Bone Immune Response to Materials, Part II: Copper and Polyetheretherketone (PEEK) Compared to Titanium at 10 and 28 Days in Rabbit Tibia
Abstract
1. Introduction
2. Materials and Methods
2.1. Surgical Procedure
2.2. Gene Expression Analysis—qPCR
2.2.1. mRNA Isolation
2.2.2. Amplification Process
2.3. Decalcified Bone Histology
2.4. Statistical Analysis
3. Results
3.1. Gene Expression Analysis
3.1.1. Days
3.1.2. 28 Days
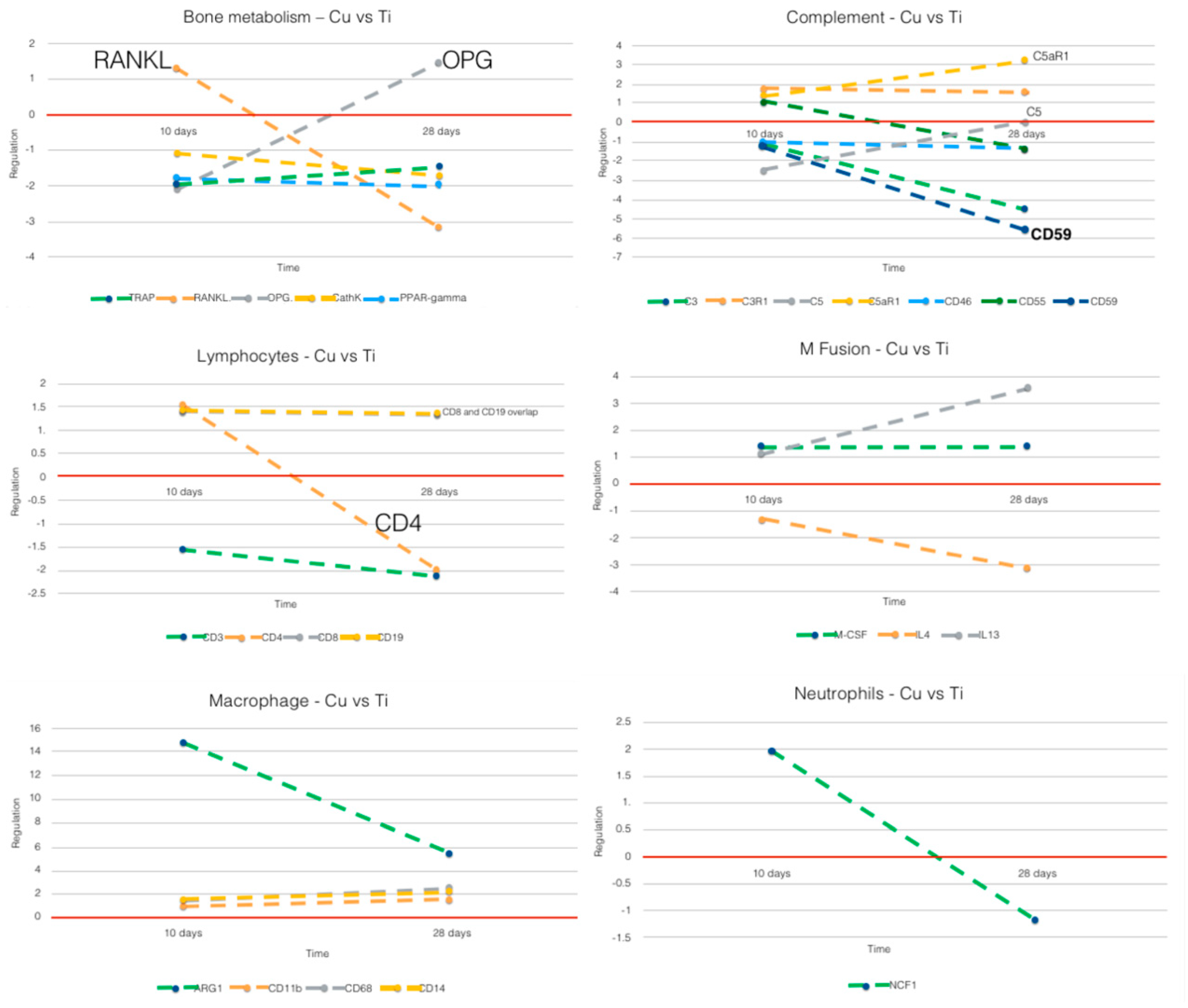
3.2. Comparative Analysis of Gene Expression: 10 vs. 28 Days
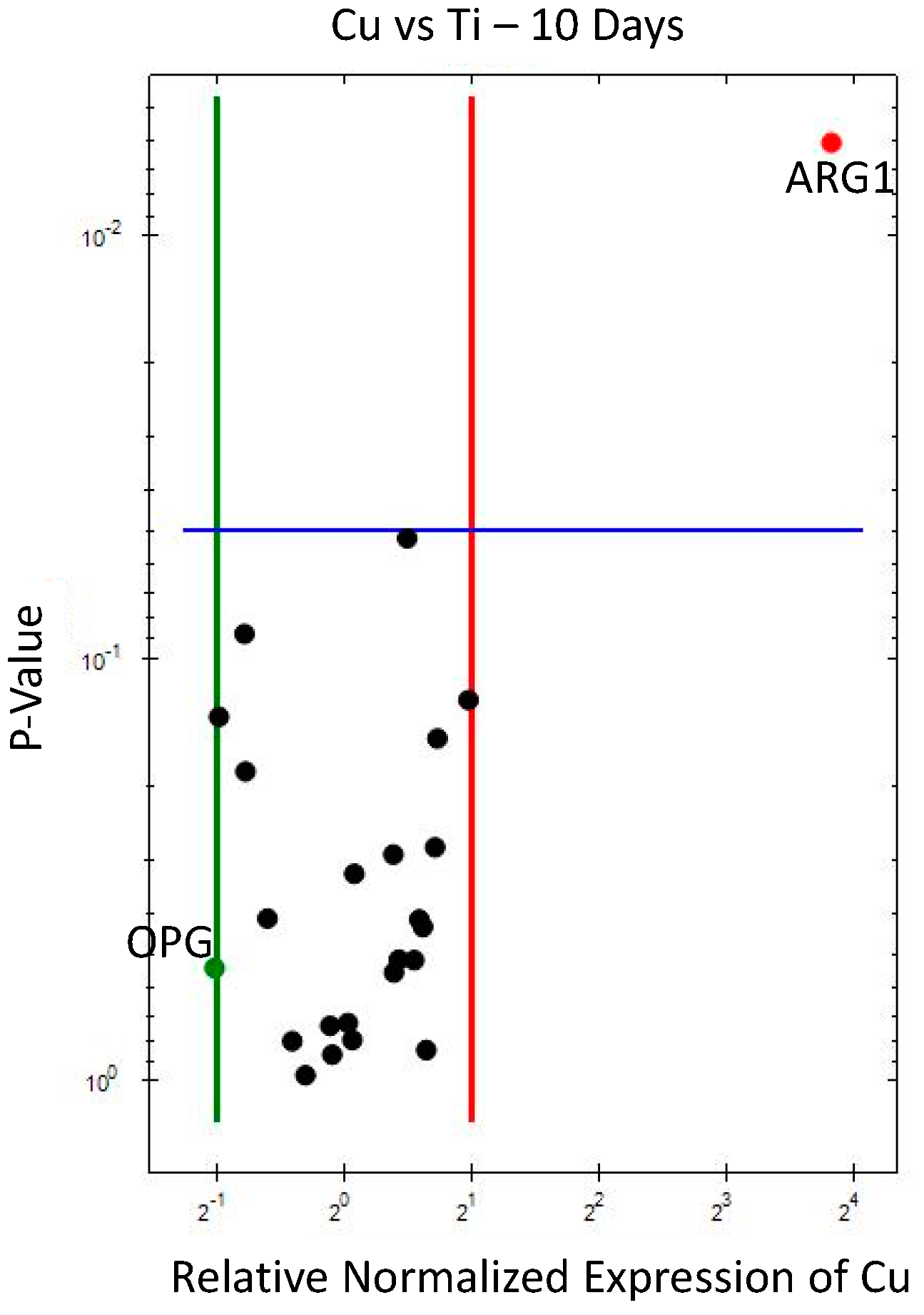
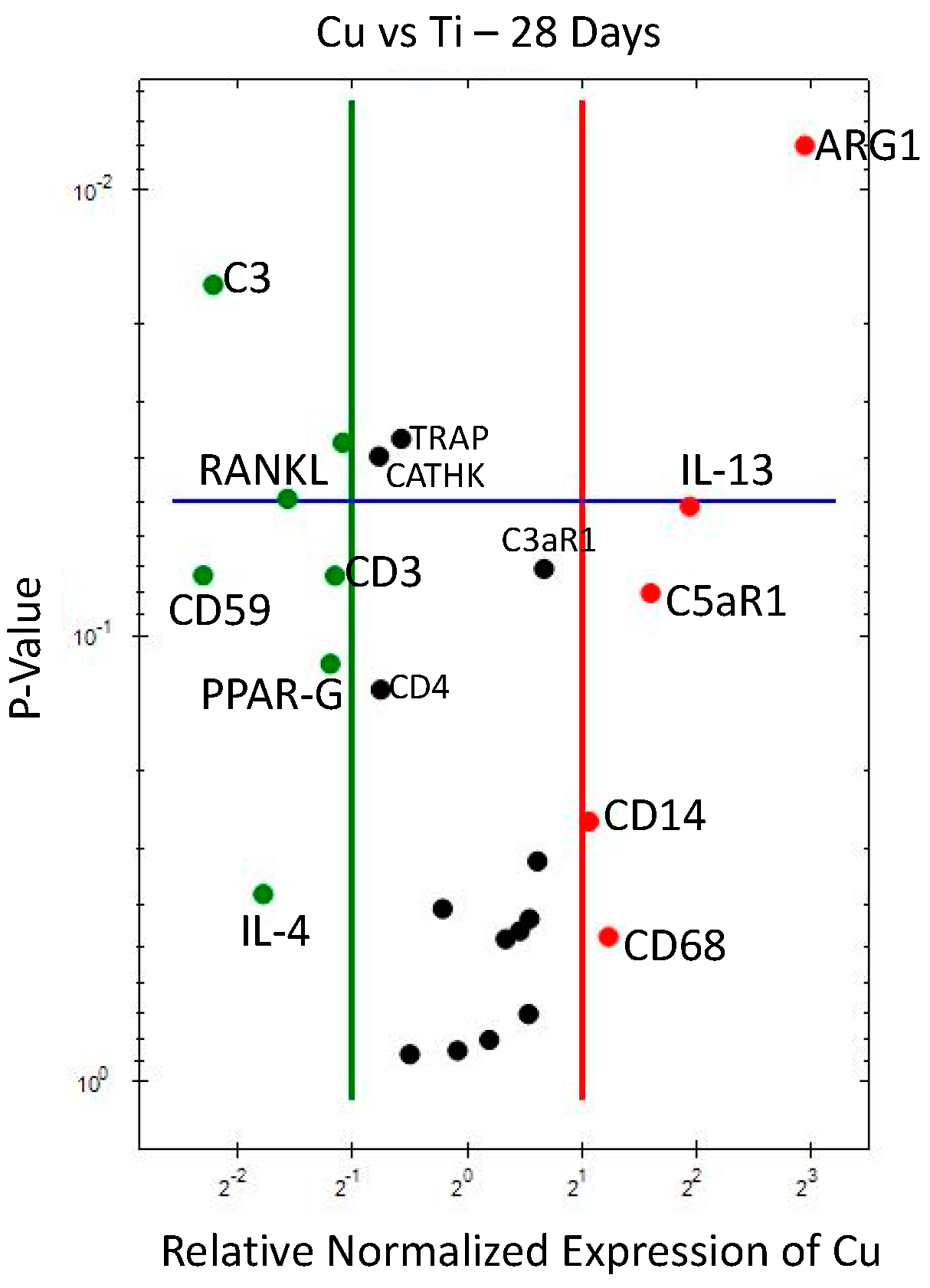
3.2.1. Cu vs. Ti (Figure 6)
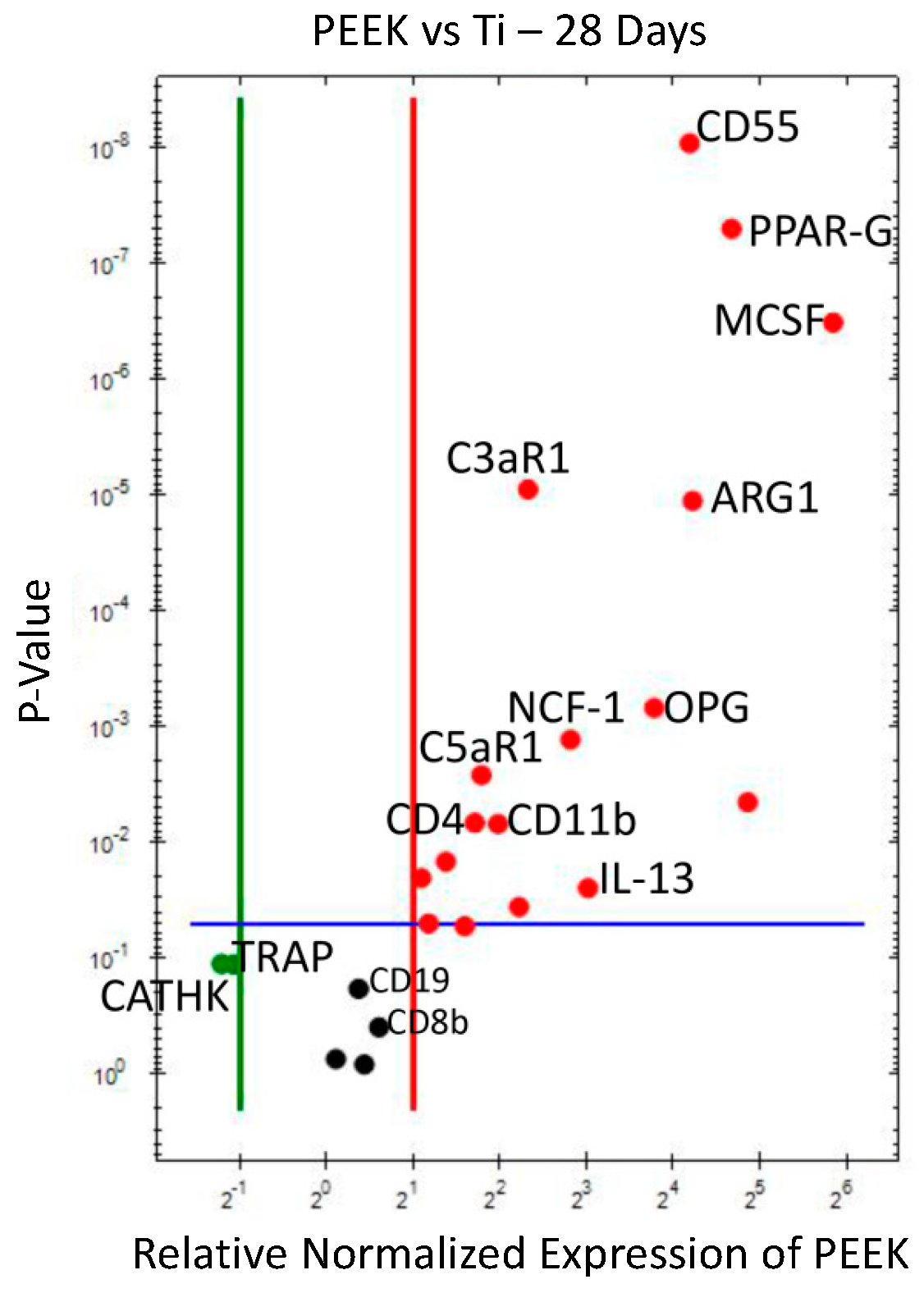
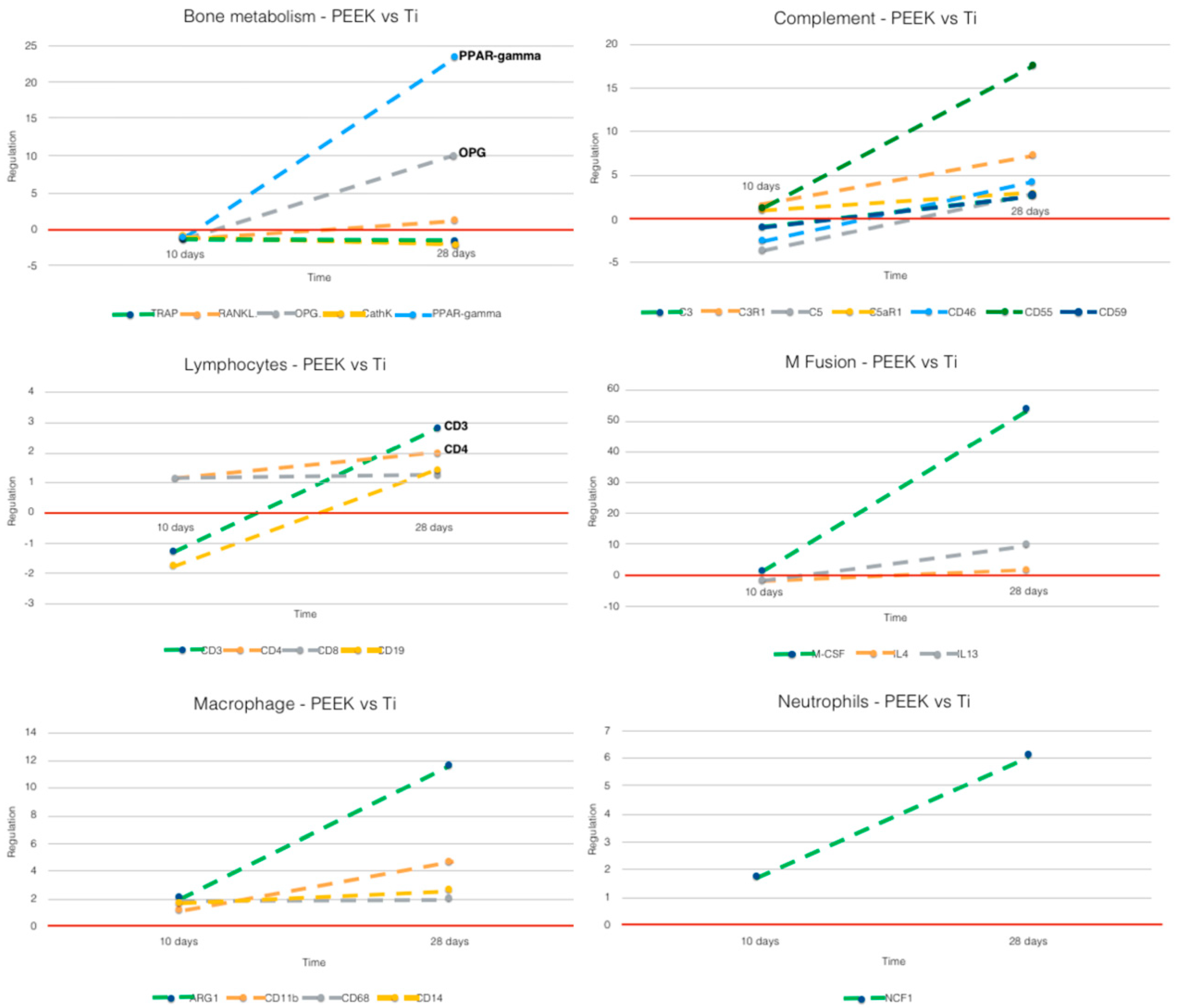
3.2.2. PEEK vs. Ti (Figure 7)
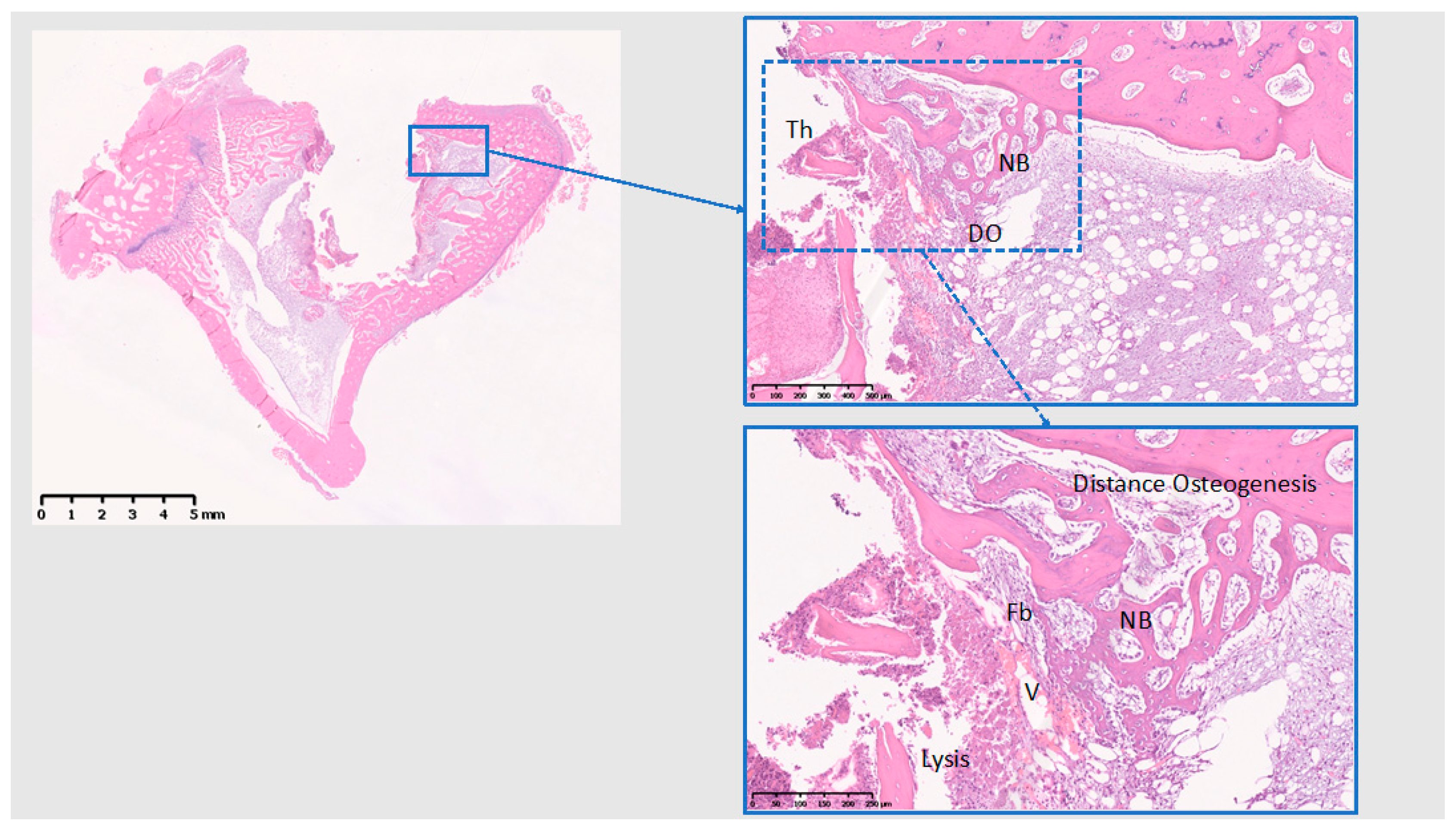
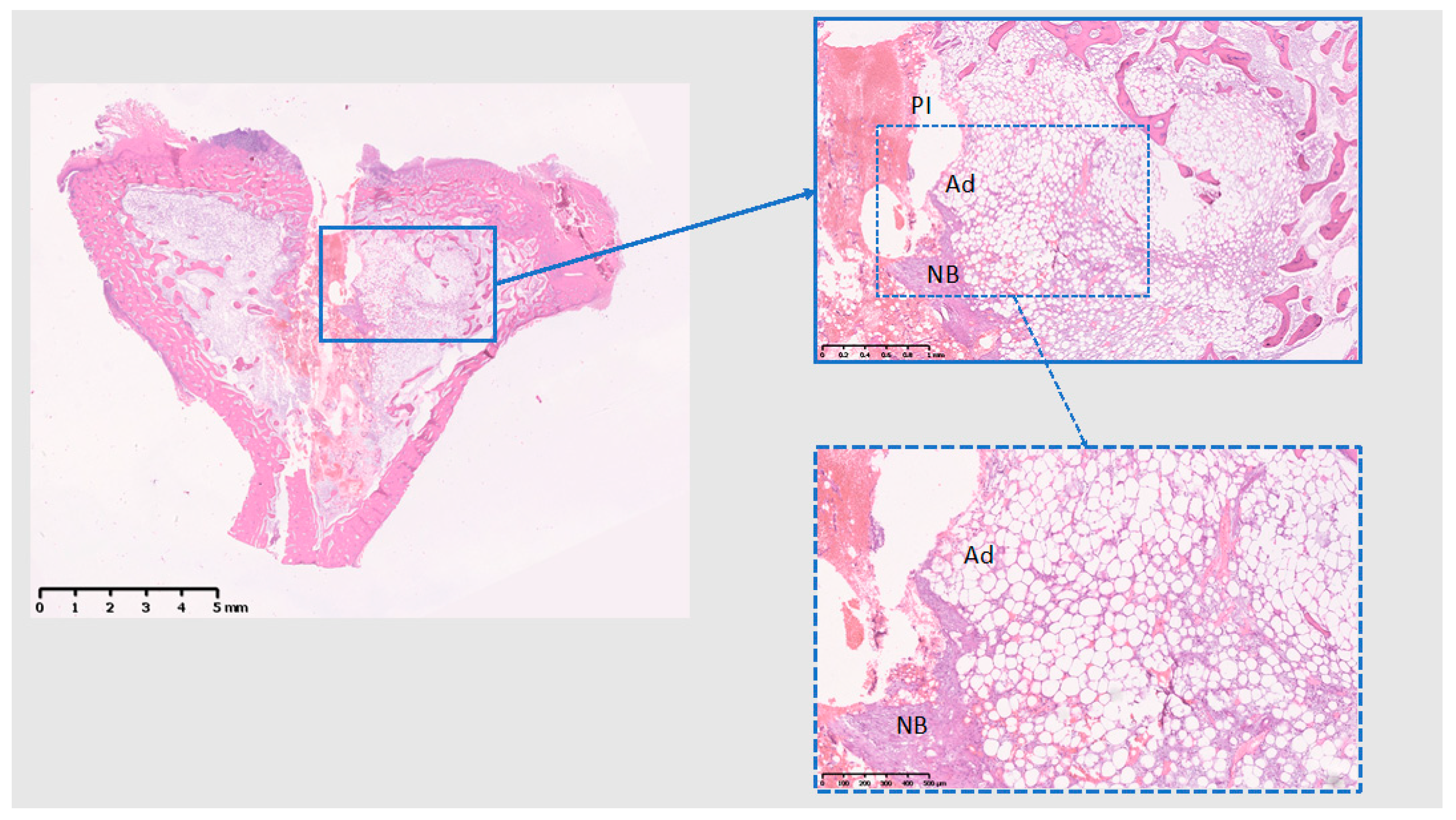
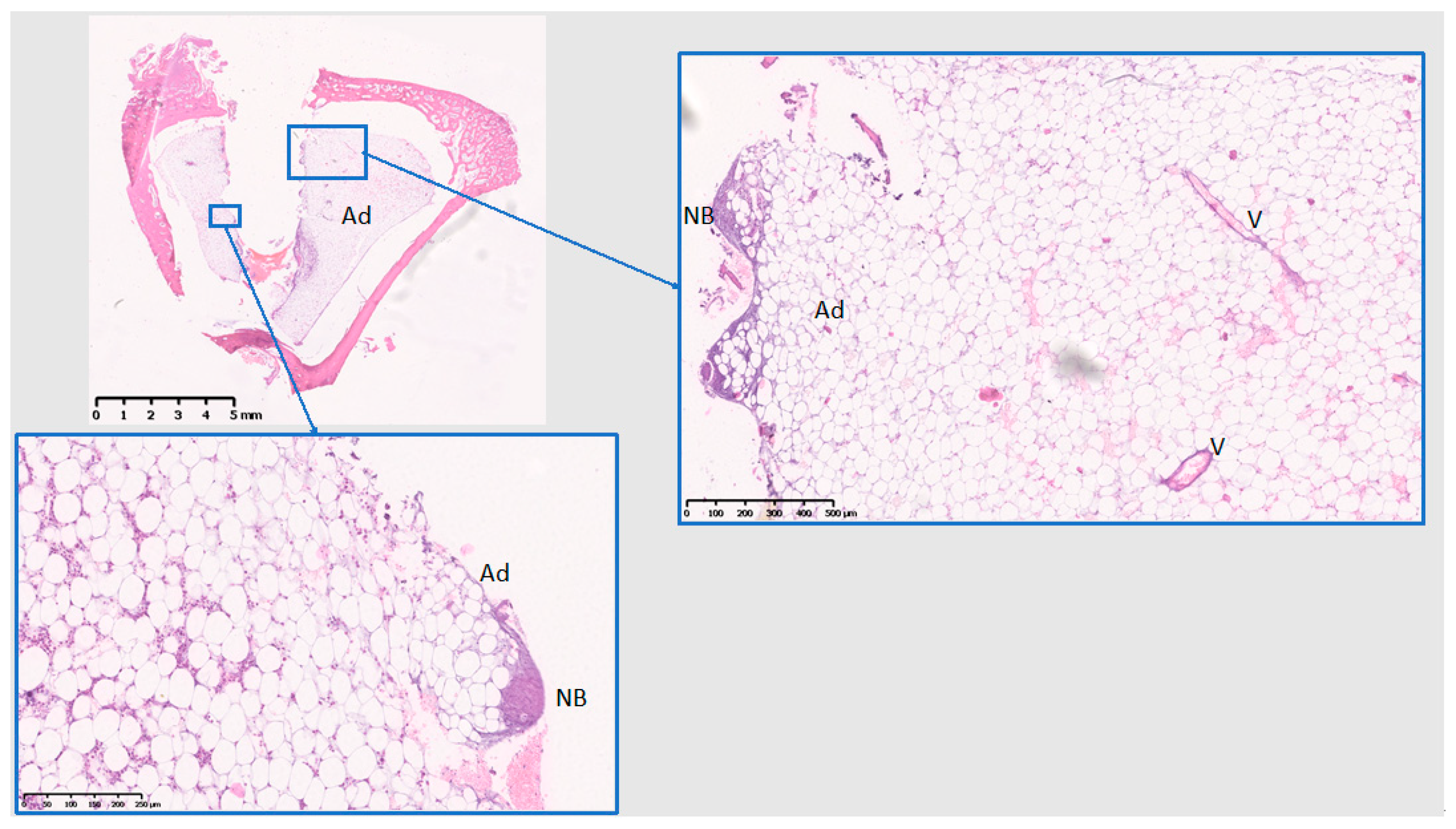
3.3. Histological Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Albrektsson, T.; Brånemark, P.-I.; Hansson, H.-A.; Lindström, J. Osseointegrated titanium implants: Requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man. Acta Orthop. Scand. 1981, 52, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Wennerberg, A.; Albrektsson, T. On implant surfaces: A review of current knowledge and opinions. Int. J. Oral Maxillofac. Implants 2010, 25, 63–74. [Google Scholar] [PubMed]
- Wennerberg, A.; Albrektsson, T.; Andersson, B. Bone tissue response to commercially pure titanium implants blasted with fine and coarse particles of aluminum oxide. Int. J. Oral Maxillofac. Implants 1996, 11, 38–45. [Google Scholar] [PubMed]
- Wennerberg, A.; Albrektsson, T.; Lausmaa, J. Torque and histomorphometric evaluation of c.p. titanium screws blasted with 25- and 75-μm-sized particles of Al2O3. J. Biomed. Mater. Res. 1996, 30, 251–260. [Google Scholar] [CrossRef]
- Buser, D.; Broggini, N.; Wieland, M.; Schenk, R.K.; Denzer, A.J.; Cochran, D.L.; Hoffmann, B.; Lussi, A.; Steinemann, S.G. Enhanced bone apposition to a chemically modified SLA titanium surface. J. Dent. Res. 2004, 83, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Ellingsen, J.E.; Johansson, C.B.; Wennerberg, A.; Holmén, A. Improved retention and bone-to-implant contact with fluoride-modified titanium implants. Int. J. Oral Maxillofac. Implants 2004, 19, 659–666. [Google Scholar] [PubMed]
- Chrcanovic, B.R.; Kisch, J.; Albrektsson, T.; Wennerberg, A. A retrospective study on clinical and radiological outcomes of oral implants in patients followed up for a minimum of 20 years. Clin. Implant Dent. Relat. Res. 2018, 20, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Friberg, B.; Gröndahl, K.; Lekholm, U.; Brånemark, P.-I. Long-term follow-up of severely atrophic edentulous mandibles reconstructed with short brånemark implants. Clin. Implant Dent. Relat. Res. 2000, 2, 184–189. [Google Scholar] [CrossRef]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef]
- Goodman, S.B. Wear particles, periprosthetic osteolysis and the immune system. Biomaterials 2007, 28, 5044–5048. [Google Scholar] [CrossRef]
- Trindade, R.; Albrektsson, T.; Tengvall, P.; Wennerberg, A. Foreign body reaction to biomaterials: On mechanisms for buildup and breakdown of osseointegration. Clin. Implant Dent. Relat. Res. 2016, 18, 192–203. [Google Scholar] [CrossRef] [PubMed]
- Trindade, R.; Albrektsson, T.; Galli, S.; Prgomet, Z.; Tengvall, P.; Wennerberg, A. Osseointegration and foreign body reaction: Titanium implants activate the immune system and suppress bone resorption during the first 4 weeks after implantation. Clin. Implant Dent. Relat. Res. 2018, 20, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M.; Jones, J.A. Phenotypic dichotomies in the foreign body reaction. Biomaterials 2007, 28, 5114–5120. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.H.; Kohno, Y.; Huang, J.F.; Romero-Lopez, M.; Maruyama, M.; Ueno, M.; Pajarinen, J.; Nathan, K.; Yao, Z.; Yang, F.; et al. Preconditioned or IL4-secreting mesenchymal stem cells enhanced osteogenesis at different stages. Tissue Eng. 2019. [Google Scholar] [CrossRef] [PubMed]
- Trindade, R.; Albrektsson, T.; Galli, S.; Prgomet, Z.; Tengvall, P.; Wennerberg, A. Bone immune response to materials, part I: Titanium, PEEK and copper in comparison to sham at 10 days in rabbit tibia. J. Clin. Med. 2018, 7, 526. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Dahlin, C.; Jemt, T.; Sennerby, L.; Turri, A.; Wennerberg, A. Is marginal bone loss around oral implants the result of a provoked foreign body reaction? Clin. Implant Dent. Relat. Res. 2014, 16, 155–165. [Google Scholar] [CrossRef]
- Insua, A.; Monje, A.; Wang, H.-L.; Miron, R.J. Basis of bone metabolism around dental implants during osseointegration and peri-implant bone loss. J. Biomed. Mater. Res. 2017, 105, 2075–2089. [Google Scholar] [CrossRef]
- Suska, F.; Emanuelsson, L.; Johansson, A.; Tengvall, P.; Thomsen, P. Fibrous capsule formation around titanium and copper. J. Biomed. Mater. Res. 2008, 85, 888–896. [Google Scholar] [CrossRef]
- Johansson, P.; Jimbo, R.; Kjellin, P.; Currie, F.; Chrcanovic, B.R.; Wennerberg, A. Biomechanical evaluation and surface characterization of a nano-modified surface on PEEK implants: A study in the rabbit tibia. Int. J. Nanomed. 2014, 9, 3903–3911. [Google Scholar] [CrossRef]
- Da Silva, T.A.; Zorzetto-Fernandes, A.L.V.; Cecílio, N.T.; Sardinha-Silva, A.; Fernandes, F.F.; Roque-Barreira, M.C. CD14 is critical for TLR2-mediated M1 macrophage activation triggered by N-glycan recognition. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- McNally, A.K.; Anderson, J.M. Foreign body-type multinucleated giant cells induced by interleukin-4 express select lymphocyte costimulatory molecules and are phenotypically distinct from osteoclasts and dendritic cells. Exp. Mol. Pathol. 2011, 91, 673–681. [Google Scholar] [CrossRef] [PubMed]
- Wenz, L.M.; Merritt, K.; Brown, S.A.; Moet, A.; Steffee, A.D. In vitro biocompatibility of polyetheretherketone and polysulfone composites. J. Biomed. Mater. Res. 1990, 24, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Katzer, A.; Marquardt, H.; Westendorf, J.; Wening, J.V.; von Foerster, G. Polyetheretherketone—Cytotoxicity and mutagenicity in vitro. Biomaterials 2002, 23, 1749–1759. [Google Scholar] [CrossRef]
- Vishwakarma, A.; Bhise, N.S.; Evangelista, M.B.; Rouwkema, J.; Dokmeci, M.R.; Ghaemmaghami, A.M.; Khademhosseini, A. Engineering immunomodulatory biomaterials to tune the inflammatory response. Trend. Biotechnol. 2016, 34, 470–482. [Google Scholar] [CrossRef] [PubMed]
- Yun, T.J.; Chaudhary, P.M.; Shu, G.L.; Kimble Frazer, J.; Ewings, M.K.; Schwartz, S.M.; Pascual, V.; Hood, L.E.; Clark, E.A. OPG/FDCR-1, a TNF receptor family member, is expressed in lymphoid cells and is up-regulated by ligating CD40. J. Immunol. 1998, 161, 6113–6121. [Google Scholar]
- Weitzmann, M.N.; Ofotokun, I. Physiological and pathophysiological bone turnover—Role of the immune system. Nat. Rev. Endocrinol. 2016, 12, 518–532. [Google Scholar] [CrossRef]
- Chinetti, G.; Griglio, S.; Antonucci, M.; Torra, I.P.; Delerive, P.; Majd, Z.; Fruchart, J.-C.; Chapman, J.; Najib, J.; Staels, B. Activation of proliferator-activated receptors α and γ induces apoptosis of human monocyte-derived macrophages. J. Biol. Chemist. 1998, 273, 25573–25580. [Google Scholar] [CrossRef]
- Jiang, C.; Ting, A.T.; Seed, B. PPAR-γ agonists inhibit production of monocyte inflammatory cytokines. Nature 1998, 391, 82–86. [Google Scholar] [CrossRef]
- Levine, J.A.; Jensen, M.D.; Eberhardt, N.L.; O’Brien, T. Adipocyte macrophage colony-stimulating factor is a mediator of adipose tissue growth. J. Clin. Invest. 1998, 101, 1557–1564. [Google Scholar] [CrossRef]
- Eriksson, A.; Albrektsson, T.; Magnusson, B. Assessment of bone viability after heat trauma: A histological, histochemical and vital microscopic study in the rabbit. Scand. J. Plast. Reconstr. Surg. 1984, 18, 261–268. [Google Scholar] [CrossRef]
- Ambrosi, T.H.; Scialdone, A.; Graja, A.; Gohlke, S.; Jank, A.-M.; Bocian, C.; Woelk, L.; Fan, H.; Logan, D.W.; Schurmann, A.; et al. Adipocyte accumulation in the bone marrow during obesity and aging impairs stem cell-based hematopoietic and bone regeneration. Cell Stem Cell 2017, 20, 771–784. [Google Scholar] [CrossRef] [PubMed]
- Kawanashi, N.; Yano, H.; Yokogawa, Y.; Suzuki, K. Exercise training inhibits inflammation in adipose tissue via both suppression of macrophage infiltration and acceleration of phenotypic switching from M1 to M2 macrophages in high-fat-diet-induced obese mice. Exerc. Immunol. Rev. 2016, 16, 105–118. [Google Scholar]
- Bianco, P.; Riminucci, M.; Gronthos, S.; Robey, P.G. Bone marrow stromal stem cells: Nature, biology, and potential applications. Stem Cells 2001, 19, 180–192. [Google Scholar] [CrossRef] [PubMed]





| Primer | Forward Sequence | Reverse Sequence | Accession No./Transcript ID |
|---|---|---|---|
| NCF-1 | TTCATCCGCCACATTGCCC | GTCCTGCCACTTCACCAAGA | NM_001082102.1 |
| CD68 | TTTCCCCAGCTCTCCACCTC | CGATGATGAGGGGCACCAAG | ENSOCUT00000010382 |
| CD11b | TTCAACCTGGAGACTGAGAACAC | TCAAACTGGACCACGCTCTG | ENSOCUT00000001589 |
| CD14 | TCTGAAAATCCTGGGCTGGG | TTCATTCCCGCGTTCCGTAG | ENSOCUT00000004218 |
| ARG1 | GGATCATTGGAGCCCCTTTCTC | TCAAGCAGACCAGCCTTTCTC | NM_001082108.1 |
| IL-4 | CTACCTCCACCACAAGGTGTC | CCAGTGTAGTCTGTCTGGCTT | ENSOCUT00000024099 |
| IL-13 | GCAGCCTCGTATCCCCAG | GGTTGACGCTCCACACCA | ENSOCUT00000000154 |
| M-CSF | GGAACTCTCGCTCAGGCTC | ACATTCTTGATCTTCTCCAGCAAC | ENSOCUT00000030714 |
| OPG | TGTGTGAATGCGAGGAAGGG | AACTGTATTCCGCTCTGGGG | ENSOCUT00000011149 |
| RANKL | GAAGGTTCATGGTTCGATCTGG | CCAAGAGGACAGGCTCACTTT | ENSOCUT00000024354 |
| TRAP | TTACTTCAGTGGCGTGCAGA | CGATCTGGGCTGAGACGTTG | NM_001081988.1 |
| CathK | GGAACCGGGGCATTGACTCT | TGTACCCTCTGCATTTGGCTG | NM_001082641.1 |
| PPAR-γ | CAAGGCGAGGGCGATCTT | ATGCGGATGGCGACTTCTTT | NM_001082148.1 |
| C3 | ACTCTGTCGAGAAGGAACGGG | CCTTGATTTGTTGATGCTGGCTG | NM_001082286.1 |
| C3aR1 | CATGTCAGTCAACCCCTGCT | GCGAATGGTTTTGCTCCCTG | ENSOCUT00000007435 |
| CD46 | TCCTGCTGTTCACTTTCTCGG | CATGTTCCCATCCTTGTTTACACTT | ENSOCUT00000033915 |
| CD55 | TGGTGTTGGGTGGAGTGACC | AGAGTGAAGCCTCTGTTGCATT | ENSOCUT00000031985 |
| CD59 | ACCACTGTCTCCTCCCAAGT | GCAATCTTCATACCGCCAACA | NM_001082712.1 |
| C5 | TCCAAAACTCTGCAACCTTAACA | AAATGCTTTGACACAACTTCCA | ENSOCUT00000005683 |
| C5aR1 | ACGTCAACTGCTGCATCAACC | AGGCTGGGGAGAGACTTGC | ENSOCUT00000029180 |
| CD3 | CCTGGGGACAGGAAGATGATGAC | CAGCACCACACGGGTTCCA | NM_001082001.1 |
| CD4 | CAACTGGAAACATGCGAACCA | TTGATGACCAGGGGGAAAGA | NM_001082313.2 |
| CD8 | GGCGTCTACTTCTGCATGACC | GAACCGGCACACTCTCTTCT | ENSOCUT00000009383 |
| CD19 | GGATGTATGTCTGTCGCCGT | AAGCAAAGCCACAACTGGAA | ENSOCUT00000028895 |
| GAPDH | GGTGAAGGTCGGAGTGAACGG | CATGTAGACCATGTAGTGGAGGTCA | NM_001082253.1 |
| ACT-β | TCATTCCAAATATCGTGAGATGCC | TACACAAATGCGATGCTGCC | NM_001101683.1 |
| LDHA | TGCAGACAAGGAACAGTGGA | CCCAGGTAGTGTAGCCCTT | NM_001082277.1 |
| Biological Entity | Gene |
|---|---|
| Neutrophil | NCF-1 |
| Macrophage | CD68, CD11b, CD14, ARG1 |
| Macrophage fusion | IL-4, IL-13, M-CSF |
| Bone resorption | OPG, RANKL, TRAP, CathK, PPAR-γ |
| Complement | Activation: C3, C3aR1, C5, C5aR1; Inhibition: CD46, CD55, CD59 |
| T lymphocytes | CD3, CD4, CD8 |
| B-lymphocytes | CD19 |
| Reference genes | GAPDH, ACT-β, LDHA |
| Marker | Regulation | p Value |
|---|---|---|
| PPAR-G | –1.72 | 0.087477 |
| TRAP | –1.98 | 0.137344 |
| OPG | –2.03 | 0.539750 |
| CD3 | –1.52 | 0.411951 |
| IL-4 | –1.71 | 0.185082 |
| ARG1 | 14.17 | 0.006031 |
| NCF1 | 1.96 | 0.125414 |
| C3aR1 | 1.66 | 0.154634 |
| CD14 | 1.50 | 0.414019 |
| CD4 | 1.53 | 0.431624 |
| CD19 | 1.64 | 0.279592 |
| Marker | Regulation | p Value |
|---|---|---|
| PPAR-G | –1.57 | 0.550176 |
| TRAP | –1.68 | 0.008821 |
| OPG | –1.70 | 0.089695 |
| CD19 | –2.14 | 0.111496 |
| IL-4 | –2.14 | 0.251881 |
| ARG1 | 1.71 | 0.361937 |
| NCF1 | 1.50 | 0.333874 |
| CD68 | 1.62 | 0.556273 |
| Marker | Regulation | p-Value |
|---|---|---|
| C3 | –4.64 | 0.016332 |
| CD59 | –4.93 | 0.073238 |
| RANKL | –2.96 | 0.049318 |
| PPAR-G | –2.29 | 0.115578 |
| TRAP | –1.49 | 0.036164 |
| CATH-K | –1.70 | 0.039611 |
| CD3 | –2.22 | 0.073334 |
| CD4 | –1.69 | 0.132057 |
| IL-4 | –3.43 | 0.379695 |
| ARG1 | 7.69 | 0.007955 |
| CD14 | 2.09 | 0.260868 |
| CD68 | 2.35 | 0.473322 |
| C5aR1 | 3.03 | 0.080240 |
| C3aR1 | 2.25 | 0.084210 |
| IL-13 | 3.84 | 0.051296 |
| Marker | Regulation | p-Value |
|---|---|---|
| TRAP | –2.09 | 0.112708 |
| CATHK | –2.31 | 0.111423 |
| CD55 | 18.29 | 0.000000 |
| C3aR1 | 8.31 | 0.000818 |
| C5aR1 | 3.46 | 0.002609 |
| CD46 | 4.68 | 0.035842 |
| CD59 | 3.03 | 0.052696 |
| ARG1 | 18.72 | 0.000011 |
| CD11b | 3.95 | 0.006903 |
| CD14 | 2.14 | 0.020201 |
| NCF-1 | 7.04 | 0.001291 |
| CD3 | 2.60 | 0.014589 |
| CD4 | 3.28 | 0.006753 |
| CD8b | 1.53 | 0.393394 |
| CD19 | 1.30 | 0.182925 |
| MCSF | 57.55 | 0.000000 |
| IL-13 | 8.11 | 0.024702 |
| PPAR-G | 25.54 | 0.000000 |
| OPG | 13.77 | 0.000687 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trindade, R.; Albrektsson, T.; Galli, S.; Prgomet, Z.; Tengvall, P.; Wennerberg, A. Bone Immune Response to Materials, Part II: Copper and Polyetheretherketone (PEEK) Compared to Titanium at 10 and 28 Days in Rabbit Tibia. J. Clin. Med. 2019, 8, 814. https://doi.org/10.3390/jcm8060814
Trindade R, Albrektsson T, Galli S, Prgomet Z, Tengvall P, Wennerberg A. Bone Immune Response to Materials, Part II: Copper and Polyetheretherketone (PEEK) Compared to Titanium at 10 and 28 Days in Rabbit Tibia. Journal of Clinical Medicine. 2019; 8(6):814. https://doi.org/10.3390/jcm8060814
Chicago/Turabian StyleTrindade, Ricardo, Tomas Albrektsson, Silvia Galli, Zdenka Prgomet, Pentti Tengvall, and Ann Wennerberg. 2019. "Bone Immune Response to Materials, Part II: Copper and Polyetheretherketone (PEEK) Compared to Titanium at 10 and 28 Days in Rabbit Tibia" Journal of Clinical Medicine 8, no. 6: 814. https://doi.org/10.3390/jcm8060814
APA StyleTrindade, R., Albrektsson, T., Galli, S., Prgomet, Z., Tengvall, P., & Wennerberg, A. (2019). Bone Immune Response to Materials, Part II: Copper and Polyetheretherketone (PEEK) Compared to Titanium at 10 and 28 Days in Rabbit Tibia. Journal of Clinical Medicine, 8(6), 814. https://doi.org/10.3390/jcm8060814






