Serotonergic, Dopaminergic, and Noradrenergic Modulation of Erotic Stimulus Processing in the Male Human Brain
Abstract
1. Introduction
2. Conceptualizing Sexual Behavior and Neural Responses
3. Neuromodulation of Sexual Responses
4. Challenges
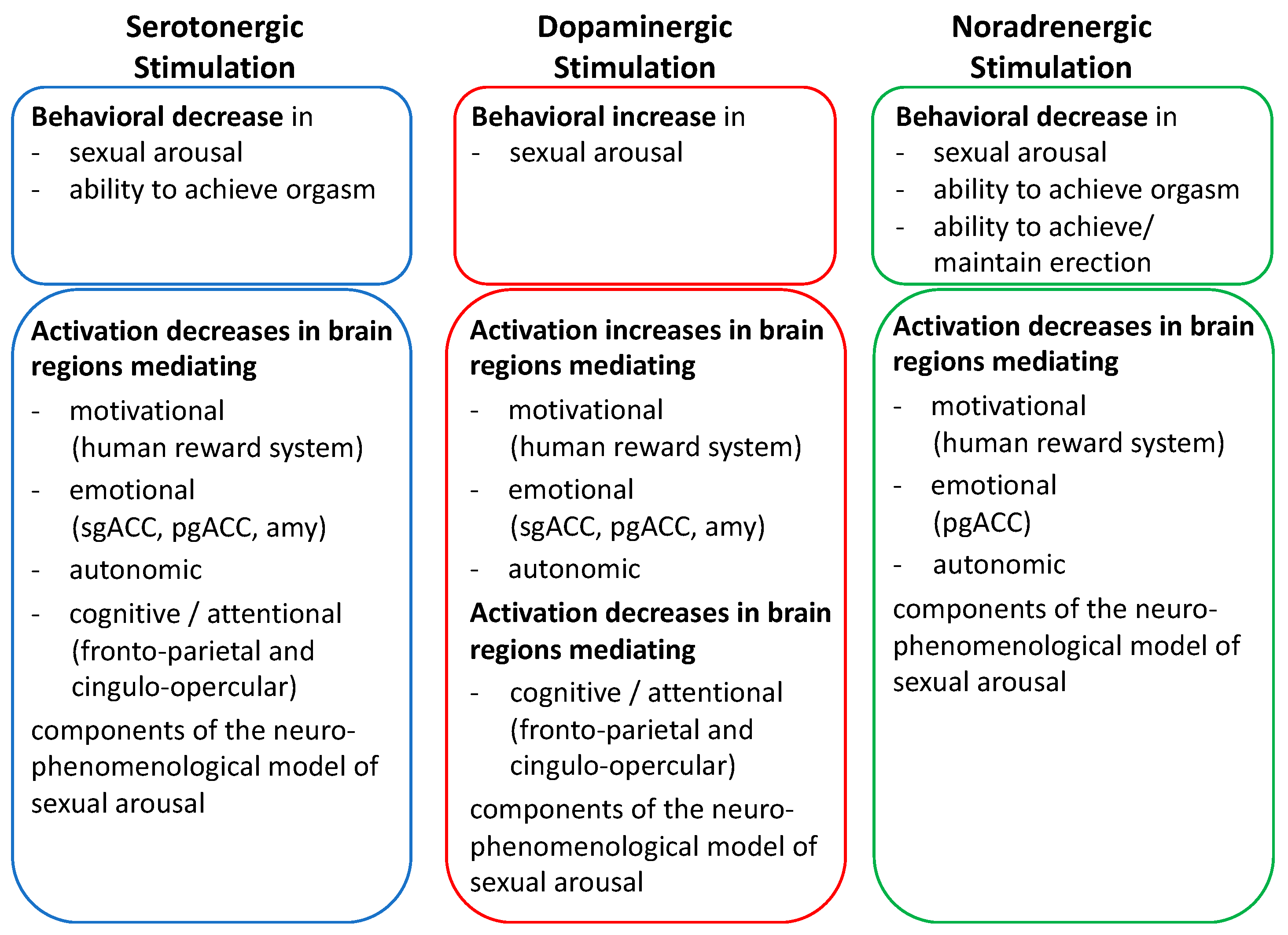
5. Serotonergic, Dopaminergic, and Noradrenergic Neuromodulation of Sexual Responses
6. Perspectives
7. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- McKenna, K. The brain is the master organ in sexual function: Central nervous system control of male and female sexual function. Int. J. Impot. Res. 1999, 1, 48–55. [Google Scholar] [CrossRef]
- Stoléru, S.; Fonteille, V.; Cornélis, C.; Joyal, C.; Moulier, V. Functional neuroimaging studies of sexual arousal and orgasm in healthy men and women: A review and meta-analysis. Neurosci. Biobehav. Rev. 2012, 36, 1481–1509. [Google Scholar] [CrossRef]
- Redouté, J.; Stoléru, S.; Grégoire, M.C.; Costes, N.; Cinotti, L.; Lavenne, F.; Le Bars, D.; Forest, M.G.; Pujol, J.F. Brain processing of visual sexual stimuli in human males. Hum. Brain Mapp. 2000, 11, 162–177. [Google Scholar] [CrossRef]
- Georgiadis, J.R.; Kringelbach, M.L.; Pfaus, J.G. Sex for fun: A synthesis of human and animal neurobiology. Nat. Rev. Urol. 2012, 9, 486–498. [Google Scholar] [CrossRef] [PubMed]
- Bossini, L.; Fortini, V.; Casolaro, I.; Caterini, C.; Koukouna, D.; Cecchini, F.; Benbow, J.; Fagiolini, A. Sexual dysfunctions, psychiatric diseases and quality of life: A review. Psychiatr. Pol. 2014, 48, 715–726. [Google Scholar] [PubMed]
- Waldinger, M.D. Psychiatric disorders and sexual dysfunction. Handb. Clin. Neurol. 2015, 130, 469–489. [Google Scholar] [PubMed]
- Montejo, A.L.; Llorca, G.; Izquierdo, J.A.; Rico-Villademoros, F. Incidence of sexual dysfunction associated with antidepressant agents: A prospective multicenter study of 1022 outpatients. Spanish Working Group for the Study of Psychotropic-Related Sexual Dysfunction. J. Clin. Psychiatry 2001, 62, 10–21. [Google Scholar] [PubMed]
- Masters, W.J.; Johnson, V.E. Human Sexual Response, 1st ed.; Brown, Little: Boston, MA, USA, 1966; ISBN 9781483198064. [Google Scholar]
- Kaplan, H.S. Disorders of Sexual Desire and Other New Concepts and Techniques in sex Therapy/Helen Singer Kaplan; Kaplan, H.S., Ed.; New Sex Therapy; Brunner/Mazel: New York, NY, USA, 1979; ISBN 0876302126. [Google Scholar]
- Georgiadis, J.R.; Kringelbach, M.L. The human sexual response cycle: Brain imaging evidence linking sex to other pleasures. Prog. Neurobiol. 2012, 98, 49–81. [Google Scholar] [CrossRef] [PubMed]
- Stoléru, S.; Gregoire, M.C.; Gerard, D.; Decety, J.; Lafarge, E.; Cinotti, L.; Lavenne, F.; Le Bars, D.; Vernet-Maury, E.; Rada, H.; et al. Neuroanatomical correlates of visually evoked sexual arousal in human males. Arch. Sex. Behav. 1999, 28, 1–21. [Google Scholar] [CrossRef]
- Poeppl, T.B.; Langguth, B.; Laird, A.R.; Eickhoff, S.B. The functional neuroanatomy of male psychosexual and physiosexual arousal: A quantitative meta-analysis. Hum. Brain Mapp. 2014, 35, 1404–1421. [Google Scholar] [CrossRef]
- Bühler, M.; Vollstädt-Klein, S.; Klemen, J.; Smolka, M.N. Does erotic stimulus presentation design affect brain activation patterns? Event-related vs. blocked fMRI designs. Behav. Brain Funct. 2008, 4, 30. [Google Scholar] [CrossRef]
- Ferretti, A.; Caulo, M.; Del Gratta, C.; Di Matteo, R.; Merla, A.; Montorsi, F.; Pizzella, V.; Pompa, P.; Rigatti, P.; Rossini, P.M.; et al. Dynamics of male sexual arousal: Distinct components of brain activation revealed by fMRI. Neuroimage 2005, 26, 1086–1096. [Google Scholar] [CrossRef]
- Cera, N.; di Pierro, E.D.; Sepede, G.; Gambi, F.; Perrucci, M.G.; Merla, A.; Tartaro, A.; del Gratta, C.; Galatioto Paradiso, G.; Vicentini, C.; et al. The Role of Left Superior Parietal Lobe in Male Sexual Behavior: Dynamics of Distinct Components Revealed by fMRI. J. Sex. Med. 2012, 9, 1602–1612. [Google Scholar] [CrossRef]
- Georgiadis, J.R.; Kortekaas, R.; Kuipers, R.; Nieuwenburg, A.; Pruim, J.; Reinders, A.A.T.S.; Holstege, G. Regional cerebral blood flow changes associated with clitorally induced orgasm in healthy women. Eur. J. Neurosci. 2006, 24, 3305–3316. [Google Scholar] [CrossRef]
- Gizewski, E.R.; Krause, E.; Karama, S.; Baars, A.; Senf, W.; Forsting, M. There are differences in cerebral activation between females in distinct menstrual phases during viewing of erotic stimuli: A fMRI study. Exp. Brain Res. 2006, 174, 101–108. [Google Scholar] [CrossRef]
- Arnow, B.A.; Millheiser, L.; Garrett, A.; Lake Polan, M.; Glover, G.H.; Hill, K.R.; Lightbody, A.; Watson, C.; Banner, L.; Smart, T.; et al. Women with hypoactive sexual desire disorder compared to normal females: A functional magnetic resonance imaging study. Neuroscience 2009, 158, 484–502. [Google Scholar] [CrossRef]
- Beauregard, M.; Lévesque, J.; Bourgouin, P. Neural Correlates of Conscious Self-Regulation of Emotion. J. Neurosci. 2001, 21, RC165. [Google Scholar] [CrossRef]
- Rodriguez, G.; Sack, A.T.; Dewitte, M.; Schuhmann, T. Inhibit My Disinhibition: The Role of the Inferior Frontal Cortex in Sexual Inhibition and the Modulatory Influence of Sexual Excitation Proneness. Front. Hum. Neurosci. 2018, 12, 300. [Google Scholar] [CrossRef]
- Mallick, H.N.; Tandon, S.; Jagannathan, N.R.; Gulia, K.K.; Kumar, V.M. Brain areas activated after ejaculation in healthy young human subjects. Indian J. Physiol. Pharmacol. 2007, 51, 81–85. [Google Scholar]
- Brackett, N.L.; Iuvone, P.M.; Edwards, D.A. Midbrain lesions, dopamine and male sexual behavior. Behav. Brain Res. 1986, 20, 231–240. [Google Scholar] [CrossRef]
- Zahran, A.R.; Simmerman, N.; Carrier, S.; Vachon, P. Erectile dysfunction occurs following substantia nigra lesions in the rat. Int. J. Impot. Res. 2001, 13, 255–260. [Google Scholar] [CrossRef]
- La Torre, A.; Giupponi, G.; Duffy, D.; Conca, A. Sexual dysfunction related to psychotropic drugs: A critical review—Part I: Antidepressants. Pharmacopsychiatry 2013, 46, 191–199. [Google Scholar] [CrossRef]
- La Torre, A.; Conca, A.; Duffy, D.; Giupponi, G.; Pompili, M.; Grözinger, M. Sexual dysfunction related to psychotropic drugs: A critical review part II: Antipsychotics. Pharmacopsychiatry 2013, 46, 201–208. [Google Scholar] [CrossRef]
- La Torre, A.; Giupponi, G.; Duffy, D.M.; Pompili, M.; Grözinger, M.; Kapfhammer, H.P.; Conca, A. Sexual dysfunction related to psychotropic drugs: A critical reviewpart III: Mood stabilizers and anxiolytic drugs. Pharmacopsychiatry 2014, 47, 1–6. [Google Scholar] [CrossRef]
- Pfaus, J.G. Pathways of sexual desire. J. Sex. Med. 2009, 6, 1506–1533. [Google Scholar] [CrossRef]
- Hull, E.M.; Muschamp, J.W.; Sato, S. Dopamine and serotonin: Influences on male sexual behavior. Physiol. Behav. 2004, 83, 291–307. [Google Scholar] [CrossRef]
- Clayton, A.H. The pathophysiology of hypoactive sexual desire disorder in women. Int. J. Gynecol. Obstet. 2010, 110, 7–11. [Google Scholar] [CrossRef]
- Modell, J.G.; May, R.S.; Katholi, C.R. Effect of bupropion-sr on orgasmic dysfunction in nondepressed subjects: A pilot study. J. Sex Marital Ther. 2000, 26, 231–240. [Google Scholar]
- Voon, V.; Fernagut, P.O.; Wickens, J.; Baunez, C.; Rodriguez, M.; Pavon, N.; Juncos, J.L.; Obeso, J.A.; Bezard, E. Chronic dopaminergic stimulation in Parkinson’s disease: From dyskinesias to impulse control disorders. Lancet Neurol. 2009, 8, 1140–1149. [Google Scholar] [CrossRef]
- Dossenbach, M.; Dyachkova, Y.; Pirildar, S.; Anders, M.; Khalil, A.; Araszkiewicz, A.; Shakhnovich, T.; Akram, A.; Pecenak, J.; McBride, M.; et al. Effects of atypical and typical antipsychotic treatments on sexual function in patients with schizophrenia: 12-month results from the Intercontinental Schizophrenia Outpatient Health Outcomes (IC-SOHO) study. Eur. Psychiatry 2006, 21, 251–258. [Google Scholar] [CrossRef]
- Haddad, P.M.; Sharma, S.G. Adverse effects of atypical antipsychotics: Differential risk and clinical implications. CNS Drugs 2007, 21, 911–936. [Google Scholar] [CrossRef]
- Park, Y.W.; Kim, Y.; Lee, J.H. Antipsychotic-induced sexual dysfunction and its management. World J. Mens. Health 2012, 30, 153–159. [Google Scholar] [CrossRef]
- Montejo, A.L.; Calama, J.; Rico-Villademoros, F.; Montejo, L.; Gonzalez-Garcia, N.; Perez, J. A Real-World Study on Antidepressant-Associated Sexual Dysfunction in 2144 Outpatients: The SALSEX I Study. Arch. Sex. Behav. 2019, 1–11. [Google Scholar] [CrossRef]
- Santana, Y.; Montejo, A.L.; Martin, J.; LLorca, G.; Bueno, G.; Blazquez, J.L. Understanding the Mechanism of Antidepressant-Related Sexual Dysfunction: Inhibition of Tyrosine Hydroxylase in Dopaminergic Neurons after Treatment with Paroxetine but Not with Agomelatine in Male Rats. J. Clin. Med. 2019, 8, 133. [Google Scholar] [CrossRef]
- Allard, J.; Truitt, W.A.; McKenna, K.E.; Coolen, L.M. Spinal cord control of ejaculation. World J. Urol. 2005, 23, 119–126. [Google Scholar] [CrossRef]
- Serretti, A.; Chiesa, A. Treatment-emergent sexual dysfunction related to antidepressants: A meta-analysis. J. Clin. Psychopharmacol. 2009, 29, 259–266. [Google Scholar] [CrossRef]
- Williams, V.S.L.; Baldwin, D.S.; Hogue, S.L.; Fehnel, S.E.; Hollis, K.A.; Edin, H.M. Estimating the Prevalence and Impact of Antidepressant-Induced Sexual Dysfunction in 2 European Countries A Cross-Sectional Patient Survey. J. Clin. Psychiatry 2006, 67, 204–210. [Google Scholar] [CrossRef]
- Williams, V.S.L.; Edin, H.M.; Hogue, S.L.; Fehnel, S.E.; Baldwin, D.S. Prevalence and impact of antidepressant-associated sexual dysfunction in three European countries: Replication in a cross-sectional patient survey. J. Psychopharmacol. 2010, 24, 489–496. [Google Scholar] [CrossRef]
- Clayton, A.; Kornstein, S.; Prakash, A.; Mallinckrodt, C.; Wohlreich, M. Changes in sexual functioning associated with duloxetine, escitalopram, and placebo in the treatment of patients with major depressive disorder. J. Sex. Med. 2007, 4, 917–929. [Google Scholar] [CrossRef]
- Berwian, I.M.; Walter, H.; Seifritz, E.; Huys, Q.J.M. Predicting relapse after antidepressant withdrawal—A systematic review. Psychol. Med. 2016, 47, 426–437. [Google Scholar] [CrossRef]
- Langworth, S.; Bodlund, O.; Agren, H. Efficacy and tolerability of reboxetine compared with citalopram: A double-blind study in patients with major depressive disorder. J. Clin. Psychopharmacol. 2006, 26, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Clayton, A.H.; Zajecka, J.; Ferguson, J.M.; Filipiak-Reisner, J.K.; Brown, M.T.; Schwartz, G.E. Lack of sexual dysfunction with the selective noradrenaline reuptake inhibitor reboxetine during treatment for major depressive disorder. Int. Clin. Psychopharmacol. 2003, 18, 151–156. [Google Scholar]
- Krüger, T.H.C.; Haake, P.; Chereath, D.; Knapp, W.; Janssen, O.E.; Exton, M.S.; Schedlowski, M.; Hartmann, U. Specificity of the neuroendocrine response to orgasm during sexual arousal in men. J. Endocrinol. 2003, 177, 57–64. [Google Scholar] [CrossRef]
- Mihailescu, C.; Mihailesku, A. Sexual dysfunction in a group of depressed female patients. Conference abstract, 21st ECNP Congress, Barcelona, Spain. Eur. Neuropsychopharmacol. 2008, 18, 341–342. [Google Scholar] [CrossRef]
- Thakurta, R.; Singh, O.; Bhattacharya, A.; Mallick, A.; Ray, P.; Sen, S.; Das, R. Nature of Sexual Dysfunctions in Major Depressive Disorder and its Impact on Quality of Life. Indian J. Psychol. Med. 2012, 34, 365–370. [Google Scholar]
- Kim, W.; Jin, B.R.; Yang, W.S.; Lee, K.U.; Juh, R.H.; Ahn, K.J.; Chung, Y.A.; Chae, J.H. Treatment with selective serotonin reuptake inhibitors and mirtapazine results in differential brain activation by visual erotic stimuli in patients with major depressive disorder. Psychiatry Investig. 2009, 6, 85–95. [Google Scholar] [CrossRef]
- Abler, B.; Seeringer, A.; Hartmann, A.; Grön, G.; Metzger, C.; Walter, M.; Stingl, J. Neural Correlates of Antidepressant-Related Sexual Dysfunction: A Placebo-Controlled fMRI Study on Healthy Males Under Subchronic Paroxetine and Bupropion. Neuropsychopharmacology 2011, 36, 1837–1847. [Google Scholar] [CrossRef]
- Labbate, L.A.; Lare, S.B. Sexual dysfunction in male psychiatric outpatients: Validity of the Massachusetts General Hospital sexual functioning questionnaire. Psychother. Psychosom. 2001, 70, 221–225. [Google Scholar] [CrossRef]
- Simmons, A.N.; Arce, E.; Lovero, K.L.; Stein, M.B.; Paulus, M.P. Subchronic SSRI administration reduces insula response during affective anticipation in healthy volunteers. Int. J. Neuropsychopharmacol. 2009, 12, 1009–1020. [Google Scholar] [CrossRef]
- Walter, M.; Bermpohl, F.; Mouras, H.; Schiltz, K.; Tempelmann, C.; Rotte, M.; Heinze, H.J.; Bogerts, B.; Northoff, G. Distinguishing specific sexual and general emotional effects in fMRI-Subcortical and cortical arousal during erotic picture viewing. Neuroimage 2008, 40, 1482–1492. [Google Scholar] [CrossRef]
- Hayes, D.J.; Greenshaw, A.J. 5-HT receptors and reward-related behaviour: A review. Neurosci. Biobehav. Rev. 2011, 35, 1419–1449. [Google Scholar] [CrossRef] [PubMed]
- Kranz, G.S.; Kasper, S.; Lanzenberger, R. Reward and the serotonergic system. Neuroscience 2010, 166, 1023–1035. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.; Patrick, C.J.; Kennealy, P.J. Role of serotonin and dopamine system interactions in the neurobiology of impulsive aggression and its comorbidity with other clinical disorders. Aggress. Violent Behav. 2008, 13, 383–395. [Google Scholar] [CrossRef] [PubMed]
- Boureau, Y.L.; Dayan, P. Opponency revisited: Competition and cooperation between dopamine and serotonin. Neuropsychopharmacology 2011, 36, 74–97. [Google Scholar] [CrossRef] [PubMed]
- Diekhof, E.K.; Gruber, O. When Desire Collides with Reason: Functional Interactions between Anteroventral Prefrontal Cortex and Nucleus Accumbens Underlie the Human Ability to Resist Impulsive Desires. J. Neurosci. 2010, 30, 1488–1493. [Google Scholar] [CrossRef]
- Abler, B.; Gron, G.; Hartmann, A.; Metzger, C.; Walter, M. Modulation of Frontostriatal Interaction Aligns with Reduced Primary Reward Processing under Serotonergic Drugs. J. Neurosci. 2012, 32, 1329–1335. [Google Scholar] [CrossRef]
- Coleman, C.C.; King, B.R.; Bolden-Watson, C.; Book, M.J.; Taylor Segraves, R.; Richard, N.; Ascher, J.; Batey, S.; Jamerson, B.; Metz, A. A placebo-controlled comparison of the effects on sexual functioning of bupropion sustained release and fluoxetine. Clin. Ther. 2001, 23, 1040–1058. [Google Scholar] [CrossRef]
- Coleman, C.C.; Cunningham, L.A.; Foster, V.J.; Batey, S.R.; Donahue, R.M.J.; Houser, T.L.; Ascher, J.A. Sexual dysfunction associated with the treatment of depression: A placebo-controlled comparison of bupropion sustained release and sertraline treatment. Ann. Clin. Psychiatry 1999, 11, 205–215. [Google Scholar] [CrossRef]
- Croft, H.; Settle, E.; Houser, T.; Batey, S.R.; Donahue, R.M.J.; Ascher, J.A. A placebo-controlled comparison of the antidepressant efficacy and effects on sexual functioning of sustained-release bupropion and sertraline. Clin. Ther. 1999, 21, 643–658. [Google Scholar] [CrossRef]
- Segraves, R.T.; Kavoussi, R.; Hughes, A.R.; Batey, S.R.; Johnston, J.A.; Donahue, R.; Ascher, J.A. Evaluation of sexual functioning depressed outpatients: A double-blind comparison of sustained-release bupropion and sertraline treatment. J. Clin. Psychopharmacol. 2000, 20, 122–128. [Google Scholar] [CrossRef]
- Georgiadis, J.R.; Farrell, M.J.; Boessen, R.; Denton, D.A.; Gavrilescu, M.; Kortekaas, R.; Renken, R.J.; Hoogduin, J.M.; Egan, G.F. Dynamic subcortical blood flow during male sexual activity with ecological validity: A perfusion fMRI study. Neuroimage 2010, 50, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Seeley, W.W.; Menon, V.; Schatzberg, A.F.; Keller, J.; Glover, G.H.; Kenna, H.; Reiss, A.L.; Greicius, M.D. Dissociable Intrinsic Connectivity Networks for Salience Processing and Executive Control. J. Neurosci. 2007, 27, 2349–2356. [Google Scholar] [CrossRef]
- Dosenbach, N.U.F.; Fair, D.A.; Cohen, A.L.; Schlaggar, B.L.; Petersen, S.E. A dual-networks architecture of top-down control. Trends Cogn. Sci. 2008, 12, 99–105. [Google Scholar] [CrossRef]
- Walter, M.; Witzel, J.; Wiebking, C.; Gubka, U.; Rotte, M.; Schiltz, K.; Bermpohl, F.; Tempelmann, C.; Bogerts, B.; Heinze, H.J.; et al. Pedophilia is Linked to Reduced Activation in Hypothalamus and Lateral Prefrontal Cortex During Visual Erotic Stimulation. Biol. Psychiatry 2007, 62, 698–701. [Google Scholar] [CrossRef]
- Lang, P.; Bradley, M.; Cuthbert, B. International Affective Picture System (IAPS): Digitized Photographs, Instruction Manual and Affective Ratings; Technical Report; University of Florida: Gainesville, FL, USA, 2005; A-6. [Google Scholar]
- Walter, M.; Matthiä, C.; Wiebking, C.; Rotte, M.; Tempelmann, C.; Bogerts, B.; Heinze, H.J.; Northoff, G. Preceding attention and the dorsomedial prefrontal cortex: Process specificity versus domain dependence. Hum. Brain Mapp. 2009, 30, 312–326. [Google Scholar] [CrossRef] [PubMed]
- Bermpohl, F.; Pascual-Leone, A.; Amedi, A.; Merabet, L.B.; Fregni, F.; Gaab, N.; Alsop, D.; Schlaug, G.; Northoff, G. Dissociable networks for the expectancy and perception of emotional stimuli in the human brain. Neuroimage 2006, 30, 588–600. [Google Scholar] [CrossRef] [PubMed]
- Herwig, U.; Abler, B.; Walter, H.; Erk, S. Expecting unpleasant stimuli—An fMRI study. Psychiatry Res. Neuroimaging 2007, 154, 1–12. [Google Scholar] [CrossRef]
- Corbetta, M.; Shulman, G.L. Control of goal-directed and stimulus-driven attention in the brain. Nat. Rev. Neurosci. 2002, 3, 201–215. [Google Scholar] [CrossRef]
- Arnsten, A.F.T. Catecholamine influences on dorsolateral prefrontal cortical networks. Biol. Psychiatry 2011, 69, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Dreher, J.C.; Guigon, E.; Burnod, Y. A model of prefrontal cortex dopaminergic modulation during the delayed alternation task. J. Cogn. Neurosci. 2002, 14, 853–865. [Google Scholar] [CrossRef]
- Graf, H.; Abler, B.; Freudenmann, R.; Beschoner, P.; Schaeffeler, E.; Spitzer, M.; Schwab, M.; Grn, G. Neural correlates of error monitoring modulated by atomoxetine in healthy volunteers. Biol. Psychiatry 2011, 69, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Lavergne, F.; Jay, T.M. A new strategy for antidepressant prescription. Front. Neurosci. 2010, 4, 192. [Google Scholar] [CrossRef] [PubMed]
- Owen, J.C.E.; Whitton, P.S. Effects of amantadine and budipine on antidepressant drug-evoked changes in extracellular dopamine in the frontal cortex of freely moving rats. Brain Res. 2006, 1117, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Riedel, W.J.; Eikmans, K.; Heldens, A.; Schmitt, J.A.J. Specific serotonergic reuptake inhibition impairs vigilance performance acutely and after subchronic treatment. J. Psychopharmacol. 2005, 19, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Wingen, M.; Kuypers, K.P.C.; van de Ven, V.; Formisano, E.; Ramaekers, J.G. Sustained attention and serotonin: A pharmaco-fMRI study. Hum. Psychopharmacol. 2008, 23, 221–230. [Google Scholar] [CrossRef]
- Whiskey, E.; Taylor, D. A review of the adverse effects and safety of noradrenergic antidepressants. J. Psychopharmacol. 2013, 27, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, D.; Bridgman, K.; Buis, C. Resolution of sexual dysfunction during double-blind treatment of major depression with reboxetine or paroxetine. J. Psychopharmacol. 2006, 20, 91–96. [Google Scholar] [CrossRef]
- Graf, H.; Wiegers, M.; Metzger, C.D.; Walter, M.; Grön, G.; Abler, B. Erotic stimulus processing under amisulpride and reboxetine: A placebo-controlled fMRI study in healthy subjects. Int. J. Neuropsychopharmacol. 2015, 18. [Google Scholar] [CrossRef]
- Haberfellner, E.M. Sexual dysfunction caused by reboxetine. Pharmacopsychiatry 2002, 35, 77–78. [Google Scholar] [CrossRef]
- Sivrioglu, E.Y.; Topaloglu, V.C.; Sarandol, A.; Akkaya, C.; Eker, S.S.; Kirli, S. Reboxetine induced erectile dysfunction and spontaneous ejaculation during defecation and micturition. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2007, 31, 548–550. [Google Scholar] [CrossRef]
- Arnow, B.A.; Desmond, J.E.; Banner, L.L.; Glover, G.H.; Solomon, A.; Polan, M.L.; Lue, T.F.; Atlas, S.W. Brain activation and sexual arousal in healthy, heterosexual males. Brain 2002, 125, 1014–1023. [Google Scholar] [CrossRef]
- Miller, E.M.; Shankar, M.U.; Knutson, B.; McClure, S.M. Dissociating motivation from reward in human striatal activity. J. Cogn. Neurosci. 2014, 26, 1075–1084. [Google Scholar] [CrossRef]
- Perrault, G.; Depoortere, R.; Morel, E.; Sanger, D.J.; Scatton, B. Psychopharmacological profile of amisulpride: An antipsychotic drug with presynaptic D2/D3 dopamine receptor antagonist activity and limbic selectivity. J. Pharmacol. Exp. Ther. 1997, 280, 73–782. [Google Scholar]
- Castelli, M.P.; Mocci, I.; Sanna, A.M.; Gessa, G.L.; Pani, L. (-)S amisulpride binds with high affinity to cloned dopamine D3 and D2 receptors. Eur. J. Pharmacol. 2001, 432, 143–147. [Google Scholar] [CrossRef]
- Härtter, S.; Hüwel, S.; Lohmann, T.; Abou El Ela, A.; Langguth, P.; Hiemke, C.; Galla, H.J. How Does the Benzamide Antipsychotic Amisulpride get into the Brain?—An in Vitro Approach Comparing Amisulpride with Clozapine. Neuropsychopharmacology 2003, 28, 1916–1922. [Google Scholar] [CrossRef]
- Smeraldi, E. Amisulpride versus fluoxetine in patients with dysthymia or major depression in partial remission. A double-blind, comparative study. J. Affect. Disord. 1998, 48, 47–56. [Google Scholar] [CrossRef]
- Montgomery, S.A. Dopaminergic deficit and the role of amisulpride in the treatment of mood disorders. Int. Clin. Psychopharmacol. 2002, 17, 9–15. [Google Scholar]
- Graf, H.; Wiegers, M.; Metzger, C.D.; Walter, M.; Grön, G.; Abler, B. Noradrenergic modulation of neural erotic stimulus perception. Eur. Neuropsychopharmacol. 2017, 27, 845–853. [Google Scholar] [CrossRef]
- El Mansari, M.; Guiard, B.P.; Chernoloz, O.; Ghanbari, R.; Katz, N.; Blier, P. Relevance of norepinephrine-dopamine interactions in the treatment of major depressive disorder. CNS Neurosci. Ther. 2010, 16, e1–e17. [Google Scholar] [CrossRef]
- Graf, H.; Metzger, C.D.; Walter, M.; Abler, B. Serotonergic antidepressants decrease hedonic signals but leave learning signals in the nucleus accumbens unaffected. Neuroreport 2016, 27, 18–22. [Google Scholar] [CrossRef]
- Graf, H.; Wiegers, M.; Metzger, C.D.; Walter, M.; Abler, B. Differential Noradrenergic Modulation of Monetary Reward and Visual Erotic Stimulus Processing. Front. Psychiatry 2018, 9, 346. [Google Scholar] [CrossRef]
- Metzger, C.D.; Walter, M.; Graf, H.; Abler, B. SSRI-related modulation of sexual functioning is predicted by pre-treatment resting state functional connectivity in healthy men. Arch. Sex. Behav. 2013, 42, 935–947. [Google Scholar] [CrossRef]
- Rupp, H.A.; Wallen, K. Sex differences in response to visual sexual stimuli: A review. Arch. Sex. Behav. 2008, 37, 206–218. [Google Scholar] [CrossRef]
- Poeppl, T.B.; Langguth, B.; Rupprecht, R.; Safron, A.; Bzdok, D.; Laird, A.R.; Eickhoff, S.B. The neural basis of sex differences in sexual behavior: A quantitative meta-analysis. Front. Neuroendocrinol. 2016, 43, 28–43. [Google Scholar] [CrossRef]
- Abler, B.; Kumpfmüller, D.; Grön, G.; Walter, M.; Stingl, J.; Seeringer, A. Neural Correlates of Erotic Stimulation under Different Levels of Female Sexual Hormones. PLoS ONE 2013, 8, e54447. [Google Scholar] [CrossRef]
- Bonenberger, M.; Groschwitz, R.C.; Kumpfmueller, D.; Groen, G.; Plener, P.L.; Abler, B. It’s all about money: Oral contraception alters neural reward processing. Neuroreport 2013, 24, 951–955. [Google Scholar] [CrossRef]
- Barth, C.; Villringer, A.; Sacher, J. Sex hormones affect neurotransmitters and shape the adult female brain during hormonal transition periods. Front. Neurosci. 2015, 9, 37. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Graf, H.; Malejko, K.; Metzger, C.D.; Walter, M.; Grön, G.; Abler, B. Serotonergic, Dopaminergic, and Noradrenergic Modulation of Erotic Stimulus Processing in the Male Human Brain. J. Clin. Med. 2019, 8, 363. https://doi.org/10.3390/jcm8030363
Graf H, Malejko K, Metzger CD, Walter M, Grön G, Abler B. Serotonergic, Dopaminergic, and Noradrenergic Modulation of Erotic Stimulus Processing in the Male Human Brain. Journal of Clinical Medicine. 2019; 8(3):363. https://doi.org/10.3390/jcm8030363
Chicago/Turabian StyleGraf, Heiko, Kathrin Malejko, Coraline Danielle Metzger, Martin Walter, Georg Grön, and Birgit Abler. 2019. "Serotonergic, Dopaminergic, and Noradrenergic Modulation of Erotic Stimulus Processing in the Male Human Brain" Journal of Clinical Medicine 8, no. 3: 363. https://doi.org/10.3390/jcm8030363
APA StyleGraf, H., Malejko, K., Metzger, C. D., Walter, M., Grön, G., & Abler, B. (2019). Serotonergic, Dopaminergic, and Noradrenergic Modulation of Erotic Stimulus Processing in the Male Human Brain. Journal of Clinical Medicine, 8(3), 363. https://doi.org/10.3390/jcm8030363



