The Combination of Patient-Reported Clinical Symptoms and an Endoscopic Score Correlates Well with Health-Related Quality of Life in Patients with Ulcerative Colitis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Symptoms and Patient-Reported Outcomes
2.3. Endoscopic Inflammation
2.4. Disease Activity According to the Symptoms and MES
2.5. Statistical Analysis
3. Results
3.1. Correlations of Symptoms, MES, and PROs
3.2. Correlations of SIBDQ and Various PROs
3.3. Association between Disease Activity and PROs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Loftus, E.V., Jr.; Davis, K.L.; Wang, C.C.; Dastani, H.; Luo, A. Treatment patterns, complications, and disease relapse in a real-world population of patients with moderate-to-severe ulcerative colitis initiating immunomodulator therapy. Inflamm. Bowel Dis. 2014, 20, 1361–1367. [Google Scholar] [CrossRef] [PubMed]
- Samuel, S.; Ingle, S.B.; Dhillon, S.; Yadav, S.; Harmsen, W.S.; Zinsmeister, A.R.; Tremaine, W.J.; Sandborn, W.J.; Loftus, E.V., Jr. Cumulative incidence and risk factors for hospitalization and surgery in a population-based cohort of ulcerative colitis. Inflamm. Bowel Dis. 2013, 19, 1858–1866. [Google Scholar] [CrossRef] [PubMed]
- Williet, N.; Sandborn, W.J.; Peyrin-Biroulet, L. Patient-reported outcomes as primary end points in clinical trials of inflammatory bowel disease. Clin. Gastroenterol. Hepatol. 2014, 12, 1246–1256. [Google Scholar] [CrossRef]
- Halpin, S.J.; Ford, A.C. Prevalence of symptoms meeting criteria for irritable bowel syndrome in inflammatory bowel disease: Systematic review and meta-analysis. Am. J. Gastroenterol. 2012, 107, 1474–1482. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research. Ulcerative Colitis Clinical Trial Endpoints Guidance for Industry. Available online: https://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM515143.pdf (accessed on 30 July 2018).
- Peyrin-Biroulet, L.; Sandborn, W.; Sands, B.E.; Reinisch, W.; Bemelman, W.; Bryant, R.V.; D’Haens, G.; Dotan, I.; Dubinsky, M.; Feagan, B.; et al. Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE): Determining Therapeutic Goals for Treat-to-Target. Am. J. Gastroenterol. 2015, 110, 1324–1338. [Google Scholar] [CrossRef]
- Shivashankar, R.; Tremaine, W.J.; Harmsen, W.S.; Loftus, E.V., Jr. Incidence and Prevalence of Crohn’s Disease and Ulcerative Colitis in Olmsted County, Minnesota from 1970 through 2010. Clin. Gastroenterol. Hepatol. 2017, 15, 857–863. [Google Scholar] [CrossRef] [PubMed]
- Loftus, C.G.; Loftus, E.V., Jr.; Harmsen, W.S.; Zinsmeister, A.R.; Tremaine, W.J.; Melton, L.J.; Sandborn, W.J. Update on the incidence and prevalence of Crohn’s disease and ulcerative colitis in Olmsted County, Minnesota, 1940-2000. Inflamm. Bowel Dis. 2007, 13, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Loftus, E.V., Jr.; Silverstein, M.D.; Sandborn, W.J.; Tremaine, W.J.; Harmsen, W.S.; Zinsmeister, A.R. Ulcerative colitis in Olmsted County, Minnesota, 1940–1993: Incidence, prevalence, and survival. Gut 2000, 46, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Jharap, B.; Sandborn, W.J.; Reinisch, W.; D’Haens, G.; Robinson, A.M.; Wang, W.; Huang, B.; Lazar, A.; Thakkar, R.B.; Colombel, J.F.; et al. Randomised clinical study: Discrepancies between patient-reported outcomes and endoscopic appearance in moderate to severe ulcerative colitis. Aliment. Pharmacol. Ther. 2015, 42, 1082–1092. [Google Scholar] [CrossRef] [PubMed]
- Irvine, E.J.; Zhou, Q.; Thompson, A.K. The Short Inflammatory Bowel Disease Questionnaire: A quality of life instrument for community physicians managing inflammatory bowel disease. CCRPT Investigators. Canadian Crohn’s Relapse Prevention Trial. Am. J. Gastroenterol. 1996, 91, 1571–1578. [Google Scholar] [PubMed]
- Kappelman, M.D.; Long, M.D.; Martin, C.; DeWalt, D.A.; Kinneer, P.M.; Chen, W.; Lewis, J.D.; Sandler, R.S. Evaluation of the patient-reported outcomes measurement information system in a large cohort of patients with inflammatory bowel diseases. Clin. Gastroenterol. Hepatol. 2014, 12, 1315–1323. [Google Scholar] [CrossRef] [PubMed]
- Stark, R.G.; Reitmeir, P.; Leidl, R.; Konig, H.H. Validity, reliability, and responsiveness of the EQ-5D in inflammatory bowel disease in Germany. Inflamm. Bowel Dis. 2010, 16, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Travis, S.; Feagan, B.G.; Peyrin-Biroulet, L.; Panaccione, R.; Danese, S.; Lazar, A.; Robinson, A.M.; Petersson, J.; Pappalardo, B.L.; Bereswill, M.; et al. Effect of Adalimumab on Clinical Outcomes and Health-related Quality of Life Among Patients with Ulcerative Colitis in a Clinical Practice Setting: Results from InspirADA. J. Crohn’s Colitis 2017, 11, 1317–1325. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, A.; Macklin, E.A.; Korzenik, J.R.; Sands, B.E. Validation of the functional assessment of chronic illness therapy-fatigue (FACIT-F) in patients with inflammatory bowel disease. Aliment. Pharmacol. Ther. 2011, 34, 1328–1336. [Google Scholar] [CrossRef] [PubMed]
- Romberg-Camps, M.J.; Bol, Y.; Dagnelie, P.C.; Hesselink-van de Kruijs, M.A.; Kester, A.D.; Engels, L.G.; van Deursen, C.; Hameeteman, W.H.; Pierik, M.; Wolters, F.; et al. Fatigue and health-related quality of life in inflammatory bowel disease: Results from a population-based study in the Netherlands: The IBD-South Limburg cohort. Inflamm. Bowel Dis. 2010, 16, 2137–2147. [Google Scholar] [CrossRef] [PubMed]
- Hinkle, D.E.; Wiersma, W.; Jurs, S.G. Applied Statistics for the Behavioral Sciences, 5th ed.; Houghton Mifflin: Boston, MA, USA, 2003. [Google Scholar]
- Theede, K.; Kiszka-Kanowitz, M.; Nordgaard-Lassen, I.; Mertz Nielsen, A. The impact of endoscopic inflammation and mucosal healing on health-related quality of life in ulcerative colitis patients. J. Crohn’s Colitis 2015, 9, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Gracie, D.J.; Williams, C.J.; Sood, R.; Mumtaz, S.; Bholah, M.H.; Hamlin, P.J.; Ford, A.C. Poor correlation between clinical disease activity and mucosal inflammation, and the role of psychological comorbidity, in inflammatory bowel disease. Am. J. Gastroenterol. 2016, 111, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Szende, A.; Janssen, B.; Cabases, J. Self-Reported Population Health: An International Perspective Based on EQ-5D; Springer: Dordrecht, The Netherlands; Heidelberg, Germany; New York, NY, USA; London, UK, 2014. [Google Scholar]
- Jowett, S.L.; Seal, C.J.; Barton, J.R.; Welfare, M.R. The short inflammatory bowel disease questionnaire is reliable and responsive to clinically important change in ulcerative colitis. Am. J. Gastroenterol. 2001, 96, 2921–2928. [Google Scholar] [CrossRef] [PubMed]

| Variables | N = 90 |
|---|---|
| Median age, years (IQR) | 45 (32–56) |
| Males, n (%) | 41 (46%) |
| White race, n (%) | 86 (96%) |
| Non-Hispanic ethnicity, n (%) | 90 (100%) |
| Mean body mass index, kg/m2 (SD) | 27.9 (6.8) |
| Smoking status, n (%) | |
| Never-smoker | 60 (67%) |
| Ex-smoker | 28 (31%) |
| Current smoker | 2 (2%) |
| Other comorbid diseases, n (%) | |
| Diabetes mellitus | 4 (4%) |
| Hypertension | 5 (6%) |
| None | 81 (90%) |
| Median UC duration, years (IQR) | 9.9 (4.2–17.2) |
| UC location, n (%) | |
| Proctitis/proctosigmoiditis | 10 (11%) |
| Left-sided colitis | 25 (28%) |
| Extensive colitis | 55 (61%) |
| UC current medication, n (%) | |
| 5-Aminosalicylates | 38 (42%) |
| Immunomodulators | 25 (28%) |
| Systemic corticosteroids | 16 (18%) |
| Biologic agents | 40 (44%) |
| None | 10 (11%) |
| ClinPRO2 | |
| Rectal bleeding, n (%) | |
| Score 0: no blood seen | 46 (51%) |
| Score 1: streaks of blood less than half the time | 20 (22%) |
| Score 2: obvious blood most of the time | 17 (19%) |
| Score 3: blood passes alone | 7 (8%) |
| Stool frequency, n (%) | |
| Score 0: normal | 27 (30%) |
| Score 1: 1–2 per day more than normal | 17 (19%) |
| Score 2: 3–4 per day more than normal | 14 (16%) |
| Score 3: ≥5 per day more than normal | 32 (35%) |
| Mayo endoscopic subscore, n (%) | |
| Score 0: normal | 10 (11%) |
| Score 1: erythema, decreased vascular pattern | 26 (29%) |
| Score 2: marked erythema, absent vascular pattern, friability, erosion | 18 (20%) |
| Score 3: spontaneous bleedings, ulceration | 36 (40%) |
| Median SIBDQ (IQR) | 48.5 (34–59) |
| Median EQ5D3L-TTO (IQR) | 0.827 (0.778–1) |
| Median WPAI (IQR) | |
| % overall work impairment | 36 (0–80) |
| Median FACIT-F (IQR) | 33.1 (13.1) |
| Median HADS-Depression (IQR) | 4 (1–7) |
| Median HADS-Anxiety (IQR) | 5 (3–9) |
| Outcomes | SIBDQ | EQ5D3L | WPAI | FACIT-F | HADS Depression | HADS Anxiety |
|---|---|---|---|---|---|---|
| MES | −0.47 * | −0.31 * | 0.44 * | −0.34 * | 0.22 † | 0.22 † |
| ClinPRO2 | −0.73 * | −0.53 * | 0.63 * | −0.62 * | 0.50 * | 0.30 * |
| Combination of ClinPRO2 and MES | −0.70 * | −0.51 * | 0.62 * | −0.58 * | 0.45* | 0.30 * |
| SIBDQ | - | 0.67 * | −0.80 * | 0.86 * | −0.75 * | −0.53 * |
| EQ5D3L | - | - | −0.48 * | 0.69 * | −0.67 * | −0.30 * |
| WPAI | - | - | - | −0.68 * | 0.51 * | 0.37 * |
| FACIT-F | - | - | - | - | −0.84 * | −0.44 * |
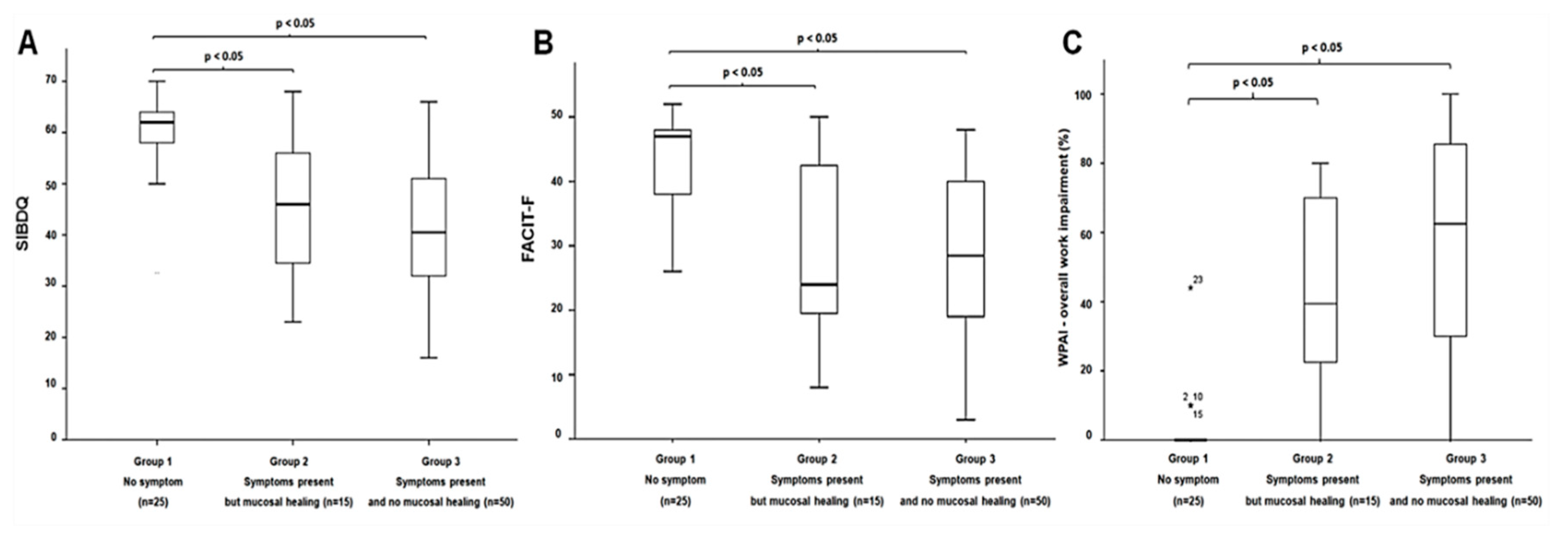
| UC Patients Categorized by ClinPRO2 and Mucosal Healing | p-Value | |||
|---|---|---|---|---|
| No Symptoms (n = 25) | Symptoms Present but MH (n = 15) | Symptoms Present and no MH (n = 50) | ||
| Median age, years (IQR) | 45 (32–63) | 49 (35–64) | 46 (31–54) | 0.38 |
| Male, n (%) | 9 (36%) | 7 (47%) | 25 (50%) | 0.24 |
| Extensive UC, n (%) | 14 (56%) | 11 (73%) | 30 (60%) | 0.78 |
| Current medication, n (%) | ||||
| 5-ASA use | 11 (44%) | 8 (53%) | 19 (38%) | 0.56 |
| Systemic corticosteroids | 1 (4%) | 2 (13%) | 13 (26%) | 0.06 |
| Immunosuppressive drugs | 7 (28%) | 6 (40%) | 12 (24%) | 0.48 |
| Biologic agents | 10 (40%) | 6 (40%) | 24 (48%) | 0.75 |
| Patient-reported outcomes, median (IQR) | ||||
| SIBDQ † | 62 (58–64) | 46 (33–56) * | 40.5 (31.8–51) * | < 0.01 |
| EQ5D3L † | 1 (0.827–1) | 0.827 (0.761–1) * | 0.816 (0.768–0.856) * | < 0.01 |
| WPAI ‡ | 0 (0–0) | 39.4 (16.9–71.7) * | 62.5 (28.0–85.8) * | < 0.01 |
| FACIT-F † | 47 (37–48.5) | 24 (19–45) * | 28.5 (18.8–40.3) * | < 0.01 |
| HADS-depression ‡ | 2 (1–3) | 6 (2–10) * | 4 (2–7.3) * | < 0.01 |
| HADS-anxiety ‡ | 4 (1–5) | 8 (4–10) * | 7 (4–10) * | < 0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aniwan, S.; Bruining, D.H.; Park, S.H.; Al-Bawardy, B.; Kane, S.V.; Coelho Prabhu, N.; Kisiel, J.B.; Raffals, L.E.; Papadakis, K.A.; Pardi, D.S.; et al. The Combination of Patient-Reported Clinical Symptoms and an Endoscopic Score Correlates Well with Health-Related Quality of Life in Patients with Ulcerative Colitis. J. Clin. Med. 2019, 8, 1171. https://doi.org/10.3390/jcm8081171
Aniwan S, Bruining DH, Park SH, Al-Bawardy B, Kane SV, Coelho Prabhu N, Kisiel JB, Raffals LE, Papadakis KA, Pardi DS, et al. The Combination of Patient-Reported Clinical Symptoms and an Endoscopic Score Correlates Well with Health-Related Quality of Life in Patients with Ulcerative Colitis. Journal of Clinical Medicine. 2019; 8(8):1171. https://doi.org/10.3390/jcm8081171
Chicago/Turabian StyleAniwan, Satimai, David H. Bruining, Sang Hyoung Park, Badr Al-Bawardy, Sunanda V. Kane, Nayantara Coelho Prabhu, John B. Kisiel, Laura E. Raffals, Konstantinos A. Papadakis, Darrell S. Pardi, and et al. 2019. "The Combination of Patient-Reported Clinical Symptoms and an Endoscopic Score Correlates Well with Health-Related Quality of Life in Patients with Ulcerative Colitis" Journal of Clinical Medicine 8, no. 8: 1171. https://doi.org/10.3390/jcm8081171




