The Thomsen-Friedenreich Antigen: A Highly Sensitive and Specific Predictor of Microsatellite Instability in Gastric Cancer
Abstract
1. Introduction
2. Material and Methods
2.1. Immunohistochemistry Profiling of Glycan Markers
2.2. Microsatellite Instability Analysis
2.3. Correlation Statistical Analysis of Glycan Markers and MSI Status
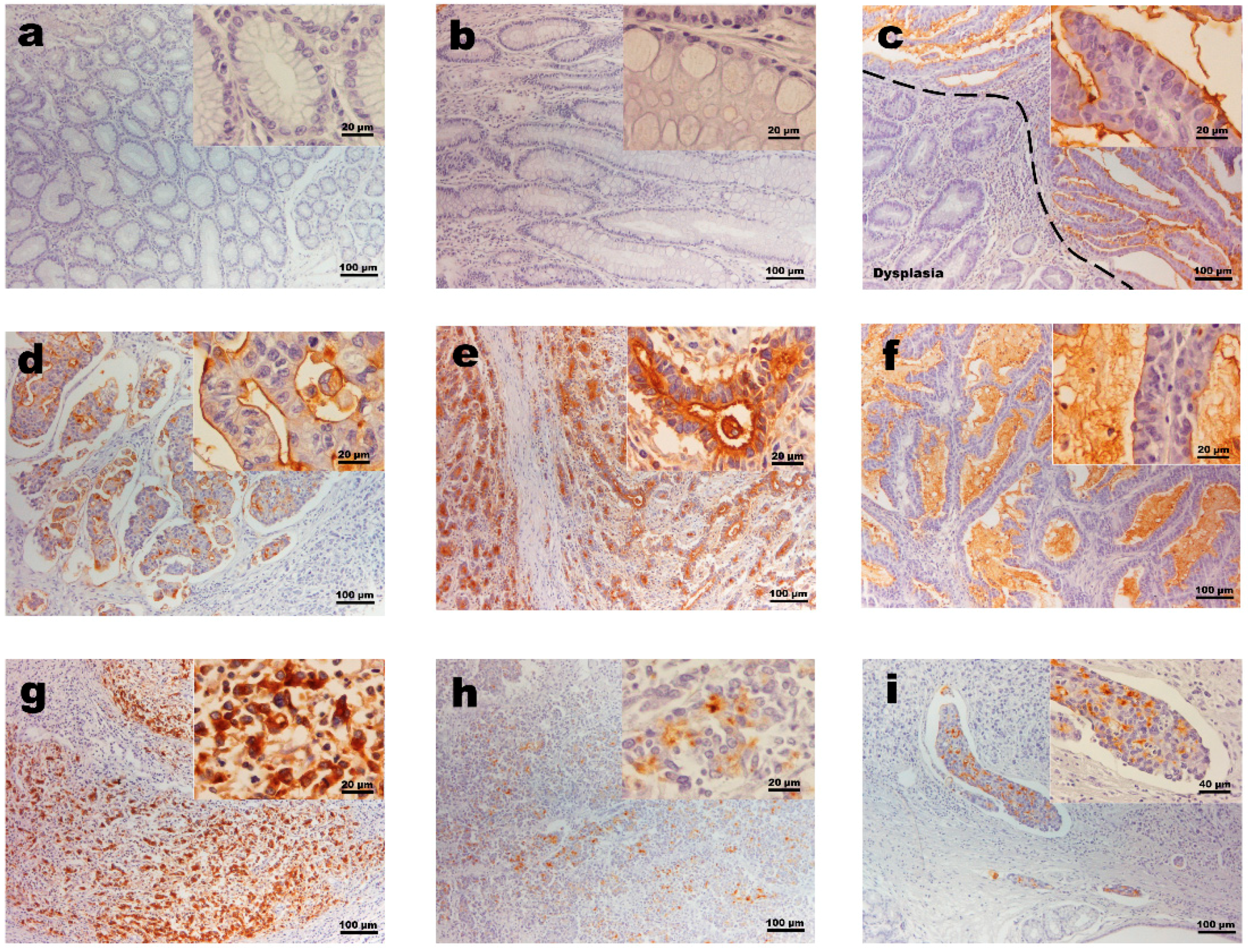
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| dMMR | mismatch repair deficiency |
| FFPE | formalin fixed paraffin embedded |
| MMR | mismatch repair |
| MSI | microsatellite instability |
| MSS | microsatellite stable |
| SLea | sialyl Lewis A |
| SLex | sialyl Lewis X |
| STn | sialyl Thomsen-nouvelle |
| TF | Thomsen-Friedenreich |
| Tn | Thomsen-nouvelle |
| VNTR | variable number of tandem repetition |
References
- Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014, 513, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Cristescu, R.; Lee, J.; Nebozhyn, M.; Kim, K.M.; Ting, J.C.; Wong, S.S.; Liu, J.; Yue, Y.G.; Wang, J.; Yu, K.; et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat. Med. 2015, 21, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Polom, K.; Boger, C.; Smyth, E.; Marrelli, D.; Behrens, H.M.; Marano, L.; Becker, T.; Lordick, F.; Rocken, C.; Roviello, F. Synchronous metastatic gastric cancer-molecular background and clinical implications with special attention to mismatch repair deficiency. Eur. J. Surg. Oncol. 2018, 44, 626–631. [Google Scholar] [CrossRef] [PubMed]
- Polom, K.; Marano, L.; Marrelli, D.; De Luca, R.; Roviello, G.; Savelli, V.; Tan, P.; Roviello, F. Meta-analysis of microsatellite instability in relation to clinicopathological characteristics and overall survival in gastric cancer. Br. J. Surg. 2018, 105, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.; Oliveira, C.; Machado, J.C.; Cirnes, L.; Tavares, J.; Carneiro, F.; Hamelin, R.; Hofstra, R.; Seruca, R.; Sobrinho-Simoes, M. MSI-L gastric carcinomas share the hmlh1 methylation status of msi-h carcinomas but not their clinicopathological profile. Lab. Investig. 2000, 80, 1915–1923. [Google Scholar] [CrossRef] [PubMed]
- Polom, K.; Marrelli, D.; Roviello, G.; Voglino, C.; Vindigni, C.; Generali, D.; Roviello, F. Single center experience on anatomy-and histopathology-based gastric cancer molecular classification. Cancer Investig. 2017, 35, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Uram, J.N.; Wang, H.; Bartlett, B.R.; Kemberling, H.; Eyring, A.D.; Skora, A.D.; Luber, B.S.; Azad, N.S.; Laheru, D.; et al. Pd-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med. 2015, 372, 2509–2520. [Google Scholar] [CrossRef] [PubMed]
- Smyth, E.C.; Wotherspoon, A.; Peckitt, C.; Gonzalez, D.; Hulkki-Wilson, S.; Eltahir, Z.; Fassan, M.; Rugge, M.; Valeri, N.; Okines, A.; et al. Mismatch repair deficiency, microsatellite instability, and survival: An exploratory analysis of the medical research council adjuvant gastric infusional chemotherapy (magic) trial. JAMA Oncol. 2017, 3, 1197–1203. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.S.; Reis, C.A. Glycosylation in cancer: Mechanisms and clinical implications. Nat. Rev. Cancer 2015, 15, 540–555. [Google Scholar] [CrossRef] [PubMed]
- Clausen, H.; Stroud, M.; Parker, J.; Springer, G.; Hakomori, S. Monoclonal antibodies directed to the blood group A associated structure, galactosyl-a: Specificity and relation to the thomsen-friedenreich antigen. Mol. Immunol. 1988, 25, 199–204. [Google Scholar] [CrossRef]
- Cao, Y.; Stosiek, P.; Springer, G.F.; Karsten, U. Thomsen-friedenreich-related carbohydrate antigens in normal adult human tissues: A systematic and comparative study. Histochem. Cell Biol. 1996, 106, 197–207. [Google Scholar] [CrossRef] [PubMed]
- David, L.; Nesland, J.M.; Clausen, H.; Carneiro, F.; Sobrinho-Simoes, M. Simple mucin-type carbohydrate antigens (tn, sialosyl-tn and t) in gastric mucosa, carcinomas and metastases. APMIS. Suppl. 1992, 27, 162–172. [Google Scholar] [PubMed]
- Reis, C.A.; Osorio, H.; Silva, L.; Gomes, C.; David, L. Alterations in glycosylation as biomarkers for cancer detection. J. Clin. Pathol. 2010, 63, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Thor, A.; Ohuchi, N.; Szpak, C.A.; Johnston, W.W.; Schlom, J. Distribution of oncofetal antigen tumor-associated glycoprotein-72 defined by monoclonal antibody b72.3. Cancer Res. 1986, 46, 3118–3124. [Google Scholar] [PubMed]
- Magnani, J.L.; Steplewski, Z.; Koprowski, H.; Ginsburg, V. Identification of the gastrointestinal and pancreatic cancer-associated antigen detected by monoclonal antibody 19-9 in the sera of patients as a mucin. Cancer Res. 1983, 43, 5489–5492. [Google Scholar] [PubMed]
- Fukushima, K.; Hirota, M.; Terasaki, P.I.; Wakisaka, A.; Togashi, H.; Chia, D.; Suyama, N.; Fukushi, Y.; Nudelman, E.; Hakomori, S. Characterization of sialosylated lewisx as a new tumor-associated antigen. Cancer Res. 1984, 44, 5279–5285. [Google Scholar] [PubMed]
- Persson, N.; Stuhr-Hansen, N.; Risinger, C.; Mereiter, S.; Polonia, A.; Polom, K.; Kovacs, A.; Roviello, F.; Reis, C.A.; Welinder, C.; et al. Epitope mapping of a new anti-tn antibody detecting gastric cancer cells. Glycobiology 2017, 27, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Marrelli, D.; Polom, K.; Pascale, V.; Vindigni, C.; Piagnerelli, R.; De Franco, L.; Ferrara, F.; Roviello, G.; Garosi, L.; Petrioli, R.; et al. Strong prognostic value of microsatellite instability in intestinal type non-cardia gastric cancer. Ann. Surg. Oncol. 2016, 23, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to pd-1 blockade. Science 2017, 357, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Springer, G.F. T and tn, general carcinoma autoantigens. Science 1984, 224, 1198–1206. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, J.W.; Wandall, H.H. Autoantibodies as biomarkers in cancer. Lab. Med. 2011, 42, 623–628. [Google Scholar] [CrossRef]
- Sindrewicz, P.; Lian, L.Y.; Yu, L.G. Interaction of the oncofetal thomsen-friedenreich antigen with galectins in cancer progression and metastasis. Front. Oncol. 2016, 6, 79. [Google Scholar] [CrossRef] [PubMed]
- Pastrello, C.; Santarosa, M.; Fornasarig, M.; Sigon, R.; Perin, T.; Giannini, G.; Boiocchi, M.; Viel, A. Muc gene abnormalities in sporadic and hereditary mucinous colon cancers with microsatellite instability. Dis. Mark. 2005, 21, 121–126. [Google Scholar] [CrossRef]
- Santos-Silva, F.; Fonseca, A.; Caffrey, T.; Carvalho, F.; Mesquita, P.; Reis, C.; Almeida, R.; David, L.; Hollingsworth, M.A. Thomsen-friedenreich antigen expression in gastric carcinomas is associated with muc1 mucin vntr polymorphism. Glycobiology 2005, 15, 511–517. [Google Scholar] [CrossRef] [PubMed]
| Patient | MSI | TF | Tn | STn | SLex | SLea | Fisher’s Exact Test | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | − | + | + | + | − | − | TF | ||||
| 2 | − | − | + | + | + | + | p = 0.00041 | + | − | Σ | |
| 3 | − | − | + | − | − | + | MSI | + | 9 | 4 | 13 |
| 4 | − | − | + | − | − | + | − | 1 | 16 | 17 | |
| 5 | − | − | + | + | − | − | Σ | 10 | 20 | 30 | |
| 6 | − | − | + | − | + | − | |||||
| 7 | − | − | + | − | − | + | Tn | ||||
| 8 | − | − | − | − | − | + | p > 0.99 | + | − | Σ | |
| 9 | − | − | + | + | − | + | MSI | + | 11 | 2 | 13 |
| 10 | − | − | + | − | − | + | − | 13 | 2 | 15 | |
| 11 | − | − | + | + | / | + | Σ | 24 | 4 | 28 | |
| 12 | − | − | / | − | − | − | |||||
| 13 | − | − | + | − | − | + | STn | ||||
| 14 | − | − | + | + | − | − | p = 0.713 | + | − | Σ | |
| 15 | − | − | / | − | − | + | MSI | + | 7 | 6 | 13 |
| 16 | − | − | + | + | − | + | − | 7 | 10 | 17 | |
| 17 | − | − | − | − | − | + | Σ | 14 | 16 | 30 | |
| 18 | + | + | + | − | − | − | |||||
| 19 | + | + | + | + | + | + | SLex | ||||
| 20 | + | + | + | − | − | − | p > 0.99 | + | − | Σ | |
| 21 | + | + | + | + | − | − | MSI | + | 2 | 11 | 13 |
| 22 | + | + | + | − | − | − | − | 2 | 14 | 16 | |
| 23 | + | + | + | + | − | + | Σ | 4 | 25 | 29 | |
| 24 | + | + | + | − | − | − | |||||
| 25 | + | + | + | + | − | − | SLea | ||||
| 26 | + | + | + | + | − | + | p = 0.063 | + | − | Σ | |
| 27 | + | − | + | + | − | − | MSI | + | 4 | 9 | 13 |
| 28 | + | − | − | − | + | + | − | 12 | 5 | 17 | |
| 29 | + | − | + | + | − | − | Σ | 16 | 14 | 30 | |
| 30 | + | − | − | − | − | − | |||||
| Variable | Categories | Total | TF Positive | TF Negative | p-Value | |||
|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |||
| 30 | 100% | 10 | 33% | 20 | 67% | |||
| Age | Median value | 72.5 | 76 | 71.5 | 0.29 | |||
| Survival in months | Median value | 44.5 | 88 | 31.5 | 0.15 | |||
| Gender | F | 13 | 43% | 5 | 50% | 8 | 40% | 0.70 |
| M | 17 | 57% | 5 | 50% | 12 | 60% | ||
| MSI status | High | 13 | 43% | 9 | 90% | 4 | 20% | 0.0004 |
| Stable | 17 | 57% | 1 | 10% | 16 | 80% | ||
| Adjuvant Therapy | No | 17 | 57% | 8 | 80% | 9 | 45% | 0.12 |
| Yes | 13 | 43% | 2 | 20% | 11 | 55% | ||
| Survival | missing value | 2 | 7% | 0 | 0% | 2 | 10% | 0.18 |
| High (≥5 years) | 13 | 43% | 7 | 70% | 6 | 30% | ||
| Medium (≥2 years & <5 years) | 5 | 17% | 1 | 10% | 4 | 20% | ||
| Low (<2 years) | 10 | 33% | 2 | 20% | 8 | 40% | ||
| GC Family History | False | 17 | 57% | 7 | 70% | 10 | 50% | 0.44 |
| True | 13 | 43% | 3 | 30% | 10 | 50% | ||
| Primary Tumor | T1 | 0 | 0% | 0 | 0% | 0 | 0% | 0.58 |
| T2 | 7 | 23% | 3 | 30% | 4 | 20% | ||
| T3 | 10 | 33% | 4 | 40% | 6 | 30% | ||
| T4 | 13 | 43% | 3 | 30% | 10 | 50% | ||
| Regional Lymph Nodes | N0 | 12 | 40% | 5 | 50% | 7 | 35% | 0.80 |
| N1 | 5 | 17% | 2 | 20% | 3 | 15% | ||
| N2 | 7 | 23% | 2 | 20% | 5 | 25% | ||
| N3a | 2 | 7% | 0 | 0% | 2 | 10% | ||
| N3b | 4 | 13% | 1 | 10% | 3 | 15% | ||
| Metastasis | M0 | 27 | 90% | 9 | 90% | 18 | 90% | 0.99 |
| M1 | 3 | 10% | 1 | 10% | 2 | 10% | ||
| Staging | I | 4 | 13% | 2 | 20% | 2 | 10% | 0.82 |
| II | 11 | 37% | 4 | 40% | 7 | 35% | ||
| III | 12 | 40% | 3 | 30% | 9 | 45% | ||
| IV | 3 | 10% | 1 | 10% | 2 | 10% | ||
| WHO | missing value | 1 | 3% | 1 | 10% | 0 | 0% | 0.37 |
| Papillary adenocarcinoma | 1 | 3% | 0 | 0% | 1 | 5% | ||
| Poorly cohesive | 11 | 37% | 3 | 30% | 8 | 40% | ||
| Signet-ring cell & mucinous | 7 | 23% | 1 | 10% | 6 | 30% | ||
| Tubular adenocarcinma | 10 | 33% | 5 | 50% | 5 | 25% | ||
| Borrman Classification | I | 2 | 7% | 0 | 0% | 2 | 10% | 0.64 |
| II | 7 | 23% | 2 | 20% | 5 | 25% | ||
| III | 17 | 57% | 7 | 70% | 10 | 50% | ||
| IV | 4 | 13% | 1 | 10% | 3 | 15% | ||
| Lauren Classification | missing value | 2 | 7% | 1 | 10% | 1 | 5% | 0.10 |
| Diffuse | 5 | 17% | 0 | 0% | 5 | 25% | ||
| Intestinal | 22 | 73% | 8 | 80% | 14 | 70% | ||
| Mixed | 1 | 3% | 1 | 10% | 0 | 0% | ||
| Tumor Site | Diffuse | 1 | 3% | 0 | 0% | 1 | 5% | 0.73 |
| Cardia | 1 | 3% | 0 | 0% | 1 | 5% | ||
| Corpus | 10 | 33% | 3 | 30% | 7 | 35% | ||
| Antrum | 18 | 60% | 7 | 70% | 11 | 55% | ||
| Vessel infiltration | missing value | 3 | 10% | 0 | 0% | 3 | 15% | 0.23 |
| FALSE | 14 | 47% | 7 | 70% | 7 | 35% | ||
| TRUE | 13 | 43% | 3 | 30% | 10 | 50% | ||
| Nerve infiltration | missing value | 6 | 20% | 3 | 30% | 3 | 15% | 0.99 |
| FALSE | 16 | 53% | 5 | 50% | 11 | 55% | ||
| TRUE | 8 | 27% | 2 | 20% | 6 | 30% | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mereiter, S.; Polom, K.; Williams, C.; Polonia, A.; Guergova-Kuras, M.; Karlsson, N.G.; Roviello, F.; Magalhães, A.; Reis, C.A. The Thomsen-Friedenreich Antigen: A Highly Sensitive and Specific Predictor of Microsatellite Instability in Gastric Cancer. J. Clin. Med. 2018, 7, 256. https://doi.org/10.3390/jcm7090256
Mereiter S, Polom K, Williams C, Polonia A, Guergova-Kuras M, Karlsson NG, Roviello F, Magalhães A, Reis CA. The Thomsen-Friedenreich Antigen: A Highly Sensitive and Specific Predictor of Microsatellite Instability in Gastric Cancer. Journal of Clinical Medicine. 2018; 7(9):256. https://doi.org/10.3390/jcm7090256
Chicago/Turabian StyleMereiter, Stefan, Karol Polom, Coralie Williams, Antonio Polonia, Mariana Guergova-Kuras, Niclas G. Karlsson, Franco Roviello, Ana Magalhães, and Celso A. Reis. 2018. "The Thomsen-Friedenreich Antigen: A Highly Sensitive and Specific Predictor of Microsatellite Instability in Gastric Cancer" Journal of Clinical Medicine 7, no. 9: 256. https://doi.org/10.3390/jcm7090256
APA StyleMereiter, S., Polom, K., Williams, C., Polonia, A., Guergova-Kuras, M., Karlsson, N. G., Roviello, F., Magalhães, A., & Reis, C. A. (2018). The Thomsen-Friedenreich Antigen: A Highly Sensitive and Specific Predictor of Microsatellite Instability in Gastric Cancer. Journal of Clinical Medicine, 7(9), 256. https://doi.org/10.3390/jcm7090256






