef]
- Boxer, R.S.; Wang, Z.; Walsh, S.J.; Hager, D.; Kenny, A.M. The utility of the 6-minute walk test as a measure of frailty in older adults with heart failure. Am. J. Geriatr. Cardiol. 2008, 17, 7–12. [Google Scholar] [CrossRef]
- Pascual, C.R.; Paredes-Galán, E.; Ferrero-Martínez, A.-I.; González-Guerrero, J.L.; Calvo, M.H.; Colino, R.M.; Torres-Torres, I.; Vilches-Moraga, A.; Galán, M.-C.; Suarez-Garcia, F.; et al. The frailty syndrome is associated with adverse health outcomes in very old patients with stable heart failure: A prospective study in six Spanish hospitals. Int. J. Cardiol. 2017, 236, 296–303. [Google Scholar] [CrossRef]
- McNallan, S.M.; Chamberlain, A.M.; Gerber, Y.; Singh, M.; Kane, R.L.; Weston, S.A.; Dunlay, S.M.; Jiang, R.; Roger, V.L. Measuring frailty in heart failure: A community perspective. Am. Heart J. 2013, 166, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Madan, S.A.; Fida, N.; Barman, P.; Sims, D.; Shin, J.; Verghese, J.; Piña, I.; Jorde, U.; Patel, S.R. Frailty Assessment in Advanced Heart Failure. J. Card. Fail. 2016, 22, 840–844. [Google Scholar] [CrossRef] [PubMed]
- Vidán, M.T.; Blaya-Novakova, V.; Sánchez, E.; Alonso, F.J.O.; Serra-Rexach, J.A.; Bueno, H. Prevalence and prognostic impact of frailty and its components in non-dependent elderly patients with heart failure. Eur. J. Heart Fail. 2016, 18, 869–875. [Google Scholar] [CrossRef] [PubMed]
- Anani, S.; Goldhaber, G.; Brom, A.; Lasman, N.; Turpashvili, N.; Shenhav-Saltzman, G.; Avaky, C.; Negru, L.; Agbaria, M.; Ariam, S.; et al. Frailty and Sarcopenia Assessment upon Hospital Admission to Internal Medicine Predicts Length of Hospital Stay and Re-Admission: A Prospective Study of 980 Patients. J. Clin. Med. 2020, 9, 2659. [Google Scholar] [CrossRef]
- Portal, D.; Hofstetter, L.; Eshed, I.; Lantsman, C.D.; Sella, T.; Urban, D.; Onn, A.; Bar, J.; Segal, G. L3 skeletal muscle index (L3SMI) is a surrogate marker of sarcopenia and frailty in non-small cell lung cancer patients. Cancer Manag. Res. 2019, 11, 2579–2588. [Google Scholar] [CrossRef]
- Vespasiani-Gentilucci, U.; De Vincentis, A.; Ferrucci, L.; Bandinelli, S.; Incalzi, R.A.; Picardi, A. Low Alanine Aminotransferase Levels in the Elderly Population: Frailty, Disability, Sarcopenia, and Reduced Survival. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2017, 73, 925–930. [Google Scholar] [CrossRef]
- Irina, G.; Refaela, C.; Brom, A.; Avia, D.; Liron, H.; Chen, A.; Gad, S. Low Blood ALT Activity and High FRAIL Questionnaire Scores Correlate with Increased Mortality and with Each Other. A Prospective Study in the Internal Medicine Department. J. Clin. Med. 2018, 7, 386. [Google Scholar] [CrossRef]
- Chung, S.M.; Moon, J.S.; Yoon, J.S.; Won, K.C.; Lee, H.W. Low alanine aminotransferase levels predict low muscle strength in older patients with diabetes: A nationwide cross-sectional study in Korea. Geriatr. Gerontol. Int. 2020, 20, 271–276. [Google Scholar] [CrossRef]
- Lasman, N.; Shalom, M.; Turpashvili, N.; Goldhaber, G.; Lifshitz, Y.; Leibowitz, E.; Berger, G.; Saltzman-Shenhav, G.; Brom, A.; Cohen, D.; et al. Baseline low ALT activity is associated with increased long-term mortality after COPD exacerbations. BMC Pulm. Med. 2020, 20, 1–6. [Google Scholar] [CrossRef]
- Itelman, E.; Segev, A.; Ahmead, L.; Leibowitz, E.; Agbaria, M.; Avaky, C.; Negro, L.; Shenhav-Saltzman, G.; Wasserstrum, Y.; Segal, G. Low ALT values amongst hospitalized patients are associated with increased risk of hypoglycemia and overall mortality: A retrospective, big-data analysis of 51 831 patients. QJM Int. J. Med. 2020. [Google Scholar] [CrossRef]
- Kashkosh, R.; Gringauz, I.; Weissmann, J.; Segal, G.; Swartzon, M.; Adunsky, A.; Justo, D. Prerehabilitation alanine aminotransferase blood levels and one-year mortality rates in older adults following hip fracture. Int. J. Rehabil. Res. 2020, 43. [Google Scholar] [CrossRef] [PubMed]
- Ramaty, E.; Maor, E.; Peltz-Sinvani, N.; Brom, A.; Grinfeld, A.; Kivity, S.; Segev, S.; Sidi, Y.; Kessler, T.; Sela, B.; et al. Low ALT blood levels predict long-term all-cause mortality among adults. A historical prospective cohort study. Eur. J. Intern. Med. 2014, 25, 919–921. [Google Scholar] [CrossRef] [PubMed]
- Kogan, M.; Klempfner, R.; Lotan, D.; Wasserstrum, Y.; Goldenberg, I.; Segal, G. Low ALT blood levels are associated with lower baseline fitness amongst participants of a cardiac rehabilitation program. J. Exerc. Sci. Fit. 2018, 16, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Irina, G.; Weismann, J.; Justo, D.; Adunsky, A.; Segal, G. Alanine aminotransferase blood levels and rehabilitation outcome in older adults following hip fracture surgery. Int. J. Rehabil. Res. 2018, 41, 41–46. [Google Scholar] [CrossRef]
- Cioffi, G.; Dahlström, U.; Filippatos, G.; Chioncel, O.; Leiro, M.C.; Drozdz, J.; Fruhwald, F.; Gullestad, L.; Logeart, D.; Fabbri, G.; et al. EURObservationalResearch Programme: Regional differences and 1-year follow-up results of the Heart Failure Pilot Survey (ESC-HF Pilot). Eur. J. Heart Fail. 2013, 15, 808–817. [Google Scholar] [CrossRef]
- Rich, M.W.; Chyun, D.A.; Skolnick, A.H.; Alexander, K.P.; Forman, D.E.; Kitzman, D.W.; Maurer, M.S.; McClurken, J.B.; Resnick, B.M.; Shen, W.K.; et al. Knowledge Gaps in Cardiovascular Care of the Older Adult Population. Circulation 2016, 133, 2103–2122. [Google Scholar] [CrossRef]
- Jha, S.R.; Hannu, M.K.; Chang, S.; Montgomery, E.; Harkess, M.; Wilhelm, K.; Hayward, C.S.; Jabbour, A.; Spratt, P.M.; Newton, P.; et al. The Prevalence and Prognostic Significance of Frailty in Patients With Advanced Heart Failure Referred for Heart Transplantation. Transplantation 2016, 100, 429–436. [Google Scholar] [CrossRef]
- Green, P.; Woglom, A.E.; Généreux, P.; Daneault, B.; Paradis, J.-M.; Schnell, S.; Hawkey, M.; Maurer, M.S.; Kirtane, A.J.; Kodali, S.; et al. The Impact of Frailty Status on Survival After Transcatheter Aortic Valve Replacement in Older Adults With Severe Aortic Stenosis. JACC Cardiovasc. Interv. 2012, 5, 974–981. [Google Scholar] [CrossRef]
- Afilalo, J.; Lauck, S.; Kim, D.H.; Lefèvre, T.; Piazza, N.; Lachapelle, K.; Martucci, G.; Lamy, A.; Labinaz, M.; Peterson, M.D.; et al. Frailty in Older Adults Undergoing Aortic Valve Replacement. J. Am. Coll. Cardiol. 2017, 70, 689–700. [Google Scholar] [CrossRef]
- Vidán, M.T.; Sánchez, E.; Ortiz, J.; Bueno, H.; Fernández-Avilés, F.; Serra-Rexach, J.A. FRAIL-HF, a Study to Evaluate the Clinical Complexity of Heart Failure in Nondependent Older Patients: Rationale, Methods and Baseline Characteristics. Clin. Cardiol. 2014, 37, 725–732. [Google Scholar] [CrossRef]
- Yang, X.; Lupón, J.; Vidán, M.T.; Ferguson, C.; Gastelurrutia, P.; Newton, P.J.; Macdonald, P.S.; Bueno, H.; Bayés-Genís, A.; Woo, J.; et al. Impact of Frailty on Mortality and Hospitalization in Chronic Heart Failure: A Systematic Review and Meta-Analysis. J. Am. Heart Assoc. 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Bellumkonda, L.; Tyrrell, D.J.; Hummel, S.L.; Goldstein, D.R. Pathophysiology of heart failure and frailty: A common inflammatory origin? Aging Cell 2017, 16, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Mann, D.L. Innate immunity and the failing heart: The cytokine hypothesis revisited. Circ. Res. 2015, 116, 1254–1268. [Google Scholar] [CrossRef] [PubMed]
- Molfino, A.; Anker, S.; Argiles, J.; Aversa, Z.; Bauer, J.; Biolo, G.; Boirie, Y.; Bosaeus, I.; Cederholm, T.; Costelli, P.; et al. Consensus definition of sarcopenia, cachexia and pre-cachexia: Joint document elaborated by Special Interest Groups (SIG) “cachexia-anorexia in chronic wasting diseases” and “nutrition in geriatrics”. Clin. Nutr. 2010, 29, 154–159. [Google Scholar] [CrossRef]
- Woo, J.; Leung, J.; Morley, J.E. Comparison of Frailty Indicators Based on Clinical Phenotype and the Multiple Deficit Approach in Predicting Mortality and Physical Limitation. J. Am. Geriatr. Soc. 2012, 60, 1478–1486. [Google Scholar] [CrossRef]
- Turner, G.; Clegg, A.P. Best practice guidelines for the management of frailty: A British Geriatrics Society, Age UK and Royal College of General Practitioners report. Age Ageing 2014, 43, 744–747. [Google Scholar] [CrossRef]
- Le Couteur, D.G.; Blyth, F.M.; Creasey, H.M.; Handelsman, D.J.; Naganathan, V.; Sambrook, P.N.; Seibel, M.J.; Waite, L.M.; Cumming, R.G. The Association of Alanine Transaminase With Aging, Frailty, and Mortality. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2010, 65A, 712–717. [Google Scholar] [CrossRef]
- Peltz-Sinvani, N.; Klempfner, R.; Ramaty, E.; Sela, B.A.; Goldenberg, I.; Segal, G. Low ALT Levels Independently Associated with 22-Year All-Cause Mortality Among Coronary Heart Disease Patients. J. Gen. Intern. Med. 2015, 31, 209–214. [Google Scholar] [CrossRef]
- Nam, J.-S.; Kim, W.-J.; An, S.-M.; Choi, D.-K.; Chin, J.-H.; Lee, E.-H.; Choi, I.-C. Age-dependent relationship between preoperative serum aminotransferase and mortality after cardiovascular surgery. Aging 2019, 11, 9060–9074. [Google Scholar] [CrossRef]
- Nikolaou, M.; Parissis, J.; Yilmaz, M.B.; Seronde, M.-F.; Kivikko, M.; Laribi, S.; Paugam-Burtz, C.; Cai, D.; Pohjanjousi, P.; Laterre, P.-F.; et al. Liver function abnormalities, clinical profile, and outcome in acute decompensated heart failure. Eur. Heart J. 2012, 34, 742–749. [Google Scholar] [CrossRef]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.; Coats, A.J.S.; Falk, V.; González-Juanatey, J.R.; Harjola, V.-P.; Jankowska, E.; et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Russ. J. Cardiol. 2017, 141, 7–81. [Google Scholar] [CrossRef]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, N.E.; Colvin, M.M.; Drazner, M.H.; Filippatos, G.S.; Fonarow, G.C.; Givertz, M.M.; et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. J. Am. Coll. Cardiol. 2017, 70, 776–803. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Vellas, B.; Van Kan, G.A.; Anker, S.D.; Bauer, J.M.; Bernabei, R.; Cesari, M.; Chumlea, W.; Doehner, W.; Evans, J.; et al. Frailty consensus: A call to action. J. Am. Med. Dir. Assoc. 2013, 14, 392–397. [Google Scholar] [CrossRef] [PubMed]
| ALT ≥ 10 IU/L (n = 7742) | ALT < 10 IU/L (n = 958) | Total (n = 8700) | p-Value | |
|---|---|---|---|---|
| Patients’ demographics | ||||
| Age—years, median (IQR) | 78.6 (69.4–85.6) | 81.8 (72.7–87.5) | 79 (69.8–85.8) | <0.001 |
| Male gender—number (%) | 4365 (56.4) | 467 (48.7) | 4832 (55.5) | <0.001 |
| Comorbidities number (%) | ||||
| HTN | 5358 (69.2) | 686 (71.6) | 6044 (69.5) | 0.138 |
| DM | 3259 (42.1) | 416 (43.4) | 3675 (42.2) | 0.453 |
| Dyslipidemia | 1842 (23.8) | 200 (20.9) | 2042 (23.5) | 0.049 |
| IHD | 3789 (48.9) | 445 (47.6) | 4245 (48.8) | 0.454 |
| Cerebrovascular disease | 1517 (19.6) | 236 (24.6) | 1753 (20.1) | <0.001 |
| Atrial fibrillation | 3237 (41.8) | 376 (39.2) | 3613 (41.5) | 0.138 |
| Dementia | 354 (4.6) | 74 (7.7) | 428 (4.9) | <0.001 |
| COPD | 1272 (16.4) | 170 (17.7) | 1442 (16.6) | 0.324 |
| Solid malignancy | 786 (10.2) | 128 (13.4) | 914 (10.5) | 0.003 |
| Hematologic malignancy | 256 (3.3) | 32 (3.3) | 288 (3.3) | >0.99 |
| CKD | 1446 (18.7) | 197 (20.6) | 1643 (18.9) | 0.173 |
| Baseline laboratory parameters median (IQR) | ||||
| ALT (IU/L) | 18 (14–25) | 8 (7–9) | 17 (12–24) | <0.001 |
| Hemoglobin (g/dL) | 11.5 (10.1–12.9) | 10.6 (9.3–11.8) | 11.4 (10–12.8) | <0.001 |
| Creatinine (mg/dL) | 1.2 (0.9–1.6) | 1.3 (1–1.9) | 1.2 (0.9–1.6) | <0.001 |
| MDRD(mL/min/1.73 m²) | 51.5 (35.7–69.1) | 43.2 (28.8–63.4) | 50.6 (34.8–68.5) | <0.001 |
| Albumin(g/dL) | 3.6 (3.3–3.9) | 3.4 (3.1–3.7) | 3.6 (3.2–3.9) | <0.001 |
| Echocardiography findings | ||||
| LVEF %, median (IQR) | 50 (30–60) | 55 (35–60) | 50 (30–60) | 0.006 |
| MR—n (%) | 0.503 | |||
| None | 437 (9.5) | 47 (8.5) | 484 (9.4) | |
| Moderate | 508 (11.0) | 48 (8.6) | 556 (10.7) | |
| Severe | 222 (4.8) | 23 (4.1) | 245 (4.7) | |
| TR—n (%) | 0.421 | |||
| None | 260 (5.6) | 36 (6.5) | 296 (5.7) | |
| Moderate | 553 (12.0) | 66 (11.9) | 619 (12.0) | |
| Severe | 307 (6.6) | 47 (8.5) | 354 (6.8) | |
| RVF—n (%) | 0.81 | |||
| Normal | 3420 (74.0) | 421 (75.9) | 3841 (74.2) | |
| Severely reduced | 249 (5.4) | 28 (5.0) | 277 (5.4) | |
| Dilated RV | 1028 (22.3) | 130 (23.4) | 1158 (22.4) | 0.576 |
| Outcome | ALT ≥ 10IU/L (n = 7742) | ALT < 10IU/L (n = 958) | Total (n = 8700) | p-Value |
|---|---|---|---|---|
| LOS—days, median (IQR) | 3 (1–6) | 4 (2–7) | 3 (1–6) | <0.001 |
| AKI—n (%) | 1205 (15.6) | 183 (19.1) | 1388 (16.0) | 0.006 |
| Maximal creatinine—mg/dL | 1.3 (1–1.9) | 1.5 (1.1–2.2) | 1.3 (1.0–1.9) | <0.001 |
| In-hospital mortality—n (%) | 304 (3.9) | 62 (6.5) | 366 (4.2) | <0.001 |
| Readmission count—median (IQR) | 2 (0–4) | 2 (0–4) | 2 (0–4) | 0.489 |
| HF readmissions—median (IQR) | 0 (0–2) | 0 (0–1) | 0 (0–2) | 0.795 |
| Total mortality—n (%) | 4764 (61.5) | 702 (73.3) | 5466 (62.8) | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Segev, A.; Itelman, E.; Avaky, C.; Negru, L.; Shenhav-Saltzman, G.; Grupper, A.; Wasserstrum, Y.; Segal, G. Low ALT Levels Associated with Poor Outcomes in 8700 Hospitalized Heart Failure Patients. J. Clin. Med. 2020, 9, 3185. https://doi.org/10.3390/jcm9103185
Segev A, Itelman E, Avaky C, Negru L, Shenhav-Saltzman G, Grupper A, Wasserstrum Y, Segal G. Low ALT Levels Associated with Poor Outcomes in 8700 Hospitalized Heart Failure Patients. Journal of Clinical Medicine. 2020; 9(10):3185. https://doi.org/10.3390/jcm9103185
Chicago/Turabian StyleSegev, Amitai, Edward Itelman, Chen Avaky, Liat Negru, Gilat Shenhav-Saltzman, Avishay Grupper, Yishay Wasserstrum, and Gad Segal. 2020. "Low ALT Levels Associated with Poor Outcomes in 8700 Hospitalized Heart Failure Patients" Journal of Clinical Medicine 9, no. 10: 3185. https://doi.org/10.3390/jcm9103185
APA StyleSegev, A., Itelman, E., Avaky, C., Negru, L., Shenhav-Saltzman, G., Grupper, A., Wasserstrum, Y., & Segal, G. (2020). Low ALT Levels Associated with Poor Outcomes in 8700 Hospitalized Heart Failure Patients. Journal of Clinical Medicine, 9(10), 3185. https://doi.org/10.3390/jcm9103185






