Melflufen: A Peptide–Drug Conjugate for the Treatment of Multiple Myeloma
Abstract
1. Introduction
2. Preclinical Development
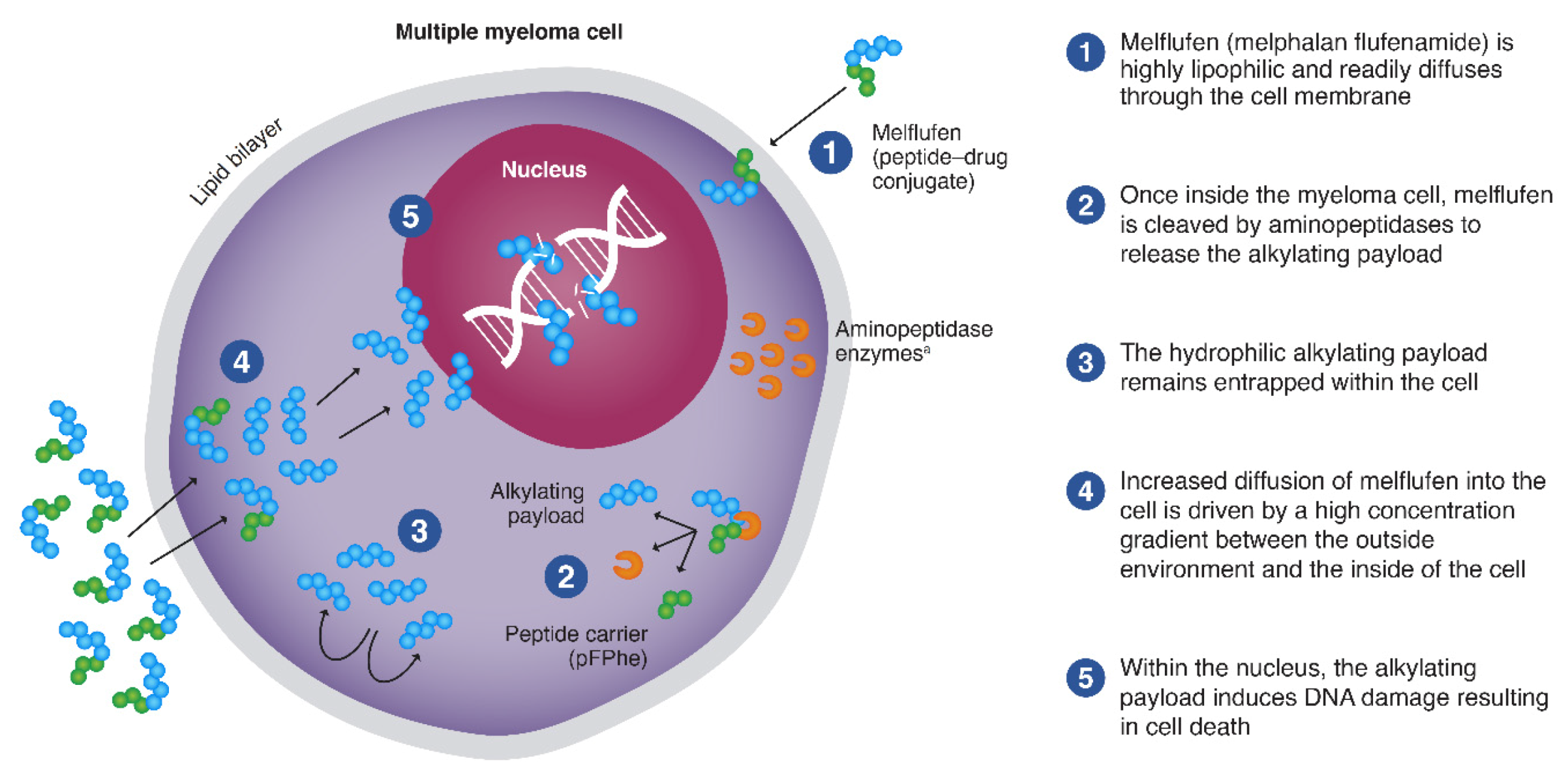
2.1. Mechanism of Action
2.2. Preclinical Anti-Tumor Activity
3. Pharmacokinetics
4. Clinical Development
4.1. Early Development in Multiple Myeloma
4.2. Efficacy and Safety of Melflufen Combination Therapies
5. Additional Clinical Development
6. Management of Melflufen
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kumar, S.K.; Dimopoulos, M.A.; Kastritis, E.; Terpos, E.; Nahi, H.; Goldschmidt, H.; Hillengass, J.; Leleu, X.; Beksac, M.; Alsina, M.; et al. Natural history of relapsed myeloma, refractory to immunomodulatory drugs and proteasome inhibitors: A multicenter IMWG study. Leukemia 2017, 31, 2443–2448. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, U.H.; Cornell, R.F.; Lakshman, A.; Gahvari, Z.J.; McGehee, E.; Jagosky, M.H.; Gupta, R.; Vamado, W.; Fiala, M.A.; Chhabra, S.; et al. Outcomes of patients with multiple myeloma refractory to CD38-targeted monoclonal antibody therapy. Leukemia 2019, 33, 2266–2275. [Google Scholar] [CrossRef] [PubMed]
- Moreau, P.; San Miguel, J.; Sonneveld, P.; Mateos, M.V.; Zamagni, E.; Avet-Loiseau, H.; Hajek, R.; Dimopoulos, M.A.; Ludwig, H.; Einsele, H.; et al. Multiple myeloma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 28, iv52–iv61. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network. NCCN Guidelines: Multiple Myeloma; Version 2. 2020. Available online: https://www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf (accessed on 23 September 2020).
- Cejalvo, M.J.; de la Rubia, J. Which therapies will move to the front line for multiple myeloma? Expert Rev. Hematol. 2017, 10, 383–392. [Google Scholar] [CrossRef]
- Moreau, P.; Attal, M.; Hulin, C.; Arnulf, B.; Belhadj, K.; Benboubker, L.; Bene, M.C.; Broijl, A.; Caillon, H.; Caillot, D.; et al. Bortezomib, thalidomide, and dexamethasone with or without daratumumab before and after autologous stem-cell transplantation for newly diagnosed multiple myeloma (CASSIOPEIA): A randomised, open-label, phase 3 study. Lancet 2019, 394, 29–38. [Google Scholar] [CrossRef]
- Facon, T.; Kumar, S.; Plesner, T.; Orlowski, R.Z.; Moreau, P.; Bahlis, N.; Basu, S.; Nahi, H.; Hulin, C.; Quach, H.; et al. Daratumumab plus lenalidomide and dexamethasone for untreated myeloma. N. Engl. J. Med. 2019, 380, 2104–2115. [Google Scholar] [CrossRef]
- Mateos, M.V.; Dimopoulos, M.A.; Cavo, M.; Suzuki, K.; Jakubowiak, A.; Knop, S.; Doyen, C.; Lucio, P.; Nagy, Z.; Kaplan, P.; et al. Daratumumab plus bortezomib, melphalan, and prednisone for untreated myeloma. N. Engl. J. Med. 2018, 378, 518–528. [Google Scholar] [CrossRef]
- Palumbo, A.; Bringhen, S.; Rossi, D.; Cavalli, M.; Larocca, A.; Ria, R.; Offidani, M.; Patriarca, F.; Nozzoli, C.; Guglielmelli, T.; et al. Bortezomib-melphalan-prednisone-thalidomide followed by maintenance with bortezomib-thalidomide compared with bortezomib-melphalan-prednisone for initial treatment of multiple myeloma: A randomized controlled trial. J. Clin. Oncol. 2010, 28, 5101–5109. [Google Scholar] [CrossRef]
- San Miguel, J.F.; Schlag, R.; Khuageva, N.K.; Dimopoulos, M.A.; Shpilberg, O.; Kropff, M.; Spicka, I.; Petrucci, M.T.; Palumbo, A.; Samoilova, O.S.; et al. Bortezomib plus melphalan and prednisone for initial treatment of multiple myeloma. N. Engl. J. Med. 2008, 359, 906–917. [Google Scholar] [CrossRef]
- Benboubker, L.; Dimopoulos, M.A.; Dispenzieri, A.; Catalano, J.; Belch, A.R.; Cavo, M.; Pinto, A.; Weisel, K.; Ludwig, H.; Bahlis, N.; et al. Lenalidomide and dexamethasone in transplant-ineligible patients with myeloma. N. Engl. J. Med. 2014, 371, 906–917. [Google Scholar] [CrossRef]
- Durie, B.G.M.; Hoering, A.; Abidi, M.H.; Rajkumar, S.V.; Epstein, J.; Kahanic, S.P.; Thakuri, M.; Reu, F.; Reynolds, C.M.; Sexton, R.; et al. Bortezomib with lenalidomide and dexamethasone versus lenalidomide and dexamethasone alone in patients with newly diagnosed myeloma without intent for immediate autologous stem-cell transplant (SWOG S0777): A randomised, open-label, phase 3 trial. Lancet 2017, 389, 519–527. [Google Scholar] [CrossRef]
- Rajkumar, S.V.; Jacobus, S.; Callander, N.S.; Fonseca, R.; Vesole, D.H.; Williams, M.E.; Abonour, R.; Siegel, D.S.; Katz, M.; Greipp, P.R.; et al. Lenalidomide plus high-dose dexamethasone versus lenalidomide plus low-dose dexamethasone as initial therapy for newly diagnosed multiple myeloma: An open-label randomised controlled trial. Lancet Oncol. 2010, 11, 29–37. [Google Scholar] [CrossRef]
- Takamatsu, H.; Iida, S.; Shibayama, H.; Shibayama, K.; Yamazaki, H.; Suzuki, K. Daratumumab, lenalidomide, and dexamethasone in Japanese patients with transplant-ineligible newly diagnosed multiple myeloma: A phase 1b study. Int. J. Hematol. 2020, 111, 692–701. [Google Scholar] [CrossRef]
- Moreau, P.; Zamagni, E.; Mateos, M.V. Treatment of patients with multiple myeloma progressing on frontline-therapy with lenalidomide. Blood Cancer J. 2019, 9, 38. [Google Scholar] [CrossRef] [PubMed]
- Chapman, M.A.; Lawrence, M.S.; Keats, J.J.; Cibulskis, K.; Sougnez, C.; Schinzel, A.C.; Harview, C.L.; Brunet, J.P.; Ahmann, G.J.; Adli, M.; et al. Initial genome sequencing and analysis of multiple myeloma. Nature 2011, 471, 467–472. [Google Scholar] [CrossRef]
- Egan, J.B.; Shi, C.X.; Tembe, W.; Christoforides, A.; Kurdoglu, A.; Sinari, S.; Middha, S.; Asmann, Y.; Schmidt, J.; Braggio, E.; et al. Whole-genome sequencing of multiple myeloma from diagnosis to plasma cell leukemia reveals genomic initiating events, evolution, and clonal tides. Blood 2012, 120, 1060–1066. [Google Scholar] [CrossRef]
- Robak, P.; Drozdz, I.; Szemraj, J.; Robak, T. Drug resistance in multiple myeloma. Cancer Treat. Rev. 2018, 70, 199–208. [Google Scholar] [CrossRef]
- Yong, K.; Delforge, M.; Driessen, C.; Fink, L.; Flinois, A.; Gonzalez-McQuire, S.; Safaei, R.; Karlin, L.; Mateos, M.V.; Raab, M.S.; et al. Multiple myeloma: Patient outcomes in real-world practice. Br. J. Haematol. 2016, 175, 252–264. [Google Scholar] [CrossRef]
- Xpovio (Selinexor) [Package Insert]; Karyopharm Therapeutics Inc.: Newton, MA, USA, 2019.
- Chari, A.; Vogl, D.T.; Gavriatopoulou, M.; Nooka, A.K.; Yee, A.J.; Huff, C.A.; Moreau, P.; Dingli, D.; Cole, C.; Lonial, S.; et al. Oral selinexor-dexamethasone for triple-class refractory multiple myeloma. N. Engl. J. Med. 2019, 381, 727–738. [Google Scholar] [CrossRef]
- Moreau, P.; Chanan-Khan, A.; Roberts, A.W.; Agarwal, A.B.; Facon, T.; Kumar, S.; Touzeau, C.; Punnoose, E.A.; Cordero, J.; Munasinghe, W.; et al. Promising efficacy and acceptable safety of venetoclax plus bortezomib and dexamethasone in relapsed/refractory MM. Blood 2017, 130, 2392–2400. [Google Scholar] [CrossRef]
- Kumar, S.; Harrison, S.; Cavo, M.; De la Rubia, J.; Popat, R.; Gasparetto, C.; Hungria, V.; Salwender, H.; Suzuki, K.; Kim, I.; et al. A phase 3 study of venetoclax or placebo in combination with bortezomib and dexamethasone in patients with relapsed/refractory multiple myeloma. Clin. Lymphoma Myeloma Leuk. 2019, 19, e31. [Google Scholar] [CrossRef]
- Cho, S.F.; Anderson, K.C.; Tai, Y.T. Targeting B Cell Maturation Antigen (BCMA) in Multiple Myeloma: Potential Uses of BCMA-Based Immunotherapy. Front. Immunol. 2018, 9, 1821. [Google Scholar] [CrossRef]
- D’Agostino, M.; Raje, N. Anti-BCMA CAR T-cell therapy in multiple myeloma: Can we do better? Leukemia 2019, 34, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Lonial, S.; Lee, H.C.; Badros, A.; Trudel, S.; Nooka, A.K.; Chari, A.; Abdallah, A.O.; Callander, N.; Lendvai, N.; Sborov, D.; et al. Belantamab mafodotin for relapsed or refractory multiple myeloma (DREAMM-2): A two-arm, randomised, open-label, phase 2 study. Lancet Oncol. 2019, 21, 207–221. [Google Scholar] [CrossRef]
- Raje, N.; Berdeja, J.; Lin, Y.; Siegel, D.; Jagannath, S.; Madduri, D.; Liedtke, M.; Rosenblatt, J.; Maus, M.V.; Turka, A.; et al. Anti-BCMA CAR T-cell therapy bb2121 in relapsed or refractory multiple myeloma. N. Engl. J. Med. 2019, 380, 1726–1737. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, D.; Ray, A.; Viktorsson, K.; Spira, J.; Paba-Prada, C.; Munshi, N.; Richardson, P.; Lewensohn, R.; Anderson, K.C. In vitro and in vivo antitumor activity of a novel alkylating agent, melphalan-flufenamide, against multiple myeloma cells. Clin. Cancer Res. 2013, 19, 3019–3031. [Google Scholar] [CrossRef] [PubMed]
- Wickstrom, M.; Nygren, P.; Larsson, R.; Harmenberg, J.; Lindberg, J.; Sjoberg, P.; Jerling, M.; Lehmann, F.; Richardson, P.; Anderson, K.; et al. Melflufen—A peptidase-potentiated alkylating agent in clinical trials. Oncotarget 2017, 8, 66641–66655. [Google Scholar] [CrossRef]
- Wickström, M.; Viktorsson, K.; Lundholm, L.; Aesoy, R.; Nygren, H.; Sooman, L.; Fryknäs, M.; Vogel, L.K.; Lewensohn, R.; Larsson, R.; et al. The alkylating prodrug J1 can be activated by aminopeptidase N, leading to a possible target directed release of melphalan. Biochem. Pharm. 2010, 79, 1281–1290. [Google Scholar] [CrossRef]
- Gullbo, J.; Wickstrom, M.; Tullberg, M.; Ehrsson, H.; Lewensohn, R.; Nygren, P.; Luthman, K.; Larsson, R. Activity of hydrolytic enzymes in tumour cells is a determinant for anti-tumour efficacy of the melphalan containing prodrug J1. J. Drug Target. 2003, 11, 355–363. [Google Scholar] [CrossRef]
- Ray, A.; Ravillah, D.; Das, D.S.; Song, Y.; Nordstrom, E.; Gullbo, J.; Richardson, P.G.; Chauhan, D.; Anderson, K.C. A novel alkylating agent Melflufen induces irreversible DNA damage and cytotoxicity in multiple myeloma cells. Br. J. Haematol. 2016, 174, 397–409. [Google Scholar] [CrossRef]
- Gullbo, J.; Dhar, S.; Luthman, K.; Ehrsson, H.; Lewensohn, R.; Nygren, P.; Larsson, R. Antitumor activity of the alkylating oligopeptides J1 (L-melphalanyl-p-L-fluorophenylalanine ethyl ester) and P2 (L-prolyl-m-L-sarcolysyl-p-L-fluorophenylalanine ethyl ester): Comparison with melphalan. Anticancer Drugs 2003, 14, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Palladini, G.; Schonland, S.; Lentzsch, S.; Cibeira, M.T.; Hajek, R.; Jaccard, A.; Jamroziak, K.; Kastritis, E.; Sanchorawala, V.; Schjesvold, F.H.; et al. OP201: A phase 1/2 study of melflufen and dexamethasone in patients with immunoglobulin light chain amyloidosis. In Proceedings of the 61st American Society of Hematology Annual Meeting, Orlando, FL, USA, 7–10 December 2019; p. 3163. [Google Scholar]
- Gullbo, J.; Tullberg, M.; Vabeno, J.; Ehrsson, H.; Lewensohn, R.; Nygren, P.; Larsson, R.; Luthman, K. Structure-activity relationship for alkylating dipeptide nitrogen mustard derivatives. Oncol. Res. 2003, 14, 113–132. [Google Scholar] [CrossRef] [PubMed]
- Wickstrom, M.; Larsson, R.; Nygren, P.; Gullbo, J. Aminopeptidase N (CD13) as a target for cancer chemotherapy. Cancer Sci. 2011, 102, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Hitzerd, S.M.; Verbrugge, S.E.; Ossenkoppele, G.; Jansen, G.; Peters, G.J. Positioning of aminopeptidase inhibitors in next generation cancer therapy. Amino Acids 2014, 46, 793–808. [Google Scholar] [CrossRef] [PubMed]
- Delforoush, M.; Strese, S.; Wickstrom, M.; Larsson, R.; Enblad, G.; Gullbo, J. In vitro and in vivo activity of melflufen (J1) in lymphoma. BMC Cancer 2016, 16, 263. [Google Scholar] [CrossRef]
- Strese, S.; Hassan, S.B.; Velander, E.; Haglund, C.; Hoglund, M.; Larsson, R.; Gullbo, J. In vitro and in vivo anti-leukemic activity of the peptidase-potentiated alkylator melflufen in acute myeloid leukemia. Oncotarget 2017, 8, 6341–6352. [Google Scholar] [CrossRef][Green Version]
- Zhao, H.; Meng, X.; Yuan, H.; Lan, M. Novel melphalan and chlorambucil derivatives of 2,2,6,6-tetramethyl-1-piperidinyloxy radicals: Synthesis, characterization, and biological evaluation in vitro. Chem. Pharm. Bull. 2010, 58, 332–335. [Google Scholar] [CrossRef][Green Version]
- Carlier, C.; Strese, S.; Viktorsson, K.; Velander, E.; Nygren, P.; Uustalu, M.; Juntti, T.; Lewensohn, R.; Larsson, R.; Spira, J.; et al. Preclinical activity of melflufen (J1) in ovarian cancer. Oncotarget 2016, 7, 59322–59335. [Google Scholar] [CrossRef]
- Wickstrom, M.; Haglund, C.; Lindman, H.; Nygren, P.; Larsson, R.; Gullbo, J. The novel alkylating prodrug J1: Diagnosis directed activity profile ex vivo and combination analyses in vitro. Investig. New Drugs 2008, 26, 195–204. [Google Scholar] [CrossRef]
- Wickstrom, M.; Johnsen, J.I.; Ponthan, F.; Segerstrom, L.; Sveinbjornsson, B.; Lindskog, M.; Lovborg, H.; Viktorsson, K.; Lewensohn, R.; Kogner, P.; et al. The novel melphalan prodrug J1 inhibits neuroblastoma growth in vitro and in vivo. Mol. Cancer 2007, 6, 2409–2417. [Google Scholar] [CrossRef]
- Byrgazov, K.; Slipicevic, A.; Lehmann, F.; Lion, T.; Kager, L.; Taschner-Mandl, S. A peptidase-potentiated alkylating agent melflufen is an effective anti-neoplastic agent in osteosarcoma. In Proceedings of the European Society of Medical Oncology (ESMO) Annual Meeting, Barcelona, Spain, 27 September–19 October 2019; p. 1726. [Google Scholar]
- Chesi, M.; Matthews, G.M.; Garbitt, V.M.; Palmer, S.E.; Shortt, J.; Lefebure, M.; Stewart, A.K.; Johnstone, R.W.; Bergsagel, P.L. Drug response in a genetically engineered mouse model of multiple myeloma is predictive of clinical efficacy. Blood 2012, 120, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Chin, M.; Sive, J.I.; Allen, C.; Roddie, C.; Chavda, S.J.; Smith, D.; Blombery, P.; Jones, K.; Ryland, G.L.; Popat, R.; et al. Prevalence and timing of TP53 mutations in del(17p) myeloma and effect on survival. Blood Cancer J. 2017, 7, e610. [Google Scholar] [CrossRef] [PubMed]
- Strese, S.; Wickstrom, M.; Fuchs, P.F.; Fryknas, M.; Gerwins, P.; Dale, T.; Larsson, R.; Gullbo, J. The novel alkylating prodrug melflufen (J1) inhibits angiogenesis in vitro and in vivo. Biochem. Pharm. 2013, 86, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Berglund, A.; Ullen, A.; Lisyanskaya, A.; Orlov, S.; Hagberg, H.; Tholander, B.; Lewensohn, R.; Nygren, P.; Spira, J.; Harmenberg, J.; et al. First-in-human, phase I/IIa clinical study of the peptidase potentiated alkylator melflufen administered every three weeks to patients with advanced solid tumor malignancies. Investig. New Drugs 2015, 33, 1232–1241. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulos, M.A.; Sonneveld, P.; Leung, N.; Merlini, G.; Ludwig, H.; Kastritis, E.; Goldschmidt, H.; Joshua, D.; Orlowski, R.Z.; Powles, R.; et al. International Myeloma Working Group Recommendations for the Diagnosis and Management of Myeloma-Related Renal Impairment. J. Clin. Oncol. 2016, 34, 1544–1557. [Google Scholar] [CrossRef] [PubMed]
- Nath, C.E.; Shaw, P.J.; Trotman, J.; Zeng, L.; Duffull, S.B.; Hegarty, G.; McLachlan, A.J.; Gurney, H.; Kerridge, I.; Kwan, Y.L.; et al. Population pharmacokinetics of melphalan in patients with multiple myeloma undergoing high dose therapy. Br. J. Clin. Pharm. 2010, 69, 484–497. [Google Scholar] [CrossRef]
- Richardson, P.; Bringhen, S.; Voorhees, P.; Plesner, T.; Mellqvist, U.H.; Reeves, B.; Paba-Prada, C.; Zubair, H.; Byrne, C.; Chauhan, D.; et al. Melflufen plus dexamethasone in relapsed and refractory multiple myeloma (O-12-M1): A multicentre, international, open-label, phase 1–2 study. Lancet Haematol. 2020, 7, e395–e407. [Google Scholar] [CrossRef]
- Bringhen, S.; Vorhees, P.M.; Plesner, T.; Mellqvist, U.H.; Reeves, B.; Sonneveld, P.; Byrne, C.; Nordstrom, E.; Harmenberg, J.; Obermuller, J.; et al. Updated progression-free survival and overall survival with melflufen and dexamethasone in patients with relapsed/refractory multiple myeloma: Results from the phase 2 study O-12-M1. In Proceedings of the 61st American Society of Hematology Annual Meeting, Orlando, FL, USA, 7–10 December 2019; p. 1839. [Google Scholar]
- Dimopoulos, M.A.; Palumbo, A.; Corradini, P.; Cavo, M.; Delforge, M.; Di Raimondo, F.; Weisel, K.C.; Oriol, A.; Hansson, M.; Vacca, A.; et al. Safety and efficacy of pomalidomide plus low-dose dexamethasone in STRATUS (MM-010): A phase 3b study in refractory multiple myeloma. Blood 2016, 128, 497–503. [Google Scholar] [CrossRef]
- San-Miguel, J.F.; Hungria, V.T.; Yoon, S.S.; Beksac, M.; Dimopoulos, M.A.; Elghandour, A.; Jedrzejczak, W.W.; Gunther, A.; Nakorn, T.N.; Siritanaratkul, N.; et al. Panobinostat plus bortezomib and dexamethasone versus placebo plus bortezomib and dexamethasone in patients with relapsed or relapsed and refractory multiple myeloma: A multicentre, randomised, double-blind phase 3 trial. Lancet Oncol. 2014, 15, 1195–1206. [Google Scholar] [CrossRef]
- Usmani, S.Z.; Weiss, B.M.; Plesner, T.; Bahlis, N.J.; Belch, A.; Lonial, S.; Lokhorst, H.M.; Voorhees, P.M.; Richardson, P.G.; Chari, A.; et al. Clinical efficacy of daratumumab monotherapy in patients with heavily pretreated relapsed or refractory multiple myeloma. Blood 2016, 128, 37–44. [Google Scholar] [CrossRef]
- Mateos, M.V.; Oriol, A.; Larocca, A.; Otero, P.R.; Bladé, J.; Cavo, M.; Hassoun, H.; Leleu, X.; Alegre, A.; Maisel, C.; et al. Clinical activity of melflufen in patients with triple-class refractory multiple myeloma and poor-risk features in an updated analysis of HORIZON (OP-106), a phase 2 study in patients with relapsed/refractory multiple myeloma refractory to pomalidomide and/or daratumumab. In Proceedings of the 61st American Society of Hematology Annual Meeting, Orlando, FL, USA, 7–10 December 2019; p. 1883. [Google Scholar]
- Ocio, E.M.; Efebera, Y.A.; Granell, M.; Hajek, R.; Maisnar, V.; Karlin, L.; Mateos, M.V.; Richardson, P.G.; Oriol, A.; Norin, S.; et al. ANCHOR (OP-104): Updated efficacy and safety from a phase 1/2 study of melflufen and dexamethasone plus bortezomib or daratumumab in patients with relapsed/refractory multiple myeloma (RRMM) refractory to an IMiD or a proteasome inhibitor (PI). Blood 2019, 133, 3124. [Google Scholar] [CrossRef]
- Schjesvold, F.; Robak, P.; Pour, L.; Aschan, J.; Sonneveld, P. OCEAN: A randomized phase III study of melphalan flufenamide + dexamethasone to treat relapsed refractory multiple myeloma. Future Oncol. 2020, 16, 631–641. [Google Scholar] [CrossRef] [PubMed]
- NIH US National Library of Medicine. ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ (accessed on 24 March 2020).
- Merlini, G.; Dispenzieri, A.; Sanchorawala, V.; Schonland, S.O.; Palladini, G.; Hawkins, P.N.; Gertz, M.A. Systemic immunoglobulin light chain amyloidosis. Nat. Rev. Dis. Primers 2018, 4, 38. [Google Scholar] [CrossRef] [PubMed]
- Varga, C.; Titus, S.E.; Toskic, D.; Comenzo, R.L. Use of novel therapies in the treatment of light chain amyloidosis. Blood Rev. 2019, 37, 100581. [Google Scholar] [CrossRef]
- Milani, P.; Palladini, G.; Merlini, G. New concepts in the treatment and diagnosis of amyloidosis. Expert Rev. Hematol. 2018, 11, 117–127. [Google Scholar] [CrossRef]
- Paner, A.; Okwuosa, T.M.; Richardson, K.J.; Libby, E.N. Triplet therapies—The new standard of care for multiple myeloma: How to manage common toxicities. Expert Rev. Hematol. 2018, 11, 957–973. [Google Scholar] [CrossRef]
- Parameswaran, R.; Lunning, M.; Mantha, S.; Devlin, S.; Hamilton, A.; Schwartz, G.; Soff, G. Romiplostim for management of chemotherapy-induced thrombocytopenia. Support. Care Cancer 2014, 22, 1217–1222. [Google Scholar] [CrossRef]
- Lakshman, A.; Abeykoon, J.P.; Kumar, S.K.; Rajkumar, S.V.; Dingli, D.; Buadi, F.K.; Gonsalves, W.I.; Leung, N.; Dispenzieri, A.; Kourelis, T.V.; et al. Efficacy of daratumumab-based therapies in patients with relapsed, refractory multiple myeloma treated outside of clinical trials. Am. J. Hematol. 2017, 92, 1146–1155. [Google Scholar] [CrossRef]
- Richardson, P.G.; San Miguel, J.F.; Moreau, P.; Hajek, R.; Dimopoulos, M.A.; Laubach, J.P.; Palumbo, A.; Luptakova, K.; Romanus, D.; Skacel, T.; et al. Interpreting clinical trial data in multiple myeloma: Translating findings to the real-world setting. Blood Cancer J. 2018, 8, 109. [Google Scholar] [CrossRef]
- Al Hamed, R.; Bazarbachi, A.H.; Malard, F.; Harousseau, J.L.; Mohty, M. Current status of autologous stem cell transplantation for multiple myeloma. Blood Cancer J. 2019, 9, 44. [Google Scholar] [CrossRef]
| Characteristic | Melflufen | Melphalan |
|---|---|---|
| Chemical structure | 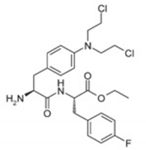 |  |
| Peak intracellular melphalan concentration * | Relative concentration: ≈80 (achieved after 15 min exposure) | Relative concentration: ≈4 (achieved after 60 min exposure) |
| Mean IC50, μM † | 0.05 | 1.5 |
| Lipophilicity (LogP) ‡ | 4.0 | −0.97 |
| Parameter, Median (Range) * | Melflufen | Melphalan |
|---|---|---|
| Cmax, ng/mL | 176 (43–1306) | 513 (320–1455) |
| tmax, h | 0.42 (0.32–0.58) | 0.58 (0.42–1.48) |
| AUC0–0.5, ng/mL*h | 40.3 (11.8–162) | 92.2 (41.1–181) |
| AUC0–∞, ng/mL*h | - | 895 (511–1503) |
| t1/2, h | - | 1.09 (0.83–1.83) |
| Clearance, L/h | - | 32.0 (19.0–56.0) |
| Study | Treatment | Patients, n | Prior Therapies, Median (Range) | Median Follow-Up, mo | ORR, % | Median PFS (95% CI), mo | Safety Profile |
|---|---|---|---|---|---|---|---|
| O-12-M1 (Phase I/II) | Melflufen + dexamethasone; median duration:4.1 mo | 45 (Phase II; evaluable for response) | 4 (2–14) | 27.9 | 31 | 5.7 (3.7–9.2) |
|
| HORIZON * (Phase II) | Melflufen + dexamethasone; median duration: 14.3 wk | 154 (125 evaluable for response) | 5 (2–12) | Not reported | 29 | 4.2 (3.7–4.9) |
|
| ANCHOR † (Phase I/II) | Melflufen (30 or 40 mg) + dexamethasone + daratumumab; median duration: 6.2 mo | 33 (30 mg, n = 6; 40 mg, n = 27) | 30 mg: 2.5 (1–3) 40 mg: 2.0 (1–4) | 6.6 | 76 | 14.3 (9.7-NR) |
|
| Melflufen (30 or 40 mg) + dexamethasone + bortezomib; median duration: 9.3 mo | 6 (30 mg or 40 mg, n = 3 each) | 2.5 (2–4) | 13.4 | 67 | NR |
|
| Study (NCT ID) | Treatment Arm | Patients | Study Status [59] |
|---|---|---|---|
| HORIZON Phase II (NCT02963493) | Melflufen + dexamethasone | Received ≥2 lines of prior therapy; refractory to pomalidomide and/or anti-CD38 monoclonal antibody |
|
| ANCHOR Phase I/II (NCT03481556) | Melflufen + dexamethasone + daratumumab or bortezomib | Relapsed/refractory; received 1–4 prior lines of therapy |
|
| OCEAN Phase III (NCT03151811) | Melflufen + dexamethasone vs. pomalidomide + dexamethasone | Relapsed/refractory; received 2–4 prior lines of therapy; refractory to lenalidomide in the last line of therapy |
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mateos, M.-V.; Bladé, J.; Bringhen, S.; Ocio, E.M.; Efebera, Y.; Pour, L.; Gay, F.; Sonneveld, P.; Gullbo, J.; Richardson, P.G. Melflufen: A Peptide–Drug Conjugate for the Treatment of Multiple Myeloma. J. Clin. Med. 2020, 9, 3120. https://doi.org/10.3390/jcm9103120
Mateos M-V, Bladé J, Bringhen S, Ocio EM, Efebera Y, Pour L, Gay F, Sonneveld P, Gullbo J, Richardson PG. Melflufen: A Peptide–Drug Conjugate for the Treatment of Multiple Myeloma. Journal of Clinical Medicine. 2020; 9(10):3120. https://doi.org/10.3390/jcm9103120
Chicago/Turabian StyleMateos, María-Victoria, Joan Bladé, Sara Bringhen, Enrique M Ocio, Yvonne Efebera, Luděk Pour, Francesca Gay, Pieter Sonneveld, Joachim Gullbo, and Paul G. Richardson. 2020. "Melflufen: A Peptide–Drug Conjugate for the Treatment of Multiple Myeloma" Journal of Clinical Medicine 9, no. 10: 3120. https://doi.org/10.3390/jcm9103120
APA StyleMateos, M.-V., Bladé, J., Bringhen, S., Ocio, E. M., Efebera, Y., Pour, L., Gay, F., Sonneveld, P., Gullbo, J., & Richardson, P. G. (2020). Melflufen: A Peptide–Drug Conjugate for the Treatment of Multiple Myeloma. Journal of Clinical Medicine, 9(10), 3120. https://doi.org/10.3390/jcm9103120










