Nutcracker Syndrome in Children: Role of Doppler Ultrasonographic Indices in Detecting the Pattern of Symptoms
Abstract
:1. Introduction
2. Material and Methods
Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- De Schepper, A. “Nutcracker” phenomenon of the renal vein and venous pathology of the left kidney. J. Belge. Radiol. 1972, 55, 507–511. [Google Scholar] [PubMed]
- Kurklinsky, A.K.; Rooke, T.W. Nutcracker phenomenon and nutcracker syndrome. Mayo Clin. Proc. 2010, 85, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Ananthan, K.; Onida, S.; Davies, A.H. Nutcracker Syndrome: An Update on Current Diagnostic Criteria and Management Guidelines. Eur. J. Vasc. Endovasc. Surg. 2017, 53, 886–894. [Google Scholar] [CrossRef] [PubMed]
- I Calàbria, H.C.; Gómez, S.Q.; Cerqueda, C.S.; de la Presa, R.B.; Miranda, A.; Alvarez-Castells, A. Nutcracker or left renal vein compression phenomenon: Multidetector computed tomography findings and clinical significance. Eur. Radiol. 2005, 15, 1745–1751. [Google Scholar] [CrossRef] [PubMed]
- Venkatachalam, S.; Bumpus, K.; Kapadia, S.R.; Gray, B.; Lyden, S.; Shishehbor, M.H. The nutcracker syndrome. Ann. Vasc. Surg. 2011, 25, 1154–1164. [Google Scholar] [CrossRef]
- Gulleroglu, K.; Gulleroglu, B.; Baskin, E. Nutcracker syndrome. World J. Nephrol. 2014, 3, 277–281. [Google Scholar] [CrossRef]
- Rudloff, U.; Holmes, R.J.; Prem, J.T.; Faust, G.R.; Moldwin, R.; Siegel, D. Mesoaortic compression of the left renal vein (nutcracker syndrome): Case reports and review of the literature. Ann. Vasc. Surg. 2006, 20, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Lim, J.W.; Cho, B.S.; Yoon, T.Y.; Oh, J.H. Nutcracker syndrome in children with orthostatic proteinuria: Diagnosis on the basis of Doppler sonography. J. Ultrasound Med. 2002, 21, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Ekim, M.; Ozçakar, Z.B.; Fitoz, S.; Soygür, T.; Yüksel, S.; Acar, B.; Yalçinkaya, F.; Arikan, N. The “nutcracker phenomenon” with orthostatic proteinuria: Case reports. Clin. Nephrol. 2006, 65, 280–283. [Google Scholar] [CrossRef] [PubMed]
- Ozçakar, Z.B.; Yalçınkaya, F.; Fitöz, S.; Cipe, G.; Soygür, T.; Ozdemir, H.; Köksoy, C. Nutcracker syndrome manifesting with severe proteinuria: A challenging scenario in a single-kidney patient. Pediatr. Nephrol. 2011, 26, 987–990. [Google Scholar] [CrossRef] [PubMed]
- Alaygut, D.; Bayram, M.; Soylu, A.; Cakmakcı, H.; Türkmen, M.; Kavukcu, S. Clinical course of children with nutcracker syndrome. Urology 2013, 82, 686–690. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.I.; Park, J.M.; Lee, J.S.; Kim, M.J. Effect of renal Doppler ultrasound on the detection of nutcracker syndrome in children with hematuria. Eur. J. Pediatr. 2007, 166, 399–404. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wu, Z.; Chen, S.; Tian, L.; Li, D.; Li, M.; Jin, W.; Zhang, H. Nutcracker syndrome—How well do we know it? Urology 2014, 83, 12–17. [Google Scholar] [CrossRef]
- Fitoz, S.; Ekim, M.; Ozcakar, Z.B.; Elhan, A.H.; Yalcinkaya, F. Nutcracker syndrome in children: The role of upright position examination and superior mesenteric artery angle measurement in the diagnosis. J. Ultrasound Med. 2007, 26, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Cho, S.W.; Kim, H.D.; Chung, J.W.; Park, J.H.; Han, M.C. Nutcracker syndrome: Diagnosis with Doppler US. Radiology 1996, 198, 93–97. [Google Scholar] [CrossRef]
- Takebayashi, S.; Ueki, T.; Ikeda, N.; Fujikawa, A. Diagnosis of the nutcracker syndrome with color Doppler sonography: Correlation with flow patterns on retrograde left renal venography. Am. J. Roentgenol. 1999, 172, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Cheon, J.E.; Kim, W.S.; Kim, I.O.; Kim, S.H.; Yeon, K.M.; Ha, I.S.; Cheong, H.I.; Choi, Y. Nutcracker syndrome in children with gross haematuria: Doppler sonographic evaluation of the left renal vein. Pediatr. Radiol. 2006, 36, 682–686. [Google Scholar] [CrossRef] [PubMed]
- Okada, M.; Tsuzuki, K.; Ito, S. Diagnosis of the nutcracker phenomenon using two-dimensional ultrasonography. Clin. Nephrol. 1998, 49, 35–40. [Google Scholar]
- Orczyk, K.; Łabętowicz, P.; Lodziński, S.; Stefańczyk, L.; Topol, M.; Polguj, M. The nutcracker syndrome—Morphology and clinical aspects of the important vascular variations: A systematic study of 112 cases. Int. Angiol. 2016, 35, 71–77. [Google Scholar] [PubMed]
- Hangge, P.T.; Gupta, N.; Khurana, A.; Quencer, K.B.; Albadawi, H.; Alzubaidi, S.J.; Knuttinen, M.G.; Naidu, S.G.; Oklu, R. Degree of Left Renal Vein Compression Predicts Nutcracker Syndrome. J. Clin. Med. 2018, 7, 107. [Google Scholar] [CrossRef] [PubMed]
- Orczyk, K.; Wysiadecki, G.; Majos, M.; Stefańczyk, L.; Topol, M.; Polguj, M. What each clinical anatomist has to know about left renal vein entrapment syndrome (Nutcracker syndrome)—A review of the most important findings. Biomed Res. Int. 2017, 2017. [Google Scholar] [CrossRef]
- Park, S.J.; Shin, J.I. Renal doppler ultrasonography in the diagnosis of nutcracker syndrome. Eur. J. Pediatr. 2013, 172, 135–136. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.I.; Park, J.M.; Lee, S.M.; Shin, Y.H.; Kim, J.H.; Lee, J.S.; Kim, M.J. Factors affecting spontaneous resolution of hematuria in childhood nutcracker syndrome. Pediatr. Nephrol. 2005, 20, 609–613. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.I.; Lee, J.S.; Kim, M.J. Re: Endovascular stent placement for the treatment of nutcracker phenomenon in three pediatric patients. J. Vasc. Interv. Radiol. 2006, 17, 1063–1064. [Google Scholar] [CrossRef]
- Kim, J.Y.; Joh, J.H.; Choi, H.Y.; Do, Y.S.; Shin, S.W.; Kim, D.I. Transposition of the left renal vein in nutcracker syndrome. Eur. J. Vasc. Endovasc. Surg. 2006, 31, 80–82. [Google Scholar] [CrossRef] [PubMed]
| Variables | Study Population (n = 44) |
|---|---|
| Age (years) | 12.8 ± 2.79 (7–18) |
| Gender, n (%) | female, 25 (56.8%), male, 19 (43.2%) |
| Clinical findings, n (%) | |
| Complaints (negative) n (%) | 27 (61.4%) |
| Incidental finding of microscopic hematuria, n (%) | 6 (13.6%) |
| Incidental finding of proteinuria, n (%) | 21 (47.7%) |
| Complaints (positive), n (%) | 17 (38.6%) |
| Loin pain, n (%) | 9 (20.5%) |
| Macroscopic hematuria, n (%) | 6 (13.6%) |
| Loin pain + macroscopic hematuria, n (%) | 2 (4.5%) |
| BMI (kg/m2) | 16.4 (14.2–26.8) |
| 24-hour urine protein excretion (n = 35) | 13 (1.6–42) |
| Individual LRV Doppler US findings | |
| Diameter at the hilar portion (mm) | 8.43 ± 3.25 |
| Diameter at the AM portion (mm) | 2 (1.1–6.1) |
| Diameter Ratio between hilar/AM | 4.36 ± 1.55 |
| PV at the AM portion (cm/s) | 144.9 ± 56.73 |
| PV at the hilar portion (cm/s) | 19.72 ± 5.09 |
| PV ratio between AM/hilar | 7.32 ± 2.68 |
| Time to diagnosis (months) | 2.50 (1–60) |
| Treatment, n | |
| Conservative | 43 |
| Surgery | 1 |
| Individual LRV Doppler US Findings | Asymptomatic * Group | Symptomatic ** Group | p |
|---|---|---|---|
| Diameter at the hilar portion (mm) | 8.93 ± 3.82 | 7.63 ± 1.89 | 0.215 |
| Diameter at the AM portion (mm) | 2 (1.20–6.10) | 2.15 (1.10–4) | 0.103 |
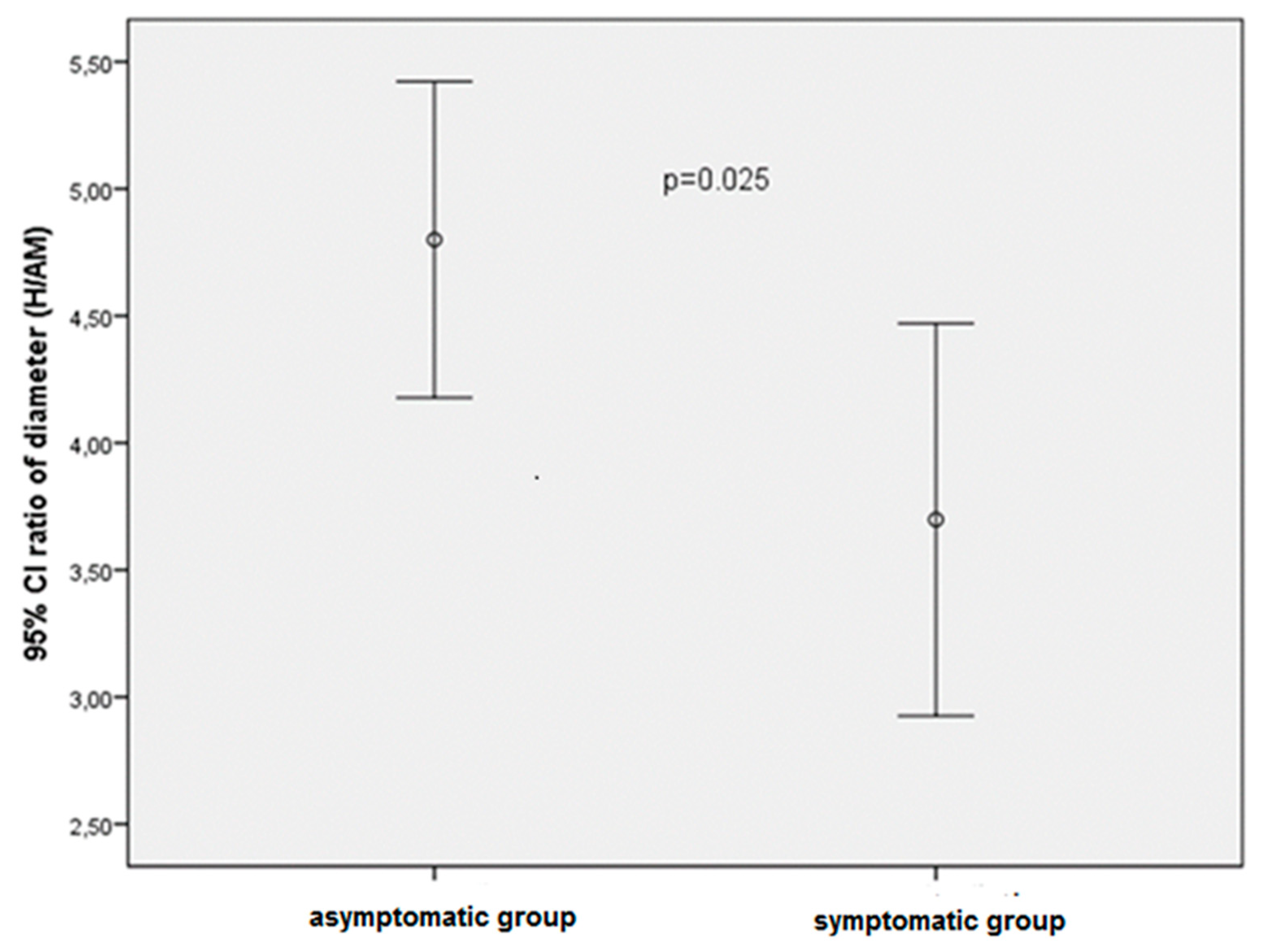
| Diameter Ratio between hilar/AM | 4.79 ± 1.47 | 3.69 ± 1.45 | 0.025 |
| PV at the AM portion (cm/s) | 150.1 ± 63.53 | 135.9 ± 43.01 | 0.118 |
| PV at the hilar portion (cm/s) | 18.77 ± 5.22 | 21.36 ± 4.55 | 0.449 |
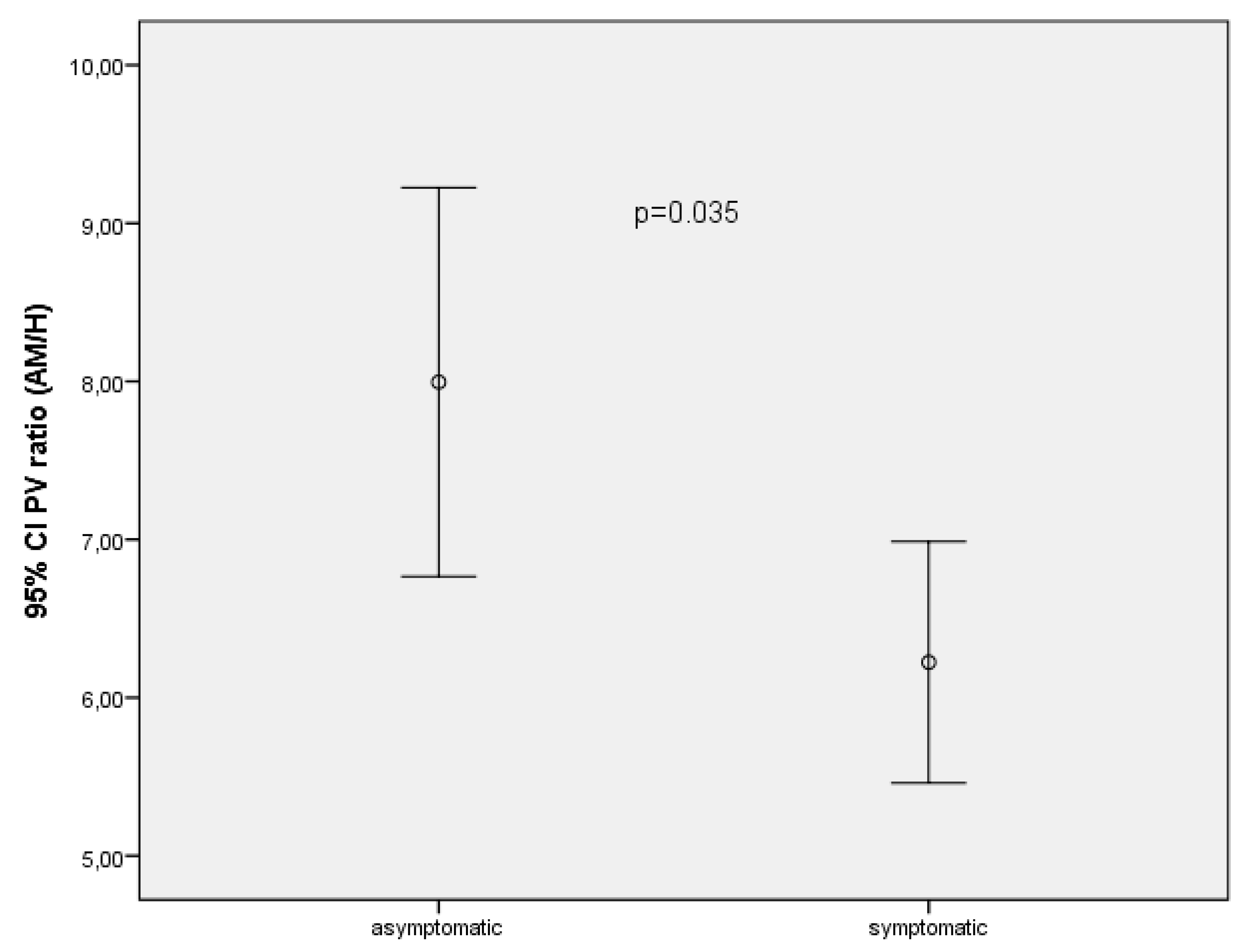
| PV ratio between AM/hilar | 7.99 ± 3.04 | 6.22 ± 1.43 | 0.035 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nalcacioglu, H.; Ceyhan Bilgici, M.; Tekcan, D.; Genc, G.; Bostanci, Y.; Yakupoglu, Y.K.; Sarikaya, S.; Ozkaya, O. Nutcracker Syndrome in Children: Role of Doppler Ultrasonographic Indices in Detecting the Pattern of Symptoms. J. Clin. Med. 2018, 7, 214. https://doi.org/10.3390/jcm7080214
Nalcacioglu H, Ceyhan Bilgici M, Tekcan D, Genc G, Bostanci Y, Yakupoglu YK, Sarikaya S, Ozkaya O. Nutcracker Syndrome in Children: Role of Doppler Ultrasonographic Indices in Detecting the Pattern of Symptoms. Journal of Clinical Medicine. 2018; 7(8):214. https://doi.org/10.3390/jcm7080214
Chicago/Turabian StyleNalcacioglu, Hulya, Meltem Ceyhan Bilgici, Demet Tekcan, Gurkan Genc, Yakup Bostanci, Yarkin Kamil Yakupoglu, Saban Sarikaya, and Ozan Ozkaya. 2018. "Nutcracker Syndrome in Children: Role of Doppler Ultrasonographic Indices in Detecting the Pattern of Symptoms" Journal of Clinical Medicine 7, no. 8: 214. https://doi.org/10.3390/jcm7080214
APA StyleNalcacioglu, H., Ceyhan Bilgici, M., Tekcan, D., Genc, G., Bostanci, Y., Yakupoglu, Y. K., Sarikaya, S., & Ozkaya, O. (2018). Nutcracker Syndrome in Children: Role of Doppler Ultrasonographic Indices in Detecting the Pattern of Symptoms. Journal of Clinical Medicine, 7(8), 214. https://doi.org/10.3390/jcm7080214





