Improving Uptake of Postnatal Checking of Blood Glucose in Women Who Had Gestational Diabetes Mellitus in Universal Healthcare Settings: A Systematic Review
Abstract
1. Introduction
- Thorough screening for GDM (prevention cannot start until the high-risk groups have been identified).
- Postnatal follow-up of all women with GDM (both short and long term) [9].
2. Methods
2.1. Search Strategy
2.2. Selection Criteria
2.2.1. Inclusion Criteria
- Barriers to postnatal screening for T2DM in women with GDM. All study types included.
- Interventions: any intervention to improve uptake of postnatal screening for T2DM screening versus any other intervention or no intervention.
2.2.2. Exclusion Criteria
2.3. Study Selection
2.4. Data Extraction
2.5. Quality Assessment
2.6. Data Synthesis and Summary Measures
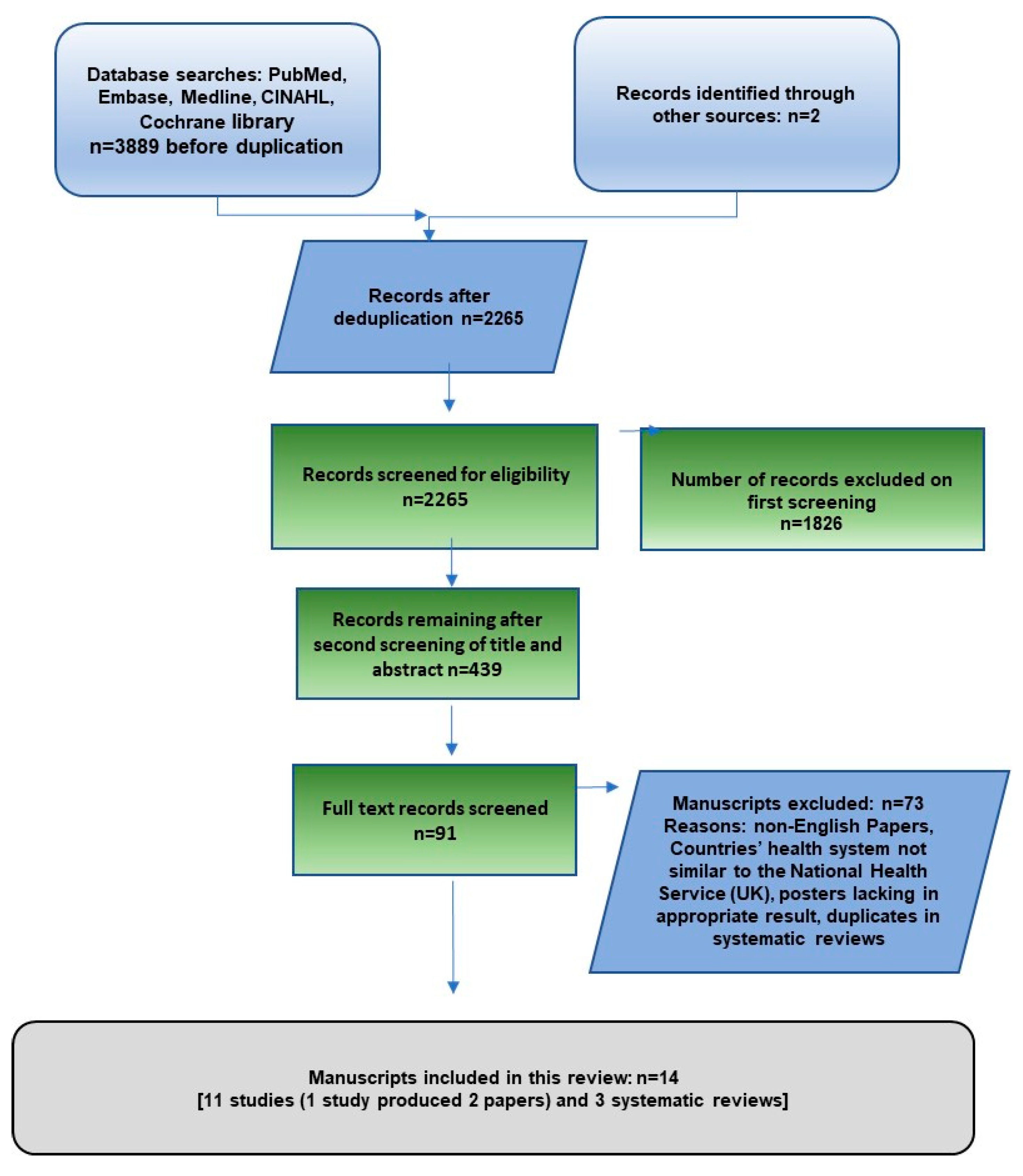
3. Results
3.1. Quality Assessment
3.2. Identified Themes
3.2.1. OGTT
3.2.2. Demands on Maternal Time
3.2.3. Education and Information
3.2.4. Risk Perception and Fear
3.2.5. Professional Knowledge/Continuity and Coordination of Care
3.2.6. Reminder Systems
4. Discussion
- The OGTT test.
- Competing demands on maternal time.
- A lack of education and information.
- Risk perception and fear.
- Knowledge amongst health care professionals.
- Problems with continuity and coordination of care, e.g., poor communication between professionals, including from secondary to primary care.
- Reminders—the intervention with the largest evidence base.
- Increasing awareness of GDM and the risk of subsequent T2DM, by education.
- A more user-friendly and convenient blood glucose test than the OGTT.
Strengths and Limitations
5. Conclusions
Implications for Future Research
Supplementary Materials
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
References
- Mulla, W.R.; Henry, T.Q.; Homko, C.J. Gestational diabetes screening after HAPO: Has anything changed? Curr. Diab. Rep. 2010, 10, 224–228. [Google Scholar] [CrossRef] [PubMed]
- Jiwani, A.; Marseille, E.; Lohse, N.; Damm, P.; Hod, M.; Kahn, J.G. Gestational diabetes mellitus: Results from a survey of country prevalence and practices. J. Matern. Fetal. Neonatal Med. 2012, 25, 600–610. [Google Scholar] [CrossRef] [PubMed]
- DeSisto, C.L.; Kim, S.Y.; Sharma, A.J. Prevalence estimates of gestational diabetes mellitus in the United States, Pregnancy Risk Assessment Monitoring System (PRAMS), 2007–2010. Prev. Chronic Dis. 2014, 11, E104. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence (NICE). Diabetes in Pregnancy: Management from Preconception to the Postnatal Period. NICE Guideline [NG3]. 2015. Available online: https://www.nice.org.uk/guidance/ng3 (accessed on 20 March 2018).
- Farrar, D. Hyperglycemia in pregnancy: Prevalence, impact, and management challenges. Int. J. Womens Health 2016, 8, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Koivusalo, S.B.; Rönö, K.; Klemetti, M.M.; Roine, R.P.; Lindström, J.; Erkkola, M.; Kaaja, R.J.; Pöyhönen-Alho, M.; Tiitinen, A.; Huvinen, E.; et al. Gestational diabetes mellitus can be prevented by lifestyle intervention: The Finnish gestational diabetes prevention study (RADIEL): A randomized controlled trial. Diabetes Care 2016, 39, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Newton, K.M.; Knopp, R.H. Gestational diabetes and the incidence of type 2 diabetes: A systematic review. Diabetes Care 2002, 25, 1862–1868. [Google Scholar] [CrossRef] [PubMed]
- Ratner, R.E. Prevention of type 2 diabetes in women with previous gestational diabetes. Diabetes Care 2007, 30 (Suppl. 2), S242–S245. [Google Scholar] [CrossRef]
- Nielsen, K.K.; Kapur, A.; Damm, P.; de Courten, M.; Bygbjerg, I.C. From screening to postpartum follow-up—The determinants and barriers for gestational diabetes mellitus (GDM) services, a systematic review. BMC Pregnancy Childbirth 2014, 14, 41. [Google Scholar] [CrossRef]
- International Association of Diabetes and Pregnancy Study Groups Consensus Panel; Metzger, B.E.; Gabbe, S.G.; Persson, B.; Buchanan, T.A.; Catalano, P.A.; Damm, P.; Dyer, A.R.; Leiva, A.D.; Hod, M.; et al. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010, 33, 676–682. [Google Scholar] [CrossRef]
- American Diabetes Association. 2. Classification and diagnosis of diabetes: Standards of medical care in diabetes 2018. Diabetes Care 2018, 41 (Suppl. 1), S13–S27. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Diabetes in Pregnancy: Management of Diabetes and Its Complications from Pre-Conception to the Postnatal Period. NICE Guideline [CG63]. 2008. Available online: https://www.nice.org.uk/guidance/cg63 (accessed on 20 March 2018).
- Clark, H.D.; van Walraven, C.; Code, C.; Karovitch, A.; Keely, E. Did publication of a clinical practice guideline recommendation to screen for type 2 diabetes in women with gestational diabetes change practice? Diabetes Care 2003, 26, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Kapaya, H.; May, L.; Jacques, R.; Madhuvrata, P. Sensitivity of postnatal fasting plasma glucose in identifying impaired glucose tolerance in women with gestational diabetes—25 Years’ data. Obstet. Med. 2017, 10, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Keely, E. An opportunity not to be missed—How do we improve postpartum screening rates for women with gestational diabetes? Diabetes Metab. Res. Rev. 2012, 28, 312–316. [Google Scholar] [CrossRef] [PubMed]
- McGovern, A.; Butler, L.; Jones, S.; van Vlymen, J.; Sadek, K.; Munro, N.; Carr, H.; de Lusignan, S. Diabetes screening after gestational diabetes in England: A quantitative retrospective cohort study. Br. J. Gen. Pract. 2014, 64, e17–e23. [Google Scholar] [CrossRef] [PubMed]
- Benhalima, K.; Damm, P.; Van Assche, A.; Mathieu, C.; Devlieger, R.; Mahmood, T.; Dunne, F. Screening for gestational diabetes in Europe: Where do we stand and how to move forward? A scientific paper commissioned by the European Board & College of Obstetrics and Gynaecology (EBCOG). Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 201, 192–196. [Google Scholar] [PubMed]
- Brownrigg, J.R.; Hughes, C.O.; Burleigh, D.; Karthikesalingam, A.; Patterson, B.O.; Holt, P.J.; Thompson, M.M.; de Lusignan, S.; Ray, K.K.; Hinchliffe, R.J. Microvascular disease and risk of cardiovascular events among individuals with type 2 diabetes: A population-level cohort study. Lancet Diabetes Endocrinol. 2016, 4, 588–597. [Google Scholar] [CrossRef]
- Goueslard, K.; Cottenet, J.; Mariet, A.S.; Sagot, P.; Petit, J.M.; Quantin, C. Early screening for type 2 diabetes following gestational diabetes mellitus in France: Hardly any impact of the 2010 guidelines. Acta Diabetol. 2017, 54, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Critical Appraisal Skills Programme (CASP). CASP Qualitative Checklist. Available online: http://www.casp-uk.net/casp-tools-checklists (accessed on 20 March 2018).
- National Institutes of Health (NIH). NIH Quality Assessment Tools. 2014. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 20 March 2018).
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Jeppesen, C.; Kristensen, J.K.; Ovesen, P.; Maindal, H.T. The forgotten risk? A systematic review of the effect of reminder systems for postpartum screening for type 2 diabetes in women with previous gestational diabetes. BMC Res. Notes 2015, 8, 373. [Google Scholar] [CrossRef]
- Van Ryswyk, E.; Middleton, P.; Shute, E.; Hague, W.; Crowther, C. Women’s views and knowledge regarding healthcare seeking for gestational diabetes in the postpartum period: A systematic review of qualitative/survey studies. Diabetes Res. Clin. Pract. 2015, 110, 109–122. [Google Scholar] [CrossRef]
- Kilgour, C.; Bogossian, F.E.; Callaway, L.; Gallois, C. Postnatal gestational diabetes mellitus follow-up: Australian women’s experiences. Women Birth 2015, 28, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Minsart, A.F.; Vander Maelen, A.; Fontaine, V.; Kirkpatrick, C. Social, medical and self-perceived factors influencing postpartum screening of diabetes after gestational diabetes. J. Obstet. Gynaecol. 2014, 34, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Sterne, V.L.; Logan, T.; Palmer, M.A. Factors affecting attendance at postpartum diabetes screening in women with gestational diabetes mellitus. Pract. Diabetes Int. 2011, 28, 64–68. [Google Scholar] [CrossRef]
- Nielsen, J.H.; Olesen, C.R.; Kristiansen, T.M.; Bak, C.K.; Overgaard, C. Reasons for women’s non-participation in follow-up screening after gestational diabetes. Women Birth 2015, 28, e157–163. [Google Scholar] [CrossRef] [PubMed]
- Van Ryswyk, E.M.; Middleton, P.F.; Hague, W.M.; Crowther, C.A. Women’s views on postpartum testing for type 2 diabetes after gestational diabetes: Six month follow-up to the DIAMIND randomised controlled trial. Prim. Care Diabetes 2016, 10, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Benhalima, K.; Verstraete, S.; Muylle, F.; Decochez, K.; Devlieger, R.; Crombrugge, P.V.; Verhaegen, A.; Wens, J.; Mathieu, C. Implementing a reminder system in the northern part of Belgium to stimulate postpartum screening for glucose intolerance in women with gestational diabetes: The “Sweet Pregnancy” Project. Int. J. Endocrinol. 2017, 2017, 3971914. [Google Scholar] [CrossRef] [PubMed]
- Carmody, L.; Egan, A.M.; Dunne, F.P. Postpartum glucose testing for women with gestational diabetes mellitus: Improving regional recall rates. Diabetes Res. Clin. Pract. 2015, 108, e38–e41. [Google Scholar] [CrossRef]
- Halperin, I.J.; Sehgal, P.; Lowe, J.; Hladunewich, M.; Wong, B.M. Increasing timely postpartum oral glucose tolerance test completion in women with gestational diabetes: A quality-improvement initiative. Can. J. Diabetes 2015, 39, 451–456. [Google Scholar] [CrossRef]
- Peticca, P.; Shah, B.R.; Shea, A.; Clark, H.D.; Malcolm, J.C.; Walker, M.; Karovitch, A.; Brazeau-Gravelle, P.; Keely, E.J. Clinical predictors for diabetes screening in the first year postpartum after gestational diabetes. Obstet. Med. 2014, 7, 116–120. [Google Scholar] [CrossRef]
- Cosson, E.; Bihan, H.; Vittaz, L.; Khiter, C.; Carbillon, L.; Faghfouri, F.; Leboeuf, D.; Dauphin, H.; Lepagnol, A.; Reach, G.; et al. Improving postpartum glucose screening after gestational diabetes mellitus: A cohort study to evaluate the multicentre IMPACT initiative. Diabet. Med. 2015, 32, 189–197. [Google Scholar] [CrossRef]
- Van Ryswyk, E.M.; Middleton, P.F.; Hague, W.M.; Crowther, C.A. Postpartum SMS reminders to women who have experienced gestational diabetes to test for Type 2 diabetes: The DIAMIND randomized trial. Diabet. Med. 2015, 32, 1368–1376. [Google Scholar] [CrossRef] [PubMed]
- Pennington, A.V.R.; O’Reilly, S.L.; Young, D.; Dunbar, J.A. Improving follow-up care for women with a history of gestational diabetes: Perspectives of GPs and patients. Aust. J. Prim. Health 2017, 23, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Pierce, M.; Modder, J.; Mortagy, I.; Springett, A.; Hughes, H.; Baldeweg, S. Missed opportunities for diabetes prevention: Post-pregnancy follow-up of women with gestational diabetes mellitus in England. Br. J. Gen. Pract. 2011, 61, e611–e619. [Google Scholar] [CrossRef] [PubMed]
- Dinglas, C.; Muscat, J.; Heo, H.; Islam, S.; Vintzileos, A. Immediate postpartum glucose tolerance testing in women with gestational diabetes: A pilot study. Am. J. Perinatol. 2017, 34, 1264–1270. [Google Scholar] [PubMed]
| Author (Year) Country Study Design Quality | Methods of Ascertainment of Barriers/Description of Intervention | Participants (Women with GDM) | Key Findings | Themes |
|---|---|---|---|---|
| Studies addressing barriers to the uptake of postnatal screening for T2DM | ||||
| Kilgour (2015) Australia. Qualitative study. Good quality. | Convergent interviews by experienced nurse and midwife, broad questions and prompts to ensure research aims addressed, approximately 40 min. | 13 | All 13 women knew of the need for a postpartum follow-up however only 7 undertook the screening. The main theme was need for information (including diagnosis of GDM; seeking GDM information; accessing specialist services; the need for postnatal screening follow-up and completing the post-natal GDM follow-up). | Risk perception; education and information; coordination of care. |
| Minsart (2014) Belgium. Observational cross-sectional survey. Fair quality. | Telephone survey about risk perception of diabetes, and reasons for avoiding screening and strategies that would have persuaded them to undertake the test. | 72 | Reasons for not attending the postpartum test included: unaware of the necessity (50%); insufficient information (42.9%); lack of time (32.1%); OGTT not convenient (17.9%); fear of diagnosis (7.2%); did not believe they were at risk anymore (3.6%); thought not necessary (25%). | Reminder systems; risk perception; education and information; coordination of care; OGTT. |
| Nielsen (2015) Denmark. Qualitative study. Good quality. | Semi-structured interviews undertaken by one researcher at woman’s home, diabetes clinic or researcher’s office. A 4-step analytical approach used to identify meaningful units and abstract and summarize content. | 7 (40 invited). | Four key themes for non-attendance: fragmented care; insufficient information; focus on physiological aspects of birth and health of baby; risk perception in everyday life. | Reminder systems; risk perception; education and information; coordination of care. |
| Sterne (2011) Australia. Observational cross-sectional interview survey/qualitative. Fair quality. | Telephone interview, approximately 10 min, standard interview form and recorded via audiotape. Questions included demographic and clinical questions and open-ended questions about barriers and facilitators to attending screening. Tick list of common barriers used. Prompts provided for potential facilitators. | 88 (187 eligible). | Barriers included lack of awareness; forgetting; inconvenience of the test; dislike of the drink for the test; fear of diabetes (also a facilitator in some). | Reminder systems; risk perception; education and information; OGTT. |
| Systematic reviews addressing barriers to the uptake of postnatal screening for T2DM | ||||
| Nielsen (2014) 19 USA, 9 Australia, 6 Canada, 1 Denmark, 1 Netherlands. Poor quality. | 58 studies included; 36 focused on postpartum follow-up; of these 5 studies addressed barriers to uptake of postpartum screening. Two studies assessed women with GDM via a survey and qualitative study respectively (3 reported perceptions of health care providers). | 344 | Barriers from women included time pressure, lost requisition, adjustment to new baby, baby’s health issues, delivery experience, feeling healthy and not in need of follow-up or fear of bad news, and experiences with medical care and services. | Reminder systems; risk perception; professional knowledge; coordination of care; demands on maternal time. |
| Van Ryswyk (2015) 12 USA, 10 Australia, 7 Canada, 3 United Kingdom, 3 Sweden, 1 Denmark, 1 France, 1 Austria, 2 Brazil, 1 Tonga, 1 Vietnam. Good quality. | 42 studies included that were qualitative or survey studies assessing barriers, facilitators and attitudes to postnatal care and follow-up including towards postpartum blood testing and use of reminders for follow-up. | 7949 | Barriers to postpartum screening included; lack of understanding around the importance; not seeing the need; forgetting; another pregnancy; lost laboratory forms; not wanting to take the test; not liking the test experience, demands on maternal time. | Reminder systems; risk perception; education and information; coordination of care; demands on maternal time; OGTT. |
| Studies addressing barriers to the uptake of postnatal screening for T2DM and interventions to increase uptake | ||||
| Van Ryswyk (2015) Van Ryswyk (2016) Australia. Cross-sectional survey and RCT. Good quality. | Intervention: text reminder to attend OGTT at 6 weeks postpartum, and further reminders at 3 and 6 months if required. Control group received 1 text reminder at 6 months. Barriers: Women sent a questionnaire that could be completed via email, post or over the telephone. | Van Ryswyk (2015)—276 (140 intervention and 136 control). Van Ryswyk (2016)—275 sent questionnaire, 208 completed. | The intervention group did not increase attendance for OGTT within 6 months postpartum compared with the control group [77.6% versus 76.8%, relative risk 1.01 (95% CI 0.89, 1.15)]. The most frequently cited barriers for non-attendance: not having enough time (73%), childcare (30%); need to focus on baby (30%); test too long (18%), perceived low risk type 2 diabetes (15%), anxiety of being diagnosed with type 2 diabetes (15%). | Reminder systems; risk perception; coordination of care; demands on maternal time; OGTT. |
| Studies of interventions to increase uptake of postnatal screening for T2DM | ||||
| Benhalima (2017) Belgium. Observational cohort study and survey. Good quality. | Automated recall via a letter and email 3 months after delivery to determine whether testing was made at 6–12 post-partum screening, where appropriate reminder letters and emails after 11 months then yearly with advice to visit GP for FPG test. Non-responders received an email/telephone call, or SMS reminder. | 5465 (500 in the survey). | 58.8% had a postpartum screening test; 2.8% of these reported having diabetes. Yearly response rates varied from 74.4% after the first year to 61.8% after the fifth year. The proportion reporting a screening test varied from 67.4% after the first year to 71.9% after the fifth year. | Reminder systems. |
| Carmody (2015) Ireland. Observational (prospective cohort). Fair quality. | Central coordinator used to facilitate attendance. Contact was written and verbal. Postdelivery women were verbally reminded of the need for a postpartum follow-up. Post 2009 a postal reminder and a telephone call was used. | 1520 | 75.6% had a postpartum OGTT. After appointment of the coordinator in 2009 there was a 12% increase in attendance on the previous year. There was a significant difference between attendance rates in 2008 compared with subsequent years (69.0% versus 77.7%, p ≤ 0.001 | Reminder systems. |
| Clark (2003) Canada. Before and after study. Fair quality. | Introduction of the CDA guideline recommending that all women with a diagnosis of gestational diabetes be screened postpartum for type 2 diabetes (OGTT 6 weeks to 6 months postpartum). | 254 (131 before and 123 after). | No women had an OGTT either before or after the guidance. 72.5% had a serum glucose in 1997 compared to 92.3% in 2000 (difference 20%, p < 0.05). | Coordination of care; OGTT. |
| Cosson (2015) France. Before and after study. Fair quality. | IMPACT initiative (health advice, care giver reminder letters) | 961 (589 before and 372 after). | The postpartum screening rate during the first 6 months postpartum was greater after (48.9%) the IMPACT campaign than before (33.3%), OR 1.7 (95% CI 1.1–2.5) | Education and information; coordination of care. |
| Halperin (2015) Canada. Before and after study. Fair quality. | 1. Improvements in physicians’ dictations 2. Patient-directed e-mail reminder systems 3. Family physician directed fax reminder systems | 300 | 44% had an OGTT within 6 months; an 11% increase to the 18 months prior to the intervention, p = 0.008. Results increased to 50% by 12 months postpartum (not significant from 42% baseline). | Reminder systems; coordination of care. |
| Peticca (2014) Canada. Observational study (retrospective cohort with intervention and no intervention). Poor quality. | A reminder package was posted within 3 months of delivery to women who attended diabetes education classes at 2 of 3 sites. Women who attended the third site received no reminder but were given education on the importance of a postpartum follow-up appointment with screening. | 546 (338 with intervention and 208 with no intervention). | Rates of OGTT completion at 12 months was 38% in those who attended clinics with reminders and 19% in those who attended clinics without reminders (p < 0.001). | Reminder systems |
| Systematic reviews of interventions to increase uptake of postnatal screening for T2DM | ||||
| Jeppesen (2015) 3 Canada, 1 USA, 1 Australia, 1 Finland. Fair quality. | 6 studies included; Reminder interventions to patients (postal, email, phone/text messages) or health professionals (also including pop-up electronic reminders and alerts or notes on medical reports) were eligible. | 1261 | All six studies included reminder systems for patients: two studies showed benefits of phone call reminders (28% attendance for OGTT versus 13.75% in control site in one study; 83.2% who completed an OGTT had received a phone reminder compared with 49.1% who did not). The number of reminders influenced the completion rate of screening tests. 80% after the first reminder, 41% of the remaining after the second reminder, and after the third reminder 28% completed the test in one study. In another study up to six reminders were sent over a 6-year period; 56.3% of women at the first reminder and 66.7% after the sixth reminder reported having an OGTT (numbers declined over time however). | Reminder systems; professional knowledge; coordination of care. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanderson, H.; Loveman, E.; Colquitt, J.; Royle, P.; Waugh, N.; Tan, B.K. Improving Uptake of Postnatal Checking of Blood Glucose in Women Who Had Gestational Diabetes Mellitus in Universal Healthcare Settings: A Systematic Review. J. Clin. Med. 2019, 8, 4. https://doi.org/10.3390/jcm8010004
Sanderson H, Loveman E, Colquitt J, Royle P, Waugh N, Tan BK. Improving Uptake of Postnatal Checking of Blood Glucose in Women Who Had Gestational Diabetes Mellitus in Universal Healthcare Settings: A Systematic Review. Journal of Clinical Medicine. 2019; 8(1):4. https://doi.org/10.3390/jcm8010004
Chicago/Turabian StyleSanderson, Helen, Emma Loveman, Jill Colquitt, Pamela Royle, Norman Waugh, and Bee Kang Tan. 2019. "Improving Uptake of Postnatal Checking of Blood Glucose in Women Who Had Gestational Diabetes Mellitus in Universal Healthcare Settings: A Systematic Review" Journal of Clinical Medicine 8, no. 1: 4. https://doi.org/10.3390/jcm8010004
APA StyleSanderson, H., Loveman, E., Colquitt, J., Royle, P., Waugh, N., & Tan, B. K. (2019). Improving Uptake of Postnatal Checking of Blood Glucose in Women Who Had Gestational Diabetes Mellitus in Universal Healthcare Settings: A Systematic Review. Journal of Clinical Medicine, 8(1), 4. https://doi.org/10.3390/jcm8010004





