Can Tumor-Infiltrating Lymphocytes (TILs) Be a Predictive Factor for Lymph Nodes Status in Both Early Stage and Locally Advanced Breast Cancer?
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Cohort
2.1.1. Type of Treatment
2.1.2. TILs Assessment
2.2. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. PST Group
3.3. SST Group
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Veronesi, U.; Cascinelli, N.; Mariani, L.; Greco, M.; Saccozzi, R.; Luini, A.; Aguilar, M.; Marubini, E. Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N. Engl. J. Med. 2002, 347, 1227–1232. [Google Scholar] [CrossRef] [PubMed]
- Veronesi, U.; Paganelli, G.; Viale, G.; Luini, A.; Zurrida, S.; Galimberti, V.; Intra, M.; Veronesi, P.; Maisonneuve, P.; Gatti, G.; et al. Sentinel-lymph-node biopsy as a staging procedure in breast cancer: Update of a randomised controlled study. Lancet Oncol. 2006, 7, 983–990. [Google Scholar] [CrossRef]
- Morrow, M.; Jagsi, R.; McLeod, M.C.; Shumway, D.; Katz, S.J. Surgeon attitudes toward the omission of axillary dissection in early breast cancer. JAMA Oncol. 2018, 4, 1511–1516. [Google Scholar] [CrossRef]
- Giuliano, A.E.; Hunt, K.K.; Ballman, K.V.; Beitsch, P.D.; Whitworth, P.W.; Blumencranz, P.W.; Leitch, A.M.; Saha, S.; McCall, L.M.; Morrow, M. Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: A randomized clinical trial. JAMA 2011, 305, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Kirova, Y.M.; Shan, S.C.; Cai, G.; Ou, D.; Cao, L.; Cai, R.; Chen, J.Y. Different radiation techniques to deliver therapeutic dose to the axilla in patients with sentinel lymph node-positive breast cancer: Doses, techniques challenges and clinical considerations. Cancer Radiother. 2018, 22, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Manca, G.; Rubello, D.; Tardelli, E.; Giammarile, F.; Mazzarri, S.; Boni, G.; Chondrogiannis, S.; Marzola, M.C.; Chiacchio, S.; Ghilli, M.; et al. Sentinel lymph node biopsy in breast cancer: Indications, contraindications, and controversies. Clin. Nucl. Med. 2016, 41, 126–133. [Google Scholar] [CrossRef]
- Takada, K.; Kashiwagi, S.; Goto, W.; Asano, Y.; Takahashi, K.; Fujita, H.; Takashima, T.; Tomita, S.; Hirakawa, K.; Ohira, M. Possibility of avoiding axillary lymph node dissection by immune microenvironment monitoring in preoperative chemotherapy for breast cancer. J. Transl. Med. 2018, 16, 318. [Google Scholar] [CrossRef]
- Hanahan, D.; Coussens, L.M. Accessories to the crime: Functions of cells recruited to the tumor microenvironment. Cancer Cell 2012, 21, 309–322. [Google Scholar] [CrossRef]
- Galluzzi, L.; Zitvogel, L.; Kroemer, G. Immunological mechanisms underneath the efficacy of cancer therapy. cancer immunology research. Cancer Immunol. Res. 2016, 4, 895–902. [Google Scholar] [CrossRef]
- Altan, M.; Kidwell, K.M.; Pelekanou, V.; Carvajal-Hausdorf, D.E.; Schalper, K.A.; Toki, M.I.; Thomas, D.G.; Sabel, M.S.; Hayes, D.F.; Rimm, D.L. Association of B7-H4, PD-L1, and tumor infiltrating lymphocytes with outcomes in breast cancer. NPJ Breast Cancer. 2018, 4, 40. [Google Scholar] [CrossRef]
- Pelekanou, V.; Carvajal-Hausdorf, D.E.; Altan, M.; Wasserman, B.; Carvajal-Hausdorf, C.; Wimberly, H.; Brown, J.; Lannin, D.; Pusztai, L.; Rimm, D.L. Effect of neoadjuvant chemotherapy on tumor-infiltrating lymphocytes and PD-L1 expression in breast cancer and its clinical significance. Breast Cancer Res. 2017, 19, 91. [Google Scholar] [CrossRef] [PubMed]
- Park, I.H.; Kong, S.Y.; Ro, J.Y.; Kwon, Y.; Kang, J.H.; Mo, H.J.; Jung, S.Y.; Lee, S.; Lee, K.S.; Kang, H.S.; et al. Prognostic implications of tumor-infiltrating lymphocytes in association with programmed death ligand 1 expression in early-stage breast cancer. Clin. Breast Cancer 2016, 16, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.R.; Glont, S.E.; Blows, F.M.; Provenzano, E.; Dawson, S.J.; Liu, B.; Hiller, L.; Dunn, J.; Poole, C.J.; Bowden, S.; et al. PD-L1 protein expression in breast cancer is rare, enriched in basal-like tumours and associated with infiltrating lymphocytes. Ann. Oncol. 2015, 26, 1488–1493. [Google Scholar] [CrossRef] [PubMed]
- Sabatier, R.; Finetti, P.; Mamessier, E.; Adelaide, J.; Chaffanet, M.; Ali, H.R.; Viens, P.; Caldas, C.; Birnbaum, D.; Bertucci, F. Prognostic and predictive value of PDL1 expression in breast cancer. Oncotarget 2015, 6, 5449–5464. [Google Scholar] [CrossRef]
- Baptista, M.Z.; Sarian, L.O.; Derchain, S.F.; Pinto, G.A.; Vassallo, J. Prognostic significance of PD-L1 and PD-L2 in breast cancer. Hum. Pathol. 2016, 47, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Van Berckelaer, C.; Rypens, C.; van Dam, P.; Pouillon, L.; Parizel, M.; Schats, K.A.; Kockx, M.; Tjalma, W.A.A.; Vermeulen, P.; van Laere, S.; et al. Infiltrating stromal immune cells in inflammatory breast cancer are associated with an improved outcome and increased PD-L1 expression. Breast Cancer Res. 2019, 21, 28. [Google Scholar] [CrossRef] [PubMed]
- Salgado, R.; Denkert, C.; Demaria, S.; Sirtaine, N.; Klauschen, F.; Pruneri, G.; Wienert, S.; Van den Eynden, G.; Baehner, F.L.; Penault-Llorca, F.; et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: Recommendations by an International TILs Working Group 2014. Ann. Oncol. 2015, 26, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Dieci, M.V.; Radosevic-Robin, N.; Fineberg, S.; van den Eynden, G.; Ternes, N.; Penault-Llorca, F.; Pruneri, G.; D’Alfonso, T.M.; Demaria, S.; Castaneda, C.; et al. Update on tumor-infiltrating lymphocytes (TILs) in breast cancer, including recommendations to assess TILs in residual disease after neoadjuvant therapy and in carcinoma in situ: A report of the International Immuno-Oncology Biomarker Working Group on Breast Cancer. Semin. Cancer Biol. 2018, 52, 16–25. [Google Scholar]
- Soysal, S.D.; Tzankov, A.; Muenst, S.E. Role of the Tumor Microenvironment in Breast Cancer. Pathobiology 2015, 82, 142–152. [Google Scholar] [CrossRef]
- Aaltomaa, S.; Lipponen, P.; Eskelinen, M.; Kosma, V.M.; Marin, S.; Alhava, E.; Syrjänen, K. Lymphocyte infiltrates as a prognostic variable in female breast cancer. Eur. J. Cancer 1992, 28, 859–864. [Google Scholar] [CrossRef]
- Kojima, Y.A.; Wang, X.; Sun, H.; Compton, F.; Covinsky, M.; Zhang, S. Reproducible evaluation of tumor-infiltrating lymphocytes (TILs) using the recommendations of International TILs Working Group 2014. Ann. Diagn. Pathol. 2018, 35, 77–79. [Google Scholar] [CrossRef] [PubMed]
- Tramm, T.; Di Caterino, T.; Jylling, A.B.; Lelkaitis, G.; Lænkholm, A.V.; Ragó, P.; Tabor, T.P.; Talman, M.M.; Vouza, E.; Scientific Committee of Pathology, Danish Breast Cancer Group (DBCG). Standardized assessment of tumor-infiltrating lymphocytes in breast cancer: An evaluation of inter-observer agreement between pathologists. Acta Oncol. 2018, 57, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Hendry, S.; Salgado, R.; Gevaert, T.; Russell, P.A.; John, T.; Thapa, B.; Christie, M.; van de Vijver, K.; Estrada, M.V.; Gonzalez-Ericsson, P.I.; et al. assessing tumor-infiltrating lymphocytes in solid tumors: A practical review for pathologists and proposal for a standardized method from the International Immunooncology Biomarkers Working Group: Part 1: Assessing the host immune response, TILs in invasive breast carcinoma and ductal carcinoma in situ, metastatic tumor deposits and areas for further research. Adv. Anat. Pathol. 2017, 24, 235–251. [Google Scholar]
- Azimi, F.; Scolyer, R.A.; Rumcheva, P.; Moncrieff, M.; Murali, R.; McCarthy, S.W.; Saw, R.P.; Thompson, J.F. Tumor-infiltrating lymphocyte grade is an independent predictor of sentinel lymph node status and survival in patients with cutaneous melanoma. J. Clin. Oncol. 2012, 30, 2678–2683. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, H.; Mori-Shiraishi, K.; Nakajima, M.; Ueki, H. Defining lymphocyte-predominant breast cancer by the proportion of lymphocyte-rich stroma and its significance in routine histopathological diagnosis. Pathol. Int. 2015, 65, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Chen, X.; Fei, X.; Huang, O.; Wu, J.; Zhu, L.; He, J.; Chen, W.; Li, Y.; Shen, K. Changes of tumor infiltrating lymphocytes after core needle biopsy and the prognostic implications in early stage breast cancer: A retrospective study. Cancer Res. Treat. 2019. [Google Scholar] [CrossRef]
- König, L.; Mairinger, F.D.; Hoffmann, O.; Bittner, A.K.; Schmid, K.W.; Kimmig, R.; Kasimir-Bauer, S.; Bankfalvi, A. Dissimilar patterns of tumor-infiltrating immune cells at the invasive tumor front and tumor center are associated with response to neoadjuvant chemotherapy in primary breast cancer. BMC Cancer 2019, 19, 120. [Google Scholar] [CrossRef]
- Hwang, H.W.; Jung, H.; Hyeon, J.; Park, Y.H.; Ahn, J.S.; Im, Y.H.; Nam, S.J.; Kim, S.W.; Lee, J.E.; Yu, J.H.; et al. A nomogram to predict pathologic complete response (pCR) and the value of tumor-infiltrating lymphocytes (TILs) for prediction of response to neoadjuvant chemotherapy (NAC) in breast cancer patients. Breast Cancer Res. Treat 2019, 173, 255–266. [Google Scholar] [CrossRef]
- Stanton, S.E.; Adams, S.; Disis, M.L. Variation in the incidence and magnitude of tumor-infiltrating lymphocytes in breast cancer subtypes: A systematic review. JAMA Oncol. 2016, 2, 1354–1360. [Google Scholar] [CrossRef]
- Ruan, M.; Tian, T.; Rao, J.; Xu, X.; Yu, B.; Yang, W.; Shui, R. Predictive value of tumor-infiltrating lymphocytes to pathological complete response in neoadjuvant treated triple-negative breast cancers. Diagn. Pathol. 2018, 13, 66. [Google Scholar] [CrossRef]
- Yang, X.; Rao, J.; Yang, W.; Shui, R. Evaluation of the predictive and prognostic values of stromal tumor-infiltrating lymphocytes in her2-positive breast cancers treated with neoadjuvant chemotherapy. Target Oncol. 2018, 13, 757–767. [Google Scholar] [CrossRef]
- Eryilmaz, M.K.; Mutlu, H.; Ünal, B.; Salim, D.K.; Musri, F.Y.; Coşkun, H.Ş. The importance of stromal and intratumoral tumor lymphocyte infiltration for pathologic complete response in patients with locally advanced breast cancer. J. Cancer Res. Ther. 2018, 14, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Dieci, M.V.; Conte, P.; Bisagni, G.; Brandes, A.A.; Frassoldati, A.; Cavanna, L.; Musolino, A.; Giotta, F.; Rimanti, A.; Garrone, O.; et al. Association of tumor-infiltrating lymphocytes with distant disease-free survival in the ShortHER randomized adjuvant trial for patients with early HER2+ breast cancer. Ann. Oncol. 2019. [Google Scholar] [CrossRef]
- Denkert, C.; von Minckwitz, G.; Darb-Esfahani, S.; Lederer, B.; Heppner, B.I.; Weber, K.E.; Budczies, J.; Huober, J.; Klauschen, F.; Furlanetto, J.; et al. Tumour-infiltrating lymphocytes and prognosis in different subtypes of breast cancer: A pooled analysis of 3771 patients treated with neoadjuvant therapy. Lancet Oncol. 2018, 19, 40–50. [Google Scholar] [CrossRef]
- Hatzis, C.; Symmans, W.F.; Zhang, Y.; Gould, R.E.; Moulder, S.L.; Hunt, K.K.; Abu-Khalaf, M.; Hofstatter, E.W.; Lannin, D.; Chagpar, A.B.; et al. Relationship between complete pathologic response to neoadjuvant chemotherapy and survival in triple-negative breast cancer. Clin. Cancer Res. 2016, 22, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Spring, L.; Greenup, R.; Niemierko, A.; Schapira, L.; Haddad, S.; Jimenez, R.; Coopey, S.; Taghian, A.; Hughes, K.S.; Isakoff, S.J.; et al. Pathologic complete response after neoadjuvant chemotherapy and long-term outcomes among young women with breast cancer. J. Natl. Compr. Cancer Netw. 2017, 15, 1216–1223. [Google Scholar] [CrossRef]
- Symmans, W.F.; Wei, C.; Gould, R.; Yu, X.; Zhang, Y.; Liu, M.; Walls, A.; Bousamra, A.; Ramineni, M.; Sinn, B.; et al. Long-term prognostic risk after neoadjuvant chemotherapy associated with residual cancer burden and breast cancer subtype. J. Clin. Oncol. 2017, 35, 1049–1060. [Google Scholar] [CrossRef]
- Asano, Y.; Kashiwagi, S.; Goto, W.; Takada, K.; Takahashi, K.; Hatano, T.; Noda, S.; Takashima, T.; Onoda, N.; Tomita, S.; et al. Prediction of survival after neoadjuvant chemotherapy for breast cancer by evaluation of tumor-infiltrating lymphocytes and residual cancer burden. BMC Cancer 2017, 17, 888. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, C.H.; Lee, Y.; Lee, J.H.; Chae, Y.S. Tumour infiltrating lymphocytes are predictors of lymph node metastasis in early gastric cancers. Pathology 2017, 49, 589–595. [Google Scholar] [CrossRef]
- Pehlivan, F.S.; Sivrikoz, O.N.; Dag, F.; Kececi, S.D.; Sanal, S.M. Distribution of CXCR4 and tumour-infiltrating lymphocytes in breast cancer subtypes; their relationship with each other, axillary lymph node involvement, and other prognostic indicators. Pol. J. Pathol. 2018, 69, 335–341. [Google Scholar] [CrossRef]
- Moossdorff, M.; Nakhlis, F.; Hu, J.; Barry, W.T.; Losk, K.; Haskett, C.; Smidt, M.L.; King, T.A. The potential impact of AMAROS on the management of the axilla in patients with clinical t1-2n0 breast cancer undergoing primary total mastectomy. Ann. Surg. Oncol. 2018, 25, 2612–2619. [Google Scholar] [CrossRef] [PubMed]
- Grossmith, S.; Nguyen, A.; Hu, J.; Plichta, J.K.; Nakhlis, F.; Cutone, L.; Dominici, L.; Golshan, M.; Duggan, M.; Carter, K.; et al. Multidisciplinary management of the axilla in patients with cT1-T2 N0 breast cancer undergoing primary mastectomy: Results from a prospective single-institution series. Ann. Surg. Oncol. 2018, 25, 3527–3534. [Google Scholar] [CrossRef] [PubMed]
- He, K.W.; Sun, J.J.; Liu, Z.B.; Zhuo, P.Y.; Ma, Q.H.; Liu, Z.Y.; Yu, Z.Y. Prognostic significance of lymphatic vessel invasion diagnosed by D2-40 in Chinese invasive breast cancers. Medicine 2017, 96. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhang, D.; Gong, M.; Wen, L.; Liao, C.; Zou, L. High lymphatic vessel density and presence of lymphovascular invasion both predict poor prognosis in breast cancer. BMC Cancer 2017, 17, 335. [Google Scholar] [CrossRef] [PubMed]
- Schoppmann, S.F.; Bayer, G.; Aumayr, K.; Taucher, S.; Geleff, S.; Rudas, M.; Kubista, E.; Hausmaninger, H.; Samonigg, H.; Gnant, M.; et al. Prognostic value of lymphangiogenesis and lymphovascular invasion in invasive breast cancer. Ann. Surg. 2004, 240, 306–312. [Google Scholar] [CrossRef]
- Sønderstrup, I.M.H.; Jensen, M.B.; Ejlertsen, B.; Eriksen, J.O.; Gerdes, A.M.; Kruse, T.A.; Larsen, M.J.; Thomassen, M.; Laenkholm, A.V. Evaluation of tumor-infiltrating lymphocytes and association with prognosis in BRCA-mutated breast cancer. Acta Oncol. 2019, 58, 363–370. [Google Scholar] [CrossRef]
- Takada, K.; Kashiwagi, S.; Asano, Y.; Goto, W.; Takahashi, K.; Fujita, H.; Takashima, T.; Tomita, S.; Hirakawa, K.; Ohira, M. Clinical verification of the relationship between smoking and the immune microenvironment of breast cancer. J. Transl. Med. 2019, 17, 13. [Google Scholar] [CrossRef]
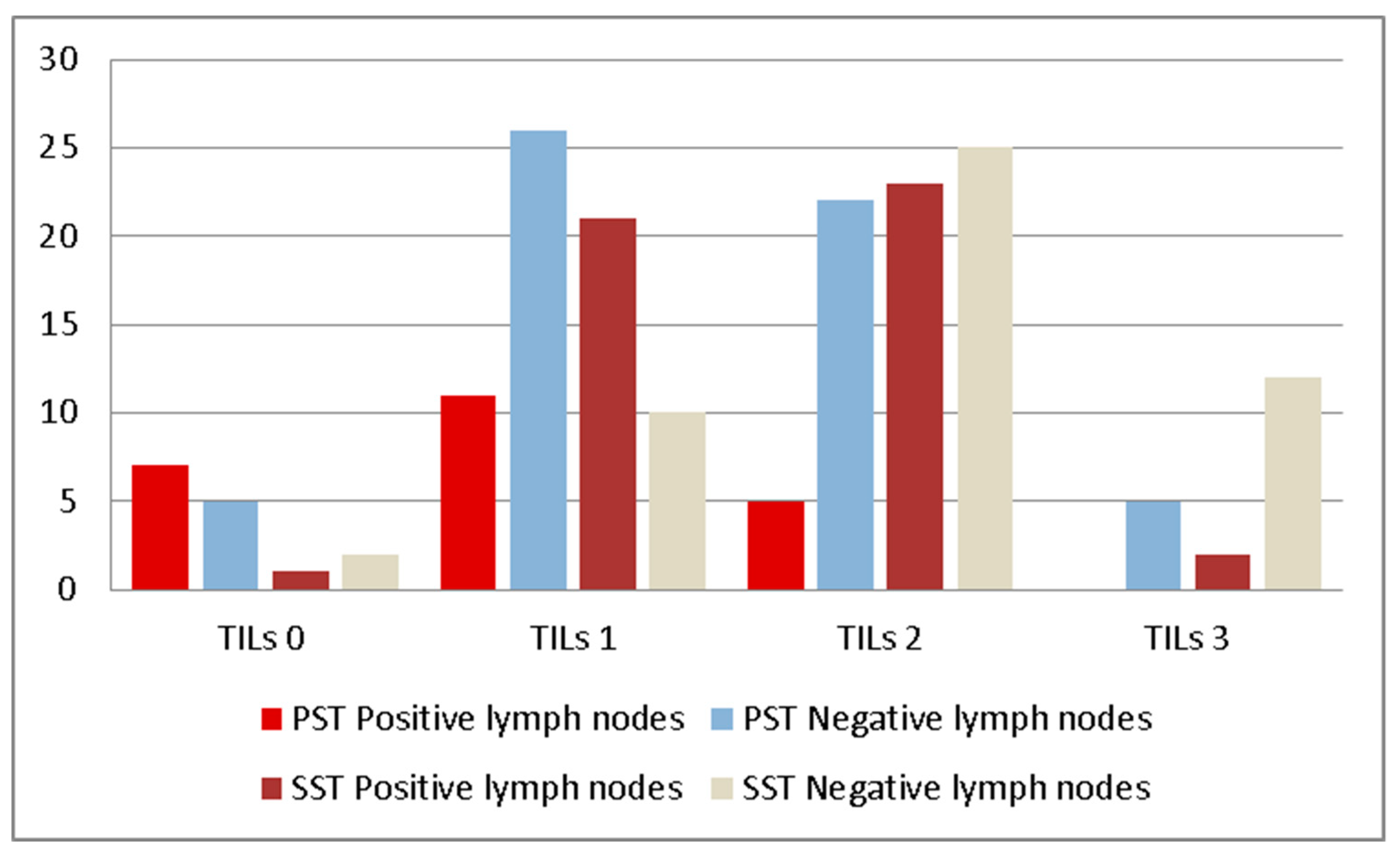
| Group PST (n = 76) | Group SST (n = 96) | ||
|---|---|---|---|
| Age–median (range) in years | 51.81 (30–73) | 54.93 (27–76) | |
| Tumor size (cT) | T1 | 57 (75.00%) | 8 (8.33%) |
| T2 | 19 (25.00%) | 42 (43.75%) | |
| T3 | - | 25 (26.04%) | |
| T4 | - | 21 (21.87%) | |
| Lymph node status (cN) | N0 | 76 (100%) | 3 (3.12%) |
| N1 | - | 65 (67.70%) | |
| N2 | - | 25 (26.04%) | |
| N3 | - | 3 (3.12%) | |
| Histological subtype | Ductal | 58 (76.31%) | 88 (91.66%) |
| Lobular | 4 (5.26%) | 3 (3.12%) | |
| Mucinous | 4 (5.26%) | 4 (4.16%) | |
| Other | 10 (13.15%) | 1 (1.04%) | |
| Estrogen receptors positivity | 72 (94.73% | 62 (64.58%) | |
| Progesterone receptor positivity | 64 (84.21%) | 52 (54.16%) | |
| HER2 positivity | 11 (14.47%) | 29 (30.20%) | |
| Ki67 > 20% | 23 (30.26%) | 60 (62.50%) | |
| Intrinsic subtype (IHC4) | Luminal A | 45 (59.21%) | 23 (23.95%) |
| Luminal B HER2 - | 16 (21.05%) | 19 (19.79%) | |
| Luminal B HER2+ | 11 (14.47%) | 20 (20.83%) | |
| HER2 over expression | 0 (0.00%) | 9 (9.37%) | |
| Triple negative | 4 (5.26%) | 25 (26.04%) | |
| Lymphovascular invasion | 10 (13.15%) | 36 (37.50% | |
| TILs | 0 | 12 (15.78%) | 3 (3.12%) |
| 1 | 37 (48.68%) | 31 (32.29%) | |
| 2 | 22 (28.94%) | 48 (50.00%) | |
| 3 | 5 (6.57%) | 14 (14.58%) | |
| TILs | Positive SLN/no. of Cases n (%) | Type of Metastasis | Positive Nodes after ALND/ no. of Cases n (%) |
|---|---|---|---|
| 0 | 7/12 (58.33%) | 5M, 1m, 1 ITC | 3/12 (25.00%) |
| 1 | 11/37 (29.72%) | 9M, 1 m, 1 ITC | 3/37 (8.10%) |
| 2 | 5/22 (22.72%) | 4M, 1 m | 0/22 (0.00%) |
| 3 | 0/5 (0.00%) | - | - |
| TILs | Positive Nodes after ALND/ no. of Cases n (%) | Type of Metastasis |
|---|---|---|
| 0 | 1/3 (33.33%) | 1M |
| 1 | 21/31 (67.74%) | 20M, 1 m |
| 2 | 23/48 (47.91%) | 20M, 2 m, 1 ITC |
| 3 | 2/14 (14.28%) | 1M, 1 m |
| TILs | pCR/no. of Cases n (%) | MPG (Mean) | RCB (Mean) |
|---|---|---|---|
| 0 | 1/3 (33.33%) | 4 | 1.33 |
| 1 | 7/31 (22.58%) | 3.03 | 2.25 |
| 2 | 7/48 (14.58%) | 3.22 | 1.79 |
| 3 | 10/14 (71.42%) | 4.42 | 0.50 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caziuc, A.; Schlanger, D.; Amarinei, G.; Dindelegan, G.C. Can Tumor-Infiltrating Lymphocytes (TILs) Be a Predictive Factor for Lymph Nodes Status in Both Early Stage and Locally Advanced Breast Cancer? J. Clin. Med. 2019, 8, 545. https://doi.org/10.3390/jcm8040545
Caziuc A, Schlanger D, Amarinei G, Dindelegan GC. Can Tumor-Infiltrating Lymphocytes (TILs) Be a Predictive Factor for Lymph Nodes Status in Both Early Stage and Locally Advanced Breast Cancer? Journal of Clinical Medicine. 2019; 8(4):545. https://doi.org/10.3390/jcm8040545
Chicago/Turabian StyleCaziuc, Alexandra, Diana Schlanger, Giorgiana Amarinei, and George Calin Dindelegan. 2019. "Can Tumor-Infiltrating Lymphocytes (TILs) Be a Predictive Factor for Lymph Nodes Status in Both Early Stage and Locally Advanced Breast Cancer?" Journal of Clinical Medicine 8, no. 4: 545. https://doi.org/10.3390/jcm8040545
APA StyleCaziuc, A., Schlanger, D., Amarinei, G., & Dindelegan, G. C. (2019). Can Tumor-Infiltrating Lymphocytes (TILs) Be a Predictive Factor for Lymph Nodes Status in Both Early Stage and Locally Advanced Breast Cancer? Journal of Clinical Medicine, 8(4), 545. https://doi.org/10.3390/jcm8040545





