Is Pelvic Plexus Block Superior to Periprostatic Nerve Block for Pain Control during Transrectal Ultrasonography-Guided Prostate Biopsy? A Double-Blind, Randomized Controlled Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Eligibility Criteria for Participants
2.3. Sample Size Estimation and Randomization
2.4. Technique for Biopsy Procedures
2.5. Blinding and Assessment
2.6. Statistical Methodology
3. Results
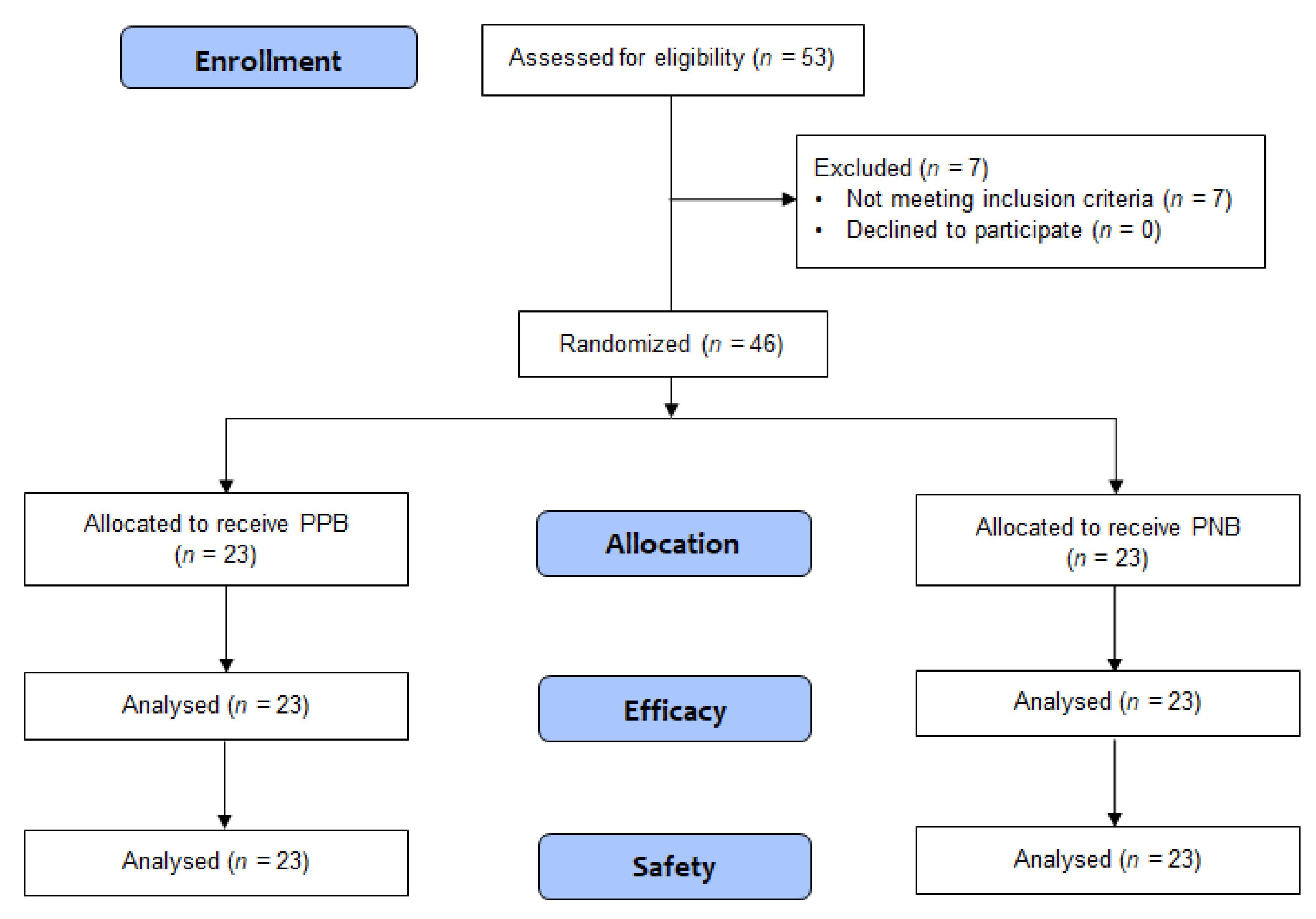
3.1. Patient Flow, Recruitment, and Number Analyzed
3.2. Outcomes
3.3. Complications
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef]
- Patel, A.R.; Jones, J.S. Optimal biopsy strategies for the diagnosis and staging of prostate cancer. Curr. Opin. Urol. 2009, 19, 232–237. [Google Scholar] [CrossRef]
- Yan, P.; Wang, X.Y.; Huang, W.; Zhang, Y. Local anesthesia for pain control during transrectal ultrasound-guided prostate biopsy: A systematic review and meta-analysis. J. Pain Res. 2016, 9, 787–796. [Google Scholar] [CrossRef]
- Li, M.; Wang, Z.; Li, H.; Yang, J.; Rao, K.; Wang, T.; Wang, S.; Liu, J. Local anesthesia for transrectal ultrasound-guided biopsy of the prostate: A meta-analysis. Sci. Rep. 2017, 7, 40421. [Google Scholar] [CrossRef]
- Kaver, I.; Mabjeesh, N.J.; Matzkin, H. Randomized prospective study of periprostatic local anesthesia during transrectal ultrasound-guided prostate biopsy. Urology 2002, 59, 405–408. [Google Scholar] [CrossRef]
- Otunctemur, A.; Dursun, M.; Besiroglu, H.; Can Polat, E.; Cakir, S.S.; Ozbek, E.; Karadeniz, T. The effectivity of periprostatic nerve blockade for the pain control during transrectal ultrasound guided prostate biopsy. Arch. Ital. Urol. Androl. 2013, 85, 69–72. [Google Scholar] [CrossRef]
- Pareek, G.; Armenakas, N.A.; Fracchia, J.A. Periprostatic nerve blockade for transrectal ultrasound guided biopsy of the prostate: A randomized, double-blind, placebo controlled study. J. Urol. 2001, 166, 894–897. [Google Scholar] [CrossRef]
- Lynn, N.N.; Collins, G.N.; Brown, S.C.; O’Reilly, P.H. Periprostatic nerve block gives better analgesia for prostatic biopsy. BJU Int. 2002, 90, 424–426. [Google Scholar] [CrossRef] [PubMed]
- Berger, A.P.; Frauscher, F.; Halpern, E.J.; Spranger, R.; Steiner, H.; Bartsch, G.; Horninger, W. Periprostatic administration of local anesthesia during transrectal ultrasound-guided biopsy of the prostate: A randomized, double-blind, placebo-controlled study. Urology 2003, 61, 585–588. [Google Scholar] [CrossRef]
- Stirling, B.N.; Shockley, K.F.; Carothers, G.G.; Maatman, T.J. Comparison of local anesthesia techniques during transrectal ultrasound-guided biopsies. Urology 2002, 60, 89–92. [Google Scholar] [CrossRef]
- Wu, C.L.; Carter, H.B.; Naqibuddin, M.; Fleisher, L.A. Effect of local anesthetics on patient recovery after transrectal biopsy. Urology 2001, 57, 925–929. [Google Scholar] [CrossRef]
- Akpinar, H.; Tufek, I.; Atug, F.; Esen, E.H.; Kural, A.R. Doppler ultrasonography-guided pelvic plexus block before systematic needle biopsy of the prostate: A prospective randomized study. Urology 2009, 74, 267–271.e261. [Google Scholar] [CrossRef] [PubMed]
- Cantiello, F.; Cicione, A.; Autorino, R.; Cosentino, C.; Amato, F.; Damiano, R. Pelvic plexus block is more effective than periprostatic nerve block for pain control during office transrectal ultrasound guided prostate biopsy: A single center, prospective, randomized, double arm study. J. Urol. 2012, 188, 417–421. [Google Scholar] [CrossRef]
- Jindal, T.; Mukherjee, S.; Sinha, R.K.; Kamal, M.R.; Ghosh, N.; Saha, B.; Mitra, N.; Sharma, P.K.; Mandal, S.N.; Karmakar, D. Transrectal ultrasonography (TRUS)-guided pelvic plexus block to reduce pain during prostate biopsy: A randomised controlled trial. BJU Int. 2015, 115, 892–896. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Nash, P.A.; Bruce, J.E.; Indudhara, R.; Shinohara, K. Transrectal ultrasound guided prostatic nerve blockade eases systematic needle biopsy of the prostate. J. Urol. 1996, 155, 607–609. [Google Scholar] [CrossRef]
- Hollabaugh, R.S., Jr.; Dmochowski, R.R.; Steiner, M.S. Neuroanatomy of the male rhabdosphincter. Urology 1997, 49, 426–434. [Google Scholar] [CrossRef]
- Hergan, L.; Kashefi, C.; Parsons, J.K. Local anesthetic reduces pain associated with transrectal ultrasound-guided prostate biopsy: A meta-analysis. Urology 2007, 69, 520–525. [Google Scholar] [CrossRef]
- Chung, B.I.; Sommer, G.; Brooks, J.D. Anatomy of the Lower Urinary Tract and Male Genitalia. Available online: https://abdominalkey.com/anatomy-of-the-lower-urinary-tract-and-male-genitalia/ (accessed on 31 January 2019).
- Schunemann, H.J.; Oxman, A.D.; Brozek, J.; Glasziou, P.; Jaeschke, R.; Vist, G.E.; Williams, J.W., Jr.; Kunz, R.; Craig, J.; Montori, V.M.; et al. Grading quality of evidence and strength of recommendations for diagnostic tests and strategies. BMJ 2008, 336, 1106–1110. [Google Scholar] [CrossRef]
- Clark, T.; Berger, U.; Mansmann, U. Sample size determinations in original research protocols for randomised clinical trials submitted to UK research ethics committees: Review. BMJ 2013, 346, f1135. [Google Scholar] [CrossRef]
- Altman, D.G. Statistics and ethics in medical research: III How large a sample? Br. Med. J. 1980, 281, 1336–1338. [Google Scholar] [CrossRef] [PubMed]
- Price, D.D.; Bush, F.M.; Long, S.; Harkins, S.W. A comparison of pain measurement characteristics of mechanical visual analogue and simple numerical rating scales. Pain 1994, 56, 217–226. [Google Scholar] [CrossRef]
- Duncan, G.H.; Bushnell, M.C.; Lavigne, G.J. Comparison of verbal and visual analogue scales for measuring the intensity and unpleasantness of experimental pain. Pain 1989, 37, 295–303. [Google Scholar] [CrossRef]
- Joyce, C.R.; Zutshi, D.W.; Hrubes, V.; Mason, R.M. Comparison of fixed interval and visual analogue scales for rating chronic pain. Eur. J. Clin. Pharmacol. 1975, 8, 415–420. [Google Scholar] [CrossRef]
- Klimek, L.; Bergmann, K.C.; Biedermann, T.; Bousquet, J.; Hellings, P.; Jung, K.; Merk, H.; Olze, H.; Schlenter, W.; Stock, P.; et al. Visual analogue scales (VAS): Measuring instruments for the documentation of symptoms and therapy monitoring in cases of allergic rhinitis in everyday health care: Position Paper of the German Society of Allergology (AeDA) and the German Society of Allergy and Clinical Immunology (DGAKI), ENT Section, in collaboration with the working group on Clinical Immunology, Allergology and Environmental Medicine of the German Society of Otorhinolaryngology, Head and Neck Surgery (DGHNOKHC). Allergo J. Int. 2017, 26, 16–24. [Google Scholar] [PubMed]
| Variable | All | PPB Group | PNB Group | p-Value |
|---|---|---|---|---|
| Number of patients (n) | 46 | 23 | 23 | |
| Age, years | 68.5 ± 5.71 | 67.35 ± 1.28 | 69.65 ± 1.07 | 0.261 |
| Prostate volume, mL | 37.98 ± 15.76 | 37.8 ± 2.88 | 38.14 ± 3.71 | 0.692 |
| PSA level, ng/mL | 9.33 ± 10.14 | 8.47 ± 1.53 | 10.18 ± 2.59 | 0.886 |
| Procedure time, min | 13.15 ± 3.19 | 12.52 ± 3.39 | 13.78 ± 2.91 | 0.154 |
| Pain Scores | All | PPB Group | PNB Group | p-Value |
|---|---|---|---|---|
| VAS-1: local anesthetic procedure | 2.48 ± 1.66 | 2.30 ± 1.52 | 2.65 ± 1.79 | 0.537 |
| VAS-2: probe insertion | 2.61 ± 2.05 | 2.83 ± 2.21 | 2.39 ± 1.90 | 0.569 |
| VAS-3: sampling procedures | 2.85 ± 1.98 | 2.83 ± 2.03 | 2.87 ± 1.96 | 0.867 |
| VAS-4: 15 min post procedures | 1.33 ± 1.46 | 1.39 ± 1.47 | 1.26 ± 1.48 | 0.631 |
| Study | Study Design | Treatment Arms (Number of Patients) | Age (Mean ± SD) | Anesthetic Methods (PPB and PNB) | Intrarectal Lidocaine Gel Instillation | Number of Biopsy Cores | Injection Site of PNB | Time Interval between Anesthesia and Sampling | Sampling VAS (Mean ± SD) | Complications |
|---|---|---|---|---|---|---|---|---|---|---|
| Present study | RCT, double-blind † | (1) PPB (23) (2) PNB (23) | (1) PPB: 67.35 ± 1.28 (2) PNB: 69.65 ± 1.07 | 2% lidocaine (2.5 mL on each side) | Yes | 12 | Base | 5 min | (1) PPB: 2.85 ± 1.98 (2) PNB: 2.87 ± 1.96 | No major complications |
| Akpinar et al. [12] | RCT, double-blind ‡ | (1) PPB (40) (2) PNB (40) | (1) PPB: 64.9 ± 9.3 (2) PNB: 61.7 ± 9.5 | 2% lidocaine (2 mL on each side) | No | 12 | Base | NA | (1) PPB: 2.7 ± 1.95 (2) PNB: 4.97 ± 2.16 | No major complications |
| Cantinello et al. [13] | RCT, double-blind ‡ | (1) PPB (90) (2) PNB (90) | (1) PPB: 63.7 ± 5.4 (2) PNB: 63.2 ± 5.5 | 1% lidocaine + 0.75% naropin (2.5 mL on each side) | Yes | 12 | Base | 5 min | (1) PPB: 2.28 ± 0.84 (2) PNB: 3.37 ± 0.78 | No major complications |
| Jindal et al. [14] | RCT, double-blind ‡ | (1) PPB (47) (2) PNB (46) | (1) PPB: 68.7 ± 9.1 (2) PNB: 67.3 ± 9.5 | 2% lidocaine (2.5 mL on each side) | Yes | 12 | Base | 10 min | (1) PPB: 2.9 ± 0.84 (2) PNB: 4.0 ± 0.85 | PNB group: high-grade fever (1) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, D.K.; Hah, Y.S.; Kim, J.W.; Koo, K.C.; Lee, K.S.; Hong, C.H.; Chung, B.H.; Cho, K.S. Is Pelvic Plexus Block Superior to Periprostatic Nerve Block for Pain Control during Transrectal Ultrasonography-Guided Prostate Biopsy? A Double-Blind, Randomized Controlled Trial. J. Clin. Med. 2019, 8, 557. https://doi.org/10.3390/jcm8040557
Kim DK, Hah YS, Kim JW, Koo KC, Lee KS, Hong CH, Chung BH, Cho KS. Is Pelvic Plexus Block Superior to Periprostatic Nerve Block for Pain Control during Transrectal Ultrasonography-Guided Prostate Biopsy? A Double-Blind, Randomized Controlled Trial. Journal of Clinical Medicine. 2019; 8(4):557. https://doi.org/10.3390/jcm8040557
Chicago/Turabian StyleKim, Do Kyung, Yoon Soo Hah, Jong Won Kim, Kyo Chul Koo, Kwang Suk Lee, Chang Hee Hong, Byung Ha Chung, and Kang Su Cho. 2019. "Is Pelvic Plexus Block Superior to Periprostatic Nerve Block for Pain Control during Transrectal Ultrasonography-Guided Prostate Biopsy? A Double-Blind, Randomized Controlled Trial" Journal of Clinical Medicine 8, no. 4: 557. https://doi.org/10.3390/jcm8040557
APA StyleKim, D. K., Hah, Y. S., Kim, J. W., Koo, K. C., Lee, K. S., Hong, C. H., Chung, B. H., & Cho, K. S. (2019). Is Pelvic Plexus Block Superior to Periprostatic Nerve Block for Pain Control during Transrectal Ultrasonography-Guided Prostate Biopsy? A Double-Blind, Randomized Controlled Trial. Journal of Clinical Medicine, 8(4), 557. https://doi.org/10.3390/jcm8040557





