Lifetime Psychotic Symptoms, Subthreshold Depression and Cognitive Impairment as Barriers to Functional Recovery in Patients with Bipolar Disorder
Abstract
1. Introduction
2. Experimental Section
2.1. Participants
2.2. Clinical and Functional Assessments
2.3. Neuropsychological Assessment
2.4. Statistical Analyses
3. Results
3.1. Demographic and Clinical Features
3.2. Neuropsychological Performance
3.3. Identifying Factors Associated with Functional Recovery
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Whiteford, H.A.; Degenhardt, L.; Rehm, J.; Baxter, A.J.; Ferrari, A.J.; Erskine, H.E.; Charlson, F.J.; Norman, R.E.; Flaxman, A.D.; Johns, N.; et al. Global burden of disease attributable to mental and substance use disorders: Findings from the Global Burden of Disease Study 2010. Lancet 2013, 382, 1575–1586. [Google Scholar] [CrossRef]
- Vieta, E.; Berk, M.; Schulze, T.G.; Carvalho, A.F.; Suppes, T.; Calabrese, J.R.; Gao, K.; Miskowiak, K.W.; Grande, I. Bipolar disorders. Nat. Rev. Dis. Primers 2018, 4, 18008. [Google Scholar] [CrossRef] [PubMed]
- Vieta, E.; Torrent, C. Functional remediation: The pathway from remission to recovery in bipolar disorder. World Psychiatry 2016, 15, 288–289. [Google Scholar] [CrossRef] [PubMed]
- MacQueen, G.M.; Young, L.T.; Joffe, R.T. A review of psychosocial outcome in patients with bipolar disorder. Acta Psychiatr. Scand. 2001, 103, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Solé, B.; Bonnin, C.M.; Jiménez, E.; Torrent, C.; Torres, I.; Varo, C.; Valls, E.; Montejo, L.; Gómez-Ocaña, C.; Tomioka, Y.; et al. Heterogeneity of functional outcomes in patients with bipolar disorder: A cluster-analytic approach. Acta Psychiatr. Scand. 2018, 137, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Bonnín, C.M.; Martínez-Arán, A.; Reinares, M.; Valentí, M.; Solé, B.; Jiménez, E.; Montejo, L.; Vieta, E.; Rosa, A.R. Thresholds for severity, remission and recovery using the functioning assessment short test (FAST) in bipolar disorder. J. Affect. Disord. 2018, 240, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Moreno, J.; Bonnin, C.M.; González-Pinto, A.; Amann, B.L.; Solé, B.; Balanzá-Martinez, V.; Arango, C.; Jiménez, E.; Tabarés-Seisdedos, R.; Garcia-Portilla, M.P.; et al. Factors associated with poor functional outcome in bipolar disorder: Sociodemographic, clinical, and neurocognitive variables. Acta Psychiatr. Scand. 2018, 138, 145–154. [Google Scholar] [CrossRef]
- Rosa, A.R.; Sánchez-Moreno, J.; Martínez-Aran, A.; Salamero, M.; Torrent, C.; Reinares, M.; Comes, M.; Colom, F.; Van Riel, W.; Ayuso-Mateos, J.L.; et al. Validity and reliability of the Functioning Assessment Short Test (FAST) in bipolar disorder. Clin. Pract. Epidemiol. Ment. Health 2007, 3, 5. [Google Scholar] [CrossRef] [PubMed]
- Vieta, E. Bipolar units and programmes: Are they really needed? World Psychiatry 2011, 10, 152. [Google Scholar] [CrossRef]
- Salagre, E.; Dodd, S.; Aedo, A.; Rosa, A.; Amoretti, S.; Pinzon, J.; Reinares, M.; Berk, M.; Kapczinski, F.P.; Vieta, E.; et al. Toward Precision Psychiatry in Bipolar Disorder: Staging 2.0. Front. Psychiatry 2018, 9, 641. [Google Scholar] [CrossRef]
- Colom, F.; Vieta, E.; Martínez-Arán, A.; Garcia-Garcia, M.; Reinares, M.; Torrent, C.; Goikolea, J.M.; Banús, S.; Salamero, M. [Spanish version of a scale for the assessment of mania: Validity and reliability of the Young Mania Rating Scale]. Med. Clin. (Barc.) 2002, 119, 366–371. [Google Scholar] [CrossRef]
- Young, R.C.; Biggs, J.T.; Ziegler, V.E.; Meyer, D.A. A rating scale for mania: Reliability, validity and sensitivity. Br. J. Psychiatry 1978, 133, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Cordero Villafáfila, A.; Ramos-Brieva, J.A. [Factor structure of the Castillian version of the Hamilton Rating Scale for Depression]. Actas Luso Esp. Neurol. Psiquiatr. Cienc. Afines 1986, 14, 339–342. [Google Scholar] [PubMed]
- Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 1960, 23, 56–62. [Google Scholar] [CrossRef] [PubMed]
- First, M.B.; Spitzer, R.; Gibbon, M. Structured Clinical Interview for DSM-IV Axis I Disorders; Biometric Research Department, Ed.; American Psychiatric Press Inc.: Whashington, DC, USA, 1997. [Google Scholar]
- Wechsler, D. The Wechsler Adult Intelligence Scale—III (WAIS-III); The Psychological Corporation: San Antonio, TX, USA, 1997. [Google Scholar]
- Delis, D.C.; Kramer, J.H.; Kaplan, E. California Verbal Learning Test; Psychological Corporation: New York, NY, USA, 1987. [Google Scholar]
- Heaton, R.K. Wisconsin Card Sorting Test Manual; Psychological Asessment Resources: Odessa, FL, USA, 1981. [Google Scholar]
- Golden, C. Stroop Color and Word Test; Stoelting: Chicago, IL, USA, 1978. [Google Scholar]
- Reitan, R. Validity of the rail making test as a indication of organic brain damage. Percept. Mot. Skills 1958, 8, 271–276. [Google Scholar] [CrossRef]
- Benton, A.; Hamsher, K. Multilingual Aphasia Examination; University of Iowa: Iowa City, IA, USA, 1976. [Google Scholar]
- Rey, A. Test de Copia de una Figura Compleja. MANUAL Adaptación Española; TEA Ediciones: Madrid, Spain, 1997. [Google Scholar]
- Dunayevich, E.; Keck, P.E. Prevalence and description of psychotic features in bipolar mania. Curr. Psychiatry Rep. 2000, 2, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Keck, P.E.; McElroy, S.L.; Havens, J.R.; Altshuler, L.L.; Nolen, W.A.; Frye, M.A.; Suppes, T.; Denicoff, K.D.; Kupka, R.; Leverich, G.S.; et al. Psychosis in bipolar disorder: Phenomenology and impact on morbidity and course of illness. Compr. Psychiatry 2003, 44, 263–269. [Google Scholar] [CrossRef]
- Manji, H.K.; Quiroz, J.A.; Payne, J.L.; Singh, J.; Lopes, B.P.; Viegas, J.S.; Zarate, C.A. The underlying neurobiology of bipolar disorder. World Psychiatry 2003, 2, 136–146. [Google Scholar]
- Vieta, E.; Salagre, E.; Grande, I.; Carvalho, A.F.; Fernandes, B.S.; Berk, M.; Birmaher, B.; Tohen, M.; Suppes, T. Early Intervention in Bipolar Disorder. Am. J. Psychiatry 2018, 175, 411–426. [Google Scholar] [CrossRef]
- Bowie, C.R.; Best, M.W.; Depp, C.; Mausbach, B.T.; Patterson, T.L.; Pulver, A.E.; Harvey, P.D. Cognitive and functional deficits in bipolar disorder and schizophrenia as a function of the presence and history of psychosis. Bipolar Disord. 2018, 20, 604–613. [Google Scholar] [CrossRef]
- Nehme, E.; Obeid, S.; Hallit, S.; Haddad, C.; Salame, W.; Tahan, F. Impact of psychosis in bipolar disorder during manic episodes. Int. J. Neurosci. 2018, 128, 1128–1134. [Google Scholar] [CrossRef] [PubMed]
- Levy, B.; Medina, A.M.; Weiss, R.D. Cognitive and psychosocial functioning in bipolar disorder with and without psychosis during early remission from an acute mood episode: A comparative longitudinal study. Compr. Psychiatry 2013, 54, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Tohen, M.; Waternaux, C.M.; Tsuang, M.T. Outcome in Mania. A 4-year prospective follow-up of 75 patients utilizing survival analysis. Arch. Gen. Psychiatry 1990, 47, 1106–1111. [Google Scholar] [CrossRef] [PubMed]
- Van Riel, W.G.; Vieta, E.; Martinez-Aran, A.; Haro, J.M.; Bertsch, J.; Reed, C.; Van Os, J. For The Emblem Advisory Board Chronic mania revisited: Factors associated with treatment non-response during prospective follow-up of a large European cohort (EMBLEM). World J. Biol. Psychiatry 2008, 9, 313–320. [Google Scholar] [CrossRef]
- Carlson, G.A.; Kotov, R.; Chang, S.-W.; Ruggero, C.; Bromet, E.J. Early determinants of four-year clinical outcomes in bipolar disorder with psychosis. Bipolar Disord. 2012, 14, 19–30. [Google Scholar] [CrossRef]
- Bora, E.; Yücel, M.; Pantelis, C. Neurocognitive markers of psychosis in bipolar disorder: A meta-analytic study. J. Affect. Disord. 2010, 127, 1–9. [Google Scholar] [CrossRef]
- Sánchez-Morla, E.M.; López-Villarreal, A.; Jiménez-López, E.; Aparicio, A.I.; Martínez-Vizcaíno, V.; Roberto, R.-J.; Vieta, E.; Santos, J.-L. Impact of number of episodes on neurocognitive trajectory in bipolar disorder patients: A 5-year follow-up study. Psychol. Med. 2018, 49, 1299–1307. [Google Scholar] [CrossRef]
- Jiménez-López, E.; Sánchez-Morla, E.M.; Aparicio, A.I.; López-Villarreal, A.; Martínez-Vizcaíno, V.; Rodriguez-Jimenez, R.; Vieta, E.; Santos, J.L. Psychosocial functioning in patients with psychotic and non-psychotic bipolar I disorder. A comparative study with individuals with schizophrenia. J. Affect. Disord. 2018, 229, 177–185. [Google Scholar] [CrossRef]
- Goldberg, J.F.; Harrow, M.; Grossman, L.S. Course and outcome in bipolar affective disorder: A longitudinal follow- up study. Am. J. Psychiatry 1995, 152, 379–384. [Google Scholar]
- Burton, C.Z.; Ryan, K.A.; Kamali, M.; Marshall, D.F.; Harrington, G.; McInnis, M.G.; Tso, I.F. Psychosis in bipolar disorder: Does it represent a more “severe” illness? Bipolar Disord. 2018, 20, 18–26. [Google Scholar] [CrossRef]
- Tost, H.; Ruf, M.; Schmäl, C.; Schulze, T.G.; Knorr, C.; Vollmert, C.; Bößhenz, K.; Ende, G.; Meyer-Lindenberg, A.; Henn, F.A.; et al. Prefrontal-temporal gray matter deficits in bipolar disorder patients with persecutory delusions. J. Affect. Disord. 2010, 120, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Geddes, J.R.; Goodwin, G.M.; Rendell, J.; Azorin, J.-M.; Cipriani, A.; Ostacher, M.J.; Morriss, R.; Alder, N.; Juszczak, E. Lithium plus valproate combination therapy versus monotherapy for relapse prevention in bipolar I disorder (BALANCE): A randomised open-label trial. Lancet 2010, 375, 385–395. [Google Scholar] [PubMed]
- Popovic, D.; Reinares, M.; Goikolea, J.M.; Bonnin, C.M.; Gonzalez-Pinto, A.; Vieta, E. Polarity index of pharmacological agents used for maintenance treatment of bipolar disorder. Eur. Neuropsychopharmacol. 2012, 22, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Grande, I.; Berk, M.; Birmaher, B.; Vieta, E. Bipolar disorder. Lancet 2016, 387, 1561–1572. [Google Scholar] [CrossRef]
- Colom, F.; Vieta, E.; Martinez-Aran, A.; Reinares, M.; Goikolea, J.M.; Benabarre, A.; Torrent, C.; Comes, M.; Corbella, B.; Parramon, G.; et al. A randomized trial on the efficacy of group psychoeducation in the prophylaxis of recurrences in bipolar patients whose disease is in remission. Arch. Gen. Psychiatry 2003, 60, 402–407. [Google Scholar] [CrossRef] [PubMed]
- Reinares, M.; Sánchez-Moreno, J.; Fountoulakis, K.N. Psychosocial interventions in bipolar disorder: What, for whom, and when. J. Affect. Disord. 2014, 156, 46–55. [Google Scholar] [CrossRef]
- Bonnín, C.M.; Martínez-Arán, A.; Torrent, C.; Pacchiarotti, I.; Rosa, A.R.; Franco, C.; Murru, A.; Sanchez-Moreno, J.; Vieta, E. Clinical and neurocognitive predictors of functional outcome in bipolar euthymic patients: A long-term, follow-up study. J. Affect. Disord. 2010, 121, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Bonnín, C.M.; Sánchez-Moreno, J.; Martínez-Arán, A.; Solé, B.; Reinares, M.; Rosa, A.R.; Goikolea, J.M.; Benabarre, A.; Ayuso-Mateos, J.L.; Ferrer, M.; et al. Subthreshold symptoms in bipolar disorder: Impact on neurocognition, quality of life and disability. J. Affect. Disord. 2012, 136, 650–659. [Google Scholar] [CrossRef]
- Murru, A.; Pacchiarotti, I.; Verdolini, N.; Reinares, M.; Torrent, C.; Geoffroy, P.-A.; Bellivier, F.; Llorca, P.-M.; Vieta, E.; Samalin, L. Modifiable and non-modifiable factors associated with functional impairment during the inter-episodic periods of bipolar disorder. Eur. Arch. Psychiatry Clin. Neurosci. 2017, 268, 749–755. [Google Scholar] [CrossRef]
- Gitlin, M.J.; Mintz, J.; Sokolski, K.; Hammen, C.; Altshuler, L.L. Subsyndromal depressive symptoms after symptomatic recovery from mania are associated with delayed functional recovery. J. Clin. Psychiatry 2011, 72, 692–697. [Google Scholar] [CrossRef]
- Samalin, L.; Boyer, L.; Murru, A.; Pacchiarotti, I.; Reinares, M.; Bonnin, C.M.; Torrent, C.; Verdolini, N.; Pancheri, C.; de Chazeron, I.; et al. Residual depressive symptoms, sleep disturbance and perceived cognitive impairment as determinants of functioning in patients with bipolar disorder. J. Affect. Disord. 2017, 210, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Samalin, L.; de Chazeron, I.; Vieta, E.; Bellivier, F.; Llorca, P.-M. Residual symptoms and specific functional impairments in euthymic patients with bipolar disorder. Bipolar Disord. 2016, 18, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Gitlin, M.J.; Miklowitz, D.J. The difficult lives of individuals with bipolar disorder: A review of functional outcomes and their implications for treatment. J. Affect. Disord. 2017, 209, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Reinares, M.; Papachristou, E.; Harvey, P.; Mar Bonnín, C.; Sánchez-Moreno, J.; Torrent, C.; Ayuso-Mateos, J.L.; Ploubidis, G.B.; Vieta, E.; Frangou, S. Towards a clinical staging for bipolar disorder: Defining patient subtypes based on functional outcome. J. Affect. Disord. 2013, 144, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Soni, A.; Singh, P.; Shah, R.; Bagotia, S. Impact of Cognition and Clinical Factors on Functional Outcome in Patients with Bipolar Disorder. East Asian Arch. Psychiatry 2017, 27, 26–34. [Google Scholar] [PubMed]
- Best, M.W.; Bowie, C.R.; Naiberg, M.R.; Newton, D.F.; Goldstein, B.I. Neurocognition and psychosocial functioning in adolescents with bipolar disorder. J. Affect. Disord. 2017, 207, 406–412. [Google Scholar] [CrossRef]
- Judd, L.L.; Akiskal, H.S.; Schettler, P.J.; Endicott, J.; Leon, A.C.; Solomon, D.A.; Coryell, W.; Maser, J.D.; Keller, M.B. Psychosocial Disability in the Course of Bipolar I and II Disorders. Arch. Gen. Psychiatry 2005, 62, 1322–1330. [Google Scholar] [CrossRef] [PubMed]
- Radua, J.; Grunze, H.; Amann, B.L. Meta-Analysis of the Risk of Subsequent Mood Episodes in Bipolar Disorder. Psychother. Psychosom. 2017, 86, 90–98. [Google Scholar] [CrossRef]
- Weinstock, L.M.; Miller, I.W. Psychosocial predictors of mood symptoms 1 year after acute phase treatment of bipolar I disorder. Compr. Psychiatry 2010, 51, 497–503. [Google Scholar] [CrossRef]
- Jiménez, E.; Arias, B.; Mitjans, M.; Goikolea, J.M.; Ruíz, V.; Brat, M.; Sáiz, P.A.; García-Portilla, M.P.; Burón, P.; Bobes, J.; et al. Clinical features, impulsivity, temperament and functioning and their role in suicidality in patients with bipolar disorder. Acta Psychiatr. Scand. 2016, 133, 266–276. [Google Scholar] [CrossRef]
- Michalak, E.E.; Yatham, L.N.; Lam, R.W. Quality of life in bipolar disorder: A review of the literature. Health Qual. Life Outcomes 2005, 3, 72. [Google Scholar] [CrossRef] [PubMed]
- Vieta, E.; Garriga, M. Adjunctive antidepressants in bipolar depression. Lancet Psychiatry 2016, 3, 1095–1096. [Google Scholar] [CrossRef]
- Pacchiarotti, I.; Bond, D.J.; Baldessarini, R.J.; Nolen, W.A.; Grunze, H.; Licht, R.W.; Post, R.M.; Berk, M.; Goodwin, G.M.; Sachs, G.S.; et al. The International Society for Bipolar Disorders (ISBD) Task Force Report on Antidepressant Use in Bipolar Disorders. Am. J. Psychiatry 2013, 170, 1249–1262. [Google Scholar] [CrossRef] [PubMed]
- Garriga, M.; Solé, E.; González-Pinto, A.; Selva-Vera, G.; Arranz, B.; Amann, B.L.; Saiz-Ruiz, J.; Pérez-Blanco, J.; Vieta, E. Efficacy of quetiapine XR vs. placebo as concomitant treatment to mood stabilizers in the control of subthreshold symptoms of bipolar disorder: Results from a pilot, randomized controlled trial. Eur. Neuropsychopharmacol. 2017, 27, 959–969. [Google Scholar] [CrossRef] [PubMed]
- Novo, P.; Landin-Romero, R.; Radua, J.; Vicens, V.; Fernandez, I.; Garcia, F.; Pomarol-Clotet, E.; McKenna, P.J.; Shapiro, F.; Amann, B.L. Eye movement desensitization and reprocessing therapy in subsyndromal bipolar patients with a history of traumatic events: A randomized, controlled pilot-study. Psychiatry Res. 2014, 219, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Rosa, A.R.; Mercadé, C.; Sánchez-Moreno, J.; Solé, B.; Bonnin, C.D.M.; Torrent, C.; Grande, I.; Sugranyes, G.; Popovic, D.; Salamero, M.; et al. Validity and reliability of a rating scale on subjective cognitive deficits in bipolar disorder (COBRA). J. Affect. Disord. 2013, 150, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Sylvia, L.G.; Salcedo, S.; Bernstein, E.E.; Baek, J.H.; Nierenberg, A.A.; Deckersbach, T. Nutrition, Exercise, and Wellness Treatment in bipolar disorder: Proof of concept for a consolidated intervention. Int. J. Bipolar Disord. 2013, 1, 24. [Google Scholar] [CrossRef] [PubMed]
- Hearing, C.M.; Chang, W.C.; Szuhany, K.L.; Deckersbach, T.; Nierenberg, A.A.; Sylvia, L.G. Physical Exercise for Treatment of Mood Disorders: A Critical Review. Curr. Behav. Neurosci. Reports 2016, 3, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Team Sky. A Wininning Advantage; Team Sky: Manchester, UK, 2010. [Google Scholar]
- Nierenberg, A.A.; Hearing, C.M.; Sande Mathias, I.; Young, L.T.; Sylvia, L.G. Getting to wellness: The potential of the athletic model of marginal gains for the treatment of bipolar disorder. Aust. N. Z. J. Psychiatry 2015, 49, 1207–1214. [Google Scholar] [CrossRef]
- Reinares, M.; Martínez-Arán, A.; Vieta, E. Psychotherapy for Bipolar Disorders: An Integrative Approach; Cambridge University Press: Cambridge, UK, 2019. [Google Scholar]
- Bourne, C.; Aydemir, Ö.; Balanzá-Martínez, V.; Bora, E.; Brissos, S.; Cavanagh, J.T.O.; Clark, L.; Cubukcuoglu, Z.; Dias, V.V.; Dittmann, S.; et al. Neuropsychological testing of cognitive impairment in euthymic bipolar disorder: An individual patient data meta-analysis. Acta Psychiatr. Scand. 2013, 128, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Tse, S.; Chan, S.; Ng, K.L.; Yatham, L.N. Meta-analysis of predictors of favorable employment outcomes among individuals with bipolar disorder. Bipolar Disord. 2014, 16, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Tabarés-Seisdedos, R.; Balanzá-Martínez, V.; Sánchez-Moreno, J.; Martinez-Aran, A.; Salazar-Fraile, J.; Selva-Vera, G.; Rubio, C.; Mata, I.; Gómez-Beneyto, M.; Vieta, E. Neurocognitive and clinical predictors of functional outcome in patients with schizophrenia and bipolar I disorder at one-year follow-up. J. Affect. Disord. 2008, 109, 286–299. [Google Scholar] [CrossRef] [PubMed]
- Bonnín, C.D.M.; González-Pinto, A.; Solé, B.; Reinares, M.; González-Ortega, I.; Alberich, S.; Crespo, J.M.; Salamero, M.; Vieta, E.; Martínez-Arán, A.; et al. Verbal memory as a mediator in the relationship between subthreshold depressive symptoms and functional outcome in bipolar disorder. J. Affect. Disord. 2014, 160, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Martino, D.J.; Marengo, E.; Igoa, A.; Scápola, M.; Ais, E.D.; Perinot, L.; Strejilevich, S.A. Neurocognitive and symptomatic predictors of functional outcome in bipolar disorders: A prospective 1 year follow-up study. J. Affect. Disord. 2009, 116, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Mora, E.; Portella, M.J.; Forcada, I.; Vieta, E.; Mur, M. Persistence of cognitive impairment and its negative impact on psychosocial functioning in lithium-treated, euthymic bipolar patients: A 6-year follow-up study. Psychol. Med. 2013, 43, 1187–1196. [Google Scholar] [CrossRef]
- Andreou, C.; Bozikas, V.P. The predictive significance of neurocognitive factors for functional outcome in bipolar disorder. Curr. Opin. Psychiatry 2013, 26, 54–59. [Google Scholar] [CrossRef]
- Dias, V.V.; Brissos, S.; Frey, B.N.; Kapczinski, F. Insight, quality of life and cognitive functioning in euthymic patients with bipolar disorder. J. Affect. Disord. 2008, 110, 75–83. [Google Scholar] [CrossRef]
- Mur, M.; Portella, M.J.; Martínez-Arán, A.; Pifarré, J.; Vieta, E. Long-term stability of cognitive impairment in bipolar disorder: A 2-year follow-up study of lithium-treated euthymic bipolar patients. J. Clin. Psychiatry 2008, 69, 712–719. [Google Scholar] [CrossRef]
- Bonnín, C.M.; Torrent, C.; Goikolea, J.M.; Reinares, M.; Solé, B.; Valentí, M.; Sánchez-Moreno, J.; Hidalgo, D.; Tabarés-Seisdedos, R.; Martínez-Arán, A.; et al. The impact of repeated manic episodes and executive dysfunction on work adjustment in bipolar disorder. Eur. Arch. Psychiatry Clin. Neurosci. 2014, 264, 247–254. [Google Scholar] [CrossRef]
- O’Donnell, L.A.; Deldin, P.J.; Grogan-Kaylor, A.; McInnis, M.G.; Weintraub, J.; Ryan, K.A.; Himle, J.A. Depression and executive functioning deficits predict poor occupational functioning in a large longitudinal sample with bipolar disorder. J. Affect. Disord. 2017, 215, 135–142. [Google Scholar] [CrossRef]
- Miguélez-Pan, M.; Pousa, E.; Cobo, J.; Duño, R. Cognitive executive performance influences functional outcome in euthymic type I bipolar disorder outpatients. Psicothema 2014, 26, 166–173. [Google Scholar] [PubMed]
- Martino, D.J.; Igoa, A.; Marengo, E.; Scápola, M.; Strejilevich, S.A. Neurocognitive Impairments and Their Relationship With Psychosocial Functioning in Euthymic Bipolar II Disorder. J. Nerv. Ment. Dis. 2011, 199, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Sarapas, C.; Shankman, S.A.; Harrow, M.; Faull, R.N. Attention/Processing Speed Prospectively Predicts Social Impairment 18 Years Later in Mood Disorders. J. Nerv. Ment. Dis. 2013, 201, 824–827. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, Z.; Burton, C.Z.; Vella, L.; Twamley, E.W. Neuropsychological predictors of performance-based measures of functional capacity and social skills in individuals with severe mental illness. J. Psychiatr. Res. 2018, 102, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Martino, D.J.; Strejilevich, S.A.; Scápola, M.; Igoa, A.; Marengo, E.; Ais, E.D.; Perinot, L. Heterogeneity in cognitive functioning among patients with bipolar disorder. J. Affect. Disord. 2008, 109, 149–156. [Google Scholar] [CrossRef]
- Martino, D.J.; Valerio, M.P.; Szmulewicz, A.G.; Strejilevich, S.A. The effect of premorbid intelligence on neurocognitive and psychosocial functioning in bipolar disorder. J. Affect. Disord. 2017, 210, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Mora, E.; Portella, M.J.; Martinez-Alonso, M.; Teres, M.; Forcada, I.; Vieta, E.; Mur, M. The Impact of Obesity on Cognitive Functioning in Euthymic Bipolar Patients. J. Clin. Psychiatry 2017, 78, e924–e932. [Google Scholar] [CrossRef]
- Forcada, I.; Mur, M.; Mora, E.; Vieta, E.; Bartrés-Faz, D.; Portella, M.J. The influence of cognitive reserve on psychosocial and neuropsychological functioning in bipolar disorder. Eur. Neuropsychopharmacol. 2015, 25, 214–222. [Google Scholar] [CrossRef]
- Anaya, C.; Torrent, C.; Caballero, F.F.; Vieta, E.; Bonnin, C.d.M.; Ayuso-Mateos, J.L.; CIBERSAM Functional Remediation Group. Cognitive reserve in bipolar disorder: Relation to cognition, psychosocial functioning and quality of life. Acta Psychiatr. Scand. 2016, 133, 386–398. [Google Scholar] [CrossRef]
- Amoretti, S.; Bernardo, M.; Bonnin, C.M.; Bioque, M.; Cabrera, B.; Mezquida, G.; Solé, B.; Vieta, E.; Torrent, C. The impact of cognitive reserve in the outcome of first-episode psychoses: 2-year follow-up study. Eur. Neuropsychopharmacol. 2016, 26, 1638–1648. [Google Scholar] [CrossRef]
- Solé, B.; Jiménez, E.; Torrent, C.; Reinares, M.; Bonnin, C.d.M.; Torres, I.; Varo, C.; Grande, C.d.M.; Valls, E.; Salagre, E.; et al. Cognitive Impairment in Bipolar Disorder: Treatment and Prevention Strategies. Int. J. Neuropsychopharmacol. 2017, 20, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Bonnín, C.D.M.; Reinares, M.; Martínez-Arán, A.; Jiménez, E.; Sánchez-Moreno, J.; Solé, B.; Montejo, L.; Vieta, E. Improving Functioning, Quality of Life, and Well-being in Patients With Bipolar Disorder. Int. J. Neuropsychopharmacol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Yatham, L.N.; Torres, I.J.; Malhi, G.S.; Frangou, S.; Glahn, D.C.; Bearden, C.E.; Burdick, K.E.; Martínez-Arán, A.; Dittmann, S.; Goldberg, J.F.; et al. The International Society for Bipolar Disorders-Battery for Assessment of Neurocognition (ISBD-BANC). Bipolar Disord. 2010, 12, 351–363. [Google Scholar] [CrossRef] [PubMed]
- Miskowiak, K.; Burdick, K.; Martinez-Aran, A.; Bonnin, C.; Bowie, C.; Carvalho, A.; Gallagher, P.; Lafer, B.; López-Jaramillo, C.; Sumiyoshi, T.; et al. Methodological recommendations for cognition trials in bipolar disorder by the International Society for Bipolar Disorders Targeting Cognition Task Force. Bipolar Disord. 2017, 19, 614–626. [Google Scholar] [CrossRef] [PubMed]
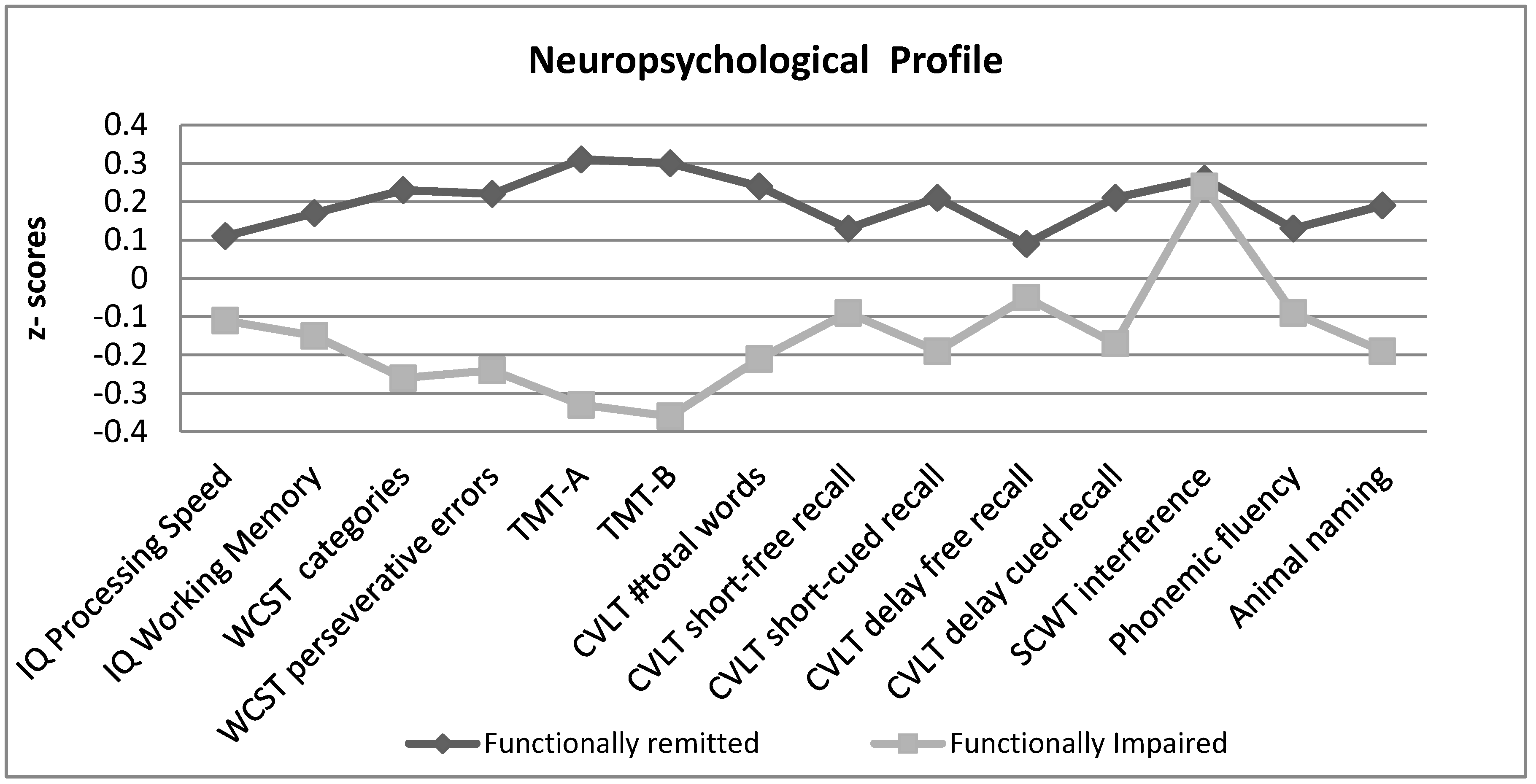
| Functionally Remitted (n = 221) Mean (SD) | Functionally Impaired (n = 199) Mean (SD) | t Student (p Value) | |
|---|---|---|---|
| Age | 38.4 (11.1) | 44.5 (10.1) | −5.85 (<0.001) |
| Years of education | 14.3 (3.3) | 14.1 (3.9) | 1.38 (0.16) |
| Estimated IQ | 108.4 (8.8) | 108.6 (9.7) | −0.16 (0.52) |
| YOUNG total score | 1.2 (1.7) | 1.4 (1.8) | −1.29 (0.19) |
| HAM-D total score | 2.1 (2.2) | 5.1 (2.9) | −11.11 (<0.001) |
| FAST total score | 10.5 (6.6) | 32.6 (8.9) | −28.5 (<0.001) |
| Chronicity (years of illness) | 14.2 (10.3) | 17.5 (11.1) | −3.1 (0.02) |
| Number of manic episodes | 2.1 (2.6) | 2.4 (3.1) | −1.10 (0.27) |
| Number of depressive episodes | 4.6 (8.3) | 6.7 (10.8) | −2.17 (0.03) |
| Number of hospitalizations | 1.6 (1.7) | 2.1 (2.4) | −2.36 (0.02) |
| n (%) | n (%) | Chi (p) | |
| Gender (women) | 116 (52.5) | 80 (40.6) | 5.9 (0.01) |
| Diagnosis (type I) | 173 (79.7) | 142 (72.1) | 3.31 (0.08) |
| Lifetime rapid cycling | 15 (9) | 26 (15.9) | 3.5 (0.07) |
| Lifetime psychotic symptoms | 142(66) | 131 (67.2) | 0.06 (0.83) |
| Family affective psychiatric history | 109 (66.9) | 118 (71.5) | 0.83 (0.40) |
| Functionally Remitted (n = 221) Mean (SD) | Functionally Impaired (n = 199) Mean (SD) | t Student (p) | |
|---|---|---|---|
| IQ Processing Speed | 104.3 (17.3) | 100.7 (14.3) | 2.2 (0.02) |
| IQ Working Memory | 100.5 (12.9) | 95.6 (14.9) | 3.5 (<0.01) |
| WCST categories | 5.4 (1.4) | 4.5 (1.9) | 5.1 (<0.01) |
| WCST perseverative errors | 11.8 (10.6) | 18.1 (14.9) | −4.8 (<0.01) |
| TMT-A | 27.8 (9.5) | 37.6 (18.1) | −6.8 (<0.01) |
| TMT-B | 70.7 (37.6) | 110.6 (75.8) | −6.7 (<0.01) |
| CVLT #total words | 56.5 (10.8) | 50.9 (13.7) | 4.6 (<0.01) |
| CVLT short-free recall | 12.1 (2.9) | 11.1 (6.1) | 2.2 (0.02) |
| CVLT short-cued recall | 13.0 (2.5) | 11.8 (3.2) | 4.2 (<0.01) |
| CVLT delay free recall | 12.6 (2.8) | 11.8 (7.3) | 1.4 (0.15) |
| CVLT delay cued recall | 13.2 (2.4) | 12.1 (3.1) | 4.1 (<0.01) |
| SCWT interference | 52.6 (6,4) | 52.3 (7.6) | 0.47 (0.63) |
| Osterrieth Rey Figure | 19.2 (4.8) | 17.3 (5.3) | 3.7 (<0.01) |
| Phonemic fluency | 36.0 (9.7) | 33.7 (10.2) | 2.3 (0.02) |
| Animal naming | 20.2 (4.5) | 18.3 (5.6) | 3.9 (<0.01) |
| Variables | Wald | p Value | OR | 95% CI |
|---|---|---|---|---|
| Hamilton Depression total score | 32.56 | <0.01 | 1.48 | 1.29–1.70 |
| Lifetime psychotic symptoms | 4.77 | 0.03 | 2.91 | 1.11–7.54 |
| Working Memory IQ | 7.57 | <0.01 | 0.95 | 0.93–0.98 |
| WCST number of categories | 4.17 | 0.04 | 0.7 | 0.50–0.98 |
| CVLT short-cued recall | 5.52 | 0.02 | 0.68 | 0.49–0.93 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonnín, C.M.; Jiménez, E.; Solé, B.; Torrent, C.; Radua, J.; Reinares, M.; Grande, I.; Ruíz, V.; Sánchez-Moreno, J.; Martínez-Arán, A.; et al. Lifetime Psychotic Symptoms, Subthreshold Depression and Cognitive Impairment as Barriers to Functional Recovery in Patients with Bipolar Disorder. J. Clin. Med. 2019, 8, 1046. https://doi.org/10.3390/jcm8071046
Bonnín CM, Jiménez E, Solé B, Torrent C, Radua J, Reinares M, Grande I, Ruíz V, Sánchez-Moreno J, Martínez-Arán A, et al. Lifetime Psychotic Symptoms, Subthreshold Depression and Cognitive Impairment as Barriers to Functional Recovery in Patients with Bipolar Disorder. Journal of Clinical Medicine. 2019; 8(7):1046. https://doi.org/10.3390/jcm8071046
Chicago/Turabian StyleBonnín, Caterina Mar, Esther Jiménez, Brisa Solé, Carla Torrent, Joaquim Radua, María Reinares, Iria Grande, Victoria Ruíz, Jose Sánchez-Moreno, Anabel Martínez-Arán, and et al. 2019. "Lifetime Psychotic Symptoms, Subthreshold Depression and Cognitive Impairment as Barriers to Functional Recovery in Patients with Bipolar Disorder" Journal of Clinical Medicine 8, no. 7: 1046. https://doi.org/10.3390/jcm8071046
APA StyleBonnín, C. M., Jiménez, E., Solé, B., Torrent, C., Radua, J., Reinares, M., Grande, I., Ruíz, V., Sánchez-Moreno, J., Martínez-Arán, A., & Vieta, E. (2019). Lifetime Psychotic Symptoms, Subthreshold Depression and Cognitive Impairment as Barriers to Functional Recovery in Patients with Bipolar Disorder. Journal of Clinical Medicine, 8(7), 1046. https://doi.org/10.3390/jcm8071046







