Nutritional Risk Screening and Assessment
Abstract
1. Introduction
2. Screening
3. Assessment
3.1. Anthropometric Measurements
3.1.1. Body Weight and Body Mass Index
3.1.2. Skinfold Measurements
- –
- Biceps skinfold (front side of the middle upper arm);
- –
- Triceps skinfold (back side of the middle upper arm);
- –
- Subscapular skinfold (under the lowest point of the shoulder blade); and
- –
- Suprailiac skinfold (above the upper bone of the hip).
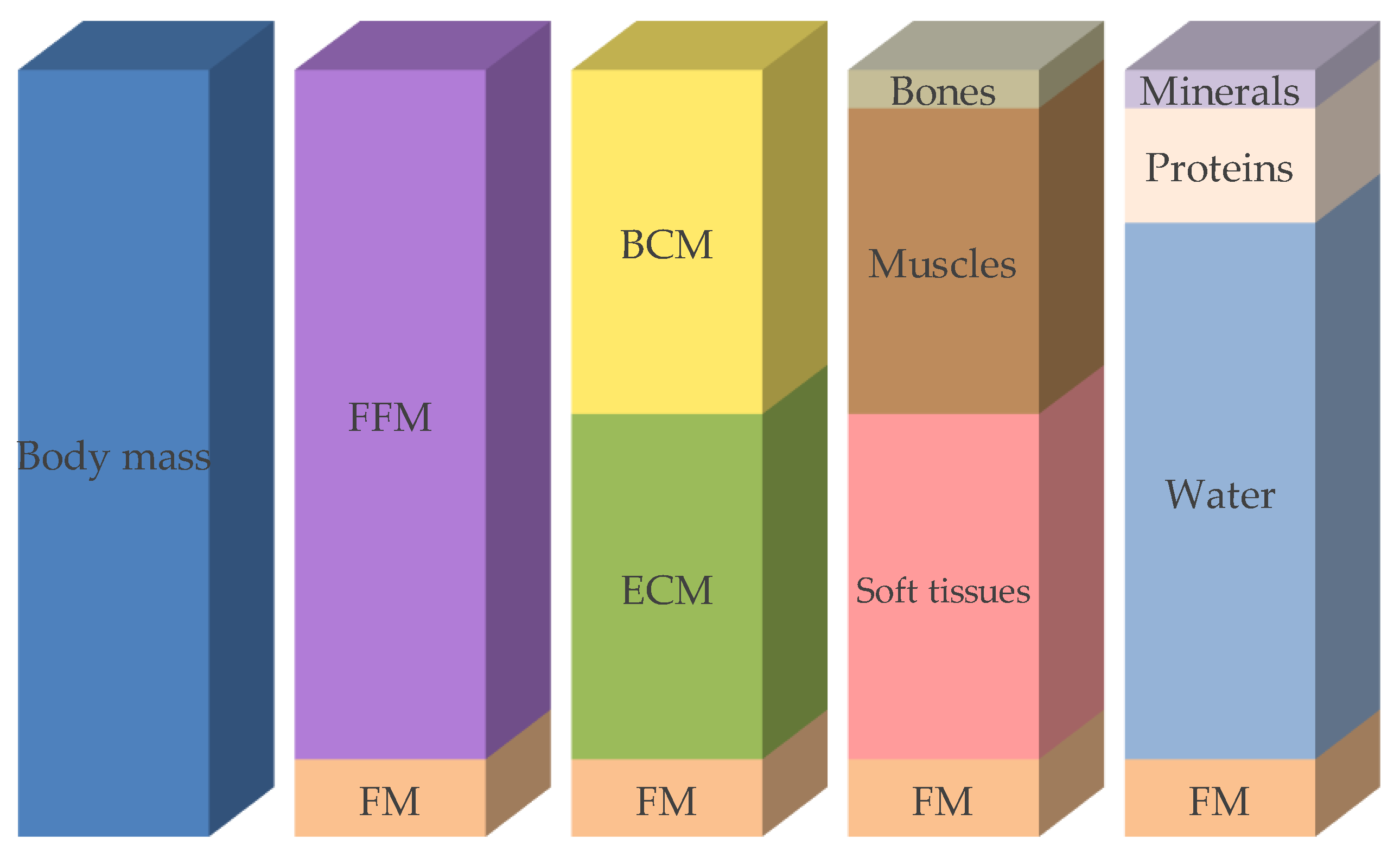
3.1.3. Body Composition
3.1.4. Bioelectrical Impedance Analysis (BIA)
3.1.5. Creatinine Height Index (CHI)
3.1.6. Dual Energy X-ray Absorptiometry (DXA)
3.1.7. Magnetic Resonance Tomography (MRT) and Computed Tomography (CT)
3.1.8. Further Methods Used to Measure Body Composition
3.2. Biochemical Analysis
3.3. Clinical Evaluation
3.3.1. Patient Clinical History
3.3.2. Physical Examination
3.3.3. Physical Function
3.3.4. Medication
3.4. Dietary History, Current Dietary Intake, and Innovative Dietary Assessment Methods
- –
- Manual dietary assessment—The user inserts all required data (e.g., portion size estimation, type of food) on a web page, smartphone app, etc. [50]. This method replaces the paper-based methods of dietary assessment into an electronic form by the use of pictures, video, text, or voice without the inclusion of automatic features.
- –
- Dietitian-supported assessment—The user takes photos of the food and sends them to the dietitian. These data are then analyzed by nutrition experts who use standardized methods (e.g., nutritional software) to estimate the corresponding amount of nutrients [51]. No automation features are usually incorporated.
- –
- Wearable devices monitoring food intake—Devices that directly measure eating behavior [52], such as detection systems which identify eating gestures (ear-based chewing and swallowing) in order to complement self-reporting of nutrient intake.
- –
- Computer-aided assessment—this includes:
- (i)
- Systems that incorporate some degree of automation. These either use bar-code readers in order to automatically recognize packaged food labels [50], or utilize smartphone applications that integrate the automatic recognition of food items. In this case, the user takes photos of the food and the system recognizes the type of food. Typically, in this situation the user needs to manually insert or select the volume/portion of the food items in order for the system to be able to translate the information into macronutrients and energy [53].
- (ii)
- Systems that are completely based on artificial intelligence. In a typical scenario, the user takes photo(s) of the food and then the system automatically and in real-time identifies the different food items (identification), recognizes the type of each of them (labeling), and creates a 3D model of each of them (3D reconstruction) [54,55,56,57,58]. Supported by food composition databases, food images are translated into nutrient values such as grams of macronutrients or calories [54,56].
3.5. Quality of Life
4. Conclusions and Outlook
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Screening | ||
| A | Has food intake declined over the past 3 months due to loss of appetite, digestive problems, chewing or swallowing difficulties? | 0 = severe decrease in food intake |
| 1 = moderate decrease in food intake | ||
| 2 = no decrease in food intake | ||
| B | Weight loss during the past 3 months | 0 = weight loss greater than 3 kg |
| 1 = does not know | ||
| 2 = weight loss between 1 and 3 kg | ||
| 3 = no weight loss | ||
| C | Mobility | 0 = bedridden or chair bound |
| 1 = able to get out of bed/chair but does not go out | ||
| 2 = goes out | ||
| D | Has suffered psychological stress or acute disease in the past 3 months? | 0 = yes |
| 2 = no | ||
| E | Neuropsychological problems | 0 = severe dementia or depression |
| 1 = mild dementia | ||
| 2 = no psychological problems | ||
| F1 | Body mass index (BMI) | 0 = BMI less than 19 |
| 1 = BMI 19 to less than 21 | ||
| 2 = BMI 21 to less than 23 | ||
| 3 = BMI 23 or greater | ||
| Screening Score(subtotal max. 14 points) | ||
| 12–14 points | Normal nutritional status | |
| 8–11 points | At risk of malnutrition | |
| 0–7 points | Malnourished | |
| For a more in-depth assessment, continue with questions G-R | ||
| Assessment | ||
| G | Lives independently (not in nursing home or hospital) | 0 = yes |
| 1 = no | ||
| H | Takes more than 3 prescription drugs per day | 0 = yes |
| 1 = no | ||
| I | Pressure sores or skin ulcers | 0 = yes |
| 1 = no | ||
| J | How many full meals does the patient eat daily? | 0 = 1 meal |
| 1 = 2 meals | ||
| 2 = 3 meals | ||
| K | Selected consumption markers for protein intake | 0.0 = if 0 or 1 yes |
| 0.5 = if 2 yes | ||
| 1.0 = if 3 yes | ||
| ● Meat, fish or poultry every day | Yes/No | |
| ● ≥1 serving of dairy products (milk, cheese, yoghurt) per day | Yes/No | |
| ● ≥2 servings of legumes or eggs per week | Yes/No | |
| L | Consumes ≥2 servings of fruit or vegetables per day? | 0 = yes |
| 1 = no | ||
| M | How much fluid (water, juice, coffee, tea, milk...) is consumed per day? | 0.0 = less than 3 cups |
| 0.5 = 3 to 5 cups | ||
| 1.0 = more than 5 cups | ||
| N | Mode of feeding | 0 = unable to eat without assistance |
| 1 = self-fed with some difficulty | ||
| 2 = self-fed without any problem | ||
| O | Self view of nutritional status | 0 = views self as being malnourished |
| 1 = is uncertain of nutritional status | ||
| 2 = views self as having no nutritional problem | ||
| P | In comparison with other people of the same age, how does the patient consider his/her health status? | 0.0 = not as good |
| 0.5 = does not know | ||
| 1.0 = as good | ||
| 2.0 = better | ||
| Q | Mid-arm circumference (MAC) in cm | 0.0 = MAC less than 21 |
| 0.5 = MAC 21 to 22 | ||
| 1.0 = MAC greater than 22 | ||
| R | Calf circumference (CC) in cm | 0 = CC less than 31 |
| 1 = CC 31 or greater | ||
| Malnutrition Indicator Score | ||
| 24–30 points | Normal nutritional status | |
| 17–23.5 points | At risk of malnutrition | |
| <17 points | Malnourished | |
References
- Sorensen, J.; Kondrup, J.; Prokopowicz, J.; Schiesser, M.; Krahenbuhl, L.; Meier, R.; Liberda, M.; EuroOOPS Study Group. EuroOOPS: An international, multicentre study to implement nutritional risk screening and evaluate clinical outcome. Clin. Nutr. 2008, 27, 340–349. [Google Scholar] [CrossRef]
- Dupertuis, Y.M.; Kossovsky, M.P.; Kyle, U.G.; Raguso, C.A.; Genton, L.; Pichard, C. Food intake in 1707 hospitalised patients: A prospective comprehensive hospital survey. Clin. Nutr. 2003, 22, 115–123. [Google Scholar] [CrossRef]
- Schwegler, I.; von Holzen, A.; Gutzwiller, J.P.; Schlumpf, R.; Muhlebach, S.; Stanga, Z. Nutritional risk is a clinical predictor of postoperative mortality and morbidity in surgery for colorectal cancer. Br. J. Surg. 2010, 97, 92–97. [Google Scholar] [CrossRef]
- Sun, Z.; Kong, X.J.; Jing, X.; Deng, R.J.; Tian, Z.B. Nutritional Risk Screening 2002 as a predictor of postoperative outcomes in patients undergoing abdominal surgery: A systematic review and meta-analysis of prospective cohort studies. PLoS ONE 2015, 10, e0132857. [Google Scholar] [CrossRef]
- Imoberdorf, R.; Meier, R.; Krebs, P.; Hangartner, P.J.; Hess, B.; Staubli, M.; Wegmann, D.; Rühlin, M.; Ballmer, P.E. Prevalence of undernutrition on admission to Swiss hospitals. Clin. Nutr. 2010, 29, 38–41. [Google Scholar] [CrossRef]
- Studley, H.O. Percentage of weight loss: A basic indicator of surgical risk in patients with chronic peptic ulcer. 1936. Nutr Hosp. 2001, 16, 141–143. [Google Scholar]
- Meguid, M.M.; Debonis, D.; Meguid, V.; Hill, L.R.; Terz, J.J. Complications of abdominal operations for malignant disease. Am. J. Surg. 1988, 156, 341–345. [Google Scholar] [CrossRef]
- Pikul, J.; Sharpe, M.D.; Lowndes, R.; Ghent, C.N. Degree of preoperative malnutrition is predictive of postoperative morbidity and mortality in liver transplant recipients. Transplantation 1994, 57, 469–472. [Google Scholar] [CrossRef]
- Soeters, P.B.; Schols, A.M. Advances in understanding and assessing malnutrition. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 487–494. [Google Scholar] [CrossRef]
- Guo, W.; Ou, G.; Li, X.; Huang, J.; Liu, J.; Wei, H. Screening of the nutritional risk of patients with gastric carcinoma before operation by NRS 2002 and its relationship with postoperative results. J. Gastroenterol. Hepatol. 2010, 25, 800–803. [Google Scholar] [CrossRef]
- Lieffers, J.R.; Bathe, O.F.; Fassbender, K.; Winget, M.; Baracos, V.E. Sarcopenia is associated with postoperative infection and delayed recovery from colorectal cancer resection surgery. Br. J. Cancer 2012, 107, 931–936. [Google Scholar] [CrossRef]
- Schiesser, M.; Kirchhoff, P.; Muller, M.K.; Schafer, M.; Clavien, P.A. The correlation of nutrition risk index, nutrition risk score, and bioimpedance analysis with postoperative complications in patients undergoing gastrointestinal surgery. Surgery 2009, 145, 519–526. [Google Scholar] [CrossRef]
- Schiesser, M.; Muller, S.; Kirchhoff, P.; Breitenstein, S.; Schafer, M.; Clavien, P.A. Assessment of a novel screening score for nutritional risk in predicting complications in gastro-intestinal surgery. Clin. Nutr. 2008, 27, 565–570. [Google Scholar] [CrossRef]
- Sungurtekin, H.; Sungurtekin, U.; Balci, C.; Zencir, M.; Erdem, E. The influence of nutritional status on complications after major intraabdominal surgery. J. Am. Coll. Nutr. 2004, 23, 227–232. [Google Scholar] [CrossRef]
- Donini, L.M.; Savina, C.; Rosano, A.; Cannella, C. Systematic review of nutritional status evaluation and screening tools in the elderly. J. Nutr. Health. Aging 2007, 11, 421–432. [Google Scholar]
- Foley, N.C.; Salter, K.L.; Robertson, J.; Teasell, R.W.; Woodbury, M.G. Which reported estimate of the prevalence of malnutrition after stroke is valid? Stroke 2009, 40, E66–E74. [Google Scholar] [CrossRef]
- Bauer, J.M.; Kaiser, M.J.; Sieber, C.C. Evaluation of nutritional status in older persons: Nutritional screening and assessment. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 8–13. [Google Scholar] [CrossRef]
- Schindler, K.; Pernicka, E.; Laviano, A.; Howard, P.; Schutz, T.; Bauer, P.; Grecu, I.; Jonkers, C.; Kondrup, J.; Ljungqvist, O.; et al. How nutritional risk is assessed and managed in European hospitals: A survey of 21,007 patients findings from the 2007–2008 cross-sectional nutritionDay survey. Clin. Nutr. 2010, 29, 552–559. [Google Scholar] [CrossRef]
- Van Bokhorst-de van der Schueren, M.A.E.; Guaitoli, P.R.; Jansma, E.P.; de Vet, H.C.W. Nutrition screening tools: Does one size fit all? A systematic review of screening tools for the hospital setting. Clin. Nutr. 2014, 33, 39–58. [Google Scholar] [CrossRef]
- Kondrup, J.; Allison, S.P.; Elia, M.; Vellas, B.; Plauth, M. ESPEN guidelines for nutrition screening 2002. Clin. Nutr. 2003, 22, 415–421. [Google Scholar] [CrossRef]
- Kondrup, J.; Rasmussen, H.H.; Hamberg, O.; Stanga, Z. Nutritional risk screening (NRS 2002): A new method based on an analysis of controlled clinical trials. Clin. Nutr. 2003, 22, 321–336. [Google Scholar] [CrossRef]
- Schuetz, P.; Fehr, R.; Baechli, V.; Geiser, M.; Gomes, F.; Kutz, A.; Tribolet, P.; Bregenzer, T.; Braun, N.; Hoess, C.; et al. Individualized nutritional support in medical inpatients at nutritional risk: A randomized clinical trial. Lancet 2019, 393, 2312–2321. [Google Scholar] [CrossRef]
- Weekes, C.E.; Elia, M.; Emery, P.W. The development, validation and reliability of a nutrition screening tool based on the recommendations of the British, Association for Parenteral and Enteral Nutrition (BAPEN). Clin. Nutr. 2004, 23, 1104–1112. [Google Scholar]
- Anthony, P.S. Nutrition screening tools for hospitalized patients. Nutr. Clin. Pract. 2008, 23, 373–382. [Google Scholar] [CrossRef]
- Raslan, M.; Gonzalez, M.C.; Dias, M.C.; Nascimento, M.; Castro, M.; Marques, P.; Segatto, S.; Torrinhas, R.S.; Cecconello, I.; Waitzberg, D.L.; et al. Comparison of nutritional risk screening tools for predicting clinical outcomes in hospitalized patients. Nutrition 2010, 26, 721–726. [Google Scholar] [CrossRef]
- Charney, P. Nutrition screening vs. nutrition assessment: How do they differ? Nutr. Clin. Pract. 2008, 23, 366–372. [Google Scholar] [CrossRef]
- Detsky, A.S.; McLaughlin, J.R.; Baker, J.P.; Johnston, N.; Whittaker, S.; Mendelson, R.A.; Jeejeebhoy, K.N. What is subjective global assessment of nutritional status? 1987. Classical article. Nutr. Hosp. 2008, 23, 400–407. [Google Scholar]
- Koom, W.S.; Ahn, S.D.; Song, S.Y.; Lee, C.G.; Moon, S.H.; Chie, E.K.; et al. Nutritional status of patients treated with radiotherapy as determined by subjective global assessment. Radiat. Oncol. J. 2012, 30, 132–139. [Google Scholar] [CrossRef]
- National Kidney Foundation. KDOQI Clinical Practice Guidelines for Nutrition in Chronic Renal Failure 2000. Available online: https://kidneyfoundation.cachefly.net/professionals/KDOQI/guidelines_nutrition/nut_a09.html (accessed on 24 June 2019).
- British Dietetic Association. Parenteral and Enteral Nutrition Group. A Pocket Guide to Clinical Nutrition, 4th ed.; Parenteral and Enteral Nutrition Group of the British Dietetic Association: Birmingham, UK, 2011. [Google Scholar]
- Han, T.S.; Lean, M.E. Lower leg length as an index of stature in adults. Int. J. Obes. Relat. Metab. Disord. 1996, 20, 21–27. [Google Scholar]
- Reeves, S.L.; Varakamin, C.; Henry, C.J. The relationship between arm-span measurement and height with special reference to gender and ethnicity. Eur. J. Clin. Nutr. 1996, 50, 398–400. [Google Scholar]
- Maastricht UMC+. Nutritional Assessment Body Composition Skinfold Measurements 2019. Available online: https://nutritionalassessment.mumc.nl/en/skinfold-measurements (accessed on 12 July 2019).
- Bosy-Westphal, A.; Kromeyer-Hausschild, K.; Pirlich, M.; Schlattmann, A.; Scholz, G. Body composition analysis—What can be measured with practical value? Aktuel Ernahrungsmed. 2004, 1, 189–195. [Google Scholar]
- Kyle, U.; Bosaeus, I.; De Lorenzo, A.; Deurenberg, P.; Elia, M.; Gomez, J.; Heitmann, B.L.; Kent-Smith, L.; Melchior, J.C.; Pirlich, M.; et al. Bioelectrical impedance analysis—Part I: Review of principles and methods. Clin. Nutr. 2004, 23, 1226–1243. [Google Scholar] [CrossRef]
- Kyle, U.; Bosaeus, I.; De Lorenzo, A.; Deurenberg, P.; Elia, M.; Manuel Gomez, J.; Lilienthal Heitmann, B.; Kent-Smith, L.; Melchior, J.C.; Pirlich, M.; et al. Bioelectrical impedance analysis—Part II: Utilization in clinical practice. Clin. Nutr. 2004, 23, 1430–1453. [Google Scholar] [CrossRef]
- Forbes, G.B.; Bruining, G.J. Urinary creatinine excretion and lean body mass. Am. J. Clin. Nutr. 1976, 29, 1359–1366. [Google Scholar] [CrossRef]
- Stratton, R.J.; Green, C.J.; Elia, M. Disease-Related Malnutrition: An Evidence-Based Approach to Treatment; CABI Publishing: Wallingford, UK, 2003. [Google Scholar]
- MacDonald, A.J.; Greig, C.A.; Baracos, V. The advantages and limitations of cross-sectional body composition analysis. Curr. Opin. Support Palliat. Care. 2011, 5, 342–349. [Google Scholar] [CrossRef]
- Pirlich, M.; Norman, K. Bestimmung des Ernährungszustands (inkl. Bestimmung der Körperzusammensetzung und ernährungsmedizinisches Screening) in Biesalski, rnährungsmedizin; Georg Thieme Verlag KG: Stuttgart, Germany, 2018. [Google Scholar]
- Leuenberger, M.S.; Joray, M.L.; Kurmann, S.; Stanga, Z. How to assess the nutritional status of my patient. Praxis (Bern 1994) 2012, 101, 307–315. [Google Scholar] [CrossRef]
- Esper, D.H. Utilization of nutrition-focused physical assessment in identifying micronutrient deficiencies. Nutr. Clin. Pract. 2015, 30, 194–202. [Google Scholar] [CrossRef]
- Norman, K.; Stobaus, N.; Gonzalez, M.C.; Schulzke, J.D.; Pirlich, M. Hand grip strength: Outcome predictor and marker of nutritional status. Clin. Nutr. 2011, 30, 135–142. [Google Scholar] [CrossRef]
- Studenski, S.; Perera, S.; Wallace, D.; Chandler, J.M.; Duncan, P.W.; Rooney, E.; Fox, M.; Guralnik, J.M. Physical performance measures in the clinical setting. J. Am. Geriatr. Soc. 2003, 51, 314–322. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Dietary Assessment: A Resource Guide to Method Selection and Application in Low Resource Settings; Food and Agriculture Organization of the United Nations: Rome, Italy, 2018. [Google Scholar]
- Forster, H.; Walsh, M.C.; Gibney, M.J.; Brennan, L.; Gibney, E.R. Personalised nutrition: The role of new dietary assessment methods. Proc. Nutr. Soc. 2016, 75, 96–105. [Google Scholar] [CrossRef]
- Gemming, L.; Utter, J.; Ni Mhurchu, C. Image-assisted dietary assessment: A systematic review of the evidence. J. Acad. Nutr. Diet. 2015, 115, 64–77. [Google Scholar] [CrossRef]
- Illner, A.K.; Freisling, H.; Boeing, H.; Huybrechts, I.; Crispim, S.P.; Slimani, N. Review and evaluation of innovative technologies for measuring diet in nutritional epidemiology. Int. J. Epidemiol. 2012, 41, 1187–1203. [Google Scholar] [CrossRef]
- Stumbo, P.J. New technology in dietary assessment: A review of digital methods in improving food record accuracy. Proc. Nutr. Soc. 2013, 72, 70–76. [Google Scholar] [CrossRef]
- Archundia Herrera, M.C.; Chan, C.B. Narrative Review of New Methods for Assessing Food and Energy Intake. Nutrients 2018, 10, 1064. [Google Scholar] [CrossRef]
- Martin, C.K.; Correa, J.B.; Han, H.; Allen, H.R.; Rood, J.C.; Champagne, C.M.; Gunturk, B.K.; Bray, G.A. Validity of the Remote Food Photography Method (RFPM) for estimating energy and nutrient intake in near real-time. Obesity (Silver Spring) 2012, 20, 891–899. [Google Scholar] [CrossRef]
- Dong, Y.; Hoover, A.; Scisco, J.; Muth, E. A new method for measuring meal intake in humans via automated wrist motion tracking. Appl Psychophysiol. Biofeedback 2012, 37, 205–215. [Google Scholar] [CrossRef]
- Kawano, Y.; Yanai, K. FoodCam: A real-time food recognition system on a smartphone. Multimed. Tool. Appl. 2015, 74, 5263–5287. [Google Scholar] [CrossRef]
- Anthimopoulos, M.; Dehais, J.; Shevchik, S.; Ransford, B.H.; Duke, D.; Diem, P.; Mougiakakou, S. Computer vision-based carbohydrate estimation for type 1 patients with diabetes using smartphones. J. Diabetes Sci. Technol. 2015, 9, 507–515. [Google Scholar] [CrossRef]
- Bally, L.; Dehais, J.; Nakas, C.T.; Anthimopoulos, M.; Laimer, M.; Rhyner, D.; Rosenberg, G.; Zueger, T.; Diem, P.; Mougiakakou, S.; et al. Carbohydrate Estimation Supported by the GoCARB System in Individuals With Type 1 Diabetes: A Randomized Prospective Pilot Study. Diabetes Care 2017, 40, e6–e7. [Google Scholar] [CrossRef]
- Dehais, J.; Anthimopoulos, M.; Shevchik, S.; Mougiakakou, S. Two-view 3D reconstruction for food volume estimation. IEEE Trans. Multimed. 2017, 19, 1090–1099. [Google Scholar] [CrossRef]
- Rhyner, D.; Loher, H.; Dehais, J.; Anthimopoulos, M.; Shevchik, S.; Botwey, R.H.; Duke, D.; Stettler, C.; Diem, P.; Mougiakakou, S.; et al. Carbohydrate Estimation by a Mobile Phone-Based System Versus Self-Estimations of Individuals With Type 1 Diabetes Mellitus: A Comparative Study. J. Med. Int. Res. 2016, 18, e101. [Google Scholar] [CrossRef]
- Vasiloglou, M.F.; Mougiakakou, S.; Aubry, E.; Bokelmann, A.; Fricker, R.; Gomes, F.; Guntermann, C.; Meyer, A.; Studerus, D.; Stanga, Z. A Comparative Study on Carbohydrate Estimation: GoCARB vs. Dietitians. Nutrients 2018, 10, 741. [Google Scholar] [CrossRef]
- Ambrosini, G.L.; Hurworth, M.; Giglia, R.; Trapp, G.; Strauss, P. Feasibility of a commercial smartphone application for dietary assessment in epidemiological research and comparison with 24-h dietary recalls. Nutr. J. 2018, 17, 5. [Google Scholar] [CrossRef]
- Bucher Della Torre, S.; Carrard, I.; Farina, E.; Danuser, B.; Kruseman, M. Development and Evaluation of e-CA, an Electronic Mobile-Based Food Record. Nutrients 2017, 9, 76. [Google Scholar]
- Bejar, L.M.; Sharp, B.N.; Garcia-Perea, M.D. The e-EPIDEMIOLOGY Mobile Phone App for Dietary Intake Assessment: Comparison with a Food Frequency Questionnaire. JMIR Res. Protoc. 2016, 5, e208. [Google Scholar] [CrossRef]
- Poslusna, K.; Ruprich, J.; de Vries, J.H.; Jakubikova, M.; van’t Veer, P. Misreporting of energy and micronutrient intake estimated by food records and 24 hour recalls, control and adjustment methods in practice. Br. J. Nutr. 2009, 101, S73–85. [Google Scholar] [CrossRef]
- Beasley, J.; Riley, W.T.; Jean-Mary, J. Accuracy of a PDA-based dietary assessment program. Nutrition 2005, 21, 672–677. [Google Scholar] [CrossRef]
- Casperson, S.L.; Sieling, J.; Moon, J.; Johnson, L.; Roemmich, J.N.; Whigham, L. A mobile phone food record app to digitally capture dietary intake for adolescents in a free-living environment: Usability study. JMIR Mhealth Uhealth 2015, 3, e30. [Google Scholar] [CrossRef]
- Ashman, A.M.; Collins, C.E.; Brown, L.J.; Rae, K.M.; Rollo, M.E. Validation of a Smartphone Image-Based Dietary Assessment Method for Pregnant Women. Nutrients 2017, 9, 73. [Google Scholar] [CrossRef]
- Recio-Rodriguez, J.I.; Rodriguez-Martin, C.; Gonzalez-Sanchez, J.; Rodriguez-Sanchez, E.; Martin-Borras, C.; Martinez-Vizcaino, V.; Arietaleanizbeaskoa, M.S.; Magdalena-Gonzalez, O.; Fernandez-Alonso, C.; Maderuelo-Fernandez, J.A.; et al. EVIDENT Smartphone App, a New Method for the Dietary Record: Comparison With a Food Frequency Questionnaire. JMIR Mhealth Uhealth 2019, 7, e11463. [Google Scholar] [CrossRef]
- Carter, M.C.; Burley, V.J.; Nykjaer, C.; Cade, J.E. ‘My Meal Mate’ (MMM): Validation of the diet measures captured on a smartphone application to facilitate weight loss. Br. J. Nutr. 2013, 109, 539–546. [Google Scholar] [CrossRef]
- Conrad, J.; Nothlings, U. Innovative approaches to estimate individual usual dietary intake in large-scale epidemiological studies. Proc. Nutr. Soc. 2017, 76, 213–219. [Google Scholar] [CrossRef]
- Lemacks, J.L.; Adams, K.; Lovetere, A. Dietary Intake Reporting Accuracy of the Bridge2U Mobile Application Food Log Compared to Control Meal and Dietary Recall Methods. Nutrients 2019, 11, 199. [Google Scholar] [CrossRef]
- Harris, J.A.; Benedict, F.G. A Biometric Study of Human Basal Metabolism. Proc. Natl. Acad. Sci. USA 1918, 4, 370–373. [Google Scholar] [CrossRef]
- Druml, W.; Jadrna, K. Recommendations for Enteral and Parenteral Nutrition in Adults; English Edition/Pocket Version; Austrian Society of Clinical Nutrition (AKE): Vienna, Austria, 2008. [Google Scholar]
| Pre-Screening | |||
| Is the BMI of the patient < 20.5 kg/m2 | Yes | ||
| Did the patient lose weight in the past 3 months? | Yes | ||
| Was the patient’s food intake reduced in the past week? | Yes | ||
| Is the patient critically ill? | Yes | ||
| If yes to one of those questions, proceed to screening. | |||
| If no for all answers, the patient should be re-screened weekly. | |||
| Screening | |||
| Nutritional status | score | Stress metabolism (severity of the disease) | score |
| None | 0 | None | 0 |
| Mild Weight loss >5% in 3 months OR 50–75% of the normal food intake in the last week | 1 | Mild stress metabolism | 1 |
| Patient is mobile Increased protein requirement can be covered with oral nutrition Hip fracture, chronic disease especially with complications e.g., liver cirrhosis, COPD, diabetes, cancer, chronic hemodialysis | |||
| Moderate | 2 | Moderate stress metabolism | 2 |
| Weight loss >5% in 2 months OR BMI 18.5–20.5 kg/m2 AND reduced general condition OR 25–50% of the normal food intake in the last week | Patient is bedridden due to illness Highly increased protein requirement, may be covered with ONS Stroke, hematologic cancer, severe pneumonia, extended abdominal surgery | ||
| Severe Weight loss >5% in 1 month OR BMI <18.5 kg/m2 AND reduced general condition OR 0–25% of the normal food intake in the last week | 3 | Severe stress metabolism Patient is critically ill (intensive care unit) Very strongly increased protein requirement can only be achieved with (par)enteral nutrition APACHE-II >10, bone marrow transplantation, head traumas | 3 |
| Total (A) | Total (B) | ||
| Age | |||
| <70 years: 0 pt | |||
| ≥70 years: 1 pt | |||
| TOTAL = (A) + (B) + Age | |||
| ≥3 points: patient is at nutritional risk. Nutritional care plan should be set up | |||
| <3 points: repeat screening weekly | |||
| Malnutrition Universal Screening Tool (MUST) | ||||
| BMI (kg/m2) | Unintentional weight loss in the past 3–6 months | Acute illness with reduced food intake (estimated) for ≥5 days | ||
| ≥20 | 0 | ≤5% | 0 | No = 0 |
| 18.5–20.0 | 1 | 5–10% | 1 | Yes = 2 |
| ≤18.5 | 2 | ≥10% | 2 | |
| Overall Risk for Malnutrition | ||||
| Total | Risk | Procedure | Implementation | |
| 0 | Low | Routine clinical care | Clinic: weekly | |
| Nursing home: monthly | ||||
| Outpatient: yearly in at-risk patient groups, e.g., age >75 years | ||||
| 1 | Medium | Observe | Clinic, nursing home, and outpatient: | |
| Document dietary intake for 3 days. | ||||
| If adequate: little concern and repeat screening (hospital weekly, care home at least monthly, community at least every 2–3 months). | ||||
| If inadequate: clinical concern. Follow local policy, set goals, improve and increase overall nutritional intake, monitor and review care plan regularly. | ||||
| ≥2 | High | Treat | Clinic, nursing home, and outpatient: | |
| Refer to dietitian, Nutritional Support Team, or implement local policy. Set goals, improve and increase overall nutritional intake. Monitor and review care plan (hospital weekly, care home monthly, community monthly). | ||||
| Screening | ||
| A | Has food intake declined over the past 3 months due to loss of appetite, digestive problems, or chewing or swallowing difficulties? | 0 = severe decrease in food intake |
| 1 = moderate decrease in food intake | ||
| 2 = no decrease in food intake | ||
| B | Weight loss during the last 3 months | 0 = weight loss greater than 3 kg |
| 1 = does not know | ||
| 2 = weight loss between 1 and 3 kg | ||
| 3 = no weight loss | ||
| C | Mobility | 0 = bedridden or chair bound |
| 1 = able to get out of bed/chair but does not go out | ||
| 2 = goes out | ||
| D | Has the patient suffered psychological stress or acute disease in the past 3 months? | 0 = yes |
| 2 = no | ||
| E | Neuropsychological problems | 0 = severe dementia or depression |
| 1 = mild dementia | ||
| 2 = no psychological problems | ||
| F1 | Body mass index (BMI) | 0 = BMI less than 19 |
| 1 = BMI 19 to less than 21 | ||
| 2 = BMI 21 to less than 23 | ||
| 3 = BMI 23 or greater | ||
| If BMI is not available, replace question F1 with F2. Do not answer F2 if F1 is already completed. | ||
| F2 | Calf circumference (CC) in cm | 0 = CC less than 31 |
| 3 = CC 31 or greater | ||
| Screening Score | ||
| 12–14 points | Normal nutritional status | |
| 8–11 points | At risk of malnutrition | |
| 0–7 points | Malnourished | |
| Method | Target | Precision | Expenditure (Time/Apparatus) | Costs |
|---|---|---|---|---|
| Anthropometrics | FM, fat distribution, MM | ↓ | ↓ | ↓↓ |
| Bioelectrical impedance analysis | TBW, FM, FFM, BCM phase angle | ↑ | ↓ | ↓ |
| Creatinine height index | MM | ↓ | - | ↓ |
| Dual energy X-ray absorptiometry | FM, bone mineral content, soft tissues, bone density | ↑ | ↑ | ↑ |
| Magnetic resonance tomography | MM, FM, fat distribution | ↑ | ↑ | ↑↑ |
| Computed tomography | FM, fat distribution, MM | ↑ | ↑ | ↑ |
| Dilution method | TBW, FM, FFM (deuterium) ECW, ICW (bromide) | ↑ | ↑ | - |
| Potassium count | BCM, FFM, FM | ↑ | ↑ | ↑↑ |
| Neutron activation | Ca, Na, Cl, PO4, N, H, O, C | ↑ | ↑ | ↑↑ |
| Laboratory Value | Nutrition Independent Factors | Half-Life | Appropriateness to Detect Malnutrition | Appropriateness to Monitor Nutritional Therapy |
|---|---|---|---|---|
| Albumin | ↑ dehydration | 20 d | +/++ | Not appropriate due to high suggestibility and long half-life |
| ↓ inflammation, infections, trauma, heart failure, edema, liver dysfunction, nephrotic syndrome | ||||
| Not appropriate in case of anorexia and acute illness | ||||
| Transferrin | ↑ renal failure, iron status, acute hepatitis, hypoxia | 10 d | + | + |
| ↓ inflammation, chronic infections hemochromatosis, nephrotic syndrome, liver dysfunction | Low sensitivity and specificity | Concentration is independent of the energy and protein intake | ||
| Prealbumin/Transthyretin (TTR) | ↑ renal dysfunction, dehydration, corticosteroid therapy | 2 d | ++ | ++/+++ |
| Not appropriate to detect anorexia Subnormal values within one week in case of fasting | ||||
| One of the most appropriate proteins | ||||
| ↓ inflammation, hyperthyreosis, liver disease, overhydration | ||||
| Retinol binding protein (RBP) | ↑ kidney failure, alcohol abuse | 12 h | Idem prealbumin | Idem prealbumin |
| ↓ hyperthyreosis, chronic liver diseases, vitamin A deficiency, selenium deficiency | ||||
| Insulin-like growth factor 1 (IGF-1) | ↑ kidney failure | 24 h | ++ | +++ |
| More specific than retinol-binding protein and prealbumin/transthyretin | ||||
| ↓ liver diseases, severe catabolic status, age | Rapid decrease in fasting periods | |||
| Urinary creatinine | ↑ collection time >24h, infection, trauma | - | 1 mmol of creatinine is derived from 1.9 kg of skeletal muscle mass | Not appropriate, very slow |
| ↓ insufficient collection time, acute kidney failure | ||||
| Lymphocytes | ↑ healing phase after infection, hematologic diseases | - | + | Not appropriate, very slow |
| ↓ sepsis, hematologic disease, immune suppressants, steroids | Very unspecific |
| Body Region | Signs | Possible Deficiencies |
|---|---|---|
| Skin | Petechiae | Vitamins A, C |
| Purpura | Vitamins C, K | |
| Pigmentation | Niacin | |
| Edema | Protein, vitamin B1 | |
| Pallor | Folic acid, iron, biotin, vitamins B12, B6 | |
| Decubitus | Protein, energy | |
| Seborrheic dermatitis | Vitamin B6, biotin, zinc, essential fatty acids | |
| Unhealed wounds | Vitamin C, protein, zinc | |
| Nails | Pallor or white coloring Clubbing, spoon-shape, or transverse ridging/banding; excessive dryness, darkness in nails, curved nail ends | Iron, protein, vitamin B12 |
| Head/Hair | Dull/lackluster; banding/sparse; alopecia; depigmentation of hair; scaly/flaky scalp | Protein and energy, biotin, copper, essential fatty acid |
| Eyes | Pallor conjunctiva | Vitamin B12, folic acid, iron |
| Night vision impairment | Vitamin A | |
| Photophobia | Zinc | |
| Oral cavity | Glossitis | Vitamins B2, B6, B12, niacin, iron, folic acid |
| Gingivitis | Vitamin C | |
| Fissures, stomatitis | Vitamin B2, iron, protein | |
| Cheilosis | Niacin, vitamins B2, B6, protein | |
| Pale tongue | Iron, vitamin B12 | |
| Atrophied papillae | Vitamin B2, niacin, iron | |
| Nervous system | Mental confusion | Vitamins B1, B2, B12, water |
| Depression, lethargy | Biotin, folic acid, vitamin C | |
| Weakness, leg paralysis | Vitamins B1, B6, B12, pantothenic acid | |
| Peripheral neuropathy | Vitamins B2, B6, B12 | |
| Ataxia | Vitamin B12 | |
| Hyporeflexia | Vitamin B1 | |
| Muscle cramps | Vitamin B6, calcium, magnesium | |
| Fatigue | Energy, biotin, magnesium, iron |
| Macronutrient | Energy Content/g | Recommended Amount/kg Body Weight/d |
|---|---|---|
| Proteins | 4 kcal | 1.0–1.5 g |
| Carbohydrates | 4 kcal | max. 3–5 g |
| Fats | 9 kcal | 0.8–1.5 g |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reber, E.; Gomes, F.; Vasiloglou, M.F.; Schuetz, P.; Stanga, Z. Nutritional Risk Screening and Assessment. J. Clin. Med. 2019, 8, 1065. https://doi.org/10.3390/jcm8071065
Reber E, Gomes F, Vasiloglou MF, Schuetz P, Stanga Z. Nutritional Risk Screening and Assessment. Journal of Clinical Medicine. 2019; 8(7):1065. https://doi.org/10.3390/jcm8071065
Chicago/Turabian StyleReber, Emilie, Filomena Gomes, Maria F. Vasiloglou, Philipp Schuetz, and Zeno Stanga. 2019. "Nutritional Risk Screening and Assessment" Journal of Clinical Medicine 8, no. 7: 1065. https://doi.org/10.3390/jcm8071065
APA StyleReber, E., Gomes, F., Vasiloglou, M. F., Schuetz, P., & Stanga, Z. (2019). Nutritional Risk Screening and Assessment. Journal of Clinical Medicine, 8(7), 1065. https://doi.org/10.3390/jcm8071065





