Correlation between Sleep Bruxism, Stress, and Depression—A Polysomnographic Study
Abstract
1. Introduction
2. Material and Methods
2.1. Participants
2.2. Video-Polysomnography Participants’ Selection
2.3. Exclusion Criteria
2.4. Video-Polysomnographic Examination
2.5. Bruxism Assessment
2.6. Perceived Stress Scale-10
2.7. Beck’s Depression Inventory
2.8. Database
2.9. Data Analysis
3. Results
3.1. Sample Characteristics
3.2. Bruxism Episodes Index (BEI) Data Distribution
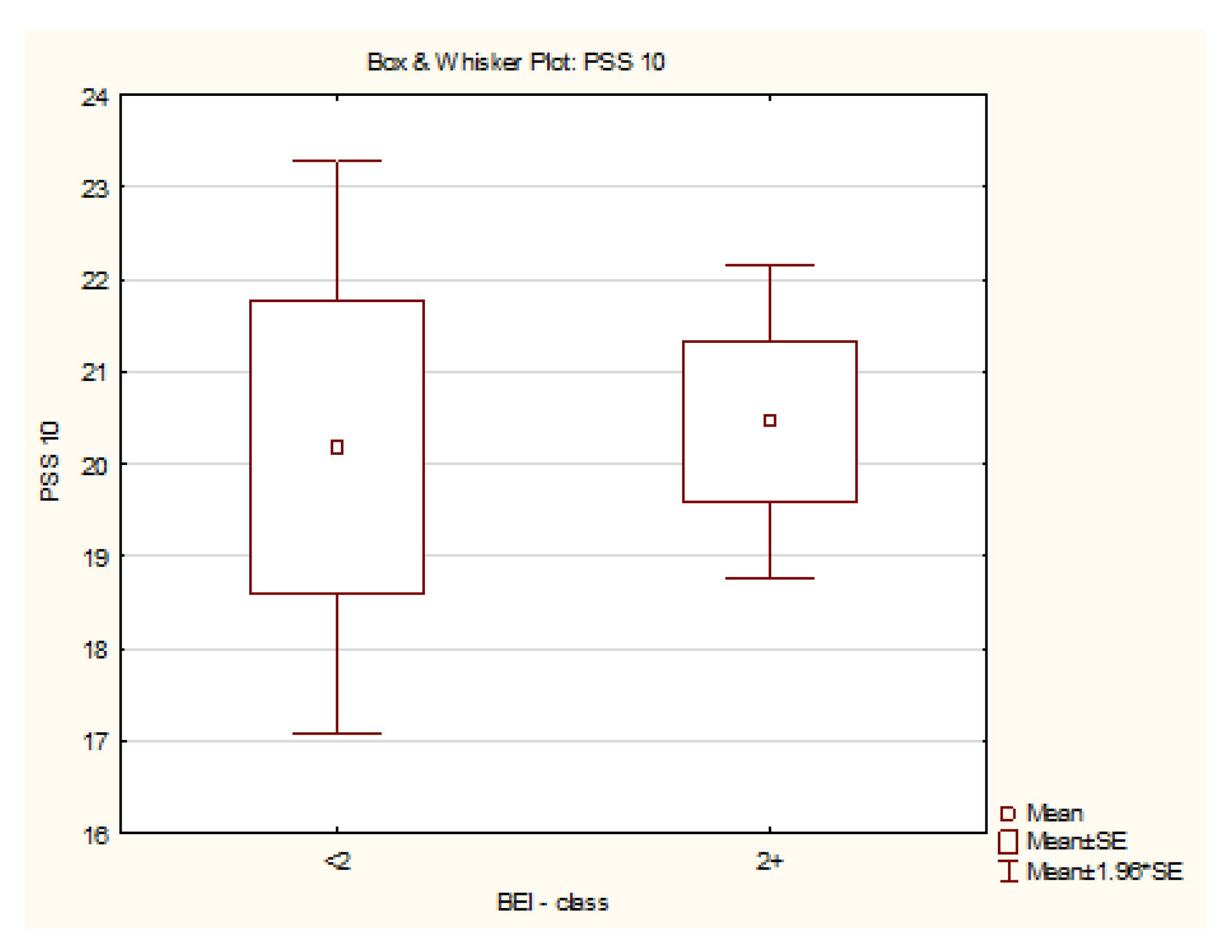
3.3. Bruxism Episodes Index (BEI) and Perceived Stress Scale–10 (PSS-10)
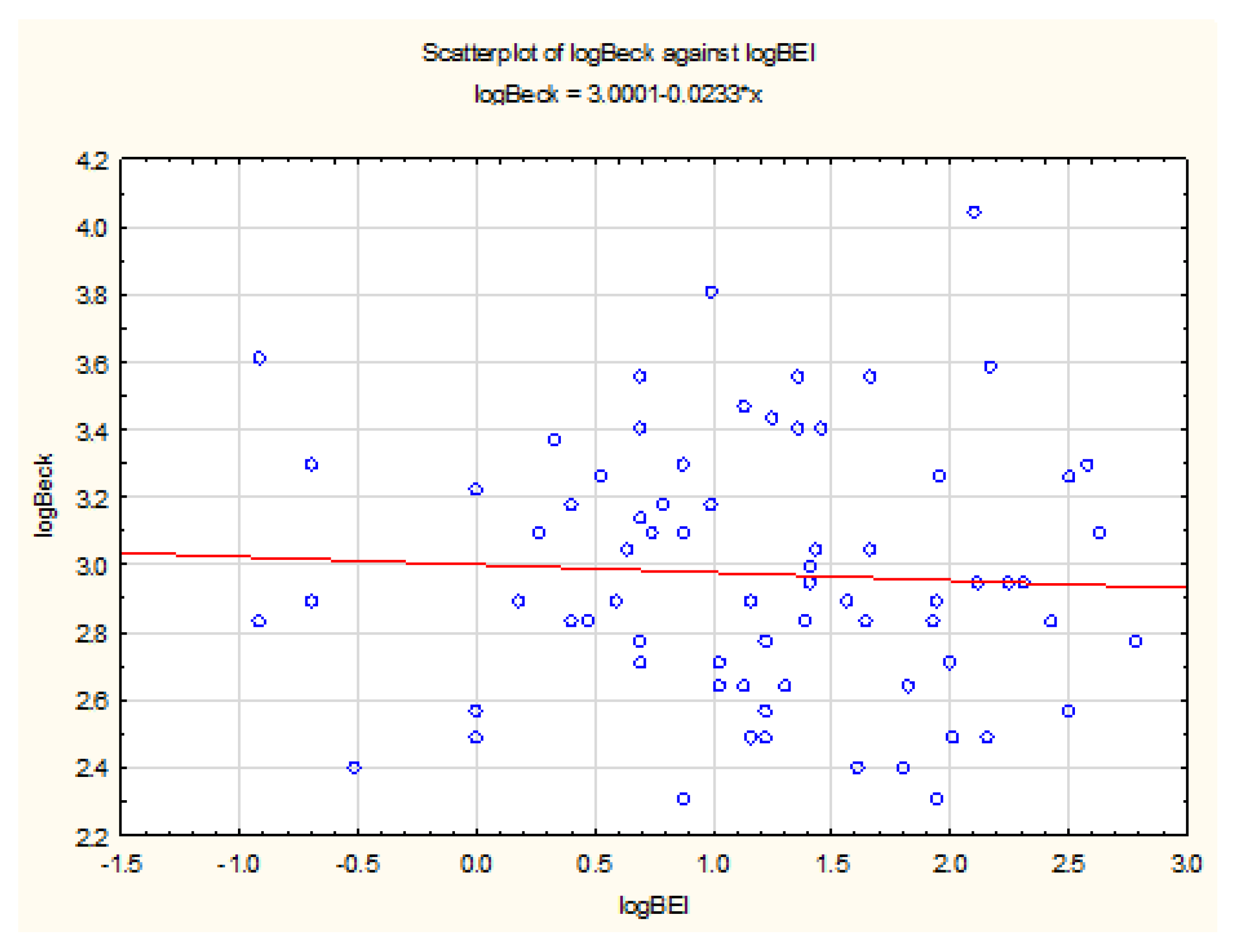
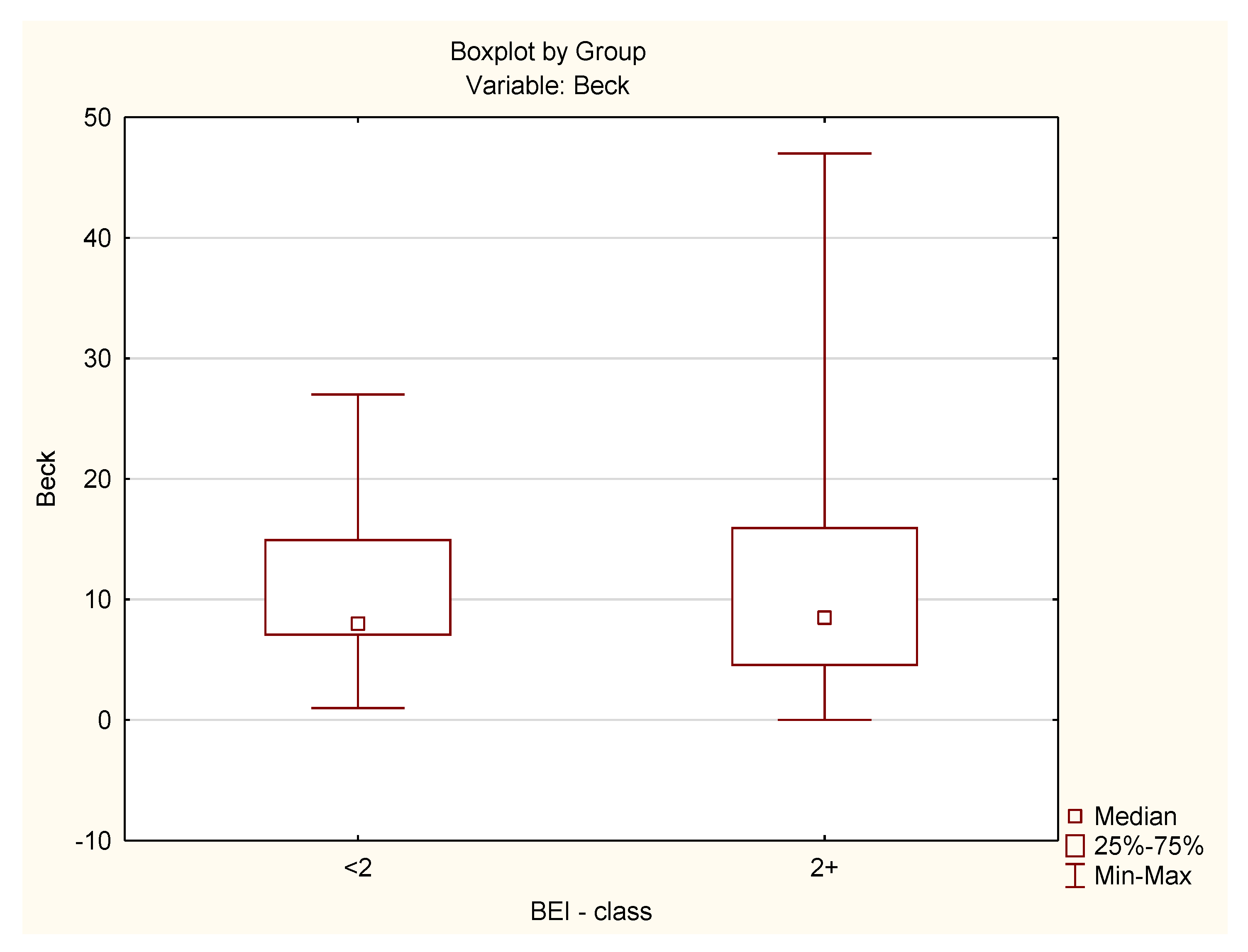
3.4. Bruxism Episodes Index (BEI) and Beck’s Depression Inventory (BDI)
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lobbezoo, F.; Ahlberg, J.; Raphael, K.G.; Wetselaar, P.; Glaros, A.G.; Kato, T.; Santiago, V.; Winocur, E.; De Laat, A.; De Leeuw, R.; et al. International consensus on the assessment of bruxism: Report of a work in progress. J. Oral Rehabil. 2018, 45, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Manfredini, D.; Serra-Negra, J.; Carboncini, F.; Lobbezoo, F. Current Concepts of Bruxism. Int. J. Prosthodont. 2017, 30, 437–438. [Google Scholar] [CrossRef] [PubMed]
- Lobbezoo, F.; Ahlberg, J.; Glaros, A.G.; Kato, T.; Koyano, K.; Lavigne, G.J.; de Leeuw, R.; Manfredini, D.; Svensson, P.; Winocur, E. Bruxism defined and graded: An international consensus. J. Oral Rehabil. 2013, 40, 2–4. [Google Scholar] [CrossRef] [PubMed]
- Macedo, C.R.; Silva, A.B.; Machado, M.A.; Saconato, H.; Prado, G.F. Occlusal splints for treating sleep bruxism (tooth grinding). Cochrane Database Syst. Rev. 2007, CD005514. [Google Scholar] [CrossRef] [PubMed]
- Castroflorio, T.; Bargellini, A.; Rossini, G.; Cugliari, G.; Rainoldi, A.; Deregibus, A. Risk factors related to sleep bruxism in children: A systematic literature review. Arch. Oral Biol. 2015, 60, 1618–1624. [Google Scholar] [CrossRef] [PubMed]
- Machado, E.; Dal-Fabbro, C.; Cunali, P.A.; Kaizer, O.B. Prevalence of sleep bruxism in children: A systematic review. Dental Press J. Orthod. 2014, 19, 54–61. [Google Scholar] [CrossRef]
- American Academy of Sleep Medicine. International Classification of Sleep Disorders, 3rd ed.; American Academy of Sleep Medicine: Westchester, NY, USA, 2014; pp. 303–311. [Google Scholar]
- World Health Organization. International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM). Available online: https://www.who.int/classifications/icd/icdonlineversions/en/ (accessed on 1 January 2017).
- De Leeuw, L.R.; Klasser, G.D. Orofacial Pain. In Guidelines for Assessment, Diagnosis, and Management, 5th ed.; Quintessence Publishing Co., Inc.: Chicago, IL, USA, 2013. [Google Scholar]
- Raphael, K.G.; Santiago, V.; Lobbezoo, F. Is bruxism a disorder or a behavior? Rethinking the international consensus on defining and grading of bruxism. J. Oral Rehabil. 2016, 43, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Winocur, E.; Uziel, N.; Lisha, T.; Goldsmith, C.; Eli, I. Self-reported bruxism—Associations with perceived stress, motivation for control, dental anxiety and gagging. J. Oral Rehabil. 2011, 38, 3–11. [Google Scholar] [CrossRef]
- Abekura, H.; Tsuboi, M.; Okura, T.; Kagawa, K.; Sadamori, S.; Akagawa, Y. Association between sleep bruxism and stress sensitivity in an experimental psychological stress task. Biomed. Res. 2011, 32, 395–399. [Google Scholar] [CrossRef]
- Gungormus, Z.; Erciyas, K. Evaluation of the relationship between anxiety and depression and bruxism. J. Int. Med. Res. 2009, 37, 547–550. [Google Scholar] [CrossRef]
- Fernandes, G.; Franco, A.L.; Siqueira, J.T.; Gonçalves, D.A.; Camparis, C.M. Sleep bruxism increases the risk for painful temporomandibular disorder, depression and non-specific physical symptoms. J. Oral Rehabil. 2012, 39, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.M. Psychometric analysis of the Ten-Item Perceived Stress Scale. Psychol. Assess. 2015, 27, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.G.; Ørnbøl, E.; Vestergaard, M.; Bech, P.; Larsen, F.B.; Lasgaard, M.; Christensen, K.S. The construct validity of the Perceived Stress Scale. J. Psychosom. Res. 2016, 84, 22–30. [Google Scholar] [CrossRef]
- González-Ramírez, M.T.; Rodríguez-Ayán, M.N.; Hernández, R.L. The perceived stress scale (PSS): Normative data and factor structure for a large-scale sample in Mexico. Span. J. Psychol. 2013, 16, E47. [Google Scholar] [CrossRef] [PubMed]
- Beck, A.T.; Ward, C.H.; Mendelson, M.; Mock, J.; Erbaugh, J. An inventory for measuring depression. Arch. Gen. Psychiatry 1961, 4, 561–571. [Google Scholar] [CrossRef]
- Richter, P.; Werner, J.; Heerlein, A.; Kraus, A.; Sauer, H. On the validity of the Beck Depression Inventory. A review. Psychopathology 1998, 31, 160–168. [Google Scholar] [CrossRef]
- Whisman, M.A.; Judd, C.M.; Whiteford, N.T.; Gelhorn, H.L. Measurement invariance of the Beck Depression Inventory-Second Edition (BDI-II) across gender, race, and ethnicity in college students. Assessment 2013, 20, 419–428. [Google Scholar] [CrossRef]
- Robinson, B.E.; Kelley, L. Concurrent validity of the Beck Depression Inventory as a measure of depression. Psychol. Rep. 1996, 79, 929–930. [Google Scholar] [CrossRef]
- Piotrowski, C. Use of the Beck Depression Inventory in clinical practice. Psychol. Rep. 1996, 79, 873–874. [Google Scholar] [CrossRef]
- Ferreira-Bacci Ado, V.; Cardoso, C.L.; Díaz-Serrano, K.V. Behavioral problems and emotional stress in children with bruxism. Braz. Dent. J. 2012, 23, 246–251. [Google Scholar] [CrossRef]
- Serra-Negra, J.M.; Paiva, S.M.; Flores-Mendoza, C.E.; Ramos-Jorge, M.L.; Pordeus, I.A. Association among stress, personality traits, and sleep bruxism in children. Pediatr. Dent. 2012, 34, e30–e34. [Google Scholar] [PubMed]
- Fluerașu, M.I.; Bocsan, I.C.; Buduru, S.; Pop, R.M.; Vesa, S.C.; Zaharia, A.; Negucioiu, M.; Iacob, S.M. The correlation between sleep bruxism, salivary cortisol, and psychological status in young, Caucasian healthy adults. Cranio 2019, 27, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, P.; Carpinelli, L.; Savarese, G. Perceived stress and bruxism in university students. BMC Res. Notes 2016, 9, 514. [Google Scholar] [CrossRef] [PubMed]
- Nakata, A.; Takahashi, M.; Ikeda, T.; Hojou, M.; Araki, S. Perceived psychosocial job stress and sleep bruxism among male and female workers. Community Dent. Oral Epidemiol. 2008, 36, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Muzalev, K.; Visscher, C.M.; Koutris, M.; Lobbezoo, F. Long-term variability of sleep bruxism and psychological stress in patients with jaw-muscle pain: Report of two longitudinal clinical cases. J. Oral Rehabil. 2018, 45, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Pierce, C.J.; Chrisman, K.; Bennett, M.E.; Close, J.M. Stress, anticipatory stress, and psychologic measures related to sleep bruxism. J. Orofac. Pain 1995, 9, 51–56. [Google Scholar]
- Ohlmann, B.; Bömicke, W.; Habibi, Y.; Rammelsberg, P.; Schmitter, M. Are there associations between sleep bruxism, chronic stress, and sleep quality? J. Dent. 2018, 74, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Uca, A.U.; Uğuz, F.; Kozak, H.H.; Gümüş, H.; Aksoy, F.; Seyithanoğlu, A.; Kurt, H.G. Antidepressant-Induced Sleep Bruxism: Prevalence, Incidence, and Related Factors. Clin. Neuropharmacol. 2015, 38, 227–230. [Google Scholar] [CrossRef]
- Fernandes, G.; Siqueira, J.T.; Godoi Gonçalves, D.A.; Camparis, C.M. Association between painful temporomandibular disorders, sleep bruxism and tinnitus. Braz. Oral Res. 2014, 28, 1–7. [Google Scholar] [CrossRef]
- Nazeri, M.; Ghahrechahi, H.R.; Pourzare, A.; Abareghi, F.; Samiee-Rad, S.; Shabani, M.; Arjmand, S.; Abazarpour, R. Role of anxiety and depression in association with migraine and myofascial pain temporomandibular disorder. Indian J. Dent. Res. 2018, 29, 583–587. [Google Scholar] [CrossRef]
- Reiter, S.; Emodi-Perlman, A.; Goldsmith, C.; Friedman-Rubin, P.; Winocur, E. Comorbidity between depression and anxiety in patients with temporomandibular disorders according to the research diagnostic criteria for temporomandibular disorders. J. Oral Facial Pain Headache. 2015, 29, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Dıraçoǧlu, D.; Yıldırım, N.K.; Saral, İ.; Özkan, M.; Karan, A.; Özkan, S.; Aksoy, C. Temporomandibular dysfunction and risk factors for anxiety and depression. J. Back Musculoskelet Rehabil. 2016, 29, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Bertoli, E.; de Leeuw, R. Prevalence of Suicidal Ideation, Depression, and Anxiety in Chronic Temporomandibular Disorder Patients. J. Oral Facial Pain Headache. 2016, 30, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Wieckiewicz, M.; Zietek, M.; Smardz, J.; Zenczak-Wieckiewicz, D.; Grychowska, N. Mental Status as a Common Factor for Masticatory Muscle Pain: A Systematic Review. Front. Psychol. 2017, 8, 646. [Google Scholar] [CrossRef] [PubMed]

| Group | Number | Mean | Median | Minimum | Maximum | Standard Deviation |
|---|---|---|---|---|---|---|
| studied group | 57 * | 20.46 | 19.00 | 9 | 34 | 6.519 |
| control group | 17 * | 20.18 | 22.00 | 7 | 30 | 6.541 |
| Group | Number | Mean | Median | Minimum | Maximum | Standard Deviation |
|---|---|---|---|---|---|---|
| studied group | 56 * | 11.11 | 8.50 | 0 | 47 | 9.230 |
| control group | 17 * | 10.71 | 8.00 | 1 | 27 | 6.734 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smardz, J.; Martynowicz, H.; Wojakowska, A.; Michalek-Zrabkowska, M.; Mazur, G.; Wieckiewicz, M. Correlation between Sleep Bruxism, Stress, and Depression—A Polysomnographic Study. J. Clin. Med. 2019, 8, 1344. https://doi.org/10.3390/jcm8091344
Smardz J, Martynowicz H, Wojakowska A, Michalek-Zrabkowska M, Mazur G, Wieckiewicz M. Correlation between Sleep Bruxism, Stress, and Depression—A Polysomnographic Study. Journal of Clinical Medicine. 2019; 8(9):1344. https://doi.org/10.3390/jcm8091344
Chicago/Turabian StyleSmardz, Joanna, Helena Martynowicz, Anna Wojakowska, Monika Michalek-Zrabkowska, Grzegorz Mazur, and Mieszko Wieckiewicz. 2019. "Correlation between Sleep Bruxism, Stress, and Depression—A Polysomnographic Study" Journal of Clinical Medicine 8, no. 9: 1344. https://doi.org/10.3390/jcm8091344
APA StyleSmardz, J., Martynowicz, H., Wojakowska, A., Michalek-Zrabkowska, M., Mazur, G., & Wieckiewicz, M. (2019). Correlation between Sleep Bruxism, Stress, and Depression—A Polysomnographic Study. Journal of Clinical Medicine, 8(9), 1344. https://doi.org/10.3390/jcm8091344







