Lung Metastases in Patients with Stage IV Pancreatic Cancer: Prevalence, Risk Factors, and Survival Impact
Abstract
1. Introduction
2. Patients and Methods
2.1. Patient Selection
2.2. Data Collection
2.3. Statistical Analysis
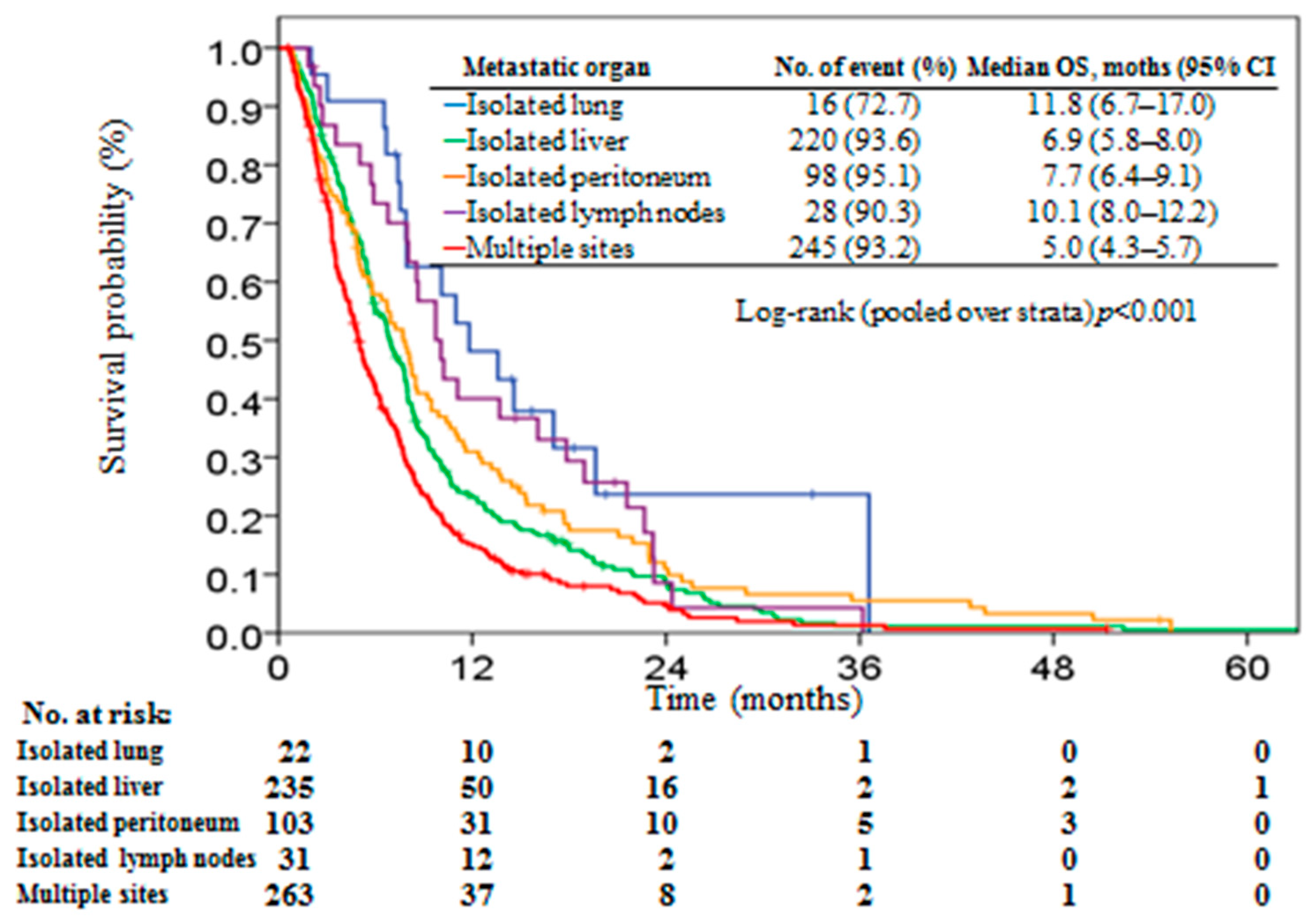
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cancer Stat Facts: Pancreatic Cancer. Available online: https://seer.cancer.gov/statfacts/html/pancreas.html (accessed on 20 July 2019).
- Health Registry Annual Report 2016. Republic of China. Republic of China: Republic of China: Bureau of Health Promotion, Department of Health, Executive Yuan. 2018. Available online: http://www.hpa.gov.tw/BHPNet/Web/Stat/Statistics.aspx (accessed on 20 July 2019).
- Conroy, T.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.L.; Gourgou-Bourgade, S.; de la Fouchardière, C.; et al. Folfirinox versus gemcitabine for metastatic pancreatic cancer. N. Engl. J. Med. 2011, 364, 1817–1825. [Google Scholar] [CrossRef] [PubMed]
- von Hoff, D.D.; Ervin, T.; Arena, F.P.; Chiorean, G.; Infante, J.; Moore, M.; Seay, T.; Tjulandin, S.A.; Ma, W.W.; Saleh, M.N.; et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N. Engl. J. Med. 2013, 369, 1691–1703. [Google Scholar] [CrossRef] [PubMed]
- Burris, H.A.; Moore, M.J.; Andersen, J.; Green, M.R.; Rothenberg, M.L.; Modiano, M.R.; Cripps, M.C.; Portenoy, R.K.; Storniolo, A.M.; Tarassoff, P.; et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: A randomized trial. J. Clin. Oncol. 1997, 15, 2403–2413. [Google Scholar] [CrossRef] [PubMed]
- Oweira, H.; Petrausch, U.; Helbling, D.; Schmidt, J.; Mannhart, M.; Mehrabi, A.; Schob, O.; Giryes, A.; Decker, M.; Abdel-Rahman, O. Prognostic value of site-specific metastases in pancreatic adenocarcinoma: A Surveillance Epidemiology and End Results database analysis. World J. Gastroenterol. 2017, 23, 1872–1880. [Google Scholar] [CrossRef] [PubMed]
- Downs-Canner, S.; Zenati, M.; Boone, B.A.; Varley, P.R.; Steve, J.; Hogg, M.E.; Zureikat, A.; Zeh, H.J.; Lee, K.K.W. The indolent nature of pulmonary metastases from ductal adenocarcinoma of the pancreas. J. Surg. Oncol. 2015, 112, 80–85. [Google Scholar] [CrossRef]
- Arnaoutakis, G.J.; Rangachari, D.; Laheru, D.A.; Iacobuzio-Donahue, C.A.; Hruban, R.H.; Herman, J.M.; Edil, B.H.; Pawlik, T.M.; Schulick, R.D.; Cameron, J.L.; et al. Pulmonary resection for isolated pancreatic adenocarcinoma metastasis: An analysis of outcomes and survival. J. Gastrointest. Surg. 2011, 15, 1611–1617. [Google Scholar] [CrossRef]
- Kruger, S.; Haas, M.; Burger, P.J.; Ormanns, S.; Modest, D.P.; Westphalen, C.B.; Michl, M.; Kleespies, A.; Angele, M.K.; Hartwig, W.; et al. Isolated pulmonary metastases define a favorable subgroup in metastatic pancreatic cancer. Pancreatology 2016, 16, 593–598. [Google Scholar] [CrossRef]
- Decoster, C.; Gilabert, M.; Autret, A.; Turrini, O.; Oziel-Taieb, S.; Poizat, F.; Giovannini, M.; Viens, P.; Iovanna, J.; Raoul, J.L. Heterogeneity of metastatic pancreatic adenocarcinoma: Lung metastasis show better prognosis than liver metastasis—A case control study. Oncotarget 2016, 7, 45649–45655. [Google Scholar] [CrossRef]
- Lovecek, M.; Skalicky, P.; Chudacek, J.; Szkorupa, M.; Svebisova, H.; Lemstrova, R.; Ehrmann, J.; Melichar, B.; Yogeswara, T.; Klos, D.; et al. Different clinical presentations of metachronous pulmonary metastases after resection of pancreatic ductal adenocarcinoma: Retrospective study and review of the literature. World J. Gastroenterol. 2017, 23, 6420–6428. [Google Scholar] [CrossRef]
- Wangjam, T.; Zhang, Z.; Zhou, X.C.; Lyer, L.; Faisal, F.; Soares, K.C.; Fishman, E.; Hruban, R.H.; Herman, J.M.; Laheru, D.; et al. Resected pancreatic ductal adenocarcinomas with recurrence limited in lung have a significantly better prognosis than those with other recurrence patterns. Oncotarget 2015, 6, 36903–36910. [Google Scholar] [CrossRef]
- Yamashita, K.; Miyamoto, A.; Hama, N.; Asaoka, T.; Maeda, S.; Omiya, H.; Takami, K.; Doki, Y.; Mori, M.; Nakamori, S. Survival impact of pulmonary metastasis as recurrence of pancreatic ductal adenocarcinoma. Dig. Surg. 2015, 32, 464–471. [Google Scholar] [CrossRef] [PubMed]
- UICC TNM. Classification of Malignant Tumours, 7th ed.; Wiley & Liss: New York, NY, USA, 2009. [Google Scholar]
- Lee, S.H.; Chang, P.H.; Chen, P.T.; Lu, C.H.; Hung, Y.S.; Tsang, N.M.; Hung, C.Y.; Chen, J.S.; Hsu, H.C.; Chen, Y.Y.; et al. Association of time interval between cancer diagnosis and initiation of palliative chemotherapy with overall survival in patients with unresectable pancreatic cancer. Cancer Med. 2019, 8, 3471–3478. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.L.; Chen, Y.Y.; Lu, C.H.; Hung, C.Y.; Kuo, Y.C.; Chen, J.S.; Hsu, H.C.; Chen, P.T.; Chang, P.H.; Hung, Y.S.; et al. Effect of S-1 on survival outcomes in 838 patients with advanced pancreatic cancer: A 7-year multicenter observational cohort study in Taiwan. Cancer Med. 2019, 8, 2085–2094. [Google Scholar] [CrossRef] [PubMed]
- Chou, W.C.; Chen, Y.Y.; Hung, C.Y.; Chen, J.S.; Lu, C.H.; Chang, P.H. Evolution of the chemotherapeutic landscape and survival outcome in patients with metastatic pancreatic cancer: A four-institute cohort study in Taiwan, 2010–2016. Cancer Manag. Res. 2019, 11, 2119–2127. [Google Scholar] [CrossRef] [PubMed]
- Pappas, S.G.; Christians, K.K.; Tolat, P.P.; Mautz, A.P.; Lal, A.; McElroy, L.; Gamblin, T.C.; Turaga, K.K.; Tsai, S.; Erickson, B.; et al. Staging chest computed tomography and positron emission tomography in patients with pancreatic adenocarcinoma: Utility or futility? HPB (Oxf.) 2014, 16, 70–74. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mehtsun, W.T.; Chipidza, F.E.; Fernández-Del Castillo, C.; Hemingway, K.; Fong, Z.V.; Chang, D.C.; Pandharipande, P.; Clark, J.W.; Allen, J.; Hong, T.S.; et al. Are staging computed tomography (CT) scans of the chest necessary in pancreatic adenocarcinoma? Ann. Surg. Oncol. 2018, 25, 3936–3942. [Google Scholar] [CrossRef] [PubMed]
- Nordback, I.; Saaristo, R.; Piironen, A.; Sand, J. Chest computed tomography in the staging of pancreatic and periampullary carcinoma. Scand. J. Gastroenterol. 2004, 39, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Poruk, K.E.; Kim, Y.; Cameron, J.L.; He, J.; Eckhauser, F.E.; Rezaee, N.; Herman, J.; Laheru, D.; Zheng, L.; Fishman, E.K.; et al. What is the significance of indeterminate pulmonary nodules in patients undergoing resection for pancreatic adenocarcinoma? J. Gastrointest. Surg. 2015, 19, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.T.; Nguyen, D.C.; Raptis, C.; Menias, C.O.; Zhou, G.; Wang-Gillam, A.; Linehan, D.C.; Hawkins, W.G.; Strasberg, S.M.; Fields, R.C. Natural history of preoperative subcentimeter pulmonary nodules in patients with resectable pancreatic adenocarcinoma: A retrospective cohort study. Ann. Surg. 2015, 261, 970–975. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.S.; Hung, C.Y.; Chang, H.; Liu, C.T.; Chen, Y.Y.; Lu, C.H.; Chang, P.H.; Hung, Y.S.; Chou, W.C. Venous thromboembolism in asian patients with pancreatic cancer following palliative chemotherapy: Low incidence but a negative prognosticator for those with early onset. Cancers (Basel) 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Hohla, F.; Hopfinger, G.; Romeder, F.; Rinnerthaler, G.; Bezan, A.; Stättner, S.; Hauser-Kronberger, C.; Ulmer, H.; Greil, R. Female gender may predict response to FOLFIRINOX in patients with unresectable pancreatic cancer: A single institution retrospective review. Int. J. Oncol. 2014, 44, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.; Mukherjee, A.; Karanam, B.; Davis, M.; Jaynes, J.; Reams, R.R.; Dean-Colomb, W.; Yates, C. African Americans with pancreatic ductal adenocarcinoma exhibit gender differences in Kaiso expression. Cancer Lett. 2016, 380, 513–522. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | N (%) |
|---|---|
| Median age, year (range) | 62 (25–88) |
| Sex | |
| male | 393 (60.1) |
| female | 261 (39.9) |
| Body mass index, Kg/m2, median (range) | 22.4 (13.0–36.2) |
| Active or ever smoker | 249 (38.1) |
| ECOG performance scale | |
| 0 or 1 | 450 (68.8) |
| 2 | 174 (26.6) |
| 3 | 30 (4.6) |
| Charlson comorbidity index | |
| 0 | 177 (27.1) |
| 1 | 227 (34.7) |
| 2 | 148 (22.6) |
| 3 | 79 (12.1) |
| 4 | 18 (2.8) |
| 5 | 5 (0.8) |
| Body weight loss, %, median (range) | 5 (0–33) |
| Primary tumor site + | |
| head | 224 (34.3) |
| body | 121 (18.5) |
| tail | 162 (24.8) |
| overlapping | 147 (22.5) |
| Tumor size, cm, median (range) + | 4.5 (1.4–14.0) |
| Previous pancreatectomy | |
| yes | 23 (3.5) |
| no | 631 (96.5) |
| T-classification, 7th AJCC + | |
| 1 | 13 (2) |
| 2 | 82 (12.5) |
| 3 | 188 (28.7) |
| 4 | 371 (56.7) |
| N-classification, 7th AJCC + | |
| 0 | 134 (20.5) |
| 1 | 520 (79.5) |
| Number of metastatic organ + | |
| 1 | 391 (59.8) |
| 2 | 191 (29.2) |
| 3 | 53 (8.1) |
| ≥4 | 19 (2.9) |
| Organ of metastases *,+ | |
| liver | 438 (67.0) |
| peritoneum | 239 (36.5) |
| distant lymph nodes | 150 (22.9) |
| lung | 98 (15.0) |
| others | 51 (6.1) |
| CEA, ng/dL | |
| ≤5 | 290 (44.3) |
| >5 | 364 (55.7) |
| CA19-9, ng/dL | |
| ≤37 | 130 (19.9) |
| >37 | 524 (80.1) |
| First-line chemotherapeutic regimen | |
| Gemcitabine monotherapy | 208 (31.8) |
| Gemcitabine plus platinum or 5-fluorouracil/TS-1 | 398 (60.9) |
| 5-fluorouracil +/- oxaliplatin +/- irinotecan | 15 (2.3) |
| TS-1 monotherapy | 32 (4.9) |
| Variable | Category | No of Lung Metastases/No of Total Patients (%) | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | p Value | Adjusted OR (95% CI) | p Value | |||
| Overall | 98/654 (15.0) | |||||
| Sex | male | 46/393 (11.7) | 1 | 1 | ||
| female | 52/261 (19.9) | 1.88 (1.22–2.89) | 0.004 | 1.90 (1.23–2.95) | 0.004 | |
| Age, year | ≤65 | 39/309 (12.6) | 1 | 1 | ||
| >65 | 59/345 (17.1) | 1.43 (0.93–2.21) | 0.09 | 1.37 (0.88–2.14) | 0.17 | |
| ECOG performance | 0 or 1 | 65/450 (14.4) | 1 | |||
| 2 | 27/174 (15.5) | 1.09 (0.67–1.77) | 0.74 | |||
| 3 | 6/30 (20) | 1.48 (0.58–3.76) | 0.41 | |||
| CCI | 0 or 1 | 57/404 (14.1) | 1 | |||
| >1 | 41/250 (16.4) | 1.19 (0.77–1.85) | 0.43 | |||
| Active or ever smoking | no | 63/405 (15.6) | 1 | |||
| yes | 35/249 (14.1) | 0.89 (0.57–1.39) | 0.60 | |||
| CEA, ng/dL | ≤5 | 46/290 (15.9) | 1 | |||
| >5 | 52/364 (14.3) | 0.88 (0.58–1.36) | 0.58 | |||
| CA19-9, ng/dL | ≤37 | 22/130 (16.9) | 1 | |||
| >37 | 76/524 (14.5) | 0.83 (0.49–1.41) | 0.49 | |||
| Body weight loss | ≤5% | 40/286 (14.0) | 1 | |||
| >5% | 47/287 (16.4) | 1.20 (0.76–1.90) | 0.43 | |||
| Primary tumor site | head | 32/224 (14.3) | 1 | |||
| body | 20/121 (16.5) | 1.19 (0.65–2.18) | 0.58 | |||
| tail | 24/162 (14.8) | 1.04 (0.59–1.85) | 0.88 | |||
| overlapping | 22/147 (15.0) | 0.59–1.90 | 0.86 | |||
| Tumor grade | well to moderate | 3/63 (4.8) | 1 | 1 | ||
| poorly or undifferentiated | 95/591 (16.1) | 3.83 (1.18–12.5) | 0.026 | 4.08 (1.24–13.4) | 0.020 | |
| T-classification | 1 or 2 | 12/95 (12.6) | 1 | |||
| 3 | 29/188 (15.4) | 1.26 (0.61–2.60) | 0.53 | |||
| 4 | 57/371 (15.4) | 1.26 (0.64–2.45) | 0.50 | |||
| N-classification | 0 | 21/134 (15.7) | 1 | |||
| 1 | 77/520 (14.8) | 0.94 (0.55–1.58) | 0.80 | |||
| T-size | <8 cm | 86/609 (14.1) | 1 | 1 | ||
| ≥8 cm | 12/45 (26.7) | 2.21 (1.10–4.45) | 0.026 | 2.23 (1.09–4.54) | 0.027 | |
| Variable | Category | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|---|
| HR (95% CI) | p Value | Adjusted HR (95% CI) | p Value | ||
| Sex | male | 1 | 1 | ||
| female | 0.84 (0.71–0.99) | 0.033 | 0.86 (0.72–1.01) | 0.86 | |
| Age, year | ≤65 | 1 | 1 | ||
| >65 | 1.30 (1.10–1.52) | 0.002 | 1.02 (0.86–1.22) | 0.80 | |
| ECOG performance | 0 or 1 | 1 | 1 | ||
| 2 | 2.50 (2.08–3.01) | <0.001 | 2.46 (2.02–2.99) | <0.001 | |
| 3 | 5.60 (3.75–8.36) | <0.001 | 5.79 (3.83–8.77) | <0.001 | |
| Body weight loss | ≤5% | 1 | |||
| >5% | 1.04 (0.87–1.23) | 0.67 | |||
| Metastatic organ | isolated lung | 1 | 1 | ||
| isolated liver | 1.96 (1.18–3.25) | 0.010 | 1.92 (1.15–3.21) | 0.013 | |
| isolated peritoneum | 1.68 (0.99–2.85) | 0.055 | 1.54 (0.90–2.62) | 0.11 | |
| isolated lymph nodes | 1.38 (0.75–2.55) | 0.31 | 1.47 (0.79–2.74) | 0.22 | |
| multiple sites | 2.66 (1.60–4.42) | <0.001 | 2.60 (1.56–4.35) | <0.001 | |
| CCI | 0 to 1 | 1 | 1 | ||
| >1 | 1.34 (1.40–1.58) | <0.001 | 1.21 (1.02–1.44) | 0.027 | |
| Active or ever smoker | no | 1 | 1 | ||
| yes | 1.16 (0.98–1.36) | 0.080 | 1.22 (1.03–1.44) | 0.021 | |
| CEA, ng/dL | ≤5 | 1 | 1 | ||
| >5 | 1.17 (0.99–1.38) | 0.053 | 1.15 (0.98–1.36) | 0.090 | |
| CA19-9, ng/dL | ≤37 | 1 | |||
| >37 | 1.03 (0.84–1.25) | 0.81 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, K.-H.; Hung, C.-Y.; Hsueh, S.-W.; Chang, P.-H.; Chen, Y.-Y.; Lu, C.-H.; Chen, P.-T.; Yeh, K.-Y.; Huang, P.-W.; Tsang, N.-M.; et al. Lung Metastases in Patients with Stage IV Pancreatic Cancer: Prevalence, Risk Factors, and Survival Impact. J. Clin. Med. 2019, 8, 1402. https://doi.org/10.3390/jcm8091402
Liu K-H, Hung C-Y, Hsueh S-W, Chang P-H, Chen Y-Y, Lu C-H, Chen P-T, Yeh K-Y, Huang P-W, Tsang N-M, et al. Lung Metastases in Patients with Stage IV Pancreatic Cancer: Prevalence, Risk Factors, and Survival Impact. Journal of Clinical Medicine. 2019; 8(9):1402. https://doi.org/10.3390/jcm8091402
Chicago/Turabian StyleLiu, Keng-Hao, Chia-Yen Hung, Shu-Wen Hsueh, Pei-Hung Chang, Yen-Yang Chen, Chang-Hsien Lu, Ping-Tsung Chen, Kun-Yun Yeh, Pei-Wei Huang, Ngan-Ming Tsang, and et al. 2019. "Lung Metastases in Patients with Stage IV Pancreatic Cancer: Prevalence, Risk Factors, and Survival Impact" Journal of Clinical Medicine 8, no. 9: 1402. https://doi.org/10.3390/jcm8091402
APA StyleLiu, K.-H., Hung, C.-Y., Hsueh, S.-W., Chang, P.-H., Chen, Y.-Y., Lu, C.-H., Chen, P.-T., Yeh, K.-Y., Huang, P.-W., Tsang, N.-M., Hung, Y.-S., & Chou, W.-C. (2019). Lung Metastases in Patients with Stage IV Pancreatic Cancer: Prevalence, Risk Factors, and Survival Impact. Journal of Clinical Medicine, 8(9), 1402. https://doi.org/10.3390/jcm8091402





