Brain Network Organization Correlates with Autistic Features in Preschoolers with Autism Spectrum Disorders and in Their Fathers: Preliminary Data from a DWI Analysis
Abstract
:1. Introduction
2. Experimental Section
2.1. Participants
2.2. Clinical Assessment
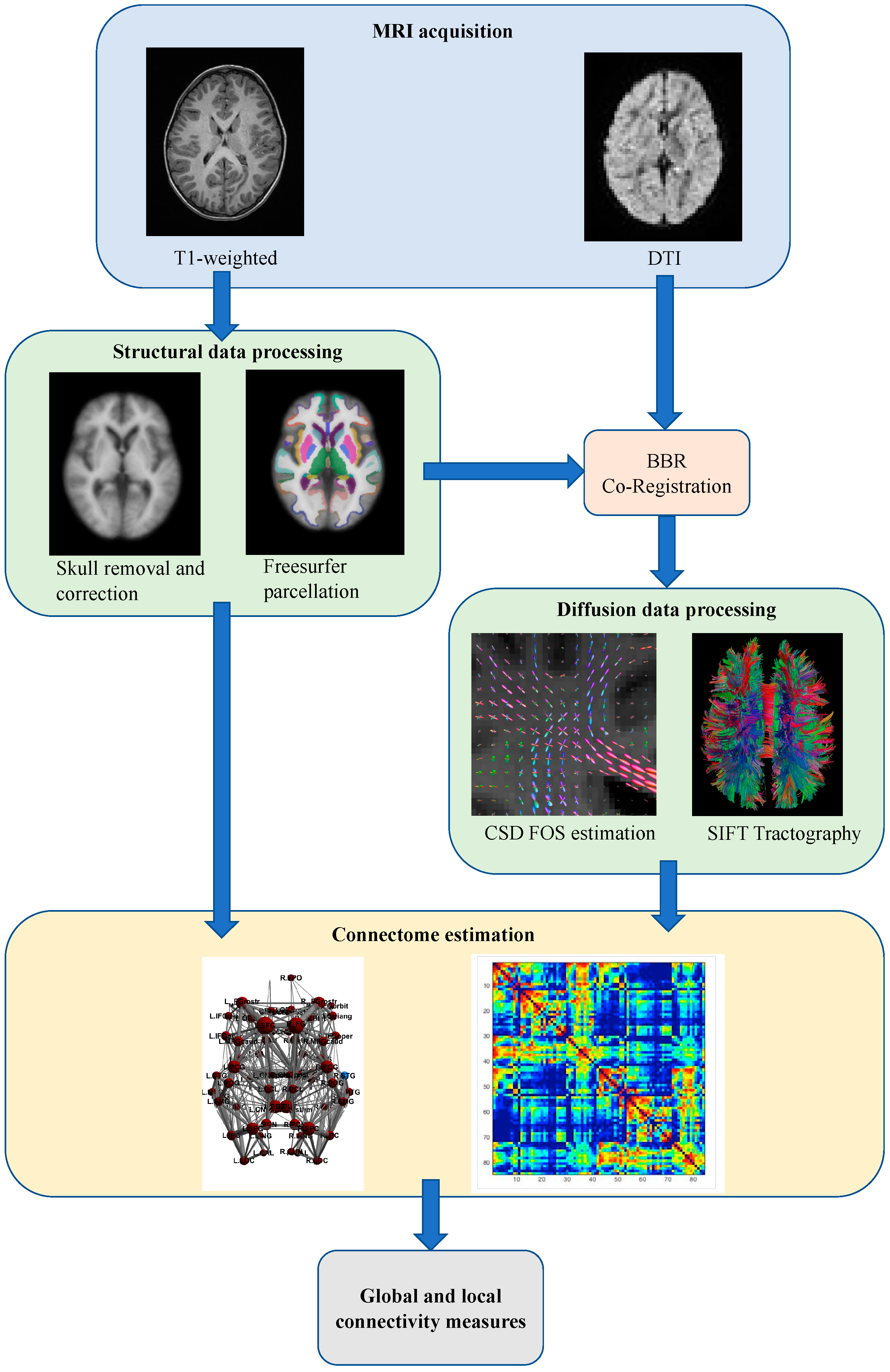
2.3. Image Acquisition
2.4. Structural Data Analysis
2.5. Diffusion Data Analysis and Connectome Construction
2.6. Graph Analysis of Connectomes
2.7. Statistical Analysis
3. Results
3.1. Correlation between ASD Symptoms and Brain Network Measures in ASD Probands
3.1.1. Global Measures
3.1.2. Nodal Measure
3.2. Correlation between BAP Traits and Brain Network Measures in Fathers of ASD Probands
3.2.1. Global Measures
3.2.2. Nodal Measures
3.3. Brain Areas Shared in Fathers and in Their ASD Probands
4. Discussion
4.1. Correlations between Autistic Severity and White Matter Measures in Children
4.2. Correlations between BAP Traits and White Matter Measures in Fathers
4.3. Overlap between Fathers and Their Children
4.4. Strengths and Limitations
4.5. Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Baio, J.; Wiggins, L.; Christensen, D.L.; Maenner, M.J.; Daniels, J.; Warren, Z.; Kurzius-Spencer, M.; Zahorodny, W.; Robinson Rosenberg, C.; White, T.; et al. Prevalence of Autism Spectrum Disorder Among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2014. MMWR Surveill. Summ. 2018, 67, 1–23. [Google Scholar] [CrossRef]
- Narzisi, A.; Posada, M.; Barbieri, F.; Chericoni, N.; Ciuffolini, D.; Pinzino, M.; Romano, R.; Scattoni, M.L.; Tancredi, R.; Calderoni, S.; et al. Prevalence of Autism Spectrum Disorder in a large Italian catchment area: A school-based population study within the ASDEU project. Epidemiol. Psychiatr. Sci. 2018, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Leventhal, B.L.; Koh, Y.J.; Fombonne, E.; Laska, E.; Lim, E.C.; Cheon, K.A.; Kim, S.J.; Kim, Y.K.; Lee, H.; et al. Prevalence of autism spectrum disorders in a total population sample. Am. J. Psychiatry 2011, 168, 904–912. [Google Scholar] [CrossRef]
- Hallmayer, J.; Cleveland, S.; Torres, A.; Phillips, J.; Cohen, B.; Torigoe, T.; Miller, J.; Fedele, A.; Collins, J.; Smith, K.; et al. Genetic heritability and shared environmental factors among twin pairs with autism. Arch. Gen. Psychiatry 2011, 68, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Tick, B.; Bolton, P.; Happé, F.; Rutter, M.; Rijsdijk, F. Heritability of autism spectrum disorders: A meta-analysis of twin studies. J. Child Psychol. Psychiatry 2016, 57, 585–595. [Google Scholar] [CrossRef]
- Anney, R.; Klei, L.; Pinto, D.; Regan, R.; Conroy, J.; Magalhaes, T.R.; Correia, C.; Abrahams, B.S.; Sykes, N.; Pagnamenta, A.T.; et al. A genome-wide scan for common alleles affecting risk for autism. Hum. Mol. Genet. 2011, 19, 4072–4082. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, A.; Zhang, R.; Yao, V.; Theesfeld, C.L.; Wong, A.K.; Tadych, A.; Volfovsky, N.; Packer, A.; Lash, A.; Troyanskaya, O.G. Genome-wide prediction and functional characterization of the genetic basis of autism spectrum disorder. Nat. Neurosci. 2016, 19, 1454. [Google Scholar] [CrossRef] [PubMed]
- Weiss, L.A.; Dan, E.A.; Gene Discovery Project of Johns Hopkins & the Autism Consortium. A genome-wide linkage and association scan reveals novel loci for autism. Nature 2009, 461, 802. [Google Scholar]
- Pinto, D.; Pagnamenta, A.T.; Klei, L.; Anney, R.; Merico, D.; Regan, R.; Conroy, J.; Magalhaes, T.R.; Correia, C.; Abrahams, B.S.; et al. Functional impact of global rare copy number variation in autism spectrum disorders. Nature 2010, 466, 368–372. [Google Scholar] [CrossRef]
- De Rubeis, S.; Buxbaum, J.D. Genetics and genomics of autism spectrum disorder: Embracing complexity. Hum. Mol. Genet. 2015, 24, R24–R31. [Google Scholar] [CrossRef]
- Iossifov, I.; O’Roak, B.J.; Sanders, S.J.; Ronemus, M.; Krumm, N.; Levy, D.; Stessman, H.A.; Witherspoon, K.T.; Vives, L.; Patterson, K.E.; et al. The contribution of de novo coding mutations to autism spectrum disorder. Nature 2014, 515, 216. [Google Scholar] [CrossRef]
- Bourgeron, T. From the genetic architecture to synaptic plasticity in autism spectrum disorder. Nat. Rev. Neurosci. 2015, 16, 551. [Google Scholar] [CrossRef]
- de la Torre-Ubieta, L.; Won, H.; Stein, J.L.; Geschwind, D.H. Advancing the understanding of autism disease mechanisms through genetics. Nat. Med. 2016, 22, 345–361. [Google Scholar] [CrossRef] [PubMed]
- Constantino, J.N.; Zhang, Y.; Frazier, T.; Abbacchi, A.M.; Law, P. Sibling Recurrence and the Genetic Epidemiology of Autism. Am. J. Psychiatry 2010, 167, 1349–1356. [Google Scholar] [CrossRef] [PubMed]
- Virkud, Y.V.; Todd, R.D.; Abbacchi, A.M.; Zhang, Y.; Constantino, J.N. Familial aggregation of quantitative autistic traits in multiplex versus simplex autism. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2009, 150, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Losh, M.; Childress, D.; Lam, K.; Piven, J. Defining key features of the broad autism phenotype: A comparison across parents of multiple- and single-incidence autism families. Am. J. Med. Genet. B Neuropsychiatr Genet. 2008, 147, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Bralten, J.; van Hulzen, K.J.; Martens, M.B.; Galesloot, T.E.; Arias Vasquez, A.; Kiemeney, L.A.; Buitelaar, J.K.; Muntjewerff, J.W.; Franke, B.; Poelmans, G. Autism spectrum disorders and autistic traits share genetics and biology. Mol. Psychiatry 2018, 23, 1205–1212. [Google Scholar] [CrossRef]
- Piven, J.; Palmer, P.; Jacobi, D.; Childress, D.; Arndt, S. Broader autism phenotype: Evidence from a family history study of multiple-incidence autism families. Am. J. Psychiatry 1997, 154, 185–190. [Google Scholar]
- Gerdts, J.; Bernier, R. The broader autism phenotype and its implications on the etiology and treatment of autism spectrum disorders. Autism Res. Treat. 2011. [Google Scholar] [CrossRef]
- Sucksmith, E.; Roth, I.; Hoekstra, R.A. Autistic traits below the clinical threshold: Re-examining the broader autism phenotype in the 21st century. Neuropsychol. Rev. 2011, 21, 360–389. [Google Scholar] [CrossRef] [PubMed]
- Rubenstein, E.; Chawla, D. Broader autism phenotype in parents of children with autism: A systematic review of percentage estimates. J. Child. Fam. Stud. 2018, 27, 1705–1720. [Google Scholar] [CrossRef]
- Rasetti, R.; Weinberger, D.R. Intermediate phenotypes in psychiatric disorders. Curr. Opin. Genet. Dev. 2011, 21, 340–348. [Google Scholar] [CrossRef]
- Volkmar, F.R.; Klin, A.; Schultz, R.; Bronen, R.; Marans, W.D.; Sparrow, S.; Cohen, D.J. Asperger’s syndrome. J. Am. Acad. Child. Adolesc. Psychiatry 1996, 35, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Billeci, L.; Calderoni, S.; Conti, E.; Gesi, C.; Carmassi, C.; Dell’Osso, L.; Cioni, G.; Muratori, F.; Guzzetta, A. The broader autism endo(phenotype): Neurostructural and neurofunctional correlates in parents of individuals with Autism Spectrum Disorders. Front. Neurosci. 2016, 10, 276. [Google Scholar] [CrossRef] [PubMed]
- Rojas, D.C.; Smith, J.A.; Benkers, T.L.; Camou, S.L.; Reite, M.L.; Rogers, S.J. Hippocampus and amygdala volumes in parents of children with autistic disorder. Am. J. Psychiatry 2004, 161, 2038–2044. [Google Scholar] [CrossRef]
- Peterson, E.; Schmidt, G.L.; Tregellas, J.R.; Winterrowd, E.; Kopelioff, L.; Hepburn, S.; Reite, M.; Rojas, D.C. A voxel-based morphometry study of gray matter in parents of children with autism. Neuroreport 2006, 17, 1289–1292. [Google Scholar] [CrossRef]
- Dawson, G.; Webb, S.J.; Wijsman, E.; Schellenberg, G.; Estes, A.; Munson, J.; Faja, S. Neurocognitive and electrophysiological evidence of altered face processing in parents of children with autism: Implications for a model of abnormal development of social brain circuitry in autism. Dev. Psychopathol. 2005, 17, 679–697. [Google Scholar] [CrossRef]
- Wilson, L.B.; Jason, R.; Tregellas, J.R.; Slason, E.; Pasko, B.E.; Hepburn, S.; Rojas, D.C. Phonological processing in first-degree relatives of individuals with autism: An fMRI study. Hum. Brain Map. 2013, 34, 1447–1463. [Google Scholar] [CrossRef]
- Greimel, E.; Schulte-Rüther, M.; Kircher, T.; Kamp-Becker, I.; Remschmidt, H.; Fink, G.R.; Herpertz-Dahlmann, B.; Konrad, K. Neural mechanisms of empathy in adolescents with autism spectrum disorder and their fathers. NeuroImage 2010, 49, 1055–1065. [Google Scholar] [CrossRef]
- Yucel, G.H.; Belger, A.; Bizzell, J.; Parlier, M.; Adolphs, R.; Piven, J. Abnormal neural activation to faces in the parents of children with autism. Cereb. Cortex 2014, 25, 4653–4666. [Google Scholar] [CrossRef] [PubMed]
- Baron-Cohen, S.; Ring, H.; Wheelwright, S.; Williams, S.; Brammer, M.; Bullmore, E. fMRI of parents of children with Asperger Syndrome: A pilot study. Brain Cogn. 2006, 61, 122–130. [Google Scholar] [CrossRef]
- Baron-Cohen, S. The extreme male brain theory of autism. Trends Cogn. Sci. 2002, 6, 248–254. [Google Scholar] [CrossRef]
- Lebel, C.; Treit, S.; Beaulieu, C. A review of diffusion MRI of typical white matter development from early childhood to young adulthood. NMR Biomed. 2019, 32, e3778. [Google Scholar] [CrossRef]
- Just, M.A.; Keller, T.A.; Malave, V.L.; Kana, R.K.; Varma, S. Autism as a neural systems disorder: A theory of frontal-posterior underconnectivity. Neurosci. Biobehav. Rev. 2012, 36, 1292–1313. [Google Scholar] [CrossRef]
- Kana, R.K.; Libero, L.E.; Moore, M.S. Disrupted cortical connectivity theory as an explanatory model for autism spectrum disorders. Phys. Life Rev. 2011, 8, 410–437. [Google Scholar] [CrossRef]
- Müller, R.A.; Fishman, I. Brain Connectivity and Neuroimaging of Social Networks in Autism. Trends Cogn. Sci. 2018, 22, 1103–1116. [Google Scholar] [CrossRef]
- Ismail, M.M.; Keynton, R.S.; Mostapha, M.M.; ElTanboly, A.H.; Casanova, M.F.; Gimel’farb, G.L.; El-Baz, A. Studying Autism Spectrum Disorder with Structural and Diffusion Magnetic Resonance Imaging: A Survey. Front. Hum. Neurosci. 2016, 10, 211. [Google Scholar] [CrossRef]
- Brothers, L. The social brain: A project for integrating primate behavior and neurophysiology in a new domain. Concepts Neurosci. 1990, 1, 27–51. [Google Scholar]
- Cheon, K.A.; Kim, Y.S.; Oh, S.H.; Park, S.Y.; Yoon, H.W.; Herrington, J.; Nair, A.; Koh, Y.J.; Jang, D.P.; Kim, Y.B.; et al. Involvement of the anterior thalamic radiation in boys with high functioning autism spectrum disorders: A diffusion tensor imaging study. Brain Res. 2011, 1417, 77–86. [Google Scholar] [CrossRef]
- Langen, M.; Leemans, A.; Johnston, P.; Ecker, C.; Daly, E.; Murphy, C.M.; Dell’acqua, F.; Durston, S.; AIMS Consortium; Murphy, D.G. Fronto-striatal circuitry and inhibitory control in autism: Findings from diffusion tensor imaging tractography. Cortex 2012, 48, 183–193. [Google Scholar] [CrossRef]
- Sivaswamy, L.; Kumar, A.; Rajan, D.; Behen, M.; Muzik, O.; Chugani, D.; Chugani, H. A diffusion tensor imaging study of the cerebellar pathways in children with autism spectrum disorder. J. Child. Neurol. 2010, 25, 1223–1231. [Google Scholar] [CrossRef]
- Thompson, A.; Murphy, D.; Dell’Acqua, F.; Ecker, C.; McAlonan, G.; Howells, H.; Baron-Cohen, S.; Lai, M.C.; Lombardo, M.V.; MRC AIMS Consortium; Catani, M. Impaired Communication Between the Motor and Somatosensory Homunculus Is Associated with Poor Manual Dexterity in Autism Spectrum Disorder. Biol. Psychiatry 2017, 81, 211–219. [Google Scholar] [CrossRef]
- Brito, A.R.; Vasconcelos, M.M.; Domingues, R.C.; Hygino da Cruz, L.C., Jr.; Rodrigues Lde, S.; Gasparetto, E.L.; Calçada, C.A. Diffusion tensor imaging findings in school-aged autistic children. J. Neuroimaging 2009, 19, 337–343. [Google Scholar] [CrossRef]
- Billeci, L.; Calderoni, S.; Tosetti, M.; Catani, M.; Muratori, F. White matter connectivity in children with autism spectrum disorders: A tract-based spatial statistics study. BMC Neurol. 2012, 12, 148. [Google Scholar] [CrossRef]
- Conti, E.; Calderoni, S.; Marchi, V.; Muratori, F.; Cioni, G.; Guzzetta, A. The first 1000 days of the autistic brain: A systematic review of diffusion imaging studies. Front. Hum. Neurosci. 2015, 9, 159. [Google Scholar] [CrossRef] [PubMed]
- Wolff, J.J.; Gu, H.; Gerig, G.; Elison, J.T.; Styner, M.; Gouttard, S.; Botteron, K.N.; Dager, S.R.; Dawson, G.; Estes, A.M.; et al. Differences in white matter fiber tract development present from 6 to 24 months in infants with autism. Am. J. Psychiatry 2012, 169, 589–600. [Google Scholar] [CrossRef]
- Wolff, J.J.; Jacob, S.; Elison, J.T. The journey to autism: Insights from neuroimaging studies of infants and toddlers. Dev. Psychopathol. 2018, 30, 479–495. [Google Scholar] [CrossRef] [PubMed]
- Pardini, M.; Garaci, F.G.; Bonzano, L.; Roccatagliata, L.; Palmieri, M.G.; Pompili, E.; Coniglione, F.; Krueger, F.; Ludovici, A.; Floris, R.; et al. White matter reduced streamline coherence in young men with autism and mental retardation. Eur. J. Neurol. 2009, 16, 1185–1190. [Google Scholar] [CrossRef]
- Nickel, K.; Tebartz van Elst, L.; Perlov, E.; Endres, D.; Müller, G.T.; Riedel, A.; Fangmeier, T.; Maier, S. Altered white matter integrity in adults with autism spectrum disorder and an IQ >100: A diffusion tensor imaging study. Acta Psychiatr. Scand. 2017, 135, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Picci, G.; Gotts, S.J.; Scherf, K.S. A theoretical rut: Revisiting and critically evaluating the generalized under/over-connectivity hypothesis of autism. Dev. Sci. 2016, 19, 524–549. [Google Scholar] [CrossRef] [PubMed]
- Bloy, L.; Ingalhalikar, M.; Batmanghelich, N.K.; Schultz, R.T.; Roberts, T.P.; Verma, R. An integrated framework for high angular resolution diffusion imaging-based investigation of structural connectivity. Brain Connect. 2012, 2, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Bullmore, E.; Sporns, O. Complex brain networks: Graph theoretical analysis of structural and functional systems. Nat. Rev. Neurosci. 2009, 10, 186–198. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.; Rutter, M.; DiLavore, P.C.; Risi, S.; Gotham, K.; Bishop, S. Autism Diagnostic Observation Schedule, 2nd ed.; Western Psychological Services: Torrance, CA, USA, 2012. [Google Scholar]
- Ruta, L.; Mazzone, D.; Mazzone, L.; Wheelwright, S.; Baron-Cohen, S. The Autism-Spectrum Quotient-Italian version: A cross-cultural confirmation of the broader autism phenotype. J. Autism Dev. Disord. 2012, 42, 625–633. [Google Scholar] [CrossRef]
- Ritchie, S.J.; Cox, S.R.; Shen, X.; Lombardo, M.V.; Reus, L.M.; Alloza, C.; Harris, M.A.; Alderson, H.L.; Hunter, S.; Neilson, E.; et al. Sex Differences in the adult human brain: Evidence from 5216 UK biobank participants. Cereb. Cortex 2017, 28, 2959–2975. [Google Scholar] [CrossRef]
- Retico, A.; Giuliano, A.; Tancredi, R.; Cosenza, A.; Apicella, F.; Narzisi, A.; Biagi, L.; Tosetti, M.; Muratori, F.; Calderoni, S. The effect of gender on the neuroanatomy of children with autism spectrum disorders: A support vector machine case-control study. Mol. Autism 2016, 7, 5. [Google Scholar] [CrossRef]
- Hus, V.; Gotham, K.; Lord, C. Standardizing ADOS domain scores: Separating severity of social affect and restricted and repetitive behaviors. J. Autism Dev. Disord. 2014, 44, 2400–2412. [Google Scholar] [CrossRef]
- First, M.B.; Williams, J.B.W.; Karg, R.S.; Spitzer, R.L. Structured Clinical Interview for DSM-5-Research Version (SCID-5 for DSM-5, Research Version; SCID-5-RV); American Psychiatric Association: Arlington, VA, USA, 2015. [Google Scholar]
- Dell’Osso, L.; Shear, M.K.; Carmassi, C.; Rucci, P.; Maser, J.D.; Frank, E.; Endicott, J.; Lorettu, L.; Altamura, C.A.; Carpiniello, B.; et al. Validity and reliability of the structured clinical interview for the trauma and loss spectrum (SCI-TALS). Clin. Pract. Epidemiol. Ment. Health 2008, 4, 2. [Google Scholar] [CrossRef]
- Dell’Osso, L.; Carmassi, C.; Rucci, P.; Conversano, C.; Shear, M.K.; Calugi, S.; Maser, J.D.; Endicott, J.; Fagiolini, A.; Cassano, G.B. A multidimensional spectrum approach to post-traumatic stress disorder: Comparison between the structured clinical interview for trauma and loss spectrum (SCI-TALS) and the self-report instrument (TALS-SR). Compr. Psychiatry 2009, 50, 485–490. [Google Scholar] [CrossRef]
- Dell’Osso, L.; Armani, A.; Rucci, P.; Frank, E.; Fagiolini, A.; Corretti, G.; Shear, M.K.; Grochocinski, V.J.; Maser, J.D.; Endicott, J.; et al. Measuring mood spectrum: Comparison of interview (SCI-MOODS) and self-report (MOODS-SR) instruments. Compr. Psychiatry 2002, 43, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Pannek, K.; Guzzetta, A.; Colditz, P.B.; Rose, S.E. Diffusion MRI of the neonate brain: Acquisition, processing and analysis techniques. Pediatr. Radiol. 2012, 42, 1169–1182. [Google Scholar] [CrossRef]
- Fischl, B. FreeSurfer. NeuroImage 2012, 62, 774–781. [Google Scholar] [CrossRef]
- Croteau-Chonka, E.C.; Dean, D.C., 3rd; Remer, J.; Dirks, H.; O’Muircheartaigh, J.; Deoni, S.C.L. Examining the relationships between cortical maturation and white matter myelination throughout early childhood. NeuroImage 2016, 125, 413–421. [Google Scholar] [CrossRef]
- Lowe, J.R.; MacLean, P.C.; Caprihan, A.; Ohls, R.K.; Qualls, C.; VanMeter, J.; Phillips, J.P. Comparison of cerebral volume in children aged 18–22 and 36–47 months born preterm and term. J. Child Neurol. 2012, 27, 172–177. [Google Scholar] [CrossRef]
- Travis, K.E.; Curran, M.M.; Torres, C.; Leonard, M.K.; Brown, T.T.; Dale, A.M.; Elman, J.L.; Halgren, E. Age-related changes in tissue signal properties within cortical areas important for word understanding in 12- to 19-month-old infants. Cereb. Cortex 2014, 24, 1948–1955. [Google Scholar] [CrossRef]
- Desikan, R.S.; Ségonne, F.; Fischl, B.; Quinn, B.T.; Dickerson, B.C.; Blacker, D.; Buckner, R.L.; Dale, A.M.; Maguire, R.P.; Hyman, B.T.; et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage 2006, 31, 968–980. [Google Scholar] [CrossRef]
- Fischl, B.; Salat, D.H.; Busa, E.; Albert, M.; Dieterich, M.; Haselgrove, C.; van der Kouwe, A.; Killiany, R.; Kennedy, D.; Klaveness, S.; et al. Whole brain segmentation: Automated labeling of neuroanatomical structures in the human brain. Neuron 2002, 33, 341–355. [Google Scholar] [CrossRef]
- Pannek, K.; Boyd, R.N.; Fiori, S.; Guzzetta, A.; Rose, S.E. Assessment of the structural brain network reveals altered connectivity in children with unilateral cerebral palsy due to periventricular white matter lesions. NeuroImage Clin. 2014, 5, 84–92. [Google Scholar] [CrossRef]
- Nam, H.; Park, H.J. Distortion correction of high b-valued and high angular resolution diffusion images using iterative simulated images. NeuroImage 2011, 57, 968–978. [Google Scholar] [CrossRef]
- Andersson, J.L.R.; Sotiropoulos, S.N. An integrated approach to correction for off-resonance effects and subject movement in diffusion MR imaging. NeuroImage 2016, 125, 1063–1078. [Google Scholar] [CrossRef]
- Sled, J.G.; Zijdenbos, A.P.; Evans, A.C. A nonparametric method for automatic correction of intensity nonuniformity in MRI data. IEEE Trans. Med. Imaging 1998, 17, 87–97. [Google Scholar] [CrossRef]
- Tournier, J.D.; Calamante, F.; Connelly, A. Determination of the appropriate b value and number of gradient directions for high-angular-resolution diffusion-weighted imaging. NMR Biomed. 2013, 26, 1775–1786. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.E.; Tournier, J.D.; Calamante, F.; Connelly, A. Anatomically-constrained tractography: Improved diffusion MRI streamlines tractography through effective use of anatomical information. NeuroImage 2012, 62, 1924–1938. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.E.; Tournier, J.D.; Calamante, F.; Connelly, A. SIFT: Spherical-deconvolution informed filtering of tractograms. NeuroImage 2013, 67, 298–312. [Google Scholar] [CrossRef] [PubMed]
- Greve, D.N.; Fischl, B. Accurate and robust brain image alignment using boundary-based registration. NeuroImage 2009, 48, 63–72. [Google Scholar] [CrossRef]
- Patenaude, B.; Smith, S.M.; Kennedy, D.N.; Jenkinson, M. A Bayesian model of shape and appearance for subcortical brain segmentation. NeuroImage 2011, 56, 907–922. [Google Scholar] [CrossRef] [PubMed]
- de Reus, M.A.; van den Heuvel, M.P. Estimating false positives and negatives in brain networks. NeuroImage 2013, 70, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Rubinov, M.; Sporns, O. Complex network measures of brain connectivity: Uses and interpretations. NeuroImage 2010, 52, 1059–1069. [Google Scholar] [CrossRef] [PubMed]
- Latora, V.; Marchiori, M. Efficient behavior of small-world networks. Phys. Rev. Lett. 2001, 87, 198701. [Google Scholar] [CrossRef] [PubMed]
- Onnela, J.P.; Saramaki, J.; Kertesz, J.; Kaski, K. Intensity and coherence of motifs in weighted complex networks. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2005, 71, 065103. [Google Scholar] [CrossRef] [PubMed]
- Watts, D.J.; Strogatz, S.H. Collective dynamics of ‘‘small-world’’ networks. Nature 1998, 393, 440–442. [Google Scholar] [CrossRef]
- Muldoon, S.F.; Bridgeford, E.W.; Bassett, D.S. Small-World Propensity and Weighted Brain Networks. Sci. Rep. 2016, 6, 22057. [Google Scholar] [CrossRef] [PubMed]
- Kintali, S. Betweenness centrality: Algorithms and lower bounds. arXiv 2008, arXiv:0809.1906v0802. [Google Scholar]
- Cao, M.; Huang, H.; Peng, Y.; Dong, Q.; He, Y. Toward Developmental Connectomics of the Human Brain. Front. Neuroanat. 2016, 10, 25. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge Academic: New York, NY, USA, 1988. [Google Scholar]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Voelbel, G.T.; Bates, M.E.; Buckman, J.F.; Pandina, G.; Hendren, R.L. Caudate Nucleus Volume and Cognitive Performance: Are they related in Childhood Psychopathology? Biol. Psychiatry 2006, 60, 942–950. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.H.; Waiter, G.D.; Perra, O.; Perrett, D.I.; Whiten, A. An fMRI study of joint attention experience. NeuroImage 2005, 25, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, A.; Ross, K.; Uddin, L.Q.; Sklar, A.B.; Castellanos, F.X.; Milham, M.P. Functional brain correlates of social and nonsocial processes in autism spectrum disorders: An activation likelihood estimation meta-analysis. Biol. Psychiatry 2009, 65, 63–74. [Google Scholar] [CrossRef]
- Keown, C.L.; Shih, P.; Nair, A.; Peterson, N.; Mulvey, M.E.; Müller, R.A. Local functional overconnectivity in posterior brain regions is associated with symptom severity in autism spectrum disorders. Cell Rep. 2013, 5, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Supekar, K.; Uddin, L.Q.; Khouzam, A.; Phillips, J.; Gaillard, W.D.; Kenworthy, L.E.; Yerys, B.E.; Vaidya, C.J.; Menon, V. Brain hyperconnectivity in children with autism and its links to social deficits. Cell Rep. 2013, 5, 738–747. [Google Scholar] [CrossRef]
- Li, H.; Xue, Z.; Ellmore, T.M.; Frye, R.E.; Wong, S.T. Network-based analysis reveals stronger local diffusion-based connectivity and different correlations with oral language skills in brains of children with high functioning autism spectrum disorders. Hum. Brain Map. 2014, 35, 396–413. [Google Scholar] [CrossRef]
- Belmonte, M.K.; Allen, G.; Beckel-Mitchener, A.; Boulanger, L.M.; Carper, R.A.; Webb, S.J. Autism and abnormal development of brain connectivity. J. Neurosci. 2004, 24, 9228–9231. [Google Scholar] [CrossRef]
- Just, M.A.; Cherkassky, V.L.; Keller, T.A.; Minshew, N.J. Cortical activation and synchronization during sentence comprehension in high-functioning autism: Evidence of underconnectivity. Brain 2004, 127, 1811–1821. [Google Scholar] [CrossRef]
- Cheng, Y.; Chou, K.H.; Chen, I.Y.; Fan, Y.T.; Decety, J.; Lin, C.P. Atypical development of white matter microstructure in adolescents with autism spectrum disorders. NeuroImage 2010, 50, 873–882. [Google Scholar] [CrossRef]
- Nair, A.; Carper, R.A.; Abbott, A.E.; Chen, C.P.; Solders, S.; Nakutin, S.; Datko, M.C.; Fishman, I.; Müller, R.A. Regional specificity of aberrant thalamocortical connectivity in autism. Hum. Brain Map. 2015, 36, 4497–4511. [Google Scholar] [CrossRef]
- Micheau, J.; Vimeney, A.; Normand, E.; Mulle, C.; Riedel, G. Impaired hippocampus-dependent spatial flexibility and sociability represent autism-like phenotypes in GluK2 mice. Hippocampus 2014, 24, 1059–1069. [Google Scholar] [CrossRef]
- Lewis, J.D.; Theilmann, R.J.; Townsend, J.; Evans, A.C. Network efficiency in autism spectrum disorder and its relation to brain overgrowth. Front. Hum. Neurosci. 2013, 7, 845. [Google Scholar] [CrossRef]
- Kim, C.; Johnson, N.F.; Cilles, S.E.; Gold, B.T. Common and distinct mechanisms of cognitive flexibility in prefrontal cortex. J. Neurosci. 2011, 31, 4771–4779. [Google Scholar] [CrossRef]
- Weng, S.J.; Wiggins, J.L.; Peltier, S.J.; Carrasco, M.; Risi, S.; Lord, C.; Monk, C.S. Alterations of resting state functional connectivity in the default network in adolescents with autism spectrum disorders. Brain Res. 2010, 1313, 202–214. [Google Scholar] [CrossRef]
- Monk, C.S.; Peltier, S.J.; Wiggins, J.L.; Weng, S.J.; Carrasco, M.; Risi, S.; Lord, C. Abnormalities of intrinsic functional connectivity in autism spectrum disorders. NeuroImage 2009, 47, 764–772. [Google Scholar] [CrossRef]
- Gibbard, C.R.; Ren, J.; Seunarine, K.K.; Clayden, J.D.; Skuse, D.H.; Clark, C.A. White matter microstructure correlates with autism trait severity in a combined clinical-control sample of high-functioning adults. NeuroImage Clin. 2013, 3, 106–114. [Google Scholar] [CrossRef]
- Hirose, K.; Miyata, J.; Sugihara, G.; Kubota, M.; Sasamoto, A.; Aso, T.; Fukuyama, H.; Murai, T.; Takahashi, H. Fiber tract associated with autistic traits in healthy adults. J. Psychiatry Res. 2014, 59, 117–124. [Google Scholar] [CrossRef]
- Iidaka, T.; Miyakoshi, M.; Harada, T.; Nakai, T. White matter connectivity between superior temporal sulcus and amygdala is associated with autistic trait in healthy humans. Neurosci. Lett. 2012, 510, 154–158. [Google Scholar] [CrossRef]
- Bradstreet, L.E.; Hecht, E.E.; King, T.Z.; Turner, J.L.; Robins, D.L. Associations between autistic traits and fractional anisotropy values in white matter tracts in a nonclinical sample of young adults. Exp. Brain Res. 2017, 235, 259–267. [Google Scholar] [CrossRef]
- Takeuchi, H.; Taki, Y.; Thyreau, B.; Sassa, Y.; Hashizume, H.; Sekiguchi, A.; Nagase, T.; Nouchi, R.; Fukushima, A.; Kawashima, R. White matter structures associated with empathizing and systemizing in young adults. NeuroImage 2013, 77, 222–236. [Google Scholar] [CrossRef]
- Jakab, A.; Emri, M.; Spisak, T.; Szeman-Nagy, A.; Beres, M.; Kis, S.A.; Molnar, P.; Berenyi, E. Autistic traits in neurotypical adults: Correlates of graph theoretical functional network topology and white matter anisotropy patterns. PLoS ONE 2013, 8, e60982. [Google Scholar] [CrossRef]
- Wallace, G.L.; Shaw, P.; Lee, N.R.; Clasen, L.S.; Raznahan, A.; Lenroot, R.K.; Martin, A.; Giedd, J.N. Distinct cortical correlates of autistic versus antisocial traits in a longitudinal sample of typically developing youth. J. Neurosci. 2012, 32, 4856–4860. [Google Scholar] [CrossRef]
- Di Martino, A.; Shehzad, Z.; Kelly, C.; Roy, A.K.; Gee, D.G.; Uddin, L.Q.; Gotimer, K.; Klein, D.F.; Castellanos, X.; Milham, M.P. Relationship between cingulo-insular functional connectivity and autistic traits in neurotypical adults. Am. J. Psychiatry 2009, 166, 891–899. [Google Scholar] [CrossRef]
- McFadden, K.L.; Hepburn, S.; Winterrowd, E.; Schmidt, G.L.; Rojas, D.C. Abnormalities in gamma-band responses to language stimuli in firstdegree relatives of children with autism spectrum disorder: An MEG study. BMC Psychiatry 2012, 12, 213. [Google Scholar] [CrossRef]
- Buard, I.; Rogers, S.J.; Hepburn, S.; Kronberg, E.; Rojas, D.C. Altered oscillation patterns and connectivity during picture naming in autism. Front. Hum. Neurosci. 2013, 7, 742. [Google Scholar] [CrossRef]
- Gökçen, E.; Petrides, K.V.; Hudry, K.; Frederickson, N.; Smillie, L.D. Sub-threshold autism traits: The role of trait emotional intelligence and cognitive flexibility. Br. J. Psychol. 2014, 105, 187–199. [Google Scholar] [CrossRef]
- Nagy, K.; Greenlee, M.W.; Kovács, G. The Lateral Occipital Cortex in the Face Perception Network: An Effective Connectivity Study. Front. Psychol. 2012, 3, 141. [Google Scholar] [CrossRef]
- Piggot, J.; Kwon, H.; Mobbs, D.; Blasey, C.; Lotspeich, L.; Menon, V.; Bookheimer, S.; Reiss, A.L. Emotional attribution in high-functioning individuals with autistic spectrum disorder: A functional imaging study. J. Am. Acad. Child Adolesc. Psychiatry 2004, 43, 473–480. [Google Scholar] [CrossRef]
- Dehaene, S.; Piazza, M.; Pinel, P.; Cohen, L. Three parietal circuits for number processing. Cogn. Neuropsychol. 2003, 20, 487–506. [Google Scholar] [CrossRef]
- Pelphrey, K.; Adolphs, R.; Morris, J.P. Neuroanatomical substrates of social cognition dysfunction in autism. Ment. Retard. Dev. Disabil. Res. Rev. 2004, 10, 259–271. [Google Scholar] [CrossRef]
- Harris, G.J.; Chabris, C.F.; Clark, J.; Urban, T.; Aharon, I.; Steele, S.; McGrath, L.; Condouris, K.; Tager-Flusberg, H. Brain activation during semantic processing in autism spectrum disorders via functional magnetic resonance imaging. Brain Cogn. 2006, 61, 54–68. [Google Scholar] [CrossRef]
- Lee, J.E.; Bigler, E.D.; Alexander, A.L.; Lazar, M.; DuBray, M.B.; Chung, M.K.; Johnson, M.; Morgan, J.; Miller, J.N.; McMahon, W.M.; et al. Diffusion tensor imaging of white matter in the superior temporal gyrus and temporal stem in autism. Neurosci. Lett. 2007, 424, 127–132. [Google Scholar] [CrossRef]
- Karnath, H.O. New insights into the functions of the superior temporal cortex. Nat. Rev. Neurosci. 2001, 2, 568–576. [Google Scholar] [CrossRef]
- Bigler, E.D.; Mortensen, S.; Neeley, E.S.; Ozonoff, S.; Krasny, L.; Johnson, M.; Lu, J.; Provencal, S.L.; McMahon, W.; Lainhart, J.E. Superior temporal gyrus, language function, and autism. Dev. Neuropsychol. 2007, 31, 217–238. [Google Scholar] [CrossRef]
- Waberski, T.D.; Kreitschmann-Andermahr, I.; Kawohl, W.; Darvas, F.; Ryang, Y.; Rodewald, M.; Gobbelé, R.; Buchner, H. Spatio-temporal source imaging reveals subcomponents of the human auditory mismatch negativity in the cingulum and right inferior temporal gyrus. Neurosci. Lett. 2001, 308, 107–110. [Google Scholar] [CrossRef]
- Okuda, J.; Fujii, T.; Ohtake, H.; Tsukiura, T.; Tanji, K.; Suzuki, K.; Kawashima, R.; Fukuda, H.; Itoh, M.; Yamadori, A. Thinking of the future and past: The roles of the frontal pole and the medial temporal lobes. NeuroImage 2003, 19, 1369–1380. [Google Scholar] [CrossRef]
- Jiao, Y.; Chen, R.; Ke, X.; Chu, K.; Lu, Z.; Herskovits, E.H. Predictive models of autism spectrum disorder based on brain regional cortical thickness. NeuroImage 2010, 50, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Sokolowski, K.; Corbin, J.G. Wired for behaviors: From development to function of innate limbic system circuitry. Front. Mol. Neurosci. 2012, 5, 55. [Google Scholar] [CrossRef] [PubMed]
- Ruggeri, B.; Sarkans, U.; Schumann, G.; Persico, A.M. Biomarkers in autism spectrum disorder: The old and the new. Psychopharmacology 2014, 231, 1201–1216. [Google Scholar] [CrossRef] [PubMed]
| ASD Children (n = 16) | |
| Age (years) | 3.1 (1.3) |
| ADOS, CSS | 6.4 (1.3) |
| ADOS, SA | 7.8 (1.4) |
| ADOS, RRB | 6.1 (1.7) |
| Performance IQ a | 83.7 (13.7) |
| Fathers of children with ASD (n = 16) | |
| Age (years) | 43.5 (5.1) |
| Autism-Spectrum Quotient (AQ), total | 13.5 (7.2) |
| AQ range | 4–28 |
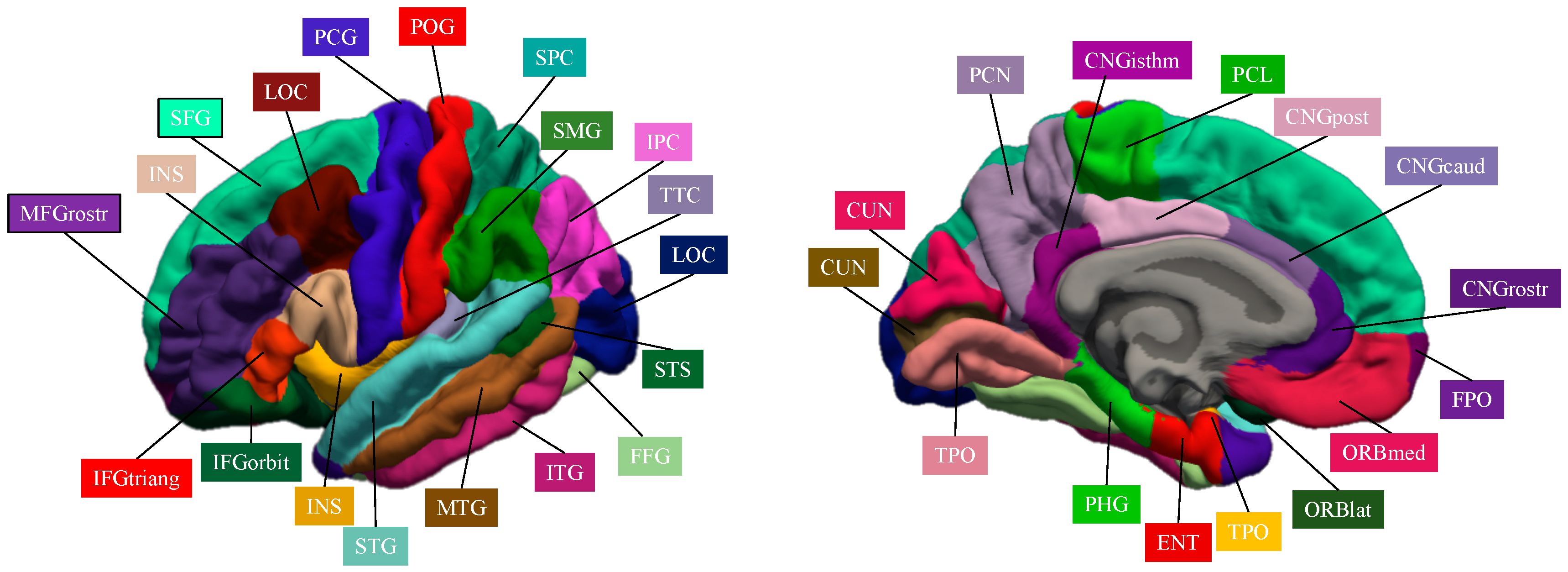
| Localization | Region | Abbreviation | Localization | Region | Abbreviation |
|---|---|---|---|---|---|
| Frontal lobe | Superior frontal gyrus | SFG | Transverse temporal cortex | TTC | |
| Middle frontal gyrus, rostral division | MFGrostr | Entorhinal cortex | ENC | ||
| Middle frontal gyrus, caudal division | MFGcaud | Temporal pole | TPO | ||
| Inferior frontal gyrus, pars opercularis | IFGoperc | Parahippocampal gyrus | PHG | ||
| Inferior frontal gyrus, pars triangularis | IFGtriang | Occipital lobe | Lateral occipital cortex | LOC | |
| Inferior frontal gyrus, pars orbitalis | IFGorbit | Lingual gyrus | LING | ||
| Orbitofrontal cortex, lateral division | ORBlat | Cuneus cortex | CUN | ||
| Orbitofrontal cortex, medial division | ORBmed | Pericalcarine cortex | CAL | ||
| Precentral gyrus | PCG | Cingulate Lobe | Cingulate cortex, rostral anterior division | CNGrostr | |
| Paracentral lobule | PCL | Cingulate cortex, caudal anterior division | CNGcaud | ||
| Frontal pole | FPO | Cingulate cortex, posterior division | CNGpost | ||
| Parietal lobe | Superior parietal cortex | SPC | Cingulate cortex, isthmus division | CNGisthm | |
| Inferior parietal cortex | IPC | Insula | Insula | INS | |
| Supramarginal gyrus | SMG | Subcortical | Thalamus | THA | |
| Postcentral gyrus | POG | Caudate nucleus | CAU | ||
| Precuneus cortex | PCN | Lenticular nucleus, putamen | PUT | ||
| Temporal lobe | Superior temporal gyrus | STG | Lenticular nucleus, pallidum | PAL | |
| Middle temporal gyrus | MTG | Hippocampus | HIP | ||
| Inferior temporal gyrus | ITG | Amygdala | AMY | ||
| Banks of the superior temporal sulcus | STS | Nucleus accumbens | ACC | ||
| Fusiform gyrus | FFG | Cerebellar cortex | CRB |
| Measure | Description |
|---|---|
| Global measures | |
| Global efficiency (EG) | Average of the inverse shortest path lengths, typically considered to be a measure of the network’s overall ability for information transfer and integrated processing. |
| Transitivity (T) | Ratio of triangles to triplets in the network. It is more robust compared to the average clustering coefficient. |
| Characteristic path length (CPL) | Average of the shortest path lengths between each pair of nodes in the network, where shortest path length is the minimum number of links that are passed through to get from one node to another node. |
| Small world propensity (SWP) | A measure of small-worldness for weighted graphs. Small-worldness is defined as a type of network that exhibits groups of highly clustered vertices (high clustering coefficient), with a limited number of edges connecting the vertex assemblies (low path length) [83]. |
| Nodal Measures | |
| Local efficiency (EL) | Efficiency of a subgraph made up by the neighborhood of the node. |
| Clustering coefficient (CC) | Weighted sum of the number of links between the nearest neighbors of the node divided by the maximum possible amount of links between the nearest neighbors. It is a measure of the percentage of the node’s neighbors that are also connected to each other. |
| Betweenness centrality (BC) | Quantifies how many shortest paths between any two nodes pass through a given node. It is a measure for how important a given node is for the efficiency of the network. |
| Children with ASD | Fathers of Children with ASD | ||
|---|---|---|---|
| Local Efficiency (LE) | |||
| Brain region | Significant interactions | Brain region | Significant interactions |
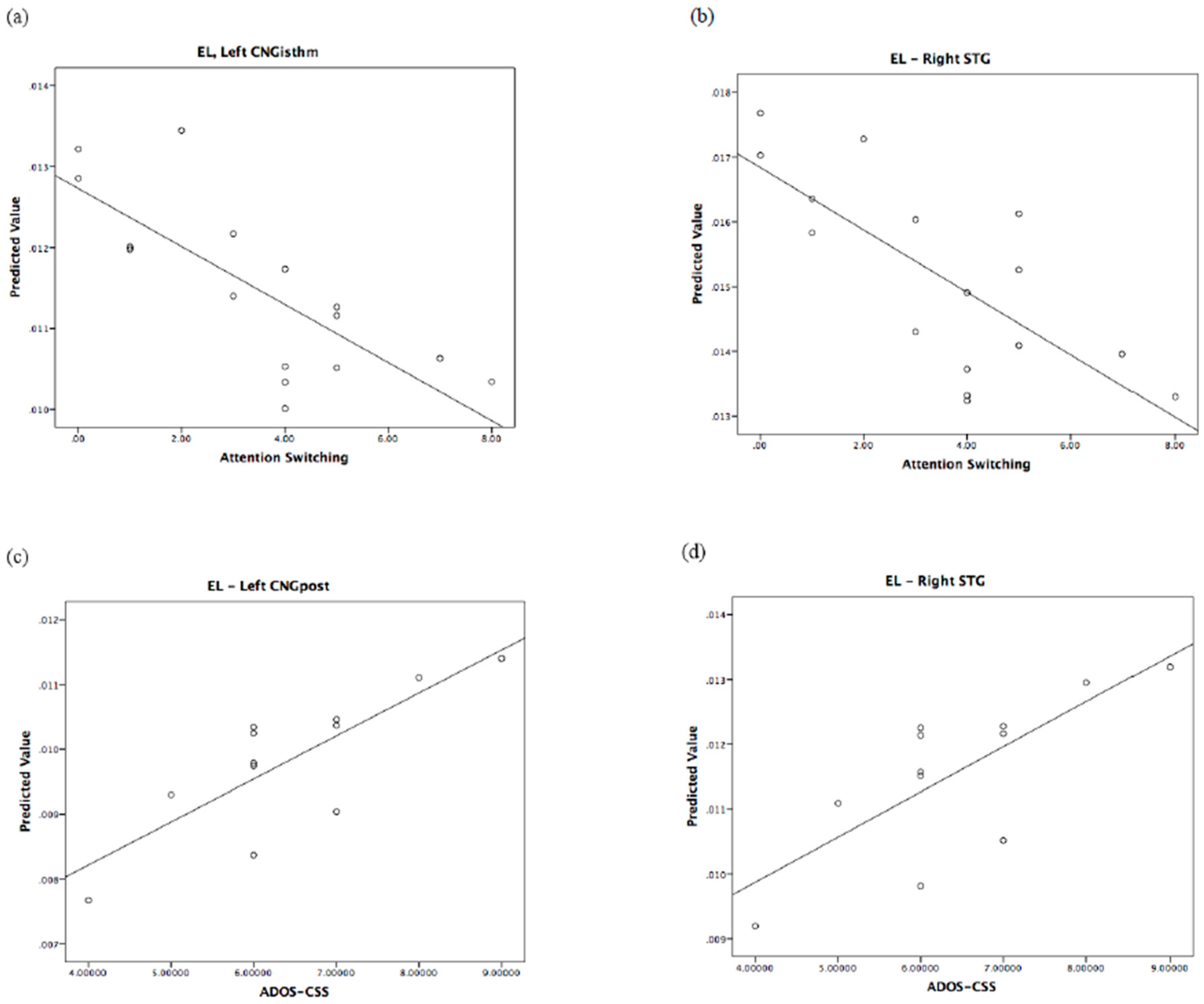
| Right PCL | ADOS-CSS: B = 0.78; F = 12.75, p = 0.006, η2 = 0.586 * | Left CNGisthm | Att. swi.: B = −1.09; F = 10.11, p = 0.01, η2 = 0.503 |
| Left CNGpost | ADOS-CSS: B = 0.66; F = 8.05, p = 0.02, η2 = 0.472 | Right STG | Att. swi.: B = −0.91; F = 5.32, p = 0.044, η2 = 0.384 |
| Right STG | ADOS-CSS: B = 0.70; F = 11.75, p = 0.008, η2 = 0.566 * Age: B = −0.54; F = 6.79, p = 0.028, η2 = 0.430 | ||
| Left CAU | SA: B = 1.10; F = 19.66, p = 0.004, η2 = 0.766 * | ||
| Cluster coefficient (CC) | |||
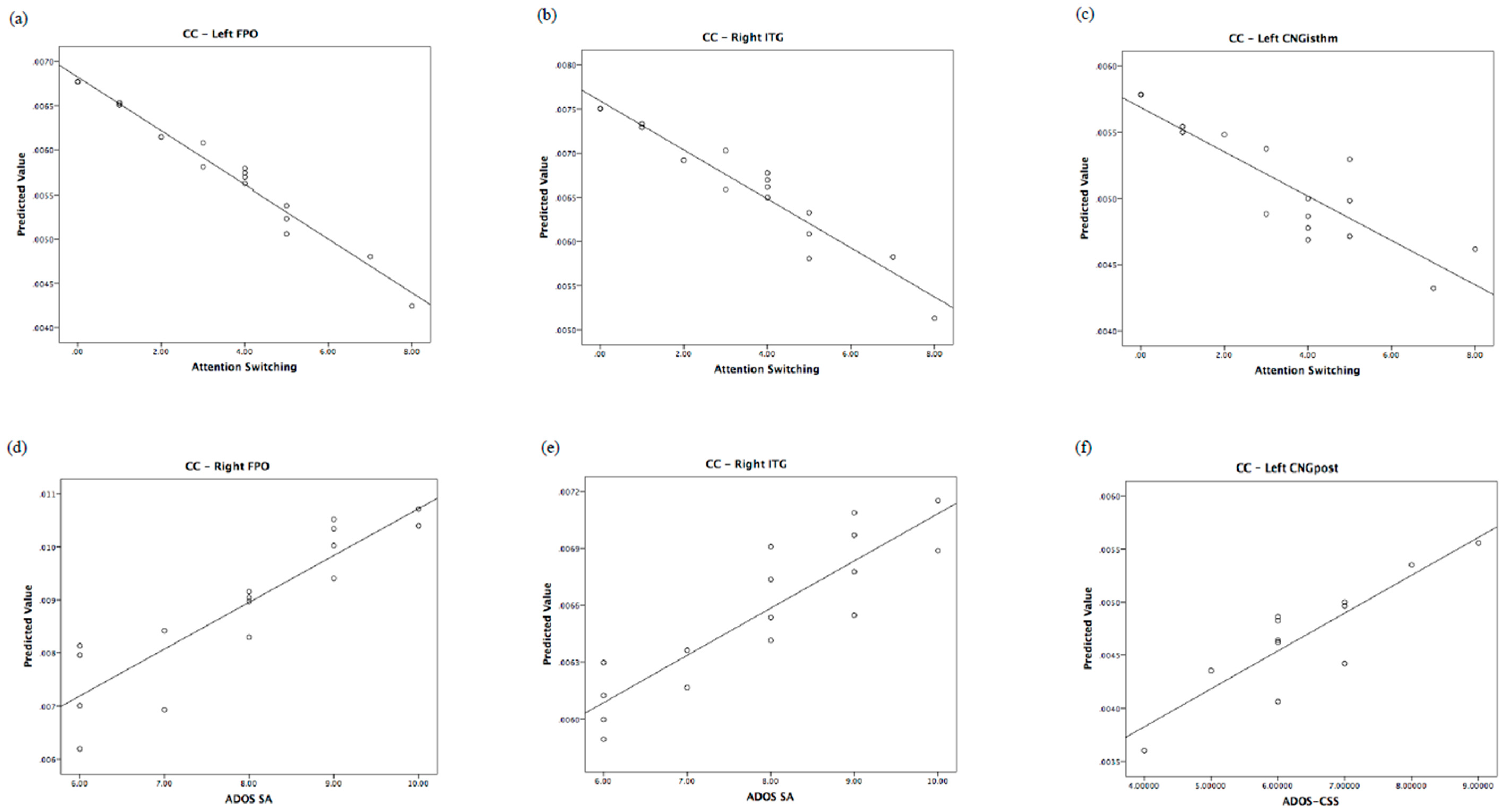
| Left CNGpost | ADOS-CSS: B = 0.76; F = 8.38, p = 0.018, η2 = 0.482 | Right SFG | Att. swi.: B = −1.10; F = 12.61, p = 0.005, η2 = 0.558 * |
| Right FPO | SA: B = 0.92; F = 10.69, p = 0.017, η2 = 0.641 | Right MFGrostr | Att. swi.: B = −1.03; F = 13.47, p = 0.004, η2 = 0.574 * |
| Right ITG | SA: B = 0.95; F = 14.21, p = 0.009, η2 = 0.703 GMDS: B = 0.59; F = 6.11, p = 0.048, η2 = 0.504 | Left FPO | Att. swi.: B = −0.82; F = 5.67, p = 0.04, η2 = 0.362 |
| Left CAU | SA: B = 1.11; F = 18.86, p = 0.005, η2 = 0.554 * | Left LOC | Att. swi.: B = −1.02; F = 10.92, p = 0.008, η2 = 0.522 * |
| Left CNGisthm | Att. swi.: B = −1.15; F = 11.89, p = 0.006, η2 = 0.543 * | ||
| Right ITG | Att. swi.: B = −0.71; F = 5.61, p = 0.039, η2 = 0.360 | ||
| Betwenness centrality (BC) | |||
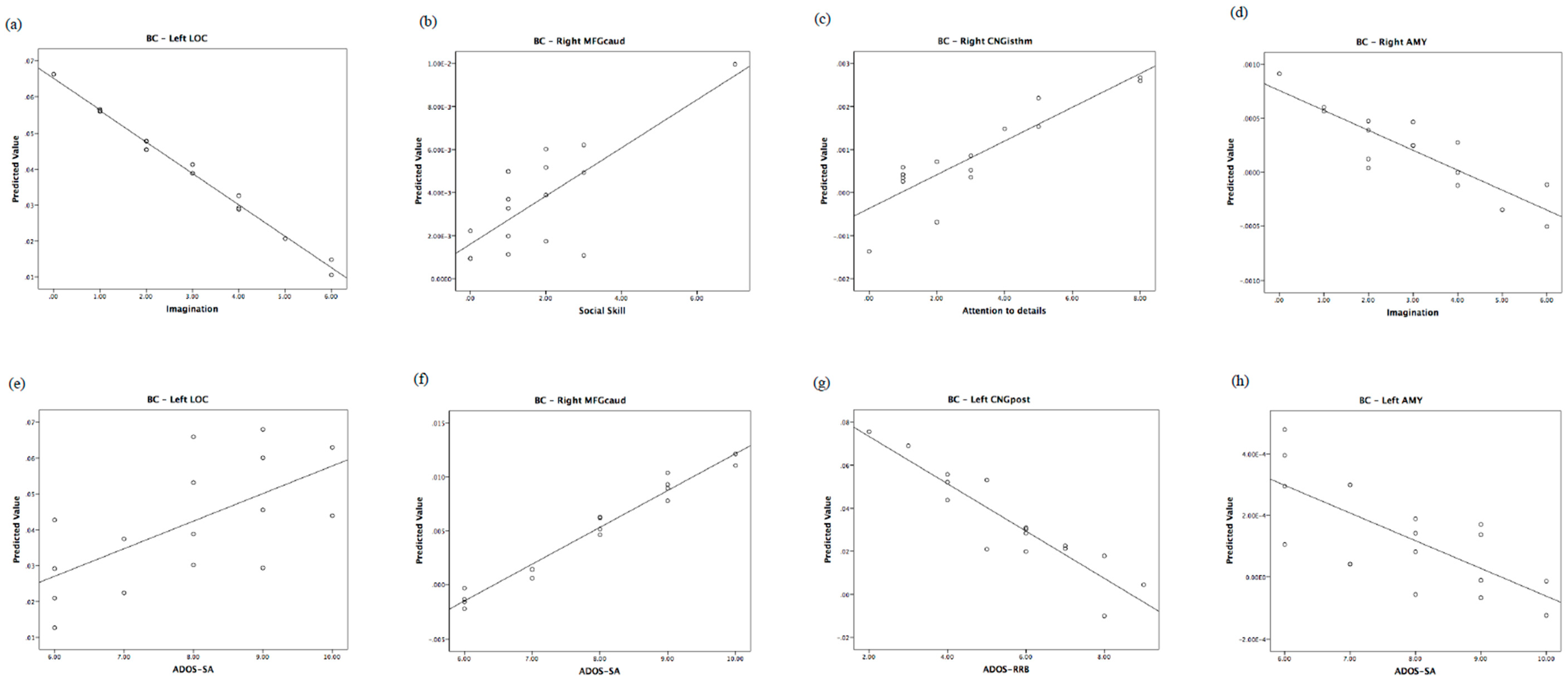
| Right MFGcaud | SA: B = 0.97, F = 6.66; p = 0.04, η2 = 0.526 | Left IFGoperc | Imm.: B = 1.00; F = 16.76, p = 0.002, η2 = 0.626 * |
| Left CNGpost | SA: B = 0.91; F = 15.35, p = 0.008, η2 = 0.719 * RRB: B = −0.87; F = 15.06, p = 0.008, η2 = 0.683 * | Left POG | Age: B = 1.14; F = 14.46, p = 0.003, η2 = 0.591 * |
| Left LOC | Imm.: B = −0.80; F = 6.41, p = 0.030, η2 = 0.391 | ||
| Right PCG | ADOS-CSS: B = 0.91; F = 14.96, p = 0.008, η2 = 0.714 * | Right MFGcaud | Soc. skills: B = 0.89; F = 6.87, p = 0.025, η2 = 0.408 |
| Left AMY | SA: B = −0.89; F = 10.41, p = 0.018, η2 = 0.662 Age: B = −0.75, p = 0.020, η2 = 0.609 | Right MTG | Soc. skills: B = 1.00; F = 15.94, p = 0.003, η2 = 0.615 * |
| Right MTG | Soc. skills: B = 1.00; F = 15.94, p = 0.003, η2 = 0.615 * | ||
| Left CNGisthm | Imm.: B = −0.70; F = 5.41, p = 0.040, η2 = 0.351 | ||
| Right THA | Soc. skills: B = 0.98; F = 16.86, p = 0.002, η2 = 0.628 * | ||
| Right AMY | Imm.: B = −0.76; F = 6.31, p = 0.030, η2 = 0.387 | ||
| Measure | Shared Brain Areas |
|---|---|
| Local efficiency (EL) | CNG, Right STG |
| Clustering coefficient (CC) | CNG, FPO, ITG |
| Betweenness centrality (BC) | CNG, LOC, MFG, AMY |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Billeci, L.; Calderoni, S.; Conti, E.; Lagomarsini, A.; Narzisi, A.; Gesi, C.; Carmassi, C.; Dell’Osso, L.; Cioni, G.; Muratori, F.; et al. Brain Network Organization Correlates with Autistic Features in Preschoolers with Autism Spectrum Disorders and in Their Fathers: Preliminary Data from a DWI Analysis. J. Clin. Med. 2019, 8, 487. https://doi.org/10.3390/jcm8040487
Billeci L, Calderoni S, Conti E, Lagomarsini A, Narzisi A, Gesi C, Carmassi C, Dell’Osso L, Cioni G, Muratori F, et al. Brain Network Organization Correlates with Autistic Features in Preschoolers with Autism Spectrum Disorders and in Their Fathers: Preliminary Data from a DWI Analysis. Journal of Clinical Medicine. 2019; 8(4):487. https://doi.org/10.3390/jcm8040487
Chicago/Turabian StyleBilleci, Lucia, Sara Calderoni, Eugenia Conti, Alessia Lagomarsini, Antonio Narzisi, Camilla Gesi, Claudia Carmassi, Liliana Dell’Osso, Giovanni Cioni, Filippo Muratori, and et al. 2019. "Brain Network Organization Correlates with Autistic Features in Preschoolers with Autism Spectrum Disorders and in Their Fathers: Preliminary Data from a DWI Analysis" Journal of Clinical Medicine 8, no. 4: 487. https://doi.org/10.3390/jcm8040487
APA StyleBilleci, L., Calderoni, S., Conti, E., Lagomarsini, A., Narzisi, A., Gesi, C., Carmassi, C., Dell’Osso, L., Cioni, G., Muratori, F., & Guzzetta, A. (2019). Brain Network Organization Correlates with Autistic Features in Preschoolers with Autism Spectrum Disorders and in Their Fathers: Preliminary Data from a DWI Analysis. Journal of Clinical Medicine, 8(4), 487. https://doi.org/10.3390/jcm8040487









