Assessment of the Minimal Targeted Biopsy Core Number per MRI Lesion for Improving Prostate Cancer Grading Prediction
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. MR Technique
2.3. Analyses
2.4. Statistics
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mottet, N.; Bellmunt, J.; Bolla, M.; Briers, E.; Cumberbatch, M.G.; De Santis, M.; Fossati, N.; Gross, T.; Henry, A.M.; Joniau, S.; et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2017, 71, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Kasivisvanathan, V.; Rannikko, A.S.; Borghi, M.; Panebianco, V.; Mynderse, L.A.; Vaarala, M.H.; Briganti, A.; Budäus, L.; Hellawell, G.; Hindley, R.G.; et al. MRI-Targeted or Standard Biopsy for Prostate-Cancer Diagnosis. N. Engl. J. Med. 2018, 378, 1767–1777. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.M.; George, A.K.; Rubin, R.; Rais-Bahrami, S.; Parnes, H.L.; Merino, M.J.; Simon, R.M.; Turkbey, B.; Choyke, P.L.; Wood, B.J.; et al. Efficiency of Prostate Cancer Diagnosis by MR/Ultrasound Fusion-Guided Biopsy vs Standard Extended-Sextant Biopsy for MR-Visible Lesions. J. Natl. Cancer Inst. 2016, 108, djw039. [Google Scholar] [CrossRef] [PubMed]
- van der Leest, M.; Cornel, E.; Israël, B.; Hendriks, R.; Padhani, A.R.; Hoogenboom, M.; Zamecnik, P.; Bakker, D.; Setiasti, A.Y.; Veltman, J.; et al. Head-to-head Comparison of Transrectal Ultrasound-guided Prostate Biopsy Versus Multiparametric Prostate Resonance Imaging with Subsequent Magnetic Resonance-guided Biopsy in Biopsy-naïve Men with Elevated Prostate-specific Antigen: A Large Prospective Multicenter Clinical Study. Eur. Urol. 2019, 75, 570–578. [Google Scholar] [PubMed]
- Rouvière, O.; Puech, P.; Renard-Penna, R.; Claudon, M.; Roy, C.; Mège-Lechevallier, F.; Decaussin-Petrucci, M.; Dubreuil-Chambardel, M.; Magaud, L.; Remontet, L.; et al. Use of prostate systematic and targeted biopsy on the basis of multiparametric MRI in biopsy-naive patients (MRI-FIRST): A prospective, multicentre, paired diagnostic study. Lancet Oncol. 2019, 20, 100–109. [Google Scholar] [CrossRef]
- Dimitroulis, P.; Rabenalt, R.; Nini, A.; Hiester, A.; Esposito, I.; Schimmöller, L.; Antoch, G.; Albers, P.; Arsov, C. Multiparametric magnetic resonance imaging/ultrasound fusion prostate biopsy: Are 2 biopsy cores per magnetic resonance imaging lesion required? J. Urol. 2018, 200, 1030–1034. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.J.; Syed, J.S.; Ghabili, K.; Hsiang, W.R.; Nguyen, K.A.; Leapman, M.S.; Sprenkle, P.C. Role of Core Number and Location in Targeted Magnetic Resonance Imaging- Ultrasound Fusion Prostate Biopsy. Eur. Urol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Schouten, M.G.; van der Leest, M.; Pokorny, M.; Hoogenboom, M.; Barentsz, J.O.; Thompson, L.C.; Fütterer, J.J. Why and Where do We Miss Significant Prostate Cancer with Multi-parametric Magnetic Resonance Imaging followed by Magnetic Resonance-guided and Transrectal Ultrasound-guided Biopsy in Biopsy-naïve Men? Eur. Urol. 2017, 71, 896–903. [Google Scholar] [CrossRef] [PubMed]
- Diamand, R.; Oderda, M.; Obeid, W.A.H.; Albisinni, S.; Van Velthoven, R.; Fasolis, G.; Simone, G.; Ferriero, M.; Roche, J.B.; Piechaud, T.; et al. A multicentric study on accurate grading of prostate cancer with systematic and MRI/US fusion targeted biopsies: Comparison with final histopathology after radical prostatectomy. World J. Urol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Ploussard, G.; Borgmann, H.; Briganti, A.; De Visschere, P.; Fütterer, J.J.; Gandaglia, G.; Heidegger, I.; Kretschmer, A.; Mathieu, R.; Ost, P.; et al. Positive pre-biopsy MRI: Are systematic biopsies still useful in addition to targeted biopsies? World J. Urol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gold, S.A.; Hale, G.R.; Bloom, J.B.; Smith, C.P.; Rayn, K.N.; Valera, V.; Wood, B.J.; Choyke, P.L.; Turkbey, B.; Pinto, P.A. Follow-up of negative MRI-targeted prostate biopsies: When are we missing cancer? World J. Urol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Radtke, J.P.; Schwab, C.; Wolf, M.B.; Freitag, M.T.; Alt, C.D.; Kesch, C.; Popeneciu, I.V.; Huettenbrink, C.; Gasch, C.; Klein, T.; et al. Multiparametric Magnetic Resonance Imaging (MRI) and MRI-Transrectal Ultrasound Fusion Biopsy for Index Tumor Detection: Correlation with Radical Prostatectomy Specimen. Eur. Urol. 2016, 70, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Le, J.D.; Tan, N.; Shkolyar, E.; Lu, D.Y.; Kwan, L.; Marks, L.S.; Huang, J.; Margolis, D.J.; Raman, S.S.; Reiter, R.E. Multifocality and prostate cancer detection by multiparametric magnetic resonance imaging: Correlation with whole-mount histopathology. Eur. Urol. 2015, 67, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Barentsz, J.O.; Richenberg, J.; Clements, R.; Choyke, P.; Verma, S.; Villeirs, G.; Rouviere, O.; Logager, V.; Fütterer, J.J. ESUR prostate MR guidelines 2012. Eur. Radiol. 2012, 22, 746–757. [Google Scholar] [CrossRef] [PubMed]
- Barentsz, J.O.; Weinreb, J.C.; Verma, S.; Thoeny, H.C.; Tempany, C.M.; Shtern, F.; Padhani, A.R.; Margolis, D.; Macura, K.J.; Haider, M.A.; et al. Synopsis of the PI-RADS v2 guidelines for multiparametric prostate magnetic resonance imaging and recommendations for use. Eur. Urol. 2016, 69, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Westhoff, N.; Siegel, F.P.; Hausmann, D.; Polednik, M.; von Hardenberg, J.; Michel, M.S.; Ritter, M. Precision of MRI/ultrasound-fusion biopsy in prostate cancer diagnosis: An ex vivo comparison of alternative biopsy techniques on prostate phantoms. World J. Urol. 2017, 35, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Ploussard, G.; Beauval, J.B.; Lesourd, M.; Almeras, C.; Assoun, J.; Aziza, R.; Gautier, J.R.; Loison, G.; Portalez, D.; Salin, A.; et al. Added Value of Concomitant Systematic and Fusion Targeted Biopsies for Grade Group Prediction Based on Radical Prostatectomy Final Pathology on Positive Magnetic Resonance Imaging. J. Urol. 2019, 202, 1182–1187. [Google Scholar] [CrossRef]
- Wibulpolprasert, P.; Raman, S.S.; Hsu, W.; Margolis, D.J.; Asvadi, N.H.; Khoshnoodi, P.; Moshksar, A.; Tan, N.; Ahuja, P.; Maehara, C.K.; et al. Detection and Localization of Prostate Cancer at 3-T Multiparametric MRI Using PI-RADS Segmentation. Am. J. Roentgenol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Covin, B.; Roumiguié, M.; Quintyn-Ranty, M.L.; Graff, P.; Khalifa, J.; Aziza, R.; Ploussard, G.; Portalez, D.; Malavaud, B. Refining the risk-stratification of transrectal biopsy-detected prostate cancer by elastic fusion registration transperineal biopsies. World J. Urol. 2018. [Google Scholar] [CrossRef]
- Cornud, F.; Roumiguié, M.; Barry de Longchamps, N.; Ploussard, G.; Bruguière, E.; Portalez, D.; Malavaud, B. Precision Matters in MR Imaging-targeted Prostate Biopsies: Evidence from a Prospective Study of Cognitive and Elastic Fusion Registration Transrectal Biopsies. Radiology 2018, 287, 534–542. [Google Scholar] [CrossRef] [PubMed]
| Grade Group on TB | Grade Group on Final Pathology (RP Specimens) | ||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4–5 | Total | |
| No cancer | 7 | 35 | 9 | 1 | 52 |
| 1 | 2 | 39 | 9 | 3 | 53 |
| 2 | 4 | 136 | 68 | 7 | 215 |
| 3 | 1 | 26 | 62 | 8 | 97 |
| 4–5 | 0 | 6 | 29 | 26 | 61 |
| Total | 14 | 242 | 177 | 55 | 478 |
| Upgrading Rate after TB | |||
|---|---|---|---|
| No n = 284 | Yes n = 194 | p-Value | |
| Age (mean), years | 65.1 | 64.6 | 0.410 |
| PSA (mean), ng/mL | 10.3 | 10.6 | 0.719 |
| PSAD (mean), ng/mL/gram | 0.27 | 0.23 | 0.297 |
| Prostate volume (mean), mL | 48.1 | 52.0 | 0.121 |
| PIRADS score (mean) | 4.2 | 3.9 | <0.001 |
| No MRI lesions (mean) | 1.37 | 1.34 | 0.632 |
| MRI lesion size (mean), mm | 12.7 | 11. | 0.017 |
| No TB cores(mean) | 4.1 | 3.2 | <0.001 |
| No TB per lesion (mean) | 3.1 | 2.6 | <0.001 |
| TB density (mean) | 0.36 | 0.30 | 0.015 |
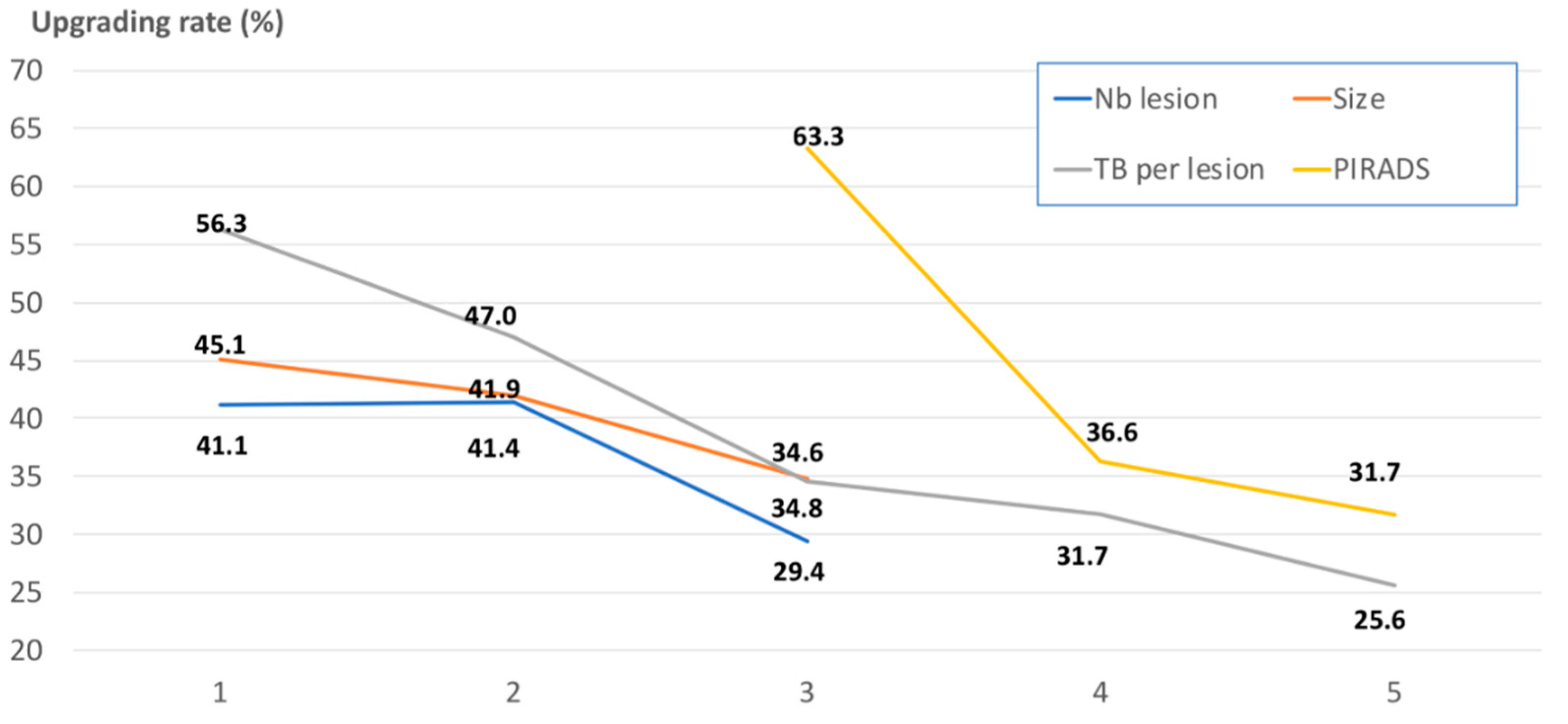
| Number | Upgrading Rate (%) after TB | |
|---|---|---|
| PIRADS score: | ||
| PIRADS 3 | 98 | 63.3 |
| PIRADS 4 | 238 | 36.6 |
| PIRADS 5 | 142 | 31.7 |
| p value | <0.001 | |
| MRI lesions: | ||
| 1 | 343 | 41.1 |
| 2 | 111 | 41.4 |
| 3–4 | 24 | 29.3 |
| p value | 0.504 | |
| MRI lesions size (mm): | ||
| <10 | 131 | 45.1 |
| 10–15 | 223 | 41.9 |
| >15 | 124 | 34.8 |
| p value | 0.254 | |
| TB core number: | ||
| 2 | 188 | 50.0 |
| 3 | 65 | 43.1 |
| 4 | 125 | 36.0 |
| 5 | 100 | 27.0 |
| p value | 0.001 | |
| TBs per lesion: | ||
| 1 | 48 | 56.3 |
| 2 | 202 | 47.0 |
| 3 | 81 | 34.6 |
| 4 | 104 | 31.7 |
| 5 or more | 43 | 25.6 |
| p value | 0.002 | |
| TB density: | ||
| <0.20 | 117 | 44.4 |
| 0.20-0.40 | 238 | 42.7 |
| ≥0.40 | 123 | 34.2 |
| p value | 0.229 |
| Upgrading Rate (%) after TB | |
|---|---|
| PIRADS 3 (n = 98) TB per lesion: | |
| 1–2 | 66.2 |
| 3 | 66.7 |
| 4 or more | 33.3 |
| PIRADS 4–5 (n = 380) TB per lesion: | |
| 1–2 | 41.0 |
| 3 | 29.0 |
| 4 or more | 29.7 |
| p-value | 0.002 |
| Upgrading Rate (%) after TB | |
|---|---|
| PIRADS score: | |
| PIRADS 3 | Ref. (Reference) |
| PIRADS 4 | 0.34 (0.20–0.57) |
| PIRADS 5 | 0.27 (0.13–0.54) |
| p-value | <0.001 |
| MRI lesions: | |
| 1 | Ref. |
| 2 | 1.29 (0.77–2.15) |
| 3–4 | 1.12 (0.41–3.01) |
| p-value | 0.620 |
| MRI lesion size (mm): | |
| <10 | Ref. |
| 10–15 | 0.77 (0.45–1.32) |
| >15 | 0.91 (0.40–2.04) |
| p-value | 0.596 |
| TBs per lesion: | |
| 2 | Ref. |
| 3–4 | 0.79 (0.42–1.46) |
| 5 or more | 0.57 (0.27–1.18) |
| p-value | 0.316 |
| TB density: | |
| <0.20 | Ref. |
| 0.20–0.40 | 0.90 (0.44–1.47) |
| ≥0.40 | 0.81 (0.33–2.44) |
| p-value | 0.725 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ploussard, G.; Beauval, J.-B.; Renard-Penna, R.; Lesourd, M.; Manceau, C.; Almeras, C.; Gautier, J.-R.; Loison, G.; Portalez, D.; Salin, A.; et al. Assessment of the Minimal Targeted Biopsy Core Number per MRI Lesion for Improving Prostate Cancer Grading Prediction. J. Clin. Med. 2020, 9, 225. https://doi.org/10.3390/jcm9010225
Ploussard G, Beauval J-B, Renard-Penna R, Lesourd M, Manceau C, Almeras C, Gautier J-R, Loison G, Portalez D, Salin A, et al. Assessment of the Minimal Targeted Biopsy Core Number per MRI Lesion for Improving Prostate Cancer Grading Prediction. Journal of Clinical Medicine. 2020; 9(1):225. https://doi.org/10.3390/jcm9010225
Chicago/Turabian StylePloussard, Guillaume, Jean-Baptiste Beauval, Raphaële Renard-Penna, Marine Lesourd, Cécile Manceau, Christophe Almeras, Jean-Romain Gautier, Guillaume Loison, Daniel Portalez, Ambroise Salin, and et al. 2020. "Assessment of the Minimal Targeted Biopsy Core Number per MRI Lesion for Improving Prostate Cancer Grading Prediction" Journal of Clinical Medicine 9, no. 1: 225. https://doi.org/10.3390/jcm9010225
APA StylePloussard, G., Beauval, J.-B., Renard-Penna, R., Lesourd, M., Manceau, C., Almeras, C., Gautier, J.-R., Loison, G., Portalez, D., Salin, A., Soulié, M., Tollon, C., Malavaud, B., & Roumiguié, M. (2020). Assessment of the Minimal Targeted Biopsy Core Number per MRI Lesion for Improving Prostate Cancer Grading Prediction. Journal of Clinical Medicine, 9(1), 225. https://doi.org/10.3390/jcm9010225







