Association between Interleukin-6 Levels and Perioperative Fatigue in Gastric Adenocarcinoma Patients
Abstract
1. Introduction
2. Methods
2.1. Instruments
2.2. Cytokine Markers and Biochemistry Assessments
2.3. Medical and Demographic Data
2.4. Statistical Analysis
3. Results
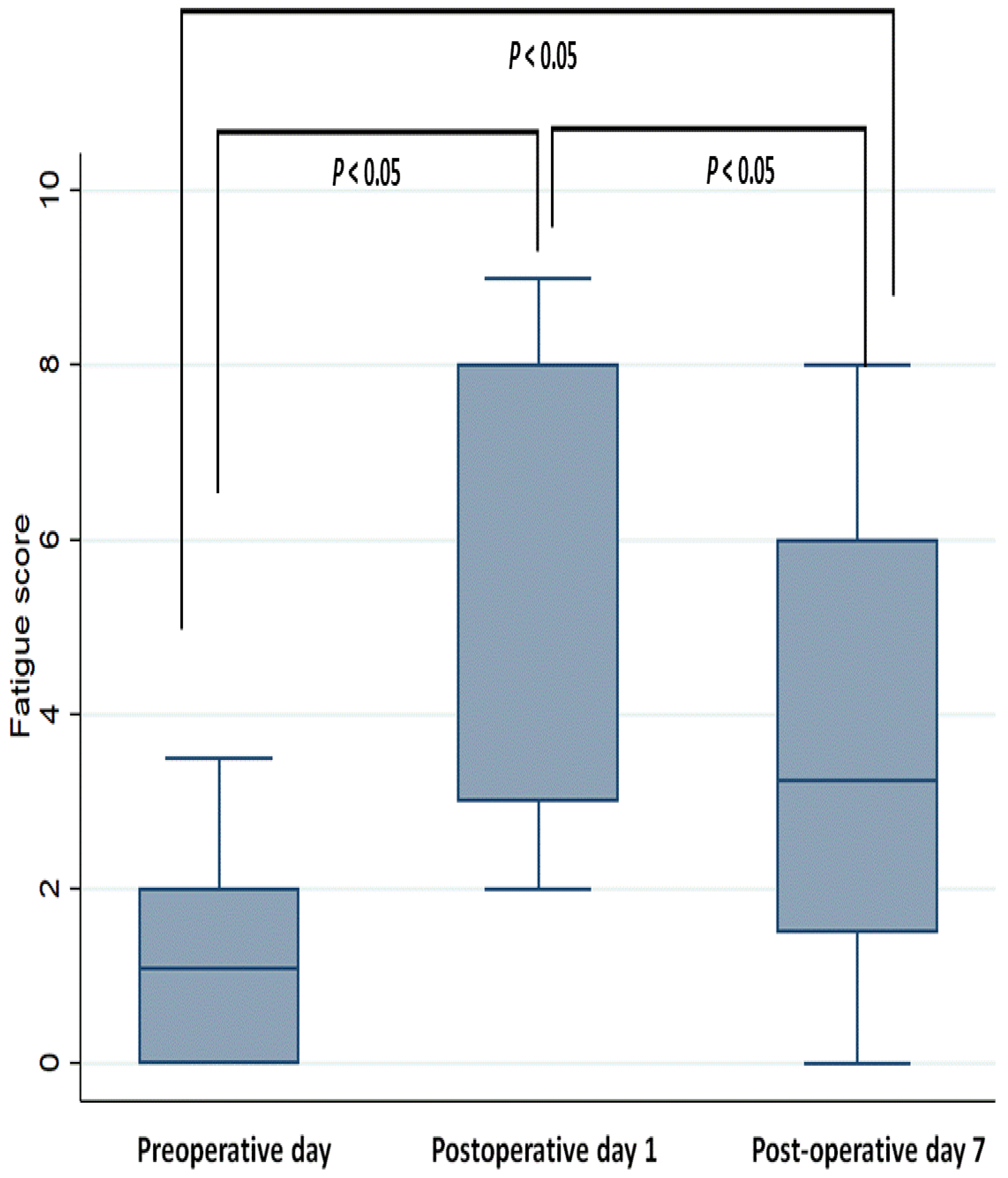
3.1. Perioperative Changes in Fatigue Scores
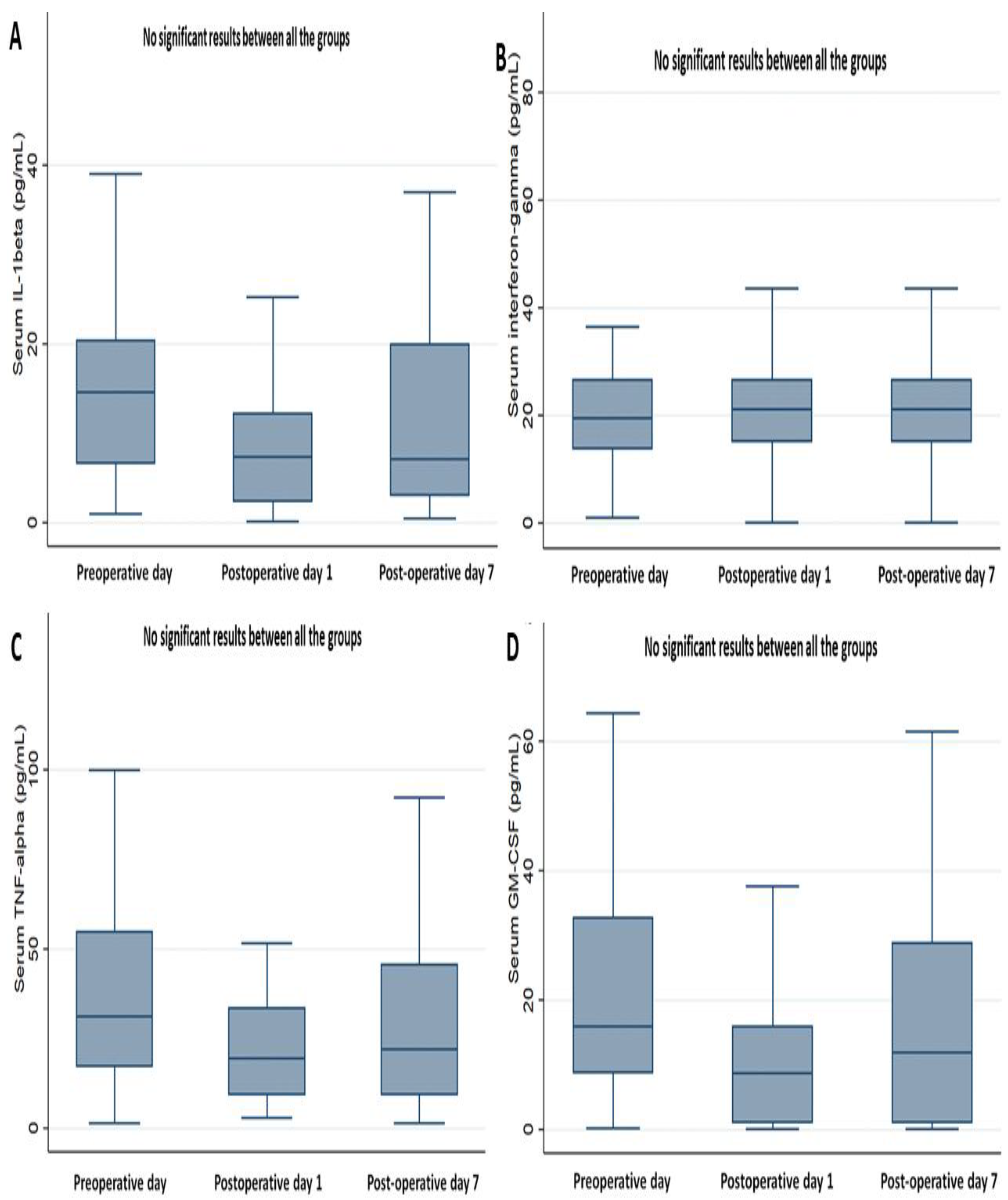
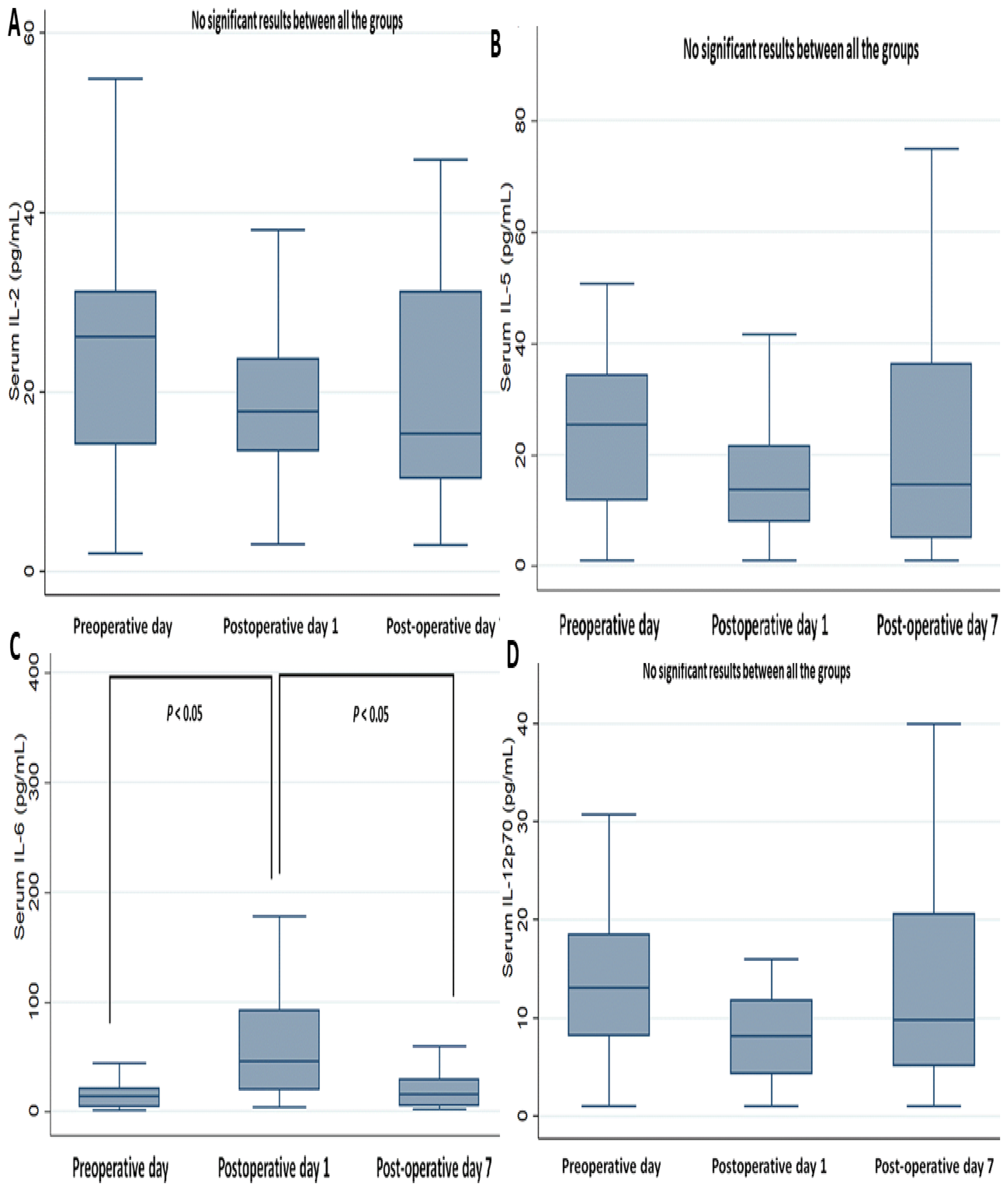
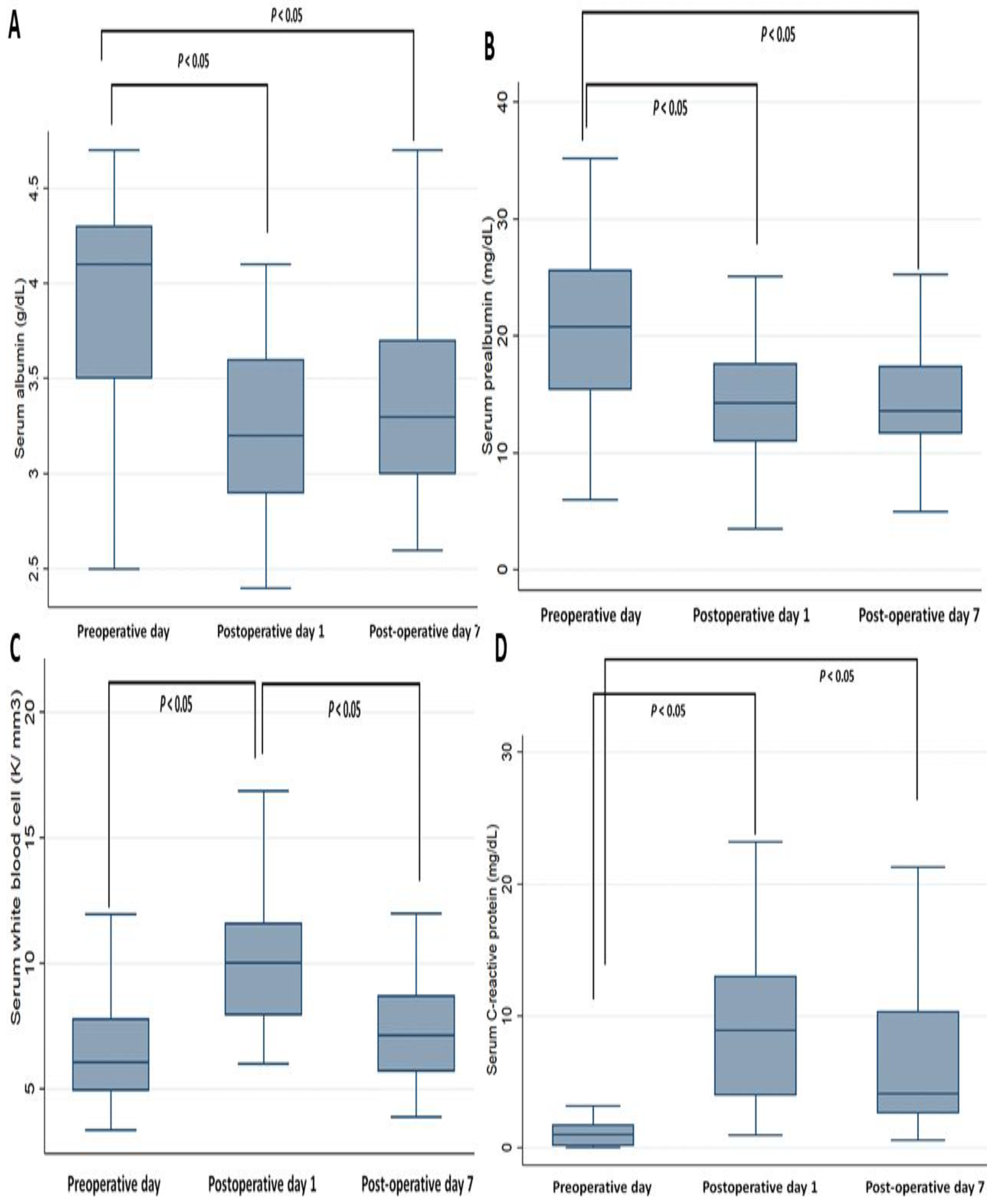
3.2. Perioperative Changes in Cytokines Markers and Biochemistry Data
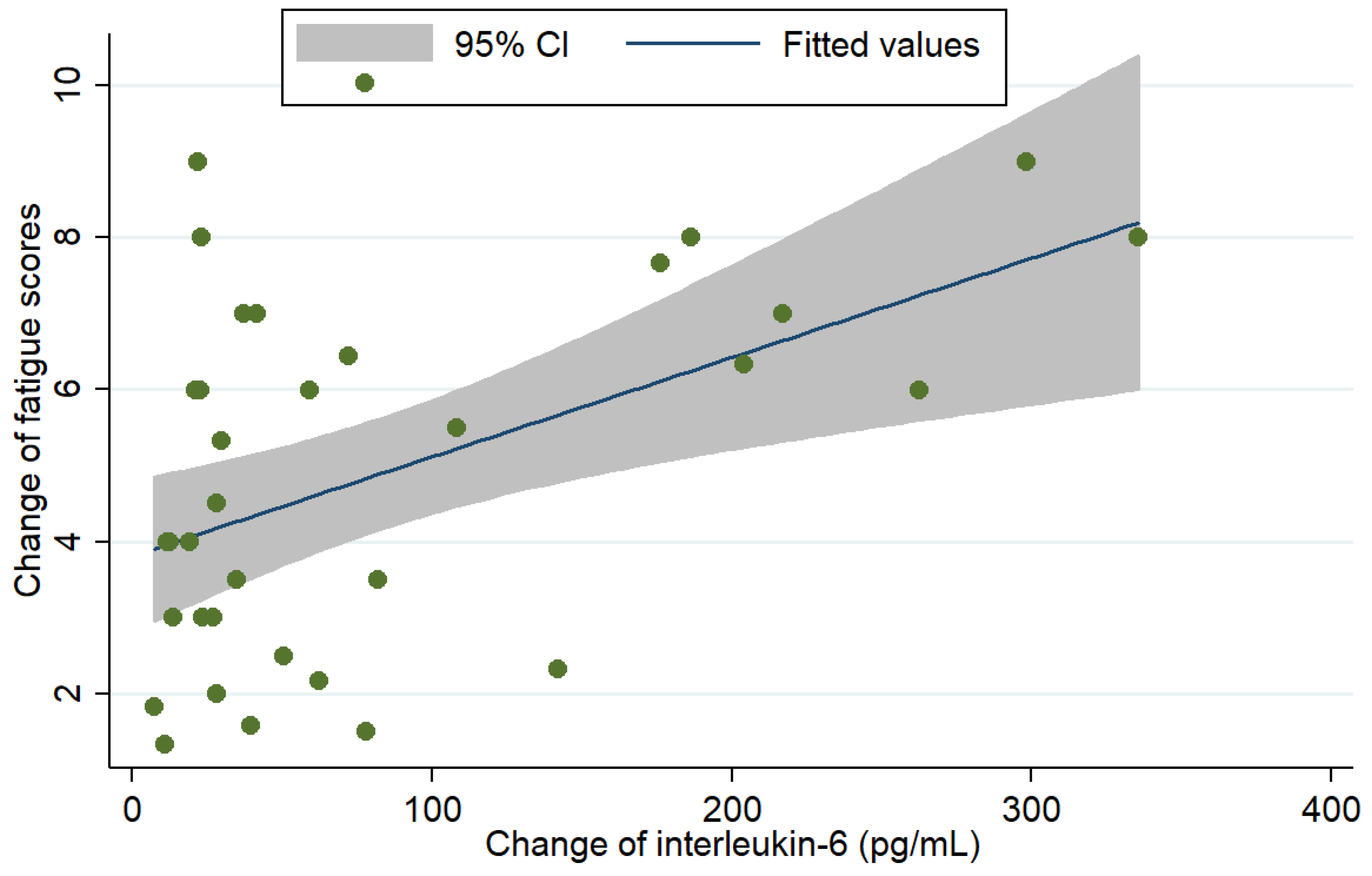
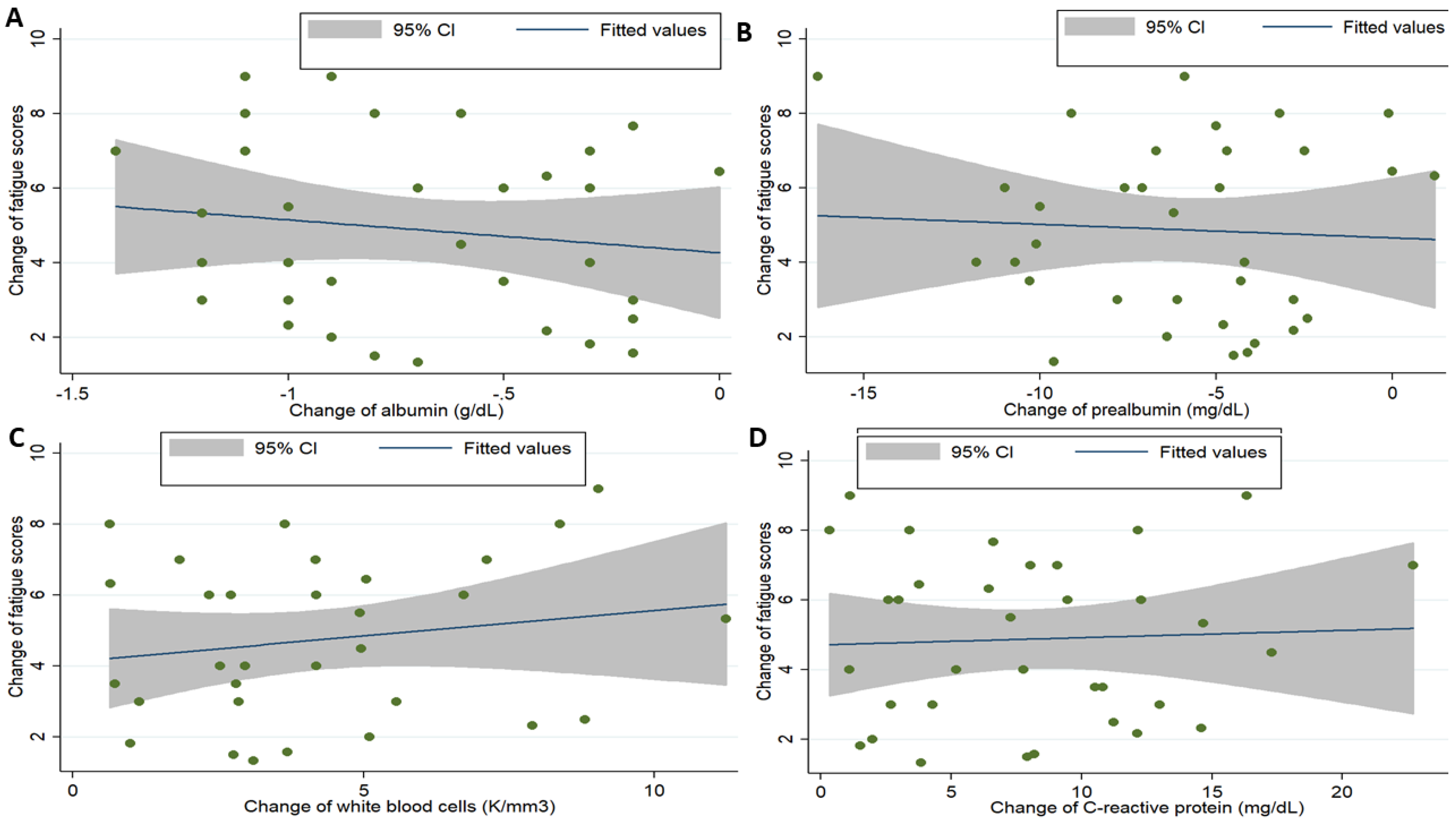
3.3. Association between Cytokines, Clinicodemographics, Biochemistry Profiles, and Fatigue
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Globocan 2012: Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012. Available online: http://globocan.iarc.fr/Default.aspx (accessed on 21 June 2012).
- Nitenberg, G.; Raynard, B. Nutritional support of the cancer patient: Issues and dilemmas. Crit. Rev. Oncol. Hematol. 2000, 34, 137–168. [Google Scholar] [CrossRef]
- Birkmeyer, J.D.; Siewers, A.E.; Finlayson, E.V.; Stukel, T.A.; Lucas, F.L.; Batista, I.; Welch, H.G.; Wennberg, D.E. Hospital volume and surgical mortality in the united states. N. Engl. J. Med. 2002, 346, 1128–1137. [Google Scholar] [CrossRef] [PubMed]
- Leaver, H.A.; Craig, S.R.; Yap, P.L.; Walker, W.S. Lymphocyte responses following open and minimally invasive thoracic surgery. Eur. J. Clin. Investig. 2000, 30, 230–238. [Google Scholar] [CrossRef]
- Rubin, G.J.; Cleare, A.; Hotopf, M. Psychological factors in postoperative fatigue. Psychosom. Med. 2004, 66, 959–964. [Google Scholar] [CrossRef]
- Rubin, G.J.; Hotopf, M. Systematic review and meta-analysis of interventions for postoperative fatigue. Br. J. Surg. 2002, 89, 971–984. [Google Scholar] [CrossRef] [PubMed]
- Christensen, T.; Bendix, T.; Kehlet, H. Fatigue and cardiorespiratory function following abdominal surgery. Br. J. Surg. 1982, 69, 417–419. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, D.; Hill, G.L. Postoperative fatigue: A prospective physiological study of patients undergoing major abdominal surgery. Aust. N. Z. J. Surg. 1991, 61, 774–779. [Google Scholar] [CrossRef]
- Schroeder, D.; Hill, G.L. Predicting postoperative fatigue: Importance of preoperative factors. World J. Surg. 1993, 17, 226–231. [Google Scholar] [CrossRef]
- Berger, A.M.; Mooney, K.; Alvarez-Perez, A.; Breitbart, W.S.; Carpenter, K.M.; Cella, D.; Cleeland, C.; Dotan, E.; Eisenberger, M.A.; Escalante, C.P.; et al. Cancer-related fatigue, version 2. 2015. J. Natl. Compr. Cancer Netw. 2015, 13, 1012–1039. [Google Scholar] [CrossRef]
- Goode, E.F.; Smyth, E.C. Immunotherapy for gastroesophageal cancer. J. Clin. Med. 2016, 5, 84. [Google Scholar] [CrossRef]
- Fearon, K.C.; Glass, D.J.; Guttridge, D.C. Cancer cachexia: Mediators, signaling, and metabolic pathways. Cell Metab. 2012, 16, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Onesti, J.K.; Guttridge, D.C. Inflammation based regulation of cancer cachexia. BioMed Res. Int. 2014, 2014, 168407. [Google Scholar] [CrossRef]
- White, J.P.; Puppa, M.J.; Narsale, A.; Carson, J.A. Characterization of the male apcmin/+ mouse as a hypogonadism model related to cancer cachexia. Biol. Open 2013, 2, 1346–1353. [Google Scholar] [CrossRef] [PubMed]
- Bonetto, A.; Aydogdu, T.; Jin, X.; Zhang, Z.; Zhan, R.; Puzis, L.; Koniaris, L.G.; Zimmers, T.A. Jak/stat3 pathway inhibition blocks skeletal muscle wasting downstream of il-6 and in experimental cancer cachexia. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E410–E421. [Google Scholar] [CrossRef]
- Ando, K.; Takahashi, F.; Motojima, S.; Nakashima, K.; Kaneko, N.; Hoshi, K.; Takahashi, K. Possible role for tocilizumab, an anti-interleukin-6 receptor antibody, in treating cancer cachexia. J. Clin. Oncol. 2013, 31, e69–e72. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, I.J.; Stephens, N.A.; MacDonald, A.J.; Skipworth, R.J.; Husi, H.; Greig, C.A.; Ross, J.A.; Timmons, J.A.; Fearon, K.C. Suppression of skeletal muscle turnover in cancer cachexia: Evidence from the transcriptome in sequential human muscle biopsies. Clin. Cancer Res. 2012, 18, 2817–2827. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, O.; Laird, B.; Aass, N.; Lea, T.; Fayers, P.; Kaasa, S.; Klepstad, P. The relationship between pro-inflammatory cytokines and pain, appetite and fatigue in patients with advanced cancer. PLoS ONE 2017, 12, e0177620. [Google Scholar] [CrossRef]
- Bower, J.E.; Ganz, P.A.; Irwin, M.R.; Kwan, L.; Breen, E.C.; Cole, S.W. Inflammation and behavioral symptoms after breast cancer treatment: Do fatigue, depression, and sleep disturbance share a common underlying mechanism? J. Clin. Oncol. 2011, 29, 3517–3522. [Google Scholar] [CrossRef]
- Bower, J.E. Cancer-related fatigue: Links with inflammation in cancer patients and survivors. Brain Behav. Immunity 2007, 21, 863–871. [Google Scholar] [CrossRef]
- Bower, J.E. Cancer-related fatigue—Mechanisms, risk factors, and treatments. Nat Rev. Clin. Oncol. 2014, 11, 597–609. [Google Scholar] [CrossRef]
- Bower, J.E.; Ganz, P.A.; Irwin, M.R.; Castellon, S.; Arevalo, J.; Cole, S.W. Cytokine genetic variations and fatigue among patients with breast cancer. J. Clin. Oncol. 2013, 31, 1656–1661. [Google Scholar] [CrossRef]
- Bower, J.E.; Ganz, P.A.; Tao, M.L.; Hu, W.; Belin, T.R.; Sepah, S.; Cole, S.; Aziz, N. Inflammatory biomarkers and fatigue during radiation therapy for breast and prostate cancer. Clin. Cancer Res. 2009, 15, 5534–5540. [Google Scholar] [CrossRef] [PubMed]
- Kemik, O.; Kemik, A.S.; Begenik, H.; Erdur, F.M.; Emre, H.; Sumer, A.; Purisa, S.; Tuzun, S.; Kotan, C. The relationship among acute-phase responce proteins, cytokines, and hormones in various gastrointestinal cancer types patients with cachectic. Hum. Exp. Toxicol. 2012, 31, 117–125. [Google Scholar] [CrossRef]
- Wang, X.S.; Hao, X.S.; Wang, Y.; Guo, H.; Jiang, Y.Q.; Mendoza, T.R.; Cleeland, C.S. Validation study of the chinese version of the brief fatigue inventory (bfi-c). J. Pain Symptom Manag. 2004, 27, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, T.R.; Wang, X.S.; Cleeland, C.S.; Morrissey, M.; Johnson, B.A.; Wendt, J.K.; Huber, S.L. The rapid assessment of fatigue severity in cancer patients: Use of the brief fatigue inventory. Cancer 1999, 85, 1186–1196. [Google Scholar] [CrossRef]
- Choi, Y.N.; Kim, Y.A.; Yun, Y.H.; Kim, S.; Bae, J.M.; Kim, Y.W.; Ryu, K.W.; Lee, J.H.; Noh, J.H.; Sohn, T.S. Suicide ideation in stomach cancer survivors and possible risk factors. Supportive Care Cancer 2014, 22, 331–337. [Google Scholar] [CrossRef]
- Hwang, I.C.; Yun, Y.H.; Kim, Y.W.; Ryu, K.W.; Kim, Y.A.; Kim, S.; Bae, J.M.; Noh, J.H.; Sohn, T.S. Factors related to clinically relevant fatigue in disease-free stomach cancer survivors and expectation-outcome consistency. Supportive Care Cancer 2014, 22, 1453–1460. [Google Scholar] [CrossRef]
- Quan, H.; Li, B.; Couris, C.M.; Fushimi, K.; Graham, P.; Hider, P.; Januel, J.M.; Sundararajan, V. Updating and validating the charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. Am. J. Epidemiol. 2011, 173, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Stratton, R.J.; Hackston, A.; Longmore, D.; Dixon, R.; Price, S.; Stroud, M.; King, C.; Elia, M. Malnutrition in hospital outpatients and inpatients: Prevalence, concurrent validity and ease of use of the ‘malnutrition universal screening tool’ (‘must’) for adults. Br. J. Nutr. 2004, 92, 799–808. [Google Scholar] [CrossRef]
- Wang, J.; Dang, P.; Raut, C.P.; Pandalai, P.K.; Maduekwe, U.N.; Rattner, D.W.; Lauwers, G.Y.; Yoon, S.S. Comparison of a lymph node ratio-based staging system with the 7th ajcc system for gastric cancer: Analysis of 18,043 patients from the seer database. Ann. Surg. 2012, 255, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Sommer, J.; Engelowski, E.; Baran, P.; Garbers, C.; Floss, D.M.; Scheller, J. Interleukin-6, but not the interleukin-6 receptor plays a role in recovery from dextran sodium sulfate-induced colitis. Int. J. Mol. Med. 2014, 34, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Narsale, A.A.; Carson, J.A. Role of interleukin-6 in cachexia: Therapeutic implications. Curr. Opin. Supportive Palliat. Care 2014, 8, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Carow, B.; Rottenberg, M.E. Socs3, a major regulator of infection and inflammation. Front. Immunol. 2014, 5, 58. [Google Scholar] [CrossRef]
- White, U.A.; Stephens, J.M. The gp130 receptor cytokine family: Regulators of adipocyte development and function. Curr. Pharm. Des. 2011, 17, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Xu, F.; Lu, T.; Duan, Z.; Zhang, Z. Interleukin-6 signaling pathway in targeted therapy for cancer. Cancer Treat. Rev. 2012, 38, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.P.; Phillips, B.E.; Smith, K.; Atherton, P.J.; Rankin, D.; Selby, A.L.; Liptrot, S.; Lund, J.; Larvin, M.; Rennie, M.J. Effect of tumor burden and subsequent surgical resection on skeletal muscle mass and protein turnover in colorectal cancer patients. Am. J. Clin. Nutr. 2012, 96, 1064–1070. [Google Scholar] [CrossRef]
- Taniguchi, K.; Karin, M. Il-6 and related cytokines as the critical lynchpins between inflammation and cancer. Semin. Immunol. 2014, 26, 54–74. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Naito, T.; Ishida, T.; Kawakami, J. Relationships between oxycodone pharmacokinetics, central symptoms, and serum interleukin-6 in cachectic cancer patients. Eur. J. Clin. Pharm. 2016, 72, 1463–1470. [Google Scholar] [CrossRef]
- Lacina, L.; Brabek, J.; Kral, V.; Kodet, O.; Smetana, K., Jr. Interleukin-6: A molecule with complex biological impact in cancer. Histol. Histopathol. 2019, 34, 125–136. [Google Scholar] [PubMed]
- Suh, S.Y.; Choi, Y.S.; Yeom, C.H.; Kwak, S.M.; Yoon, H.M.; Kim, D.G.; Koh, S.J.; Park, J.; Lee, M.A.; Lee, Y.J.; et al. Interleukin-6 but not tumour necrosis factor-alpha predicts survival in patients with advanced cancer. Supportive Care Cancer 2013, 21, 3071–3077. [Google Scholar] [CrossRef]
- Lippitz, B.E.; Harris, R.A. Cytokine patterns in cancer patients: A review of the correlation between interleukin 6 and prognosis. Oncoimmunology 2016, 5, e1093722. [Google Scholar] [CrossRef] [PubMed]
- Ebihara, N.; Matsuda, A.; Nakamura, S.; Matsuda, H.; Murakami, A. Role of the il-6 classic- and trans-signaling pathways in corneal sterile inflammation and wound healing. Investig. Ophthalmol. Vis. Sci. 2011, 52, 8549–8557. [Google Scholar] [CrossRef] [PubMed]
- Mohandas, H.; Jaganathan, S.K.; Mani, M.P.; Ayyar, M.; Rohini Thevi, G.V. Cancer-related fatigue treatment: An overview. J. Cancer Res. 2017, 13, 916–929. [Google Scholar]
- Tuca, A.; Jimenez-Fonseca, P.; Gascon, P. Clinical evaluation and optimal management of cancer cachexia. Crit. Rev. Oncol. Hematol. 2013, 88, 625–636. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.S.; Li, X.; Cao, Y.Z.; Zhang, C.C.; Yang, F.; Shi, Y.M.; Peng, L.M. Clinical studies on the treatment of cancer cachexia with megestrol acetate plus thalidomide. Chemotherapy 2012, 58, 461–467. [Google Scholar] [CrossRef]
- Ando, K.; Takahashi, F.; Kato, M.; Kaneko, N.; Doi, T.; Ohe, Y.; Koizumi, F.; Nishio, K.; Takahashi, K. Tocilizumab, a proposed therapy for the cachexia of interleukin6-expressing lung cancer. PLoS ONE 2014, 9, e102436. [Google Scholar] [CrossRef]
- Wang, C.H.; Lin, C.Y.; Chen, J.S.; Ho, C.L.; Rau, K.M.; Tsai, J.T.; Chang, C.S.; Yeh, S.P.; Cheng, C.F.; Lai, Y.L. Karnofsky performance status as a predictive factor for cancer-related fatigue treatment with astragalus polysaccharides (pg2) injection—A double blind, multi-center, randomized phase iv study. Cancers 2019, 11, 128. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.N.; Li, S.X. The relationship between nutritional risks and cancer-related fatigue in patients with colorectal cancer fast-track surgery. Cancer Nurs. 2018, 41, E41–E47. [Google Scholar] [CrossRef] [PubMed]
- Hatao, F.; Chen, K.Y.; Wu, J.M.; Wang, M.Y.; Aikou, S.; Onoyama, H.; Shimizu, N.; Fukatsu, K.; Seto, Y.; Lin, M.T. Randomized controlled clinical trial assessing the effects of oral nutritional supplements in postoperative gastric cancer patients. Langenbecks Arch. Surg. 2017, 402, 203–211. [Google Scholar] [CrossRef]
- Weijs, P.J.M.; Mogensen, K.M.; Rawn, J.D.; Christopher, K.B. Protein intake, nutritional status and outcomes in icu survivors: A single center cohort study. J. Clin. Med. 2019, 8, 43. [Google Scholar] [CrossRef]
- Niraula, A.; Witcher, K.G.; Sheridan, J.F.; Godbout, J.P. Interleukin-6 induced by social stress promotes a unique transcriptional signature in the monocytes that facilitate anxiety. Biol. Psychiatry 2019, 85, 679–689. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Value |
|---|---|
| Age, mean (SD) | 68.9 (9.7) |
| Gender | |
| Male | 22 (65%) |
| Female | 12 (35%) |
| Charlson comorbidity index scores | |
| ≤3 | 30 (88%) |
| >3 | 4 (12%) |
| Malnutrition Universal Screening Tool | |
| Low risk | 13 (38%) |
| Medium risk | 10 (30%) |
| High risk | 11 (32%) |
| The American Joint Committee on Cancer stage | |
| I | 7 (21%) |
| II | 3 (9%) |
| III | 13 (38%) |
| IV | 11 (32%) |
| Coefficients | 95% Confidence Interval | P-Value | |
|---|---|---|---|
| Change in IL-6 levels | 0.01 | (0.01, 0.02) | 0.037 |
| Age | 0.03 | (−0.06, 0.11) | 0.470 |
| Male gender (ref: female) | 0.33 | (−1.30, 1.98) | 0.678 |
| Malnutrition risk * (ref: low risk) | |||
| Medium risk | 1.42 | (−0.12, 1.56) | 0.785 |
| High risk | 2.80 | (1.45, 3.52) | 0.041 |
| AJCC stage (ref: stage I) | |||
| II | 2.58 | (−0.87, 6.04) | 0.137 |
| III | 0.97 | (−1.32, 3.27) | 0.392 |
| IV | 1.72 | (−0.57, 4.02) | 0.135 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, J.-M.; Yang, H.-T.; Ho, T.-W.; Shun, S.-C.; Lin, M.-T. Association between Interleukin-6 Levels and Perioperative Fatigue in Gastric Adenocarcinoma Patients. J. Clin. Med. 2019, 8, 543. https://doi.org/10.3390/jcm8040543
Wu J-M, Yang H-T, Ho T-W, Shun S-C, Lin M-T. Association between Interleukin-6 Levels and Perioperative Fatigue in Gastric Adenocarcinoma Patients. Journal of Clinical Medicine. 2019; 8(4):543. https://doi.org/10.3390/jcm8040543
Chicago/Turabian StyleWu, Jin-Ming, Hui-Ting Yang, Te-Wei Ho, Shiow-Ching Shun, and Ming-Tsan Lin. 2019. "Association between Interleukin-6 Levels and Perioperative Fatigue in Gastric Adenocarcinoma Patients" Journal of Clinical Medicine 8, no. 4: 543. https://doi.org/10.3390/jcm8040543
APA StyleWu, J.-M., Yang, H.-T., Ho, T.-W., Shun, S.-C., & Lin, M.-T. (2019). Association between Interleukin-6 Levels and Perioperative Fatigue in Gastric Adenocarcinoma Patients. Journal of Clinical Medicine, 8(4), 543. https://doi.org/10.3390/jcm8040543





