Effect of Sleep Quality on the Prevalence of Sarcopenia in Older Adults: A Systematic Review with Meta-Analysis
Abstract
1. Introduction
2. Experimental Section
2.1. Study Design
2.2. Search and Data Sources
2.3. Data extraction and Inclusion/Exclusion Criteria
2.4. Outcomes
2.5. Assessment of Risk of Bias
2.6. Data Synthesis and Statistical Analysis
3. Results
3.1. General Characteristics of the Studies
3.2. Quality of the Studies
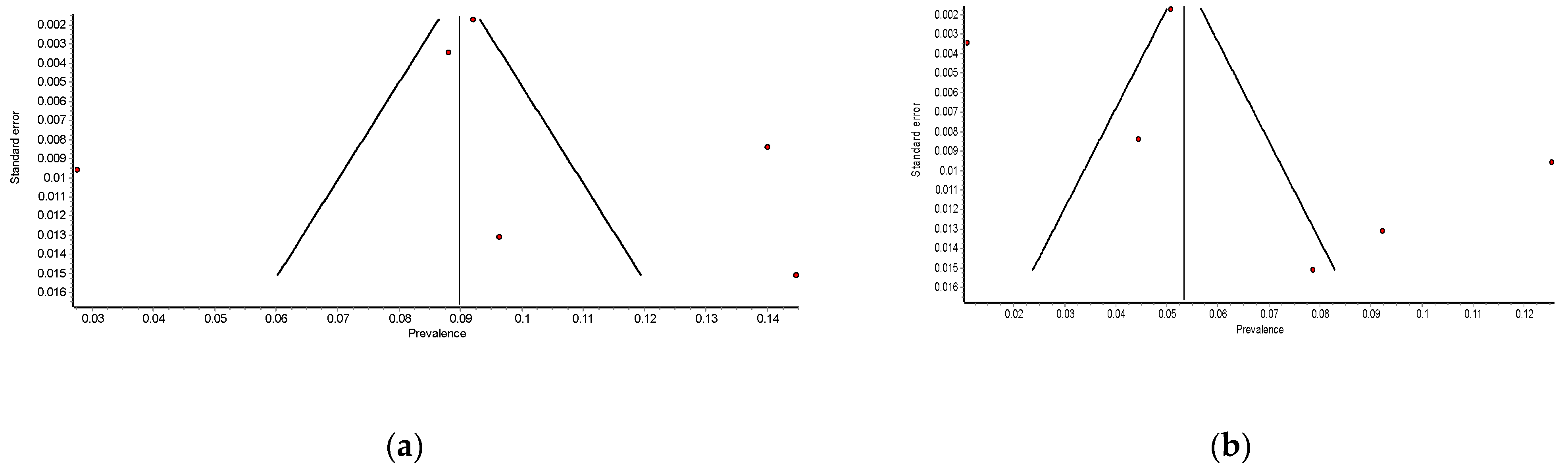
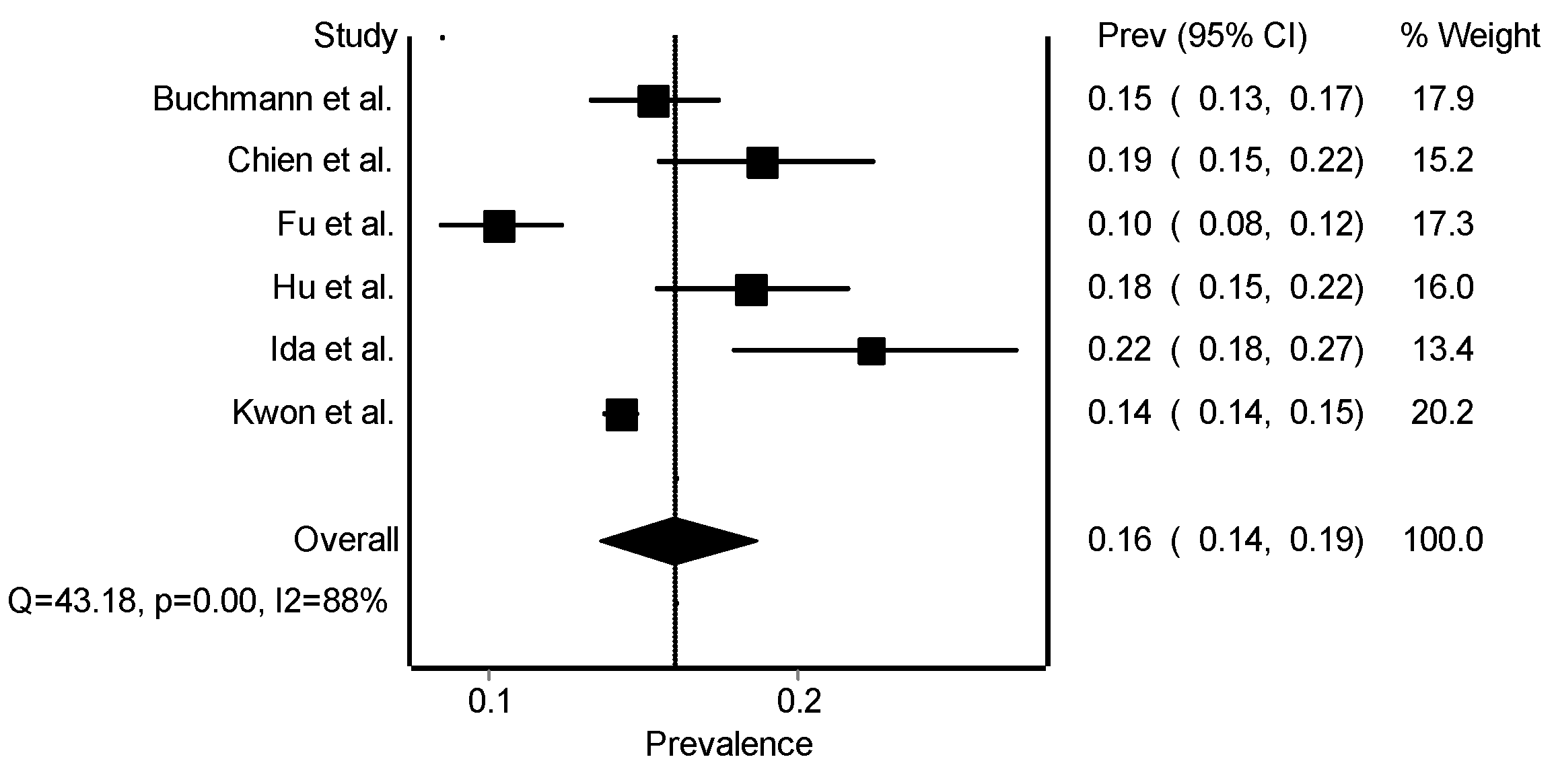
3.3. Meta-Analysis
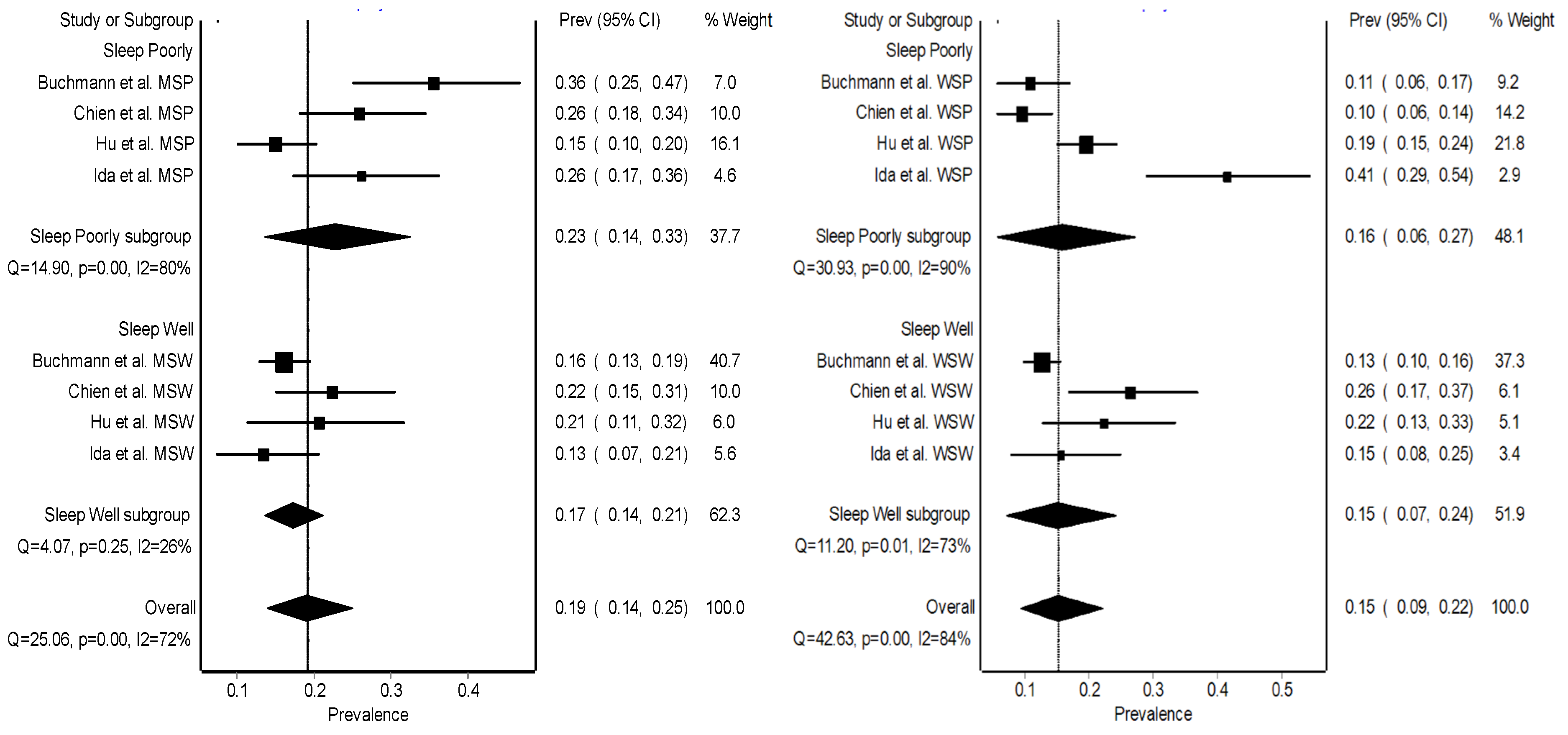
Results by Sleep Categories
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tobergte, D.R.; Curtis, S. Informe Mundial sobre el Envejecimiento y la Salud. OMS 2015. [Google Scholar]
- Shafiee, G.; Keshtkar, A.; Soltani, A.; Ahadi, Z.; Larijani, B.; Heshmat, R. Prevalence of sarcopenia in the world: A systematic review and meta-Analysis of general population studies. J. Diabetes Metab. Disord. 2017, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Antunes, A.C.; Araujo, D.A.; Verissimo, M.T.; Amaral, T.F. Sarcopenia and hospitalisation costs in older adults: A cross-Sectional study. Nutr. Diet. 2017, 74, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Beaudart, C.; Zaaria, M.; Pasleau, F.; Reginster, J.Y.; Bruyere, O. Health outcomes of sarcopenia: A systematic review and meta-Analysis. PLoS ONE 2017, 12, e0169548. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Anker, S.D.; Von Haehling, S. Prevalence, incidence, and clinical impact of sarcopenia: Facts, numbers, and epidemiology—Update 2014. J. Cachexia Sarcopenia Muscle 2014, 5, 253–259. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Rosenberg, I.H. Sarcopenia: Origins and clinical relevance. J. Nutr. 1997, 127, 990S–991S. [Google Scholar] [CrossRef]
- Lexell, J. Human aging, muscle mass, and fiber type composition. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 1995, 50, 11–16. [Google Scholar]
- Hairi, N.N.; Cumming, R.G.; Naganathan, V.; Handelsman, D.J.; Le Couteur, D.G.; Creasey, H.; Waite, L.M.; Seibel, M.J.; Sambrook, P.N. Loss of muscle strength, mass (sarcopenia), and quality (specific force) and its relationship with functional limitation and physical disability: The concord health and ageing in men project. J. Am. Geriatr. Soc. 2010, 58, 2055–2062. [Google Scholar] [CrossRef]
- Rizzoli, R.; Reginster, J.Y.; Arnal, J.F.; Bautmans, I.; Beaudart, C.; Bischoff-Ferrari, H.; Biver, E.; Boonen, S.; Brandi, M.L.; Chines, A.; et al. Quality of life in sarcopenia and frailty. Calcif. Tissue Int. 2013, 93, 101–120. [Google Scholar] [CrossRef]
- Rantanen, T. Muscle strength, disability and mortality. Scand. J. Med. Sci. Sports 2003, 13, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Arango-Lopera, V.E.; Arroyo, P.; Gutierrez-Robledo, L.M.; Perez-Zepeda, M.U.; Cesari, M. Mortality as an adverse outcome of sarcopenia. J. Nutr. Health Aging 2013, 17, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Messier, V.; Rabasa-Lhoret, R.; Barbat-Artigas, S.; Elisha, B.; Karelis, A.D.; Aubertin-Leheudre, M. Menopause and sarcopenia: A potential role for sex hormones. Maturitas 2011, 68, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Bredella, M.A. Sex Differences in Body Composition BT-Sex and Gender Factors Affecting Metabolic Homeostasis, Diabetes and Obesity; Mauvais-Jarvis, F., Ed.; Springer International Publishing: Cham, Germany, 2017; pp. 9–27. ISBN 978-3-319-70178-3. [Google Scholar]
- Janssen, I.; Heymsfield, S.B.; Wang, Z.M.; Ross, R. Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. J. Appl. Physiol. 2000, 89, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Melton, L.J.; Khosla, S.; Riggs, B.L. Epidemiology of sarcopenia. Mayo Clin. Proc. 2000, 48, 625–630. [Google Scholar] [CrossRef]
- Shimokata, H.; Shimada, H.; Satake, S.; Endo, N.; Shibasaki, K.; Ogawa, S.; Arai, H. Chapter 2 Epidemiology of sarcopenia. Geriatr. Gerontol. Int. 2018, 18, 13–22. [Google Scholar] [CrossRef]
- Dodds, R.M.; Roberts, H.C.; Cooper, C.; Sayer, A.A. The Epidemiology of Sarcopenia. J. Clin. Densitom. 2015, 27, 355–363. [Google Scholar] [CrossRef]
- Buchmann, N.; Spira, D.; Norman, K.; Demuth, I.; Eckardt, R.; Steinhagen-Thiessen, E. Sleep, Muscle Mass and Muscle Function in Older People. Dtsch. Aerzteblatt Int. 2016, 113, 253. [Google Scholar]
- Vitale, J.; Bonato, M.; La Torre, A.; Banfi, G. The Role of the Molecular Clock in Promoting Skeletal Muscle Growth and Protecting against Sarcopenia. Int. J. Mol. Sci. 2019, 20, 4318. [Google Scholar] [CrossRef]
- Hirshkowitz, M.; Whiton, K.; Albert, S.M.; Alessi, C.; Bruni, O.; DonCarlos, L.; Hazen, N.; Herman, J.; Katz, E.S.; Kheirandish-Gozal, L.; et al. National Sleep Foundation’s sleep time duration recommendations: Methodology and results summary. Sleep Health 2015, 1, 233–243. [Google Scholar] [CrossRef]
- Ancoli-Israel, S.; Cooke, J.R. Prevalence and comorbidity of insomnia and effect on functioning in elderly populations. J. Am. Geriatr. Soc. 2005, 53, S264–S271. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wing, Y.K. Sex differences in insomnia: A meta-Analysis. Sleep 2006, 29, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Dattilo, M.; Antunes, H.K.M.; Medeiros, A.; Monico Neto, M.; Souza, H.S.; Tufik, S.; De Mello, M.T. Sleep and muscle recovery: Endocrinological and molecular basis for a new and promising hypothesis. Med. Hypotheses 2011, 77, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Cappuccio, F.P.; Cooper, D.; Delia, L.; Strazzullo, P.; Miller, M.A. Sleep duration predicts cardiovascular outcomes: A systematic review and meta-Analysis of prospective studies. Eur. Heart J. 2011, 32, 1484–1492. [Google Scholar] [CrossRef]
- Tuomilehto, H.; Peltonen, M.; Partinen, M.; Lavigne, G.; Eriksson, J.G.; Herder, C.; Aunola, S.; Keinanen-Kiukaanniemi, S.; Ilanne-Parikka, P.; Uusitupa, M.; et al. Sleep duration, lifestyle intervention, and incidence of type 2 diabetes in impaired glucose tolerance: The finnish diabetes prevention study. Diabetes Care 2009, 32, 1965–1971. [Google Scholar] [CrossRef]
- Cappuccio, F.P.; Taggart, F.M.; Kandala, N.B.; Currie, A.; Peile, E.; Stranges, S.; Miller, M.A. Meta-Analysis of short sleep duration and obesity in children and adults. Sleep 2008, 31, 619–626. [Google Scholar] [CrossRef]
- Zhao, H.; Yin, J.Y.; Yang, W.S.; Qin, Q.; Li, T.T.; Shi, Y.; Deng, Q.; Wei, S.; Liu, L.; Wang, X.; et al. Sleep duration and cancer risk: A systematic review and meta-Analysis of prospective studies. Asian Pac. J. Cancer Prev. 2013, 14, 7509–7515. [Google Scholar] [CrossRef]
- Chen, H.C.; Hsu, N.W.; Chou, P. The association between sleep duration and hand grip strength in community-Dwelling older adults: The yilan study, Taiwan. Sleep 2017, 40, zsx021. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 6, e1000100. [Google Scholar]
- Barendregt, J.J.; Doi, S.A. MetaXL User Guide: Version 5.3; EpiGear International Pty Ltd.: Queensland, Australia, 2016. [Google Scholar]
- Barendregt, J.J.; Doi, S.A.; Lee, Y.Y.; Norman, R.E.; Vos, T. Meta-Analysis of prevalence. J. Epidemiol. Community Health 2013, 67, 974–978. [Google Scholar] [CrossRef]
- Colimon, K.M. Fundamentos De Epidemiologia; Diaz de Santos: Madrid, Spain, 1990; ISBN 9788578110796. [Google Scholar]
- Ida, S.; Kaneko, R.; Nagata, H.; Noguchi, Y.; Araki, Y.; Nakai, M.; Ito, S.; Ishihara, Y.; Imataka, K.; Murata, K. Association between sarcopenia and sleep disorder in older patients with diabetes. Geriatr. Gerontol. Int. 2019, 19, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T. Measuring inconsistency in meta-Analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Chien, M.Y.; Wang, L.Y.; Chen, H.C. The Relationship of Sleep Duration with Obesity and Sarcopenia in Community-Dwelling Older Adults. Gerontology 2015, 61, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Yu, X.; Zhang, W.; Han, P.; Kang, L.; Ma, Y.; Jia, L.; Yu, H.; Chen, X.; Hou, L.; et al. The Relationship Between Sleep Duration, Falls, and Muscle Mass: A Cohort Study in an Elderly Chinese Population. Rejuvenation Res. 2018. [Google Scholar] [CrossRef]
- Hu, X.; Jiang, J.; Wang, H.; Zhang, L.; Dong, B.; Yang, M. Association between sleep duration and sarcopenia among community-Dwelling older adults: A cross-Sectional study. Medicine 2017, 96, e6268. [Google Scholar] [CrossRef]
- Kwon, Y.J.; Jang, S.Y.; Park, E.C.; Cho, A.R.; Shim, J.Y.; Linton, J.A. Long sleep duration is associated with sarcopenia in Korean adults based on data from the 2008–2011 KNHANES. J. Clin. Sleep Med. 2017, 13, 1097–1104. [Google Scholar] [CrossRef]
- Stitt, T.N.; Drujan, D.; Clarke, B.A.; Panaro, F.; Timofeyva, Y.; Kline, W.O.; Gonzalez, M.; Yancopoulos, G.D.; Glass, D.J. The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-Induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol. Cell 2004, 14, 395–403. [Google Scholar] [CrossRef]
- Schaap, L.A.; Pluijm, S.M.F.; Deeg, D.J.H.; Visser, M. Inflammatory Markers and Loss of Muscle Mass (Sarcopenia) and Strength. Am. J. Med. 2006, 119, e9–e526. [Google Scholar] [CrossRef]
- Van Leeuwen, W.M.A.; Lehto, M.; Karisola, P.; Lindholm, H.; Luukkonen, R.; Sallinen, M.; Harma, M.; Porkka-Heiskanen, T.; Alenius, H. Sleep restriction increases the risk of developing cardiovascular diseases by augmenting proinflammatory responses through IL-17 and CRP. PLoS ONE 2009, 4, e4589. [Google Scholar] [CrossRef]
- Verlaan, S.; Aspray, T.J.; Bauer, J.M.; Cederholm, T.; Hemsworth, J.; Hill, T.R.; McPhee, J.S.; Piasecki, M.; Seal, C.; Sieber, C.C.; et al. Nutritional status, body composition, and quality of life in community-Dwelling sarcopenic and non-Sarcopenic older adults: A case-Control study. Clin. Nutr. 2017, 36, 267–274. [Google Scholar] [CrossRef]
- Salva, A.; Serra-Rexach, J.A.; Artaza, I.; Formiga, F.; Rojano, I.; Luque, X.; Cuesta, F.; Lopez-Soto, A.; Masanes, F.; Ruiz, D.; et al. La prevalencia de sarcopenia en residencias de Espana: Comparacion de los resultados del estudio multicentrico ELLI con otras poblaciones. Rev. Esp. Geriatr. Gerontol. 2016, 51, 260–264. [Google Scholar] [CrossRef] [PubMed]
- Piovezan, R.D.; Abucham, J.; Dos Santos, R.V.T.; Mello, M.T.; Tufik, S.; Poyares, D. The impact of sleep on age-Related sarcopenia: Possible connections and clinical implications. Ageing Res. Rev. 2015, 23, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Baumgartner, R.; Roubenoff, R.; Mayer, J.; Nair, K.S. Sarcopenia. J. Lab. Clin. Med. 2001, 137, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Schoenau, E. From mechanostat theory to development of the “functional muscle-Bone-Unit”. J. Musculoskelet. Neuronal Interact. 2005, 5, 232. [Google Scholar]
- Gregg, E.W.; Kriska, A.M.; Salamone, L.M.; Roberts, M.M.; Anderson, S.J.; Ferrell, R.E.; Kuller, L.H.; Cauley, J.A. The epidemiology of quantitative ultrasound: A review of the relationships with bone mass, osteoporosis and fracture risk. Osteoporos. Int. 1997, 7, 89–99. [Google Scholar] [CrossRef]
- Rubio-Arias, J.; Marin-Cascales, E.; Ramos-Campo, D.J.; Hernandez, A.V.; Perez-Lopez, F.R. Effect of exercise on sleep quality and insomnia in middle-Aged women: A systematic review and meta-Analysis of randomized controlled trials. Maturitas 2017, 100, 49–56. [Google Scholar] [CrossRef]
- Pagotto, V.; Silveira, E.A. Methods, diagnostic criteria, cutoff points, and prevalence of sarcopenia among older people. Sci. World J. 2014, 2014, 231312. [Google Scholar] [CrossRef]

| Extrinsic Variables | Substantive Characteristics | Methodological Characteristics | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sleep Well | Sleep Poorly | |||||||||||||||||||
| Study | Country of the Study | Sex | Alcohol | Tobacco | Level of Physical Activity | Muscle Mass | Sleep Quality | Age | Weight | Height | BMI | Type | Sex | Total | N | Cases | Control | N2 | Cases | Control |
| Buchmann et al. | Berlin | Women | 494 | 46 | Moderate | DXA | PSQI | 68 | 25.7 | Cross-sectional | M | 568 | 492 | 79 | 413 | 76 | 27 | 49 | ||
| Women | 66 | 4 | Moderate | 69 | 31.2 | W | 628 | 508 | 64 | 444 | 120 | 13 | 107 | |||||||
| Men | 424 | 59 | Moderate | 69 | 26.4 | T | 1196 | 1000 | 143 | 857 | 196 | 40 | 156 | |||||||
| Men | 99 | 7 | Moderate | 69 | 30.2 | |||||||||||||||
| Chien et al. | Taiwan | Men | Regular | BIA | PSQI and Self-report | 78.7 | 63.9 | 162.1 | 24.3 | Cross-sectional | M | 224 | 112 | 25 | 87 | 112 | 29 | 83 | ||
| Men | Regular | 77.8 | 64.1 | 163.7 | 23.9 | W | 264 | 76 | 20 | 56 | 188 | 18 | 170 | |||||||
| Men | Regular | 80 | 66.8 | 164.4 | 24.7 | T | 488 | 188 | 45 | 143 | 300 | 47 | 253 | |||||||
| Women | Regular | 74.4 | 58.3 | 151.9 | 25.3 | |||||||||||||||
| Women | Regular | 74.5 | 57.9 | 153 | 24.8 | |||||||||||||||
| Women | Regular | 76.2 | 56.6 | 152.2 | 24.3 | |||||||||||||||
| Fu et al. | China | 48.7% Men | No = 61.1% | No = 37.2% | Moderate | BIA | Self-report | 68.24 | 70.24 | 163.38 | 25.9 | Cohort study | ||||||||
| 40.5% Men | No = 62.2% | No = 38.9% | Moderate | 66.3 | 67.96 | 163.91 | 25.3 | |||||||||||||
| 37.9% Men | No = 59.5% | No = 33.7% | Moderate | 67.38 | 67.16 | 163.39 | 25.1 | T | 920 | 468 | 52 | 416 | 452 | 43 | 409 | |||||
| 54% Men | No = 64.6% | No = 28.8% | Moderate | 68.93 | 68.1 | 163.37 | 25.4 | |||||||||||||
| Hu et al. | China | Men | 57 | 62 | Moderate | DXA | Self-report | 70.8 | 23.6 | Cross-Sectional Study | M | 251 | 63 | 13 | 50 | 188 | 28 | 160 | ||
| Men | 16 | 19 | Moderate | 72.6 | 18.7 | W | 356 | 63 | 14 | 49 | 293 | 57 | 236 | |||||||
| Women | 16 | 1 | Moderate | 69.1 | 23.6 | T | 607 | 126 | 27 | 99 | 481 | 85 | 396 | |||||||
| Women | 5 | 2 | Moderate | 72.3 | 20.3 | |||||||||||||||
| Ida et al. | Japan | Men | 60% | 72.1% | Self-report | PSQI | 71.8 | 24.3 | Cross-sectional study | M | 189 | 105 | 14 | 91 | 84 | 22 | 62 | |||
| Women | 17.2% | 4,9% | 72.8 | 23.9 | W | 129 | 71 | 11 | 60 | 58 | 24 | 34 | ||||||||
| T | 318 | 176 | 25 | 151 | 142 | 46 | 96 | |||||||||||||
| Kwon et al. | Korea | Men = 5819; Women = 8118 | 4.209 | 3.579 | Regular | DXA | Self-report | 44 | Cross-sectional study | M | ||||||||||
| Men = 1339; Women = 872 | 635 | 797 | Regular | 45.2 | W | |||||||||||||||
| T | 16148 | 4938 | 819 | 4119 | 11210 | 1486 | 9724 | |||||||||||||
| Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | M.S. | Qi | ||
|---|---|---|---|---|---|---|---|---|---|
| Buchmann et al. 2016 | [19] | 1 | 1 | 2 | 3 | 1 | 2 | 10 | 1 |
| Chien et al. 2015 | [36] | 1 | 1 | 2 | 3 | 1 | 2 | 9 | 1 |
| Fu et al. 2019 | [37] | 1 | 1 | 0 | 3 | 1 | 2 | 7 | 0.8 |
| Hu et al. 2017 | [38] | 1 | 1 | 2 | 3 | 1 | 2 | 10 | 1 |
| Ida et al. 2019 | [34] | 1 | 0 | 0 | 3 | 0 | 2 | 7 | 0.6 |
| Kwon et al. 2017 | [39] | 1 | 1 | 2 | 3 | 1 | 2 | 10 | 1 |
| Model | Sarcopenia and Self-Report or PSQI | Sarcopenia and Self-Report | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Transf. | Category | Pooled | LCI | HCI | I2 (%) | Cochran’s Q | χ2 (p) | tau2/Q-Index | Pooled | LCI | HCI | I2 | Cochran’s Q | χ2 (p) | tau2/Q-Index | |
| Inverse Variance | None | SW | 0.056 | 0.053 | 0.059 | 95.786 | 118.642 | 0.000 | 0.052 | 0.049 | 0.055 | 94.549 | 55.035 | 0.000 | ||
| SP | 0.088 | 0.084 | 0.091 | 95.786 | 118.642 | 0.000 | 0.090 | 0.086 | 0.094 | 94.549 | 55.035 | 0.000 | ||||
| Logit | SW | 0.384 | 0.052 | 0.058 | 95.241 | 105.072 | 0.000 | 0.363 | 0.049 | 0.055 | 90.653 | 32.094 | 0.000 | |||
| SP | 0.616 | 0.084 | 0.092 | 95.241 | 105.072 | 0.000 | 0.637 | 0.087 | 0.095 | 90.653 | 32.094 | 0.000 | ||||
| Double arcsine | SW | 0.388 | 0.052 | 0.059 | 95.847 | 120.389 | 0.000 | 0.363 | 0.049 | 0.055 | 93.072 | 43.300 | 0.000 | |||
| SP | 0.612 | 0.084 | 0.092 | 95.847 | 120.389 | 0.000 | 0.637 | 0.087 | 0.095 | 93.072 | 43.300 | 0.000 | ||||
| Random effects | None | SW | 0.073 | 0.044 | 0.102 | 95.786 | 118.642 | 0.000 | 0.001 | 0.060 | 0.030 | 0.091 | 94.549 | 55.035 | 0.000 | 0.001 |
| SP | 0.090 | 0.061 | 0.119 | 95.786 | 118.642 | 0.000 | 0.001 | 0.093 | 0.062 | 0.124 | 94.549 | 55.035 | 0.000 | 0.001 | ||
| Logit | SW | 0.460 | 0.046 | 0.103 | 95.241 | 105.072 | 0.000 | 0.264 | 0.399 | 0.041 | 0.083 | 90.653 | 32.094 | 0.000 | 0.126 | |
| SP | 0.540 | 0.055 | 0.119 | 95.241 | 105.072 | 0.000 | 0.264 | 0.601 | 0.062 | 0.122 | 90.653 | 32.094 | 0.000 | 0.126 | ||
| Double arcsine | SW | 0.453 | 0.044 | 0.102 | 95.847 | 120.389 | 0.000 | 0.018 | 0.395 | 0.036 | 0.086 | 93.072 | 43.300 | 0.000 | 0.011 | |
| SP | 0.547 | 0.056 | 0.120 | 95.847 | 120.389 | 0.000 | 0.018 | 0.605 | 0.061 | 0.123 | 93.072 | 43.300 | 0.000 | 0.011 | ||
| Quality effects | None | SW | 0.056 | -0.001 | 0.113 | 95.786 | 118.642 | 0.000 | 1.698 | 0.052 | 0.000 | 0.104 | 94.549 | 55.035 | 0.000 | 1.356 |
| SP | 0.088 | 0.031 | 0.145 | 95.786 | 118.642 | 0.000 | 1.698 | 0.091 | 0.039 | 0.143 | 94.549 | 55.035 | 0.000 | 1.356 | ||
| Logit | SW | 0.384 | 0.024 | 0.118 | 95.241 | 105.072 | 0.000 | 1.714 | 0.362 | 0.028 | 0.093 | 90.653 | 32.094 | 0.000 | 0.804 | |
| SP | 0.616 | 0.040 | 0.182 | 95.241 | 105.072 | 0.000 | 1.714 | 0.638 | 0.051 | 0.158 | 90.653 | 32.094 | 0.000 | 0.804 | ||
| Double arcsine | SW | 0.379 | 0.011 | 0.112 | 95.847 | 120.389 | 0.000 | 1.583 | 0.353 | 0.015 | 0.096 | 93.072 | 43.300 | 0.000 | 1.013 | |
| SP | 0.621 | 0.030 | 0.155 | 95.847 | 120.389 | 0.000 | 1.583 | 0.647 | 0.042 | 0.148 | 93.072 | 43.300 | 0.000 | 1.013 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rubio-Arias, J.Á.; Rodríguez-Fernández, R.; Andreu, L.; Martínez-Aranda, L.M.; Martínez-Rodriguez, A.; Ramos-Campo, D.J. Effect of Sleep Quality on the Prevalence of Sarcopenia in Older Adults: A Systematic Review with Meta-Analysis. J. Clin. Med. 2019, 8, 2156. https://doi.org/10.3390/jcm8122156
Rubio-Arias JÁ, Rodríguez-Fernández R, Andreu L, Martínez-Aranda LM, Martínez-Rodriguez A, Ramos-Campo DJ. Effect of Sleep Quality on the Prevalence of Sarcopenia in Older Adults: A Systematic Review with Meta-Analysis. Journal of Clinical Medicine. 2019; 8(12):2156. https://doi.org/10.3390/jcm8122156
Chicago/Turabian StyleRubio-Arias, Jacobo Á., Raquel Rodríguez-Fernández, Luis Andreu, Luis M. Martínez-Aranda, Alejandro Martínez-Rodriguez, and Domingo J. Ramos-Campo. 2019. "Effect of Sleep Quality on the Prevalence of Sarcopenia in Older Adults: A Systematic Review with Meta-Analysis" Journal of Clinical Medicine 8, no. 12: 2156. https://doi.org/10.3390/jcm8122156
APA StyleRubio-Arias, J. Á., Rodríguez-Fernández, R., Andreu, L., Martínez-Aranda, L. M., Martínez-Rodriguez, A., & Ramos-Campo, D. J. (2019). Effect of Sleep Quality on the Prevalence of Sarcopenia in Older Adults: A Systematic Review with Meta-Analysis. Journal of Clinical Medicine, 8(12), 2156. https://doi.org/10.3390/jcm8122156









