T-Regulatory Cells Confer Increased Myelination and Stem Cell Activity after Stroke-Induced White Matter Injury
Abstract
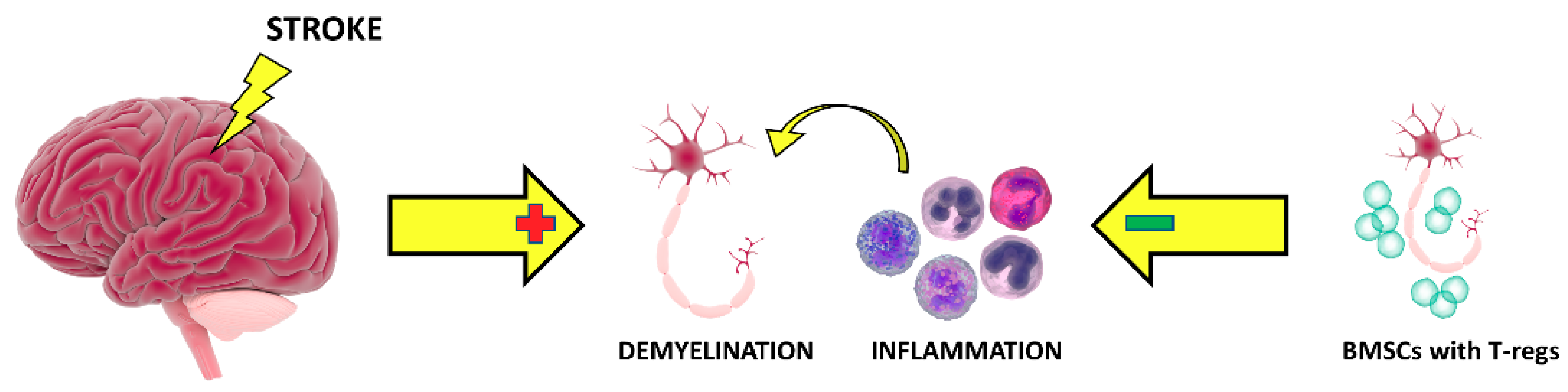
1. Introduction
2. Materials and Methods
2.1. Groups
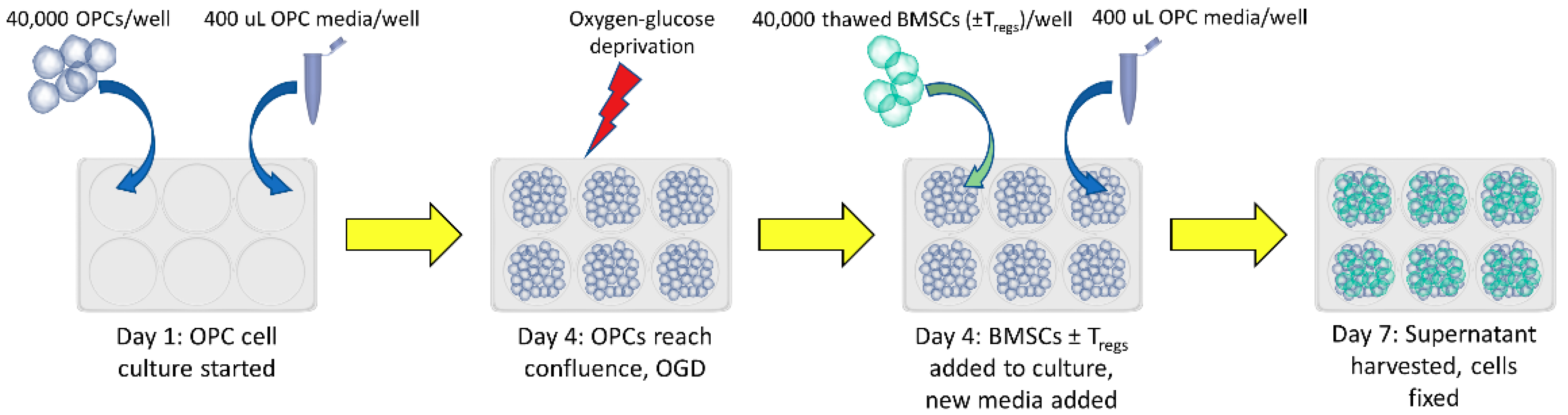
2.2. Cell Culture
2.3. Treg Depletion
2.4. Measurement of Extracellular Cytokines: IL-6, FGF-β
2.5. Immunocytochemistry
2.6. Statistics
3. Results
3.1. OGD Resulted in Decreased Cell Viability
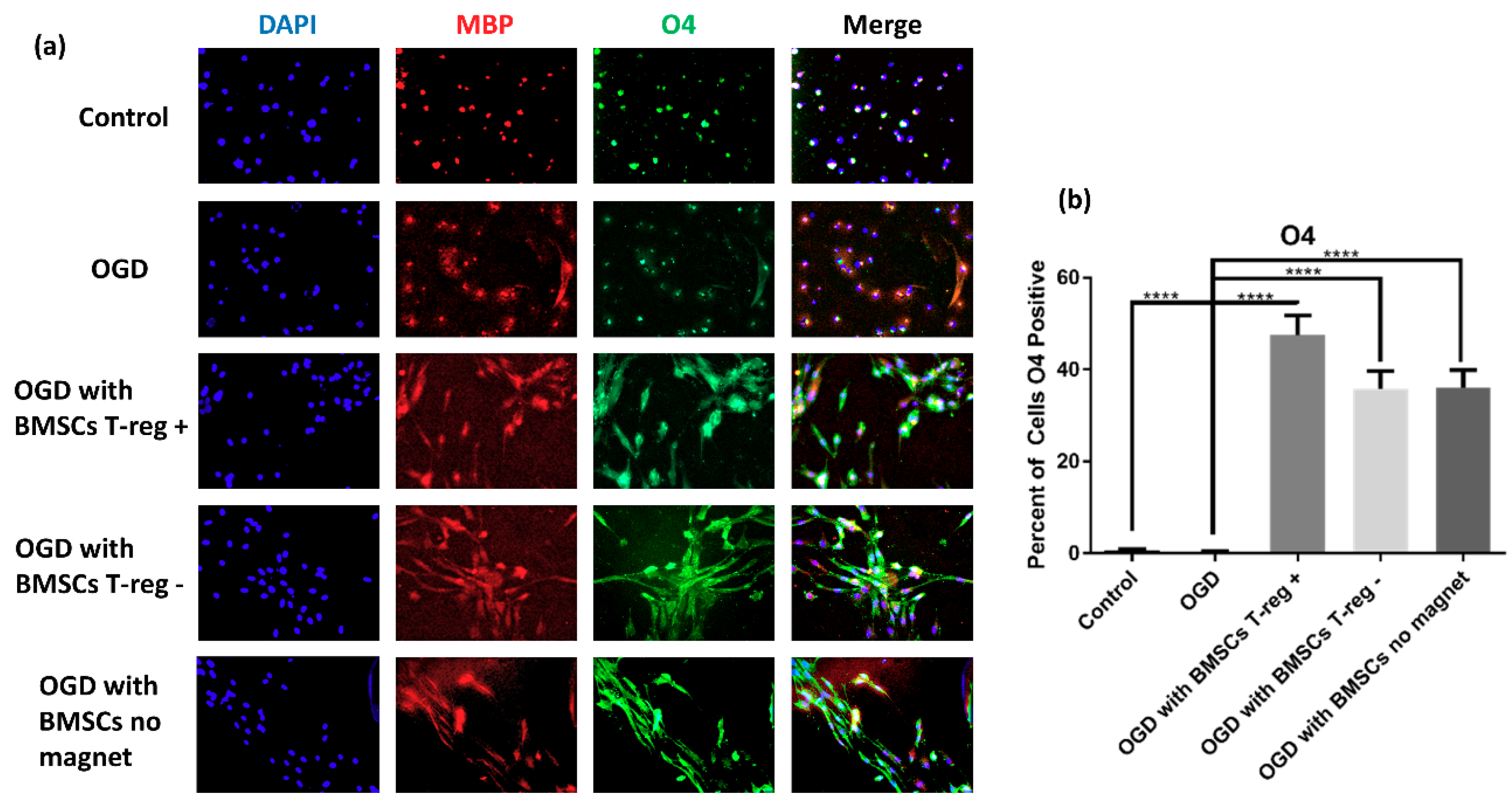
3.2. OPCs Show a More Mature Oligodendrocyte Phenotype
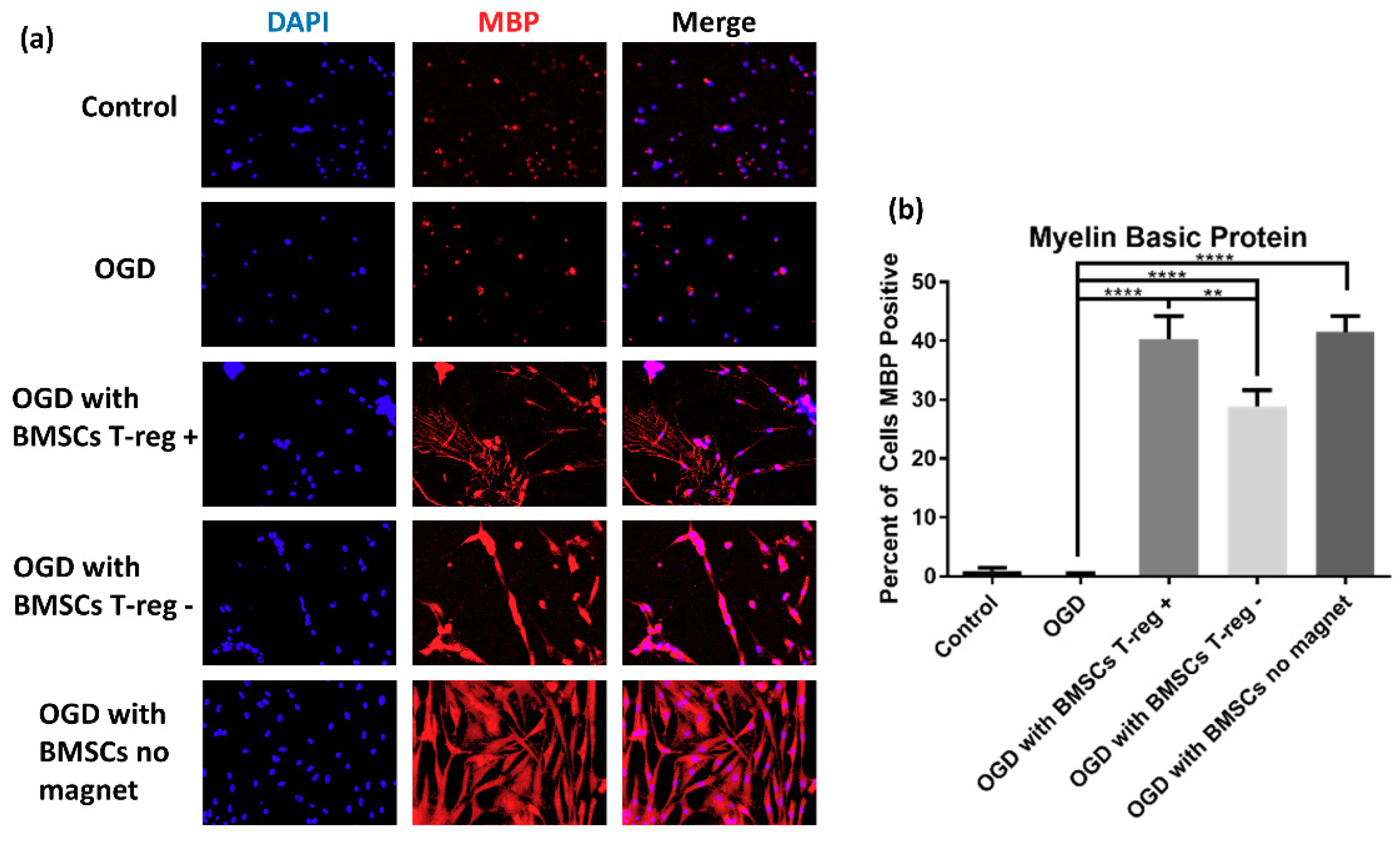
3.3. Tregs Confer Increased Myelination after OGD/R
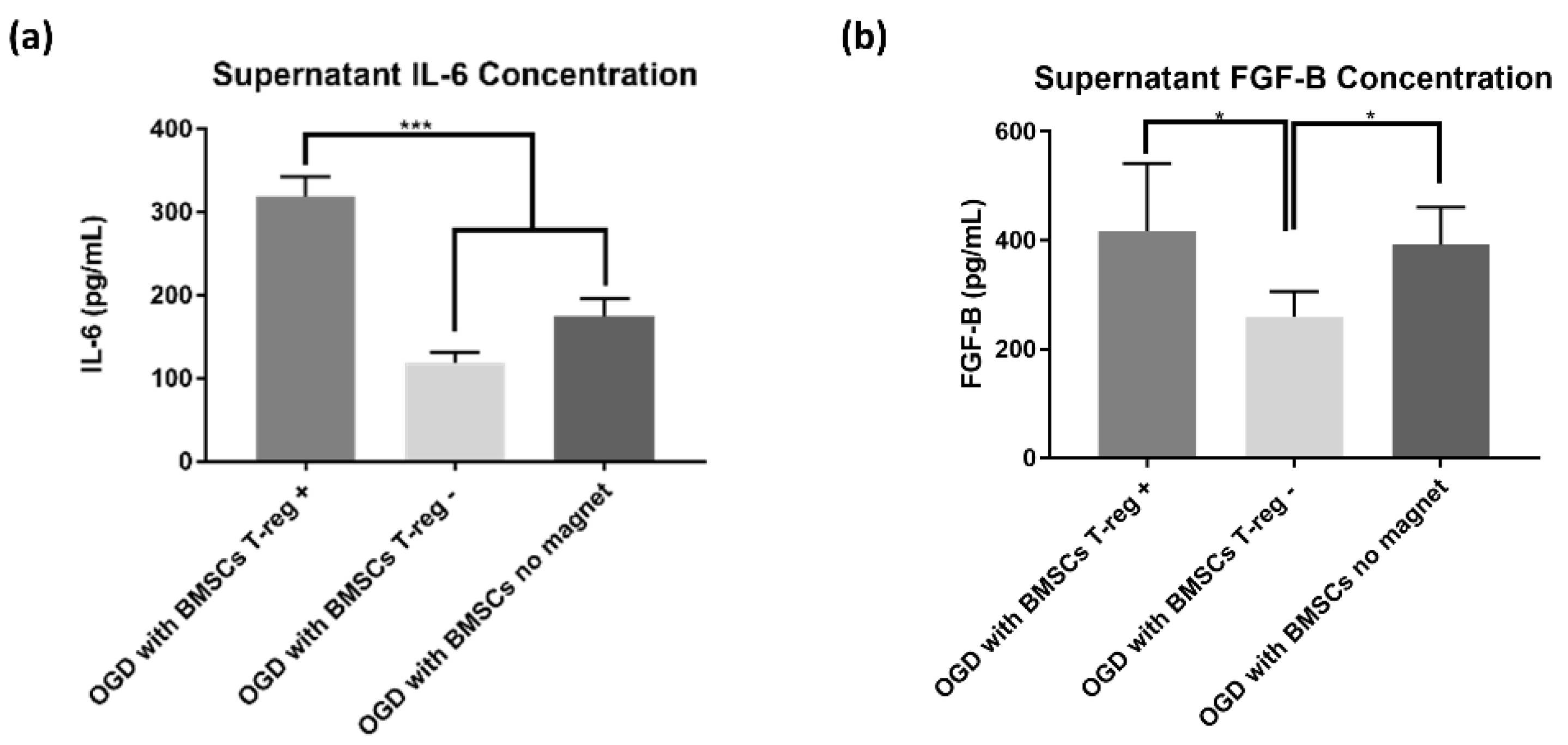
3.4. Treg+ Groups Showed Increased Extracellular Cytokines
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; Deo, R.; de Ferranti, S.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. Heart Disease and Stroke Statistics-2017 Update: A Report From the American Heart Association. Circulation 2017, 135, e146–e603. [Google Scholar]
- Mehta, S.L.; Manhas, N.; Raghubir, R. Molecular targets in cerebral ischemia for developing novel therapeutics. Brain Res. Rev. 2007, 54, 34–66. [Google Scholar] [CrossRef] [PubMed]
- Liesz, A.; Dalpke, A.; Mracsko, E.; Antoine, D.J.; Roth, S.; Zhou, W.; Yang, H.; Na, S.Y.; Akhisaroglu, M.; Fleming, T.; et al. DAMP signaling is a key pathway inducing immune modulation after brain injury. J. Neurosci. 2015, 35, 583–598. [Google Scholar] [CrossRef]
- Chamorro, A.; Meisel, A.; Planas, A.M.; Urra, X.; van de Beek, D.; Veltkamp, R. The immunology of acute stroke. Nat. Rev. Neurol. 2012, 8, 401–410. [Google Scholar] [CrossRef]
- Jin, R.; Yang, G.; Li, G. Inflammatory mechanisms in ischemic stroke: Role of inflammatory cells. J. Leukoc. Biol. 2010, 87, 779–789. [Google Scholar] [CrossRef]
- Borlongan, C.V.; Rodrigues, A.A., Jr.; Oliveira, M.C. Breaking the barrier in stroke: What should we know? A mini-review. Curr. Pharm. Des. 2012, 18, 3615–3623. [Google Scholar] [CrossRef]
- Napoli, E.; Borlongan, C.V. Recent Advances in Stem Cell-Based Therapeutics for Stroke. Transl. Stroke Res. 2016, 7, 452–457. [Google Scholar] [CrossRef]
- Napoli, E.; Borlongan, C.V. Stem Cell Recipes of Bone Marrow and Fish: Just What the Stroke Doctors Ordered. Stem Cell Rev. 2017, 13, 192–197. [Google Scholar] [CrossRef]
- Borlongan, C.V.; Glover, L.E.; Tajiri, N.; Kaneko, Y.; Freeman, T.B. The great migration of bone marrow-derived stem cells toward the ischemic brain: Therapeutic implications for stroke and other neurological disorders. Prog. Neurobiol. 2011, 95, 213–228. [Google Scholar] [CrossRef]
- Kfoury, Y.; Scadden, D.T. Mesenchymal cell contributions to the stem cell niche. Cell Stem Cell 2015, 16, 239–253. [Google Scholar] [CrossRef]
- Prockop, D.J. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 1997, 276, 71–74. [Google Scholar] [CrossRef]
- Stonesifer, C.; Corey, S.; Ghanekar, S.; Diamandis, Z.; Acosta, S.A.; Borlongan, C.V. Stem Cell Therapy for Abrogating Stroke-Induced Neuroinflammation and Relevant Secondary Cell Death Mechanisms. Prog. Neurobiol. 2017, 158, 94–131. [Google Scholar] [CrossRef]
- Glover, L.E.; Tajiri, N.; Lau, T.; Kaneko, Y.; van Loveren, H.; Borlongan, C.V. Immediate, but not delayed, microsurgical skull reconstruction exacerbates brain damage in experimental traumatic brain injury model. PLoS ONE 2012, 7, e33646. [Google Scholar] [CrossRef]
- Neal, E.G.; Acosta, S.A.; Kaneko, Y.; Ji, X.; Borlongan, C.V. Regulatory T-cells within bone marrow-derived stem cells actively confer immunomodulatory and neuroprotective effects against stroke. J. Cereb. Blood Flow Metab. 2018. [Google Scholar] [CrossRef]
- Williams, L.M.; Rudensky, A.Y. Maintenance of the Foxp3-dependent developmental program in mature regulatory T cells requires continued expression of Foxp3. Nat. Immunol. 2007, 8, 277–284. [Google Scholar] [CrossRef]
- Wang, G.; Shi, Y.; Jiang, X.; Leak, R.K.; Hu, X.; Wu, Y.; Pu, H.; Li, W.W.; Tang, B.; Wang, Y.; et al. HDAC inhibition prevents white matter injury by modulating microglia/macrophage polarization through the GSK3beta/PTEN/Akt axis. Proc. Natl. Acad. Sci. USA 2015, 112, 2853–2858. [Google Scholar] [CrossRef]
- Suenaga, J.; Hu, X.; Pu, H.; Shi, Y.; Hassan, S.H.; Xu, M.; Leak, R.K.; Stetler, R.A.; Gao, Y.; Chen, J. White matter injury and microglia/macrophage polarization are strongly linked with age-related long-term deficits in neurological function after stroke. Exp. Neurol. 2015, 272, 109–119. [Google Scholar] [CrossRef]
- Weirather, J.; Hofmann, U.D.; Beyersdorf, N.; Ramos, G.C.; Vogel, B.; Frey, A.; Ertl, G.; Kerkau, T.; Frantz, S. Foxp3+ CD4+ T cells improve healing after myocardial infarction by modulating monocyte/macrophage differentiation. Circ. Res. 2014, 115, 55–67. [Google Scholar] [CrossRef]
- Zhou, K.; Zhong, Q.; Wang, Y.C.; Xiong, X.Y.; Meng, Z.Y.; Zhao, T.; Zhu, W.Y.; Liao, M.F.; Wu, L.R.; Yang, Y.R.; et al. Regulatory T cells ameliorate intracerebral hemorrhage-induced inflammatory injury by modulating microglia/macrophage polarization through the IL-10/GSK3beta/PTEN axis. J. Cereb. Blood Flow Metab. 2017, 37, 967–979. [Google Scholar] [CrossRef]
- Dolati, S.; Ahmadi, M.; Khalili, M.; Taheraghdam, A.A.; Siahmansouri, H.; Babaloo, Z.; Aghebati-Maleki, L.; Jadidi-Niaragh, F.; Younesi, V.; Yousefi, M. Peripheral Th17/Treg imbalance in elderly patients with ischemic stroke. Neurol. Sci. 2018, 39, 647–654. [Google Scholar] [CrossRef]
- Hu, Y.; Zheng, Y.; Wu, Y.; Ni, B.; Shi, S. Imbalance between IL-17A-producing cells and regulatory T cells during ischemic stroke. Mediators Inflamm. 2014, 2014, 813045. [Google Scholar] [CrossRef]
- Wang, X.; Xuan, W.; Zhu, Z.Y.; Li, Y.; Zhu, H.; Zhu, L.; Fu, D.Y.; Yang, L.Q.; Li, P.Y.; Yu, W.F. The evolving role of neuro-immune interaction in brain repair after cerebral ischemic stroke. CNS Neurosci. Ther. 2018, 24, 1100–1114. [Google Scholar] [CrossRef] [PubMed]
- Matute, C.; Domercq, M.; Perez-Samartin, A.; Ransom, B.R. Protecting white matter from stroke injury. Stroke 2013, 44, 1204–1211. [Google Scholar] [CrossRef] [PubMed]
- Tekkok, S.B.; Goldberg, M.P. Ampa/kainate receptor activation mediates hypoxic oligodendrocyte death and axonal injury in cerebral white matter. J. Neurosci. 2001, 21, 4237–4248. [Google Scholar] [CrossRef]
- Yoshioka, A.; Yamaya, Y.; Saiki, S.; Kanemoto, M.; Hirose, G.; Beesley, J.; Pleasure, D. Non-N-methyl-D-aspartate glutamate receptors mediate oxygen–glucose deprivation-induced oligodendroglial injury. Brain Res. 2000, 854, 207–215. [Google Scholar] [CrossRef]
- Schaapsmeerders, P.; Tuladhar, A.M.; Arntz, R.M.; Franssen, S.; Maaijwee, N.A.; Rutten-Jacobs, L.C.; Schoonderwaldt, H.C.; Dorresteijn, L.D.; van Dijk, E.J.; Kessels, R.P.; et al. Remote Lower White Matter Integrity Increases the Risk of Long-Term Cognitive Impairment After Ischemic Stroke in Young Adults. Stroke 2016, 47, 2517–2525. [Google Scholar] [CrossRef]
- Correa, F.; Gauberti, M.; Parcq, J.; Macrez, R.; Hommet, Y.; Obiang, P.; Hernangomez, M.; Montagne, A.; Liot, G.; Guaza, C.; et al. Tissue plasminogen activator prevents white matter damage following stroke. J. Exp. Med. 2011, 208, 1229–1242. [Google Scholar] [CrossRef] [PubMed]
- Takase, H.; Liang, A.C.; Miyamoto, N.; Hamanaka, G.; Ohtomo, R.; Maki, T.; Pham, L.D.; Lok, J.; Lo, E.H.; Arai, K. Protective effects of a radical scavenger edaravone on oligodendrocyte precursor cells against oxidative stress. Neurosci. Lett. 2018, 668, 120–125. [Google Scholar] [CrossRef]
- Otero-Ortega, L.; Gutierrez-Fernandez, M.; Ramos-Cejudo, J.; Rodriguez-Frutos, B.; Fuentes, B.; Sobrino, T.; Hernanz, T.N.; Campos, F.; Lopez, J.A.; Cerdan, S.; et al. White matter injury restoration after stem cell administration in subcortical ischemic stroke. Stem Cell Res. Ther. 2015, 6, 121. [Google Scholar] [CrossRef]
- Baker, E.W.; Platt, S.R.; Lau, V.W.; Grace, H.E.; Holmes, S.P.; Wang, L.; Duberstein, K.J.; Howerth, E.W.; Kinder, H.A.; Stice, S.L.; et al. Induced Pluripotent Stem Cell-Derived Neural Stem Cell Therapy Enhances Recovery in an Ischemic Stroke Pig Model. Sci. Rep. 2017, 7, 10075. [Google Scholar] [CrossRef]
- Kaneko, Y.; Pappas, C.; Tajiri, N.; Borlongan, C.V. Oxytocin modulates GABAAR subunits to confer neuroprotection in stroke in vitro. Sci. Rep. 2016, 6, 35659. [Google Scholar] [CrossRef]
- Yu, X.; Wu, H.; Zhao, Y.; Guo, Y.; Chen, Y.; Dong, P.; Mu, Q.; Wang, X.; Wang, X. Bone marrow mesenchymal stromal cells alleviate brain white matter injury via the enhanced proliferation of oligodendrocyte progenitor cells in focal cerebral ischemic rats. Brain Res. 2018, 1680, 127–136. [Google Scholar] [CrossRef]
- Zhang, J.; Li, Y.; Zhang, Z.G.; Lu, M.; Borneman, J.; Buller, B.; Savant-Bhonsale, S.; Elias, S.B.; Chopp, M. Bone marrow stromal cells increase oligodendrogenesis after stroke. J. Cereb. Blood Flow Metab. 2009, 29, 1166–1174. [Google Scholar] [CrossRef]
- Maki, T.; Morancho, A.; Martinez-San Segundo, P.; Hayakawa, K.; Takase, H.; Liang, A.C.; Gabriel-Salazar, M.; Medina-Gutierrez, E.; Washida, K.; Montaner, J.; et al. Endothelial Progenitor Cell Secretome and Oligovascular Repair in a Mouse Model of Prolonged Cerebral Hypoperfusion. Stroke 2018, 49, 1003–1010. [Google Scholar] [CrossRef]
- Chen, X.; Li, Y.; Wang, L.; Katakowski, M.; Zhang, L.; Chen, J.; Xu, Y.; Gautam, S.C.; Chopp, M. Ischemic rat brain extracts induce human marrow stromal cell growth factor production. Neuropathology 2002, 22, 275–279. [Google Scholar] [CrossRef]
- Miyamoto, N.; Maki, T.; Shindo, A.; Liang, A.C.; Maeda, M.; Egawa, N.; Itoh, K.; Lo, E.K.; Lok, J.; Ihara, M.; et al. Astrocytes Promote Oligodendrogenesis after White Matter Damage via Brain-Derived Neurotrophic Factor. J. Neurosci. 2015, 35, 14002–14008. [Google Scholar] [CrossRef]
- Tsiperson, V.; Huang, Y.; Bagayogo, I.; Song, Y.; VonDran, M.W.; DiCicco-Bloom, E.; Dreyfus, C.F. Brain-derived neurotrophic factor deficiency restricts proliferation of oligodendrocyte progenitors following cuprizone-induced demyelination. ASN Neuro 2015, 7. [Google Scholar] [CrossRef]
- Lundegardh, G.; Hansson, L.E.; Nyren, O.; Adami, H.O.; Krusemo, U.B. The risk of gastrointestinal and other primary malignant diseases following gastric cancer. Acta Oncol. 1991, 30, 1–6. [Google Scholar] [CrossRef]
- Chan, A.; Yan, J.; Csurhes, P.; Greer, J.; McCombe, P. Circulating brain derived neurotrophic factor (BDNF) and frequency of BDNF positive T cells in peripheral blood in human ischemic stroke: Effect on outcome. J. Neuroimmunol. 2015, 286, 42–47. [Google Scholar] [CrossRef]
- Falcon-Urrutia, P.; Carrasco, C.M.; Lois, P.; Palma, V.; Roth, A.D. Shh Signaling through the Primary Cilium Modulates Rat Oligodendrocyte Differentiation. PLoS ONE 2015, 10, e0133567. [Google Scholar] [CrossRef]
- Ferent, J.; Zimmer, C.; Durbec, P.; Ruat, M.; Traiffort, E. Sonic Hedgehog signaling is a positive oligodendrocyte regulator during demyelination. J. Neurosci. 2013, 33, 1759–1772. [Google Scholar] [CrossRef] [PubMed]
- Maki, T.; Choi, Y.K.; Miyamoto, N.; Shindo, A.; Liang, A.C.; Ahn, B.J.; Mandeville, E.T.; Kaji, S.; Itoh, K.; Seo, J.H.; et al. A-Kinase Anchor Protein 12 Is Required for Oligodendrocyte Differentiation in Adult White Matter. Stem Cells 2018, 36, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Perigolo-Vicente, R.; Ritt, K.; Goncalves-de-Albuquerque, C.F.; Castro-Faria-Neto, H.C.; Paes-de-Carvalho, R.; Giestal-de-Araujo, E. IL-6, A1 and A2aR: A crosstalk that modulates BDNF and induces neuroprotection. Biochem. Biophys. Res. Commun. 2014, 449, 477–482. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; He, M.; Zhou, X.; Liu, J.; Hou, N.; Bin, T.; Zhang, Y.; Li, T.; Chen, J. Endogenous IL-6 of mesenchymal stem cell improves behavioral outcome of hypoxic-ischemic brain damage neonatal rats by supressing apoptosis in astrocyte. Sci. Rep. 2016, 6, 18587. [Google Scholar] [CrossRef] [PubMed]
- Lahiani, A.; Zahavi, E.; Netzer, N.; Ofir, R.; Pinzur, L.; Raveh, S.; Arien-Zakay, H.; Yavin, E.; Lazarovici, P. Human placental eXpanded (PLX) mesenchymal-like adherent stromal cells confer neuroprotection to nerve growth factor (NGF)-differentiated PC12 cells exposed to ischemia by secretion of IL-6 and VEGF. Biochim. Biophys. Acta 2015, 1853, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.H.; Zhuang, Q.X.; Shen, W.X.; Peng, Y.P.; Qiu, Y.H. Interleukin-6 reduces NMDAR-mediated cytosolic Ca(2)(+) overload and neuronal death via JAK/CaN signaling. Cell Calcium 2015, 58, 286–295. [Google Scholar] [CrossRef]
- Holla, S.; Stephen-Victor, E.; Prakhar, P.; Sharma, M.; Saha, C.; Udupa, V.; Kaveri, S.V.; Bayry, J.; Balaji, K.N. Mycobacteria-responsive sonic hedgehog signaling mediates programmed death-ligand 1- and prostaglandin E2-induced regulatory T cell expansion. Sci. Rep. 2016, 6, 24193. [Google Scholar] [CrossRef]
- Valerio, A.; Ferrario, M.; Dreano, M.; Garotta, G.; Spano, P.; Pizzi, M. Soluble interleukin-6 (IL-6) receptor/IL-6 fusion protein enhances in vitro differentiation of purified rat oligodendroglial lineage cells. Mol. Cell Neurosci. 2002, 21, 602–615. [Google Scholar] [CrossRef]
- Ganeshan, K.; Bryce, P.J. Regulatory T cells enhance mast cell production of IL-6 via surface-bound TGF-beta. J. Immunol. 2012, 188, 594–603. [Google Scholar] [CrossRef]
- Ganeshan, K.; Johnston, L.K.; Bryce, P.J. TGF-beta1 limits the onset of innate lung inflammation by promoting mast cell-derived IL-6. J. Immunol. 2013, 190, 5731–5738. [Google Scholar] [CrossRef]
- Shi, H.; Hu, X.; Leak, R.K.; Shi, Y.; An, C.; Suenaga, J.; Chen, J.; Gao, Y. Demyelination as a rational therapeutic target for ischemic or traumatic brain injury. Exp. Neurol. 2015, 272, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Abbaszadeh, H.A.; Tiraihi, T.; Delshad, A.R.; Saghedi Zadeh, M.; Taheri, T. Bone marrow stromal cell transdifferentiation into oligodendrocyte-like cells using triiodothyronine as a inducer with expression of platelet-derived growth factor alpha as a maturity marker. Iran. Biomed. J. 2013, 17, 62–70. [Google Scholar] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zarriello, S.; Neal, E.G.; Kaneko, Y.; Borlongan, C.V. T-Regulatory Cells Confer Increased Myelination and Stem Cell Activity after Stroke-Induced White Matter Injury. J. Clin. Med. 2019, 8, 537. https://doi.org/10.3390/jcm8040537
Zarriello S, Neal EG, Kaneko Y, Borlongan CV. T-Regulatory Cells Confer Increased Myelination and Stem Cell Activity after Stroke-Induced White Matter Injury. Journal of Clinical Medicine. 2019; 8(4):537. https://doi.org/10.3390/jcm8040537
Chicago/Turabian StyleZarriello, Sydney, Elliot G. Neal, Yuji Kaneko, and Cesario V. Borlongan. 2019. "T-Regulatory Cells Confer Increased Myelination and Stem Cell Activity after Stroke-Induced White Matter Injury" Journal of Clinical Medicine 8, no. 4: 537. https://doi.org/10.3390/jcm8040537
APA StyleZarriello, S., Neal, E. G., Kaneko, Y., & Borlongan, C. V. (2019). T-Regulatory Cells Confer Increased Myelination and Stem Cell Activity after Stroke-Induced White Matter Injury. Journal of Clinical Medicine, 8(4), 537. https://doi.org/10.3390/jcm8040537





