Associations of Adiposity and Diet Quality with Serum Ceramides in Middle-Aged Adults with Cardiovascular Risk Factors
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Sample
2.2. Demographic and Health-Related Data
2.3. Diet Quality Assessment
2.4. Ceramides Analysis
2.5. Ceramides Risk Score
2.6. Statistical Analysis
3. Results
3.1. Participant Characteristics by Ceramide Risk Category
3.2. Relationship between Potential Covariates and Ceramide
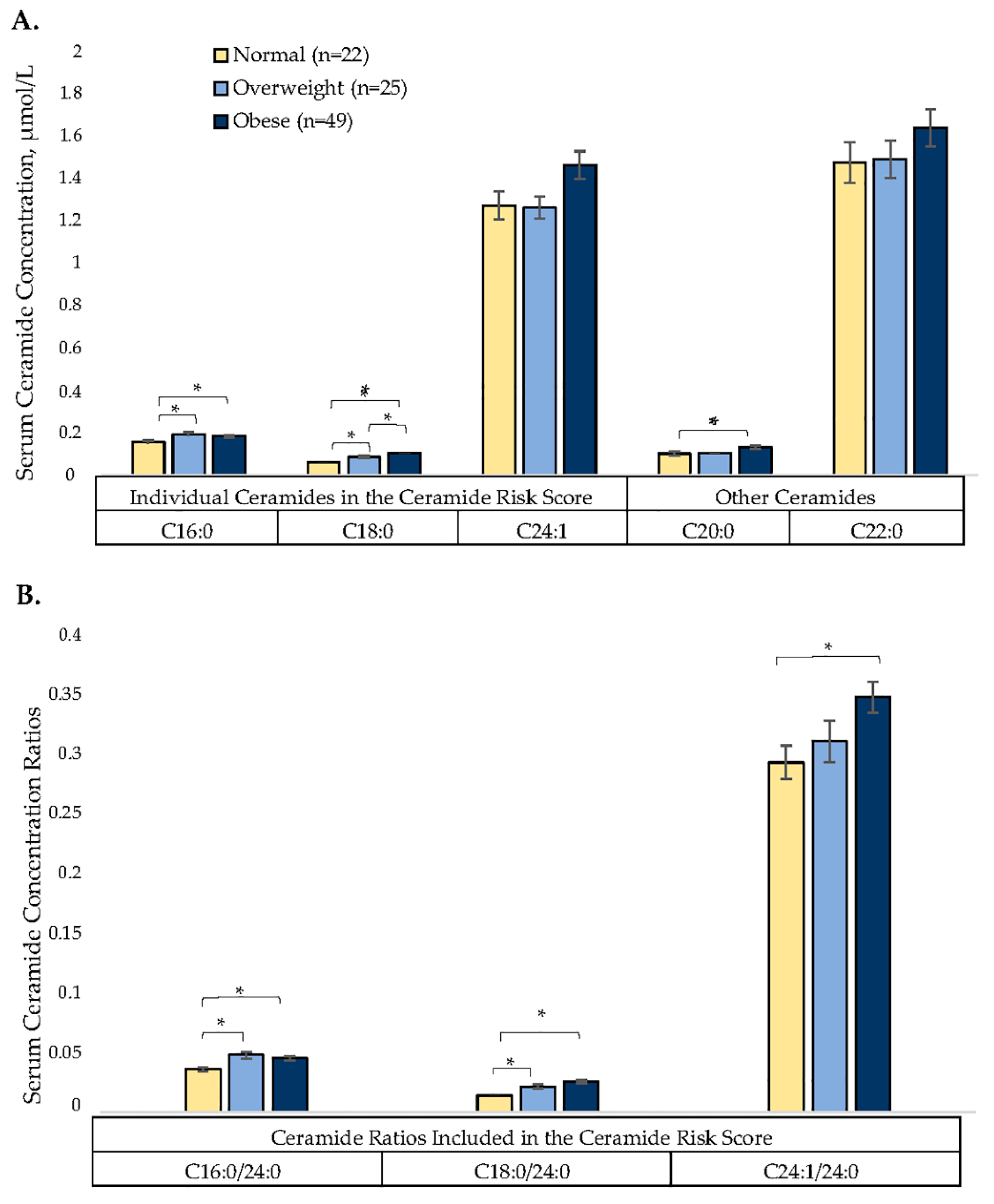
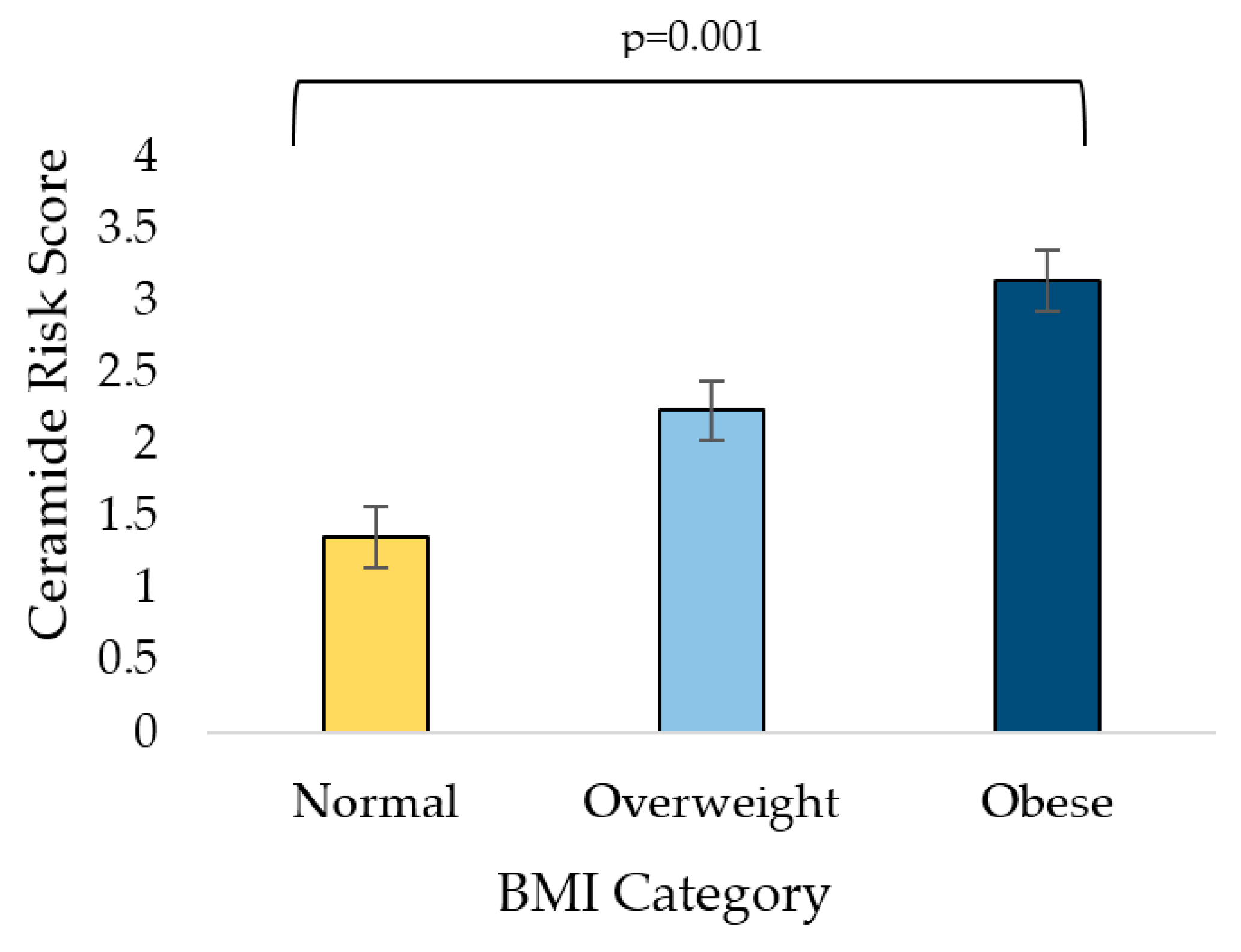
3.3. Relationship of BMI (Adiposity) and HEI (Diet Quality) with Ceramides
4. Discussion
4.1. Associations between Adiposity and Ceramides
4.2. Associations between Diet Quality and Ceramides
4.3. Limitations
4.4. Clinical implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- CDC. Prevent 1 Million Heart Attacks & Strokes. Available online: https://www.cdc.gov/vitalsigns/million-hearts/index.html (accessed on 17 December 2018).
- Wall, H.K. Vital Signs: Prevalence of Key Cardiovascular Disease Risk Factors for Million Hearts 2022—United States, 2011–2016. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 983–991. [Google Scholar] [PubMed]
- Sachdeva, A.; Cannon, C.P.; Deedwania, P.C.; Labresh, K.A.; Smith, S.C.; Dai, D.; Hernandez, A.; Fonarow, G.C. Lipid levels in patients hospitalized with coronary artery disease: An analysis of 136,905 hospitalizations in Get with the Guidelines. Am. Heart J. 2009, 157, 111–117.e2. [Google Scholar]
- Meeusen, J.W.; Donato, L.J.; Bryant, S.C.; Baudhuin, L.M.; Berger, P.B.; Jaffe, A.S. Plasma Ceramides: A Novel Predictor of Major Adverse Cardiovascular Events After Coronary Angiography. Arterioscler. Thromb. Vasc. Biol. 2018, 38. [Google Scholar] [CrossRef]
- Laaksonen, R.; Ekroos, K.; Sysi-Aho, M.; Hilvo, M.; Vihervaara, T.; Kauhanen, D.; Suoniemi, M.; Hurme, R.; März, W.; Scharnagl, H.; et al. Plasma ceramides predict cardiovascular death in patients with stable coronary artery disease and acute coronary syndromes beyond LDL-cholesterol. Eur. Heart J. 2016, 37, 1967–1976. [Google Scholar]
- Havulinna, A.S.; Sysi-Aho, M.; Hilvo, M.; Kauhanen, D.; Hurme, R.; Ekroos, K.; Salomaa, V.; Laaksonen, R. Circulating Ceramides Predict Cardiovascular Outcomes in the Population-Based FINRISK 2002 Cohort. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2424–2430. [Google Scholar]
- Cremesti, A.E.; Fischl, A.S. Current methods for the identification and quantitation of ceramides: An overview. Lipids 2000, 35, 937–945. [Google Scholar] [PubMed]
- Park, J.-W.; Park, W.-J.; Futerman, A.H. Ceramide synthases as potential targets for therapeutic intervention in human diseases. Biochim. Biophys. Acta 2014, 1841, 671–681. [Google Scholar]
- Wattenberg, B.W. The long and the short of ceramides. J. Biol. Chem. 2018, 293, 9922–9923. [Google Scholar] [PubMed]
- Bikman, B.T.; Summers, S.A. Ceramides as modulators of cellular and whole-body metabolism. J. Clin. Investig. 2011, 121, 4222–4230. [Google Scholar]
- Bismuth, J.; Lin, P.; Yao, Q.; Chen, C. Ceramide: A common pathway for atherosclerosis? Atherosclerosis 2008, 196, 497–504. [Google Scholar]
- Mundra, P.A.; Shaw, J.E.; Meikle, P.J. Lipidomic analyses in epidemiology. Int. J. Epidemiol. 2016, 45, 1329–1338. [Google Scholar]
- Summers, S.A. Could Ceramides Become the New Cholesterol? Cell Metab. 2018, 27, 276–280. [Google Scholar] [PubMed]
- Reedy, J.; Krebs-Smith, S.M.; Miller, P.E.; Liese, A.D.; Kahle, L.L.; Park, Y.; Subar, A.F. Higher diet quality is associated with decreased risk of all-cause, cardiovascular disease, and cancer mortality among older adults. J. Nutr. 2014, 144, 881–889. [Google Scholar]
- Bergman, B.C.; Brozinick, J.T.; Strauss, A.; Bacon, S.; Kerege, A.; Bui, H.H.; Sanders, P.; Siddall, P.; Kuo, M.S.; Perreault, L. Serum sphingolipids: Relationships to insulin sensitivity and changes with exercise in humans. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E398–E408. [Google Scholar]
- Tarasov, K.; Ekroos, K.; Suoniemi, M.; Kauhanen, D.; Sylvänne, T.; Hurme, R.; Gouni-Berthold, I.; Berthold, H.K.; Kleber, M.E.; Laaksonen, R.; et al. Molecular lipids identify cardiovascular risk and are efficiently lowered by simvastatin and PCSK9 deficiency. J. Clin. Endocrinol. Metab. 2014, 99, E45–E52. [Google Scholar]
- Ng, T.W.K.; Ooi, E.M.M.; Watts, G.F.; Chan, D.C.; Weir, J.M.; Meikle, P.J.; Barrett, P.H.R. Dose-dependent effects of rosuvastatin on the plasma sphingolipidome and phospholipidome in the metabolic syndrome. J. Clin. Endocrinol. Metab. 2014, 99, E2335–E2340. [Google Scholar]
- Huang, H.; Kasumov, T.; Gatmaitan, P.; Heneghan, H.M.; Kashyap, S.R.; Schauer, P.R.; Brethauer, S.A.; Kirwan, J.P. Gastric bypass surgery reduces plasma ceramide subspecies and improves insulin sensitivity in severely obese patients. Obesity 2011, 19, 2235–2240. [Google Scholar]
- Özer, H.; Aslan, İ.; Oruç, M.T.; Çöpelci, Y.; Afşar, E.; Kaya, S.; Aslan, M. Early postoperative changes of sphingomyelins and ceramides after laparoscopic sleeve gastrectomy. Lipids Health Dis. 2018, 17, 269. [Google Scholar]
- Heilbronn, L.K.; Coster, A.C.F.; Campbell, L.V.; Greenfield, J.R.; Lange, K.; Christopher, M.J.; Meikle, P.J.; Samocha-Bonet, D. The effect of short-term overfeeding on serum lipids in healthy humans. Obesity 2013, 21, E649–E659. [Google Scholar]
- Luukkonen, P.K.; Sädevirta, S.; Zhou, Y.; Kayser, B.; Ali, A.; Ahonen, L.; Lallukka, S.; Pelloux, V.; Gaggini, M.; Jian, C.; et al. Saturated Fat Is More Metabolically Harmful for the Human Liver Than Unsaturated Fat or Simple Sugars. Diabetes Care 2018, 41, 1732–1739. [Google Scholar]
- Kien, C.L.; Bunn, J.Y.; Poynter, M.E.; Stevens, R.; Bain, J.; Ikayeva, O.; Fukagawa, N.K.; Champagne, C.M.; Crain, K.I.; Koves, T.R.; et al. A lipidomics analysis of the relationship between dietary fatty acid composition and insulin sensitivity in young adults. Diabetes 2013, 62, 1054–1063. [Google Scholar]
- Wang, D.D.; Toledo, E.; Hruby, A.; Rosner, B.A.; Willett, W.C.; Sun, Q.; Razquin, C.; Zheng, Y.; Ruiz-Canela, M.; Guasch-Ferré, M.; et al. Plasma Ceramides, Mediterranean Diet, and Incident Cardiovascular Disease in the PREDIMED Trial. Circulation 2017, 135, 2028–2040. [Google Scholar]
- Segal, L.M.; Rayburn, J.; Beck, S.E. The State of Obesity: Better Policies for a Healthier America 2017; Trust for America’s Health, The Robert Wood Johnson Foundation: Washington, DC, USA, 2017; pp. 1–108. Available online: https://www.tfah.org/report-details/the-state-of-obesity-2017 (accessed on 16 April 2019).
- Lee-Kwan, S.H.; Moore, L.V.; Blanck, H.M.; Harris, D.M.; Galuska, D. Disparities in State-Specific Adult Fruit and Vegetable Consumption—United States, 2015. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 1241–1247. [Google Scholar] [PubMed]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar]
- Gutch, M.; Kumar, S.; Razi, S.M.; Gupta, K.K.; Gupta, A. Assessment of insulin sensitivity/resistance. Indian J. Endocrinol. Metab. 2015, 19, 160–164. [Google Scholar]
- National Institute of Health Diabetes & Prediabetes Tests|NIDDK. Available online: https://www.niddk.nih.gov/health-information/diagnostic-tests/diabetes-prediabetes (accessed on 30 July 2018).
- National Institute of Health. High Blood Pressure. Available online: http://www.nia.nih.gov/health/high-blood-pressure (accessed on 30 July 2018).
- Grundy, S.M.; Bryan Brewer, J.H.; Cleeman, J.I.; Sidney, C.; Smith, J.; Lenfant, C. Definition of Metabolic Syndrome. Circulation 2004, 109, 433–438. [Google Scholar]
- Ostchega, Y.; Nwankwo, T.; Sorlie, P.D.; Wolz, M.; Zipf, G. Assessing the validity of the Omron HEM-907XL oscillometric blood pressure measurement device in a National Survey environment. J. Clin. Hypertens. 2010, 12, 22–28. [Google Scholar]
- Viera, A.J.; Zhu, S.; Hinderliter, A.L.; Shimbo, D.; Person, S.D.; Jacobs, D.R. Diurnal blood pressure pattern and development of prehypertension or hypertension in young adults: The CARDIA study. J. Am. Soc. Hypertens. 2011, 5, 48–55. [Google Scholar]
- World Health Organization. Waist Circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation, Geneva, 8–11 December 2008; vi; World Health Organization: Geneva, Switzerland, 2008; ISBN 978-92-4-150149-1. [Google Scholar]
- U.S. Department of Health and Human Services and U.S. Department of Agriculture 2015–2020 Dietary Guidelines for Americans—health.gov. Available online: https://health.gov/dietaryguidelines/2015/ (accessed on 27 June 2018).
- Nutrition Coordination Center (NCC), University of Minnesota. Guide to Creating Variables, Needed to Calculate Scores for Each Component of the Healthy Eating Index-2015 (HEI-2015). Available online: http://www.ncc.umn.edu/ndsrsupport/hei2015.pdf (accessed on 16 August 2017).
- National Cancer Institute Division of Cancer Control and Population Sciences. The Healthy Eating Index Research Uses: Overview of the Methods & Calculations. Available online: https://epi.grants.cancer.gov/hei/hei-methods-and-calculations.html (accessed on 2 July 2018).
- Rico, J.E.; Bandaru, V.V.R.; Dorskind, J.M.; Haughey, N.J.; McFadden, J.W. Plasma ceramides are elevated in overweight Holstein dairy cows experiencing greater lipolysis and insulin resistance during the transition from late pregnancy to early lactation. J. Dairy Sci. 2015, 98, 7757–7770. [Google Scholar] [PubMed]
- Haughey, N.J.; Cutler, R.G.; Tamara, A.; McArthur, J.C.; Vargas, D.L.; Pardo, C.A.; Turchan, J.; Nath, A.; Mattson, M.P. Perturbation of sphingolipid metabolism and ceramide production in HIV-dementia. Ann. Neurol. 2004, 55, 257–267. [Google Scholar] [PubMed]
- Meikle, P.J.; Wong, G.; Barlow, C.K.; Weir, J.M.; Greeve, M.A.; MacIntosh, G.L.; Almasy, L.; Comuzzie, A.G.; Mahaney, M.C.; Kowalczyk, A.; et al. Plasma lipid profiling shows similar associations with prediabetes and type 2 diabetes. PLoS ONE 2013, 8, e74341. [Google Scholar]
- Mayo Clinic. Plasma Ceramides: A Novel Biomarker of Unstable Atherosclerotic Cardiovascular Disease; Mayo Foundation for Medical Education and Research: Rochester, MN, USA, 2016; pp. 1–4. Available online: https://cdn.prod-carehubs.net/n1/96e99366cea7b0de/uploads/2016/07/ceramides-brochure-final-0616.pdf (accessed on 16 April 2019).
- Lankinen, M.; Schwab, U.; Kolehmainen, M.; Paananen, J.; Nygren, H.; Seppänen-Laakso, T.; Poutanen, K.; Hyötyläinen, T.; Risérus, U.; Savolainen, M.J.; et al. A Healthy Nordic Diet Alters the Plasma Lipidomic Profile in Adults with Features of Metabolic Syndrome in a Multicenter Randomized Dietary Intervention. J. Nutr. 2016, 146, 662–672. [Google Scholar]
- Mielke, M.M.; Bandaru, V.V.R.; Han, D.; An, Y.; Resnick, S.M.; Ferrucci, L.; Haughey, N.J. Demographic and clinical variables affecting mid- to late-life trajectories of plasma ceramide and dihydroceramide species. Aging Cell 2015, 14, 1014–1023. [Google Scholar] [PubMed]
- Mantovani, A.; Bonapace, S.; Lunardi, G.; Salgarello, M.; Dugo, C.; Canali, G.; Byrne, C.D.; Gori, S.; Barbieri, E.; Targher, G. Association between plasma ceramides and inducible myocardial ischemia in patients with established or suspected coronary artery disease undergoing myocardial perfusion scintigraphy. Metabolism 2018, 85, 305–312. [Google Scholar] [PubMed]
- Haus, J.M.; Kashyap, S.R.; Kasumov, T.; Zhang, R.; Kelly, K.R.; Defronzo, R.A.; Kirwan, J.P. Plasma ceramides are elevated in obese subjects with type 2 diabetes and correlate with the severity of insulin resistance. Diabetes 2009, 58, 337–343. [Google Scholar]
- Ichi, I.; Nakahara, K.; Miyashita, Y.; Hidaka, A.; Kutsukake, S.; Inoue, K.; Maruyama, T.; Miwa, Y.; Harada-Shiba, M.; Tsushima, M.; et al. Association of ceramides in human plasma with risk factors of atherosclerosis. Lipids 2006, 41, 859–863. [Google Scholar] [PubMed]
- Peterson, L.R.; Xanthakis, V.; Duncan, M.S.; Gross, S.; Friedrich, N.; Völzke, H.; Felix, S.B.; Jiang, H.; Sidhu, R.; Nauck, M.; et al. Ceramide Remodeling and Risk of Cardiovascular Events and Mortality. J. Am. Heart Assoc. 2018, 7, e007931. [Google Scholar] [PubMed]
- Chaurasia, B.; Summers, S.A. Ceramides—Lipotoxic Inducers of Metabolic Disorders. Trends Endocrinol. Metab. TEM 2015, 26, 538–550. [Google Scholar]
- Boden, G. Obesity and Free Fatty Acids (FFA). Endocrinol. Metab. Clin. N. Am. 2008, 37, 635–646. [Google Scholar]
- Fucho, R.; Casals, N.; Serra, D.; Herrero, L. Ceramides and mitochondrial fatty acid oxidation in obesity. FASEB J. 2017, 31, 1263–1272. [Google Scholar] [PubMed]
- Iqbal, J.; Walsh, M.T.; Hammad, S.M.; Hussain, M.M. Sphingolipids and Lipoproteins in Health and Metabolic Disorders. Trends Endocrinol. Metab. TEM 2017, 28, 506–518. [Google Scholar]
- Loprinzi, P.D.; Branscum, A.; Hanks, J.; Smit, E. Healthy Lifestyle Characteristics and Their Joint Association with Cardiovascular Disease Biomarkers in US Adults. Mayo Clin. Proc. 2016, 91, 432–442. [Google Scholar] [PubMed]
- Schwingshackl, L.; Bogensberger, B.; Hoffmann, G. Diet Quality as Assessed by the Healthy Eating Index, Alternate Healthy Eating Index, Dietary Approaches to Stop Hypertension Score, and Health Outcomes: An Updated Systematic Review and Meta-Analysis of Cohort Studies. J. Acad. Nutr. Diet. 2018, 118, 74–100.e11. [Google Scholar] [PubMed]
| All N = 96 | Lower Risk 1 n = 56 | Moderate Risk 2 n = 40 | p-Value 3 | |
|---|---|---|---|---|
| Demographic Factors | ||||
| Age, year | 54.30 ± 0.47 | 53.95 ± 0.55 | 54.8 ± 0.84 | 0.40 |
| Sex, women | 55 (57.3%) | 33 (58.9%) | 22 (55.0%) | 0.70 |
| Health-Related Factors | ||||
| Adiposity | ||||
| Body Mass Index, kg/m2 | 30.85 ± 0.74 | 29.22 ± 0.92 | 33.13 ± 1.13 | 0.003 |
| Waist Circumference, cm | 103.35 ± 1.67 | 99.41 ± 2.07 | 108.87 ± 2.55 | 0.005 |
| Elevated Waist Circumference 4 | 70 (72.9%) | 37 (66.1%) | 33 (82.5%) | 0.07 |
| Waist-to-Hip Ratio, cm | 0.90 ± 0.008 | 0.89 ± 0.01 | 0.91 ± 0.01 | 0.16 |
| Elevated Waist-to-Hip Ratio 5 | 63 (65.6%) | 36 (64.3%) | 27 (67.5%) | 0.74 |
| Fat Mass Index, kg/m2 | 12.26 ± 0.55 | 11.00 ± 0.65 | 14.02 ± 0.89 | 0.004 |
| Laboratory Values | ||||
| Total Cholesterol ≥200 mg/dL | 52 (54.2%) | 30 (53.6%) | 22 (55.0%) | 0.89 |
| LDL ≥100 mg/dL | 77 (81.1%) | 43 (76.8%) | 34 (87.2%) | 0.20 |
| Low HDL 6 | 22 (22.9%) | 12 (21.4%) | 10 (25.0%) | 0.68 |
| Triglycerides ≥150 mg/dL | 18 (18.8%) | 9 (16.1%) | 9 (22.5%) | 0.43 |
| Glucose >100 mg/dL | 35 (36.5%) | 19 (33.9%) | 16 (40.0%) | 0.54 |
| Insulin >24 mg/dL | 4 (4.17%) | 1 (1.8%) | 3 (7.5%) | 0.17 |
| HOMA-IR | 2.18 ± (0.17) | 1.97 (0.19) | 2.48 (0.31) | 0.19 |
| CRP ≥8 mg/dL | 16 (16.7%) | 9 (16.1%) | 7 (17.5%) | 0.85 |
| Fibrinogen >400 mg/dL | 22 (22.9%) | 8 (14.3%) | 14 (35.0%) | 0.02 |
| Medical Conditions | ||||
| Metabolic Syndrome | 37 (38.5%) | 16 (28.6%) | 21 (52.5%) | 0.02 |
| Diabetes | 39 (40.6%) | 22 (39.3%) | 17 (42.5%) | 0.75 |
| Dyslipidemia | 88 (91.7%) | 50 (89.3%) | 38 (95.0%) | 0.32 |
| Hypertension | 37 (38.5%) | 18 (32.1%) | 19 (47.5%) | 0.13 |
| Statin use | 23 (24.0%) | 13 (23.2%) | 10 (25.0%) | 0.84 |
| Lifestyle Factors | ||||
| Former Smoker | 48 (50.0%) | 23 (41.1%) | 25 (62.5%) | 0.04 |
| HEI-2015 Diet Scores | 54.05 ± 1.45 | 55.27 ± 1.73 | 52.36 ± 2.49 | 0.34 |
| Outcome Variables | Ceramides Included in the Ceramide Risk Score | Ceramide Risk Score | Other Ceramides | ||||
|---|---|---|---|---|---|---|---|
| C18:0 | C24:1 | C16:0/24:0 | C18:0/24:0 | C20:0 | C22:0 | ||
| Unadjusted Model | |||||||
| BMI (kg/m2) | 0.80; 0.19 (<0.001) | NS | 0.36; 0.07 (0.006) | 1.02; 0.26 (<0.001) | 5.97; 0.07 (0.006) | NS | NS |
| HEI-2015 | NS | NS | NS | NS | NS | NS | −0.002; 0.06 (0.009) |
| Adjusted Models | |||||||
| BMI (kg/m2) | 0.81; 0.31 (<0.0001) | NS | NS | 0.91; 0.30 (<0.0001) | 5.58; 0.11 (0.009) | NS | NS |
| HEI-2015 | NS | NS | NS | NS | NS | NS | −0.002; 0.42 (0.02) |
| HEI Component | Ceramides in Risk Score | Other Ceramides | |||
|---|---|---|---|---|---|
| C16:0 | C24:1 | C16:0/24:0 | C20:0 | C22:0 | |
| Unadjusted Models | |||||
| Total Vegetables | −0.02; 0.04 (0.02) | NS | NS | NS | −0.03; 0.05 (0.01) |
| Whole Grains | NS | NS | NS | −0.01; 0.04 (0.03) | −0.01; 0.04 (0.03) |
| Refined Grains | NS | NS | −0.007; 0.04 (0.03) | NS | NS |
| Saturated Fats | NS | −0.01; 0.05 (0.01) | NS | −0.02; 0.06 (0.01) | −0.02; 0.08 (0.003) |
| Added Sugar | NS | NS | NS | NS | −0.01; 0.07 (0.007) |
| Adjusted Models | |||||
| Total Vegetables | NS | NS | NS | NS | −0.02; 0.44 (0.02) |
| Whole Grains | NS | NS | NS | NS | −0.007; 0.43 (0.03) |
| Saturated Fats | NS | NS | NS | NS | −0.008; 0.43 (0.03) |
| Added Sugar | NS | NS | NS | NS | −0.009; 0.44 (0.01) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drazba, M.A.; Holásková, I.; Sahyoun, N.R.; Ventura Marra, M. Associations of Adiposity and Diet Quality with Serum Ceramides in Middle-Aged Adults with Cardiovascular Risk Factors. J. Clin. Med. 2019, 8, 527. https://doi.org/10.3390/jcm8040527
Drazba MA, Holásková I, Sahyoun NR, Ventura Marra M. Associations of Adiposity and Diet Quality with Serum Ceramides in Middle-Aged Adults with Cardiovascular Risk Factors. Journal of Clinical Medicine. 2019; 8(4):527. https://doi.org/10.3390/jcm8040527
Chicago/Turabian StyleDrazba, Margaret A., Ida Holásková, Nadine R. Sahyoun, and Melissa Ventura Marra. 2019. "Associations of Adiposity and Diet Quality with Serum Ceramides in Middle-Aged Adults with Cardiovascular Risk Factors" Journal of Clinical Medicine 8, no. 4: 527. https://doi.org/10.3390/jcm8040527
APA StyleDrazba, M. A., Holásková, I., Sahyoun, N. R., & Ventura Marra, M. (2019). Associations of Adiposity and Diet Quality with Serum Ceramides in Middle-Aged Adults with Cardiovascular Risk Factors. Journal of Clinical Medicine, 8(4), 527. https://doi.org/10.3390/jcm8040527





