Outcome Prediction of Acute Kidney Injury Biomarkers at Initiation of Dialysis in Critical Units
Abstract
1. Background
2. Methods
2.1. Registration of Clinical Trials
2.2. Study Population
2.3. Data Collection
2.4. Measurements of Kidney Biomarkers
2.5. Outcome Definitions
2.6. Statistical Analysis
3. Results
3.1. Clinical Characteristics
3.2. Hospital and 90-Day Outcomes
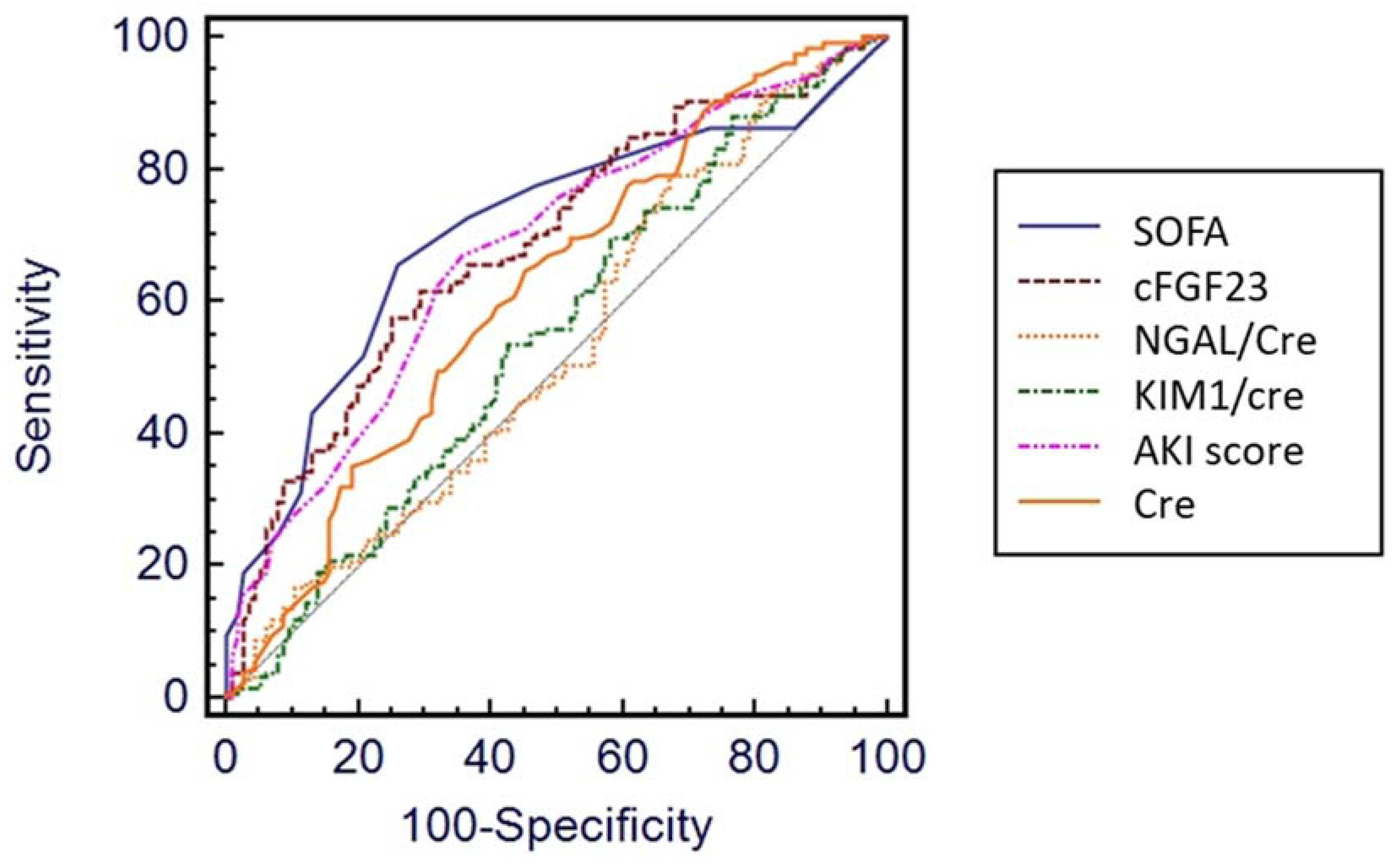
3.3. Discriminative Power of Biomarkers for 90-Day Relevant Outcomes
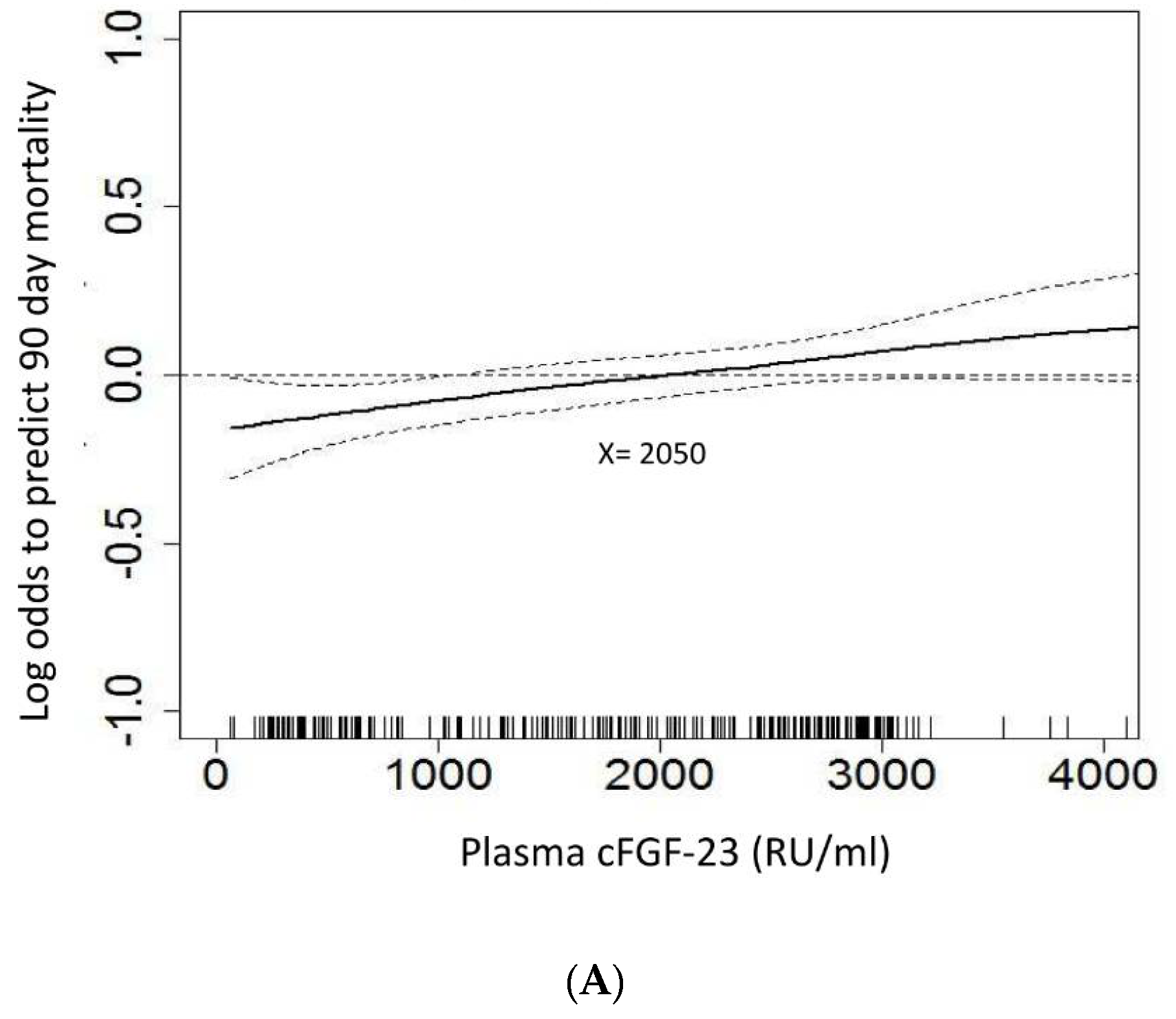
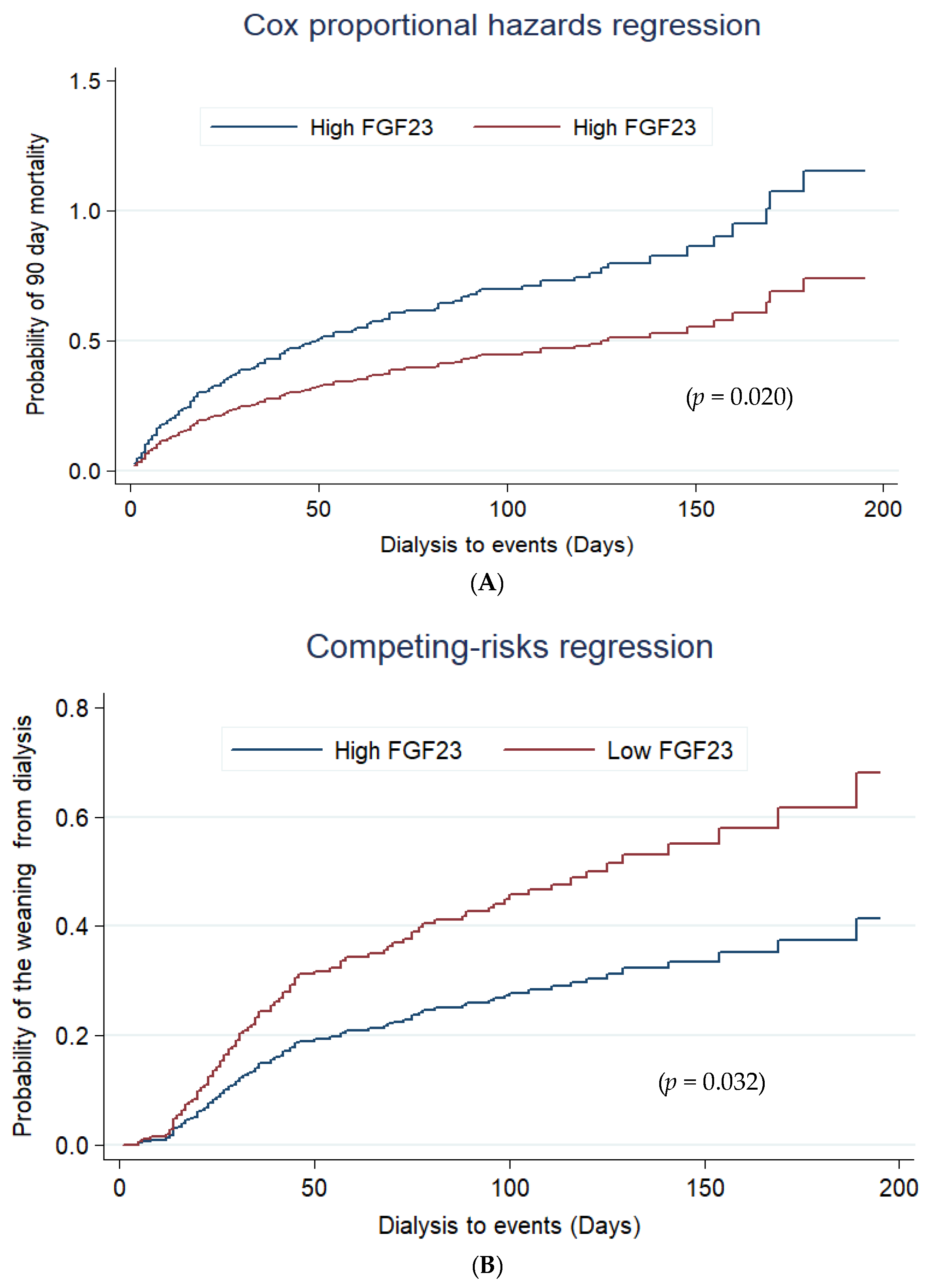
3.4. Plasma cFGF-23 and Outcome
3.5. Addition of cFGF-23 to AKI Risk Predicting Score at Start of Dialysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgment
Conflicts of Interest
Abbreviations
| AKI | acute kidney injury |
| APACHE II | Acute Physiology and Chronic Health Evaluation II |
| AUROC | area under the receiver operator characteristic curve |
| CKD | chronic kidney disease |
| ESRD | end-stage renal disease |
| ICU | intensive care unit |
| KIM-1 | Kidney Injury Molecule-1 |
| MODS | Multiple Organ Dysfunction Score |
| NGAL | neutrophil gelatinase-associated lipocalin |
| RRT | renal replacement therapy |
| RU | relative units |
| sCr | serum creatinine |
| SOFA | Sequential Organ Failure Assessment |
References
- Shiao, C.C.; Wu, P.C.; Huang, T.M.; Lai, T.S.; Yang, W.S.; Wu, C.H.; Lai, C.F.; Wu, V.C.; Chu, T.S.; Wu, K.D.; et al. Long-term remote organ consequences following acute kidney injury. Crit. Care 2015, 19, 438. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Shiao, C.C.; Chang, C.H.; Huang, T.M.; Lai, C.F.; Lin, M.C.; Chiang, W.C.; Chu, T.S.; Wu, K.D.; Ko, W.J.; et al. Long-term outcomes after dialysis-requiring acute kidney injury. Biomed. Res. Int. 2014, 2014, 365186. [Google Scholar] [CrossRef] [PubMed]
- Wald, R.; Bagshaw, S.M. The timing of renal replacement therapy initiation in acute kidney injury: Is earlier truly better? Crit. Care Med. 2014, 42, 1933–1934. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.J.; Brandtner, A.K.; Lehner, G.F.; Ulmer, H.; Bagshaw, S.M.; Wiedermann, C.J.; Joannidis, M. Biomarkers for prediction of renal replacement therapy in acute kidney injury: A systematic review and meta-analysis. Intensive Care Med. 2018, 44, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Kumpers, P.; Hafer, C.; Lukasz, A.; Lichtinghagen, R.; Brand, K.; Fliser, D.; Faulhaber-Walter, R.; Kielstein, J.T. Serum neutrophil gelatinase-associated lipocalin at inception of renal replacement therapy predicts survival in critically ill patients with acute kidney injury. Crit. Care 2010, 14, R9. [Google Scholar] [CrossRef] [PubMed]
- Zarbock, A.; Kellum, J.A.; Schmidt, C.; Van Aken, H.; Wempe, C.; Pavenstadt, H.; Boanta, A.; Gerss, J.; Meersch, M. Effect of Early vs Delayed Initiation of Renal Replacement Therapy on Mortality in Critically Ill Patients With Acute Kidney Injury: The Elain Randomized Clinical Trial. JAMA 2016, 315, 2190–2199. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Chang, C.H.; Fan, P.C.; Tian, Y.C.; Chang, M.Y.; Jenq, C.C.; Hung, C.C.; Fang, J.T.; Yang, C.W.; Chen, Y.C. Serum interleukin-18 at commencement of renal replacement therapy predicts short-term prognosis in critically ill patients with acute kidney injury. PLoS ONE 2013, 8, e66028. [Google Scholar] [CrossRef] [PubMed]
- Pike, F.; Murugan, R.; Keener, C.; Palevsky, P.M.; Vijayan, A.; Unruh, M.; Finkel, K.; Wen, X.; Kellum, J.A. Biomarker Enhanced Risk Prediction for Adverse Outcomes in Critically Ill Patients Receiving RRT. Clin. J. Am. Soc. Nephrol. 2015, 10, 1332–1339. [Google Scholar] [CrossRef] [PubMed]
- Consortium, A. Autosomal dominant hypophosphataemic rickets is associated with mutations in FGF23. Nat. Genet. 2000, 26, 345–348. [Google Scholar]
- Berndt, T.; Kumar, R. Phosphatonins and the regulation of phosphate homeostasis. Annu. Rev. Physiol. 2007, 69, 341–359. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.N.; Hassinger, A.; Price, H.; Langman, C.B. Preoperative plasma FGF23 levels predict acute kidney injury in children: Results of a pilot study. Pediatr. Nephrol. 2013, 28, 959–962. [Google Scholar] [CrossRef] [PubMed]
- Leaf, D.E.; Jacob, K.A.; Srivastava, A.; Chen, M.E.; Christov, M.; Juppner, H.; Sabbisetti, V.S.; Martin, A.; Wolf, M.; Waikar, S.S. Fibroblast Growth Factor 23 Levels Associate with AKI and Death in Critical Illness. J. Am. Soc. Nephrol. 2017, 28, 1877–1885. [Google Scholar] [CrossRef] [PubMed]
- Leaf, D.E.; Christov, M.; Juppner, H.; Siew, E.; Ikizler, T.A.; Bian, A.; Chen, G.; Sabbisetti, V.S.; Bonventre, J.V.; Cai, X.; et al. Fibroblast growth factor 23 levels are elevated and associated with severe acute kidney injury and death following cardiac surgery. Kidney Int. 2016, 89, 939–948. [Google Scholar] [CrossRef] [PubMed]
- Christov, M.; Waikar, S.S.; Pereira, R.C.; Havasi, A.; Leaf, D.E.; Goltzman, D.; Pajevic, P.D.; Wolf, M.; Juppner, H. Plasma FGF23 levels increase rapidly after acute kidney injury. Kidney Int. 2013, 84, 776–785. [Google Scholar] [CrossRef] [PubMed]
- Donate-Correa, J.; de Fuentes, M.M.; Mora-Fernandez, C.; Navarro-Gonzalez, J.F. Pathophysiological implications of fibroblast growth factor-23 and Klotho and their potential role as clinical biomarkers. Clin. Chem. 2014, 60, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Ko, W.J.; Chang, H.W.; Chen, Y.S.; Chen, Y.W.; Chen, Y.M.; Hu, F.C.; Lin, Y.H.; Tsai, P.R.; Wu, K.D. Early renal replacement therapy in patients with postoperative acute liver failure associated with acute renal failure: Effect on postoperative outcomes. J. Am. Coll. Surg. 2007, 205, 266–276. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Ko, W.J.; Chang, H.W.; Chen, Y.W.; Lin, Y.F.; Shiao, C.C.; Chen, Y.M.; Chen, Y.S.; Tsai, P.R.; Hu, F.C.; et al. Risk factors of early redialysis after weaning from postoperative acute renal replacement therapy. Intensive Care Med. 2008, 34, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Shiao, C.C.; Wu, V.C.; Li, W.Y.; Lin, Y.F.; Hu, F.C.; Young, G.H.; Kuo, C.C.; Kao, T.W.; Huang, D.M.; Chen, Y.M.; et al. Late initiation of renal replacement therapy is associated with worse outcomes in acute kidney injury after major abdominal surgery. Crit. Care 2009, 13, R171. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Wang, C.H.; Wang, W.J.; Lin, Y.F.; Hu, F.C.; Chen, Y.W.; Chen, Y.S.; Wu, M.S.; Lin, Y.H.; Kuo, C.C.; et al. Sustained low-efficiency dialysis versus continuous veno-venous hemofiltration for postsurgical acute renal failure. Am. J. Surg. 2010, 199, 466–476. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.M.; Wu, V.C.; Young, G.H.; Lin, Y.F.; Shiao, C.C.; Wu, P.C.; Li, W.Y.; Yu, H.Y.; Hu, F.C.; Lin, J.W.; et al. Preoperative proteinuria predicts adverse renal outcomes after coronary artery bypass grafting. J. Am. Soc. Nephrol. 2011, 22, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Demirjian, S.; Chertow, G.M.; Zhang, J.H.; O’Connor, T.Z.; Vitale, J.; Paganini, E.P.; Palevsky, P.M. Network VNARFT. Model to predict mortality in critically ill adults with acute kidney injury. Clin. J. Am. Soc. Nephrol. 2011, 6, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Huang, T.M.; Lai, C.F.; Shiao, C.C.; Lin, Y.F.; Chu, T.S.; Wu, P.C.; Chao, C.T.; Wang, J.Y.; Kao, T.W.; et al. Acute-on-chronic kidney injury at hospital discharge is associated with long-term dialysis and mortality. Kidney Int. 2011, 80, 1222–1230. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.F.; Ko, W.J.; Wu, V.C.; Chen, Y.S.; Chen, Y.M.; Hu, F.C.; Shiao, C.C.; Wu, M.S.; Chen, Y.W.; Li, W.Y.; et al. A modified sequential organ failure assessment score to predict hospital mortality of postoperative acute renal failure patients requiring renal replacement therapy. Blood Purif. 2008, 26, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Shiao, C.C.; Ko, W.J.; Wu, V.C.; Huang, T.M.; Lai, C.F.; Lin, Y.F.; Chao, C.T.; Chu, T.S.; Tsai, H.B.; Wu, P.C.; et al. U-curve association between timing of renal replacement therapy initiation and in-hospital mortality in postoperative acute kidney injury. PLoS ONE 2012, 7, e42952. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Lo, S.C.; Chen, Y.L.; Huang, P.H.; Tsai, C.T.; Liang, C.J.; Kuo, C.C.; Kuo, Y.S.; Lee, B.C.; Wu, E.L.; et al. Endothelial progenitor cells in primary aldosteronism: A biomarker of severity for aldosterone vasculopathy and prognosis. J. Clin. Endocrinol. Metab. 2011, 96, 3175–3183. [Google Scholar] [CrossRef] [PubMed]
- Wu, V.C.; Lai, C.F.; Shiao, C.C.; Lin, Y.F.; Wu, P.C.; Chao, C.T.; Hu, F.C.; Huang, T.M.; Yeh, Y.C.; Tsai, I.J.; et al. Effect of diuretic use on 30-day postdialysis mortality in critically ill patients receiving acute dialysis. PLoS ONE 2012, 7, e30836. [Google Scholar] [CrossRef] [PubMed]
- Hin, L.Y.; Lau, T.K.; Rogers, M.S.; Chang, A.M. Dichotomization of continuous measurements using generalized additive modelling—Application in predicting intrapartum caesarean delivery. Stat. Med. 1999, 18, 1101–1110. [Google Scholar] [CrossRef]
- Wu, V.C.; Chang, C.H.; Wang, C.Y.; Lin, Y.H.; Kao, T.W.; Lin, P.C.; Chu, T.S.; Chang, Y.S.; Chen, L.; Wu, K.D.; et al. Risk of Fracture in Primary Aldosteronism: A Population-Based Cohort Study. J. Bone Miner. Res. 2017, 32, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Hsu, R.; Hsu, C.Y.; Kordesch, K.; Nicasio, E.; Cortez, A.; McAlpine, I.; Brady, S.; Zhuo, H.; Kangelaris, K.N.; et al. FGF-23 and PTH levels in patients with acute kidney injury: A cross-sectional case series study. Ann. Intensive Care. 2011, 1, 21. [Google Scholar] [CrossRef] [PubMed]
- Leaf, D.E.; Wolf, M.; Waikar, S.S.; Chase, H.; Christov, M.; Cremers, S.; Stern, L. FGF-23 levels in patients with AKI and risk of adverse outcomes. Clin. J. Am. Soc. Nephrol. 2012, 7, 1217–1223. [Google Scholar] [CrossRef] [PubMed]
- Leaf, D.E.; Waikar, S.S.; Wolf, M.; Cremers, S.; Bhan, I.; Stern, L. Dysregulated mineral metabolism in patients with acute kidney injury and risk of adverse outcomes. Clin. Endocrinol. 2013, 79, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.R.; Katz, R.; Ix, J.H.; de Boer, I.H.; Siscovick, D.S.; Grams, M.E.; Shlipak, M.; Sarnak, M.J. Fibroblast growth factor-23 and the long-term risk of hospital-associated AKI among community-dwelling older individuals. Clin. J. Am. Soc. Nephrol. 2014, 9, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Neyra, J.A.; Moe, O.W.; Hu, M.C. Fibroblast growth factor 23 and acute kidney injury. Pediatr. Nephrol. 2015, 30, 1909–1918. [Google Scholar] [CrossRef] [PubMed]
- Shimada, T.; Muto, T.; Urakawa, I.; Yoneya, T.; Yamazaki, Y.; Okawa, K.; Takeuchi, Y.; Fujita, T.; Fukumoto, S.; Yamashita, T. Mutant FGF-23 responsible for autosomal dominant hypophosphatemic rickets is resistant to proteolytic cleavage and causes hypophosphatemia in vivo. Endocrinology 2002, 143, 3179–3182. [Google Scholar] [CrossRef] [PubMed]
- Spichtig, D.; Zhang, H.; Mohebbi, N.; Pavik, I.; Petzold, K.; Stange, G.; Saleh, L.; Edenhofer, I.; Segerer, S.; Biber, J.; et al. Renal expression of FGF23 and peripheral resistance to elevated FGF23 in rodent models of polycystic kidney disease. Kidney Int. 2014, 85, 1340–1350. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.; Durlacher, K.; Silver, J.; Naveh-Many, T.; Levi, R. The fibroblast growth factor receptor mediates the increased FGF23 expression in acute and chronic uremia. Am. J. Physiol. Renal Physiol. 2016, 310, F217–F221. [Google Scholar] [CrossRef] [PubMed]
- Faul, C.; Amaral, A.P.; Oskouei, B.; Hu, M.C.; Sloan, A.; Isakova, T.; Gutierrez, O.M.; Aguillon-Prada, R.; Lincoln, J.; Hare, J.M.; et al. FGF23 induces left ventricular hypertrophy. J. Clin. Investig. 2011, 121, 4393–4408. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.H.; Fan, P.C.; Chang, M.Y.; Tian, Y.C.; Hung, C.C.; Fang, J.T.; Yang, C.W.; Chen, Y.C. Acute kidney injury enhances outcome prediction ability of sequential organ failure assessment score in critically ill patients. PLoS ONE 2014, 9, e109649. [Google Scholar] [CrossRef] [PubMed]
| All | 90-Day Survival | 90-Day Mortality | p | 90-Day Composite Outcome (−) | 90-Day Composite Outcome (+) | p | |
|---|---|---|---|---|---|---|---|
| (n = 257) | (n = 122) | (n = 135) | (n = 76) | (n = 181) | |||
| Patient characteristics | |||||||
| Age | 65.7 ± 16.6 | 63.4 ± 16.0 | 67.8 ± 16.9 | 0.035 | 61.3 ± 17.5 | 67.6 ± 15.9 | 0.005 |
| Gender (male (%)) | 167 (65.0%) | 82 (67.2%) | 85 (63.0%) | 0.514 | 54 (71.1%) | 113 (62.4%) | 0.200 |
| Baseline creatinine (mg/dL) | 2.0 ± 1.6 | 2.5 ± 1.9 | 1.5 ± 1.1 | <0.001 | 1.8 ± 1.3 | 2.1 ± 1.7 | 0.220 |
| eGFR (MDRD) (mL/min/1.73 m2) | 55.6 ± 41.0 | 48.3 ± 44.2 | 62.2 ± 36.9 | 0.006 | 63.3 ± 47.6 | 52.3 ± 37.6 | 0.428 |
| Co-morbidities | |||||||
| Diabetes mellitus | 115 (44.7%) | 61 (50.0%) | 54 (40.0%) | 0.132 | 33 (43.4%) | 82 (45.3%) | 0.891 |
| Cirrhosis | 9 (3.5%) | 3 (2.5%) | 6 (4.4%) | 0.505 | 2 (2.6%) | 7 (3.9%) | 0.999 |
| COPD | 15 (5.8%) | 5 (4.1%) | 10 (7.4%) | 0.297 | 5 (6.6%) | 10 (5.5%) | 0.777 |
| CAD | 54 (21.0%) | 24 (19.7%) | 30 (22.2%) | 0.648 | 18 (23.7%) | 36 (19.9%) | 0.505 |
| CVA | 24 (9.3%) | 9 (7.4%) | 15 (11.1%) | 0.392 | 4 (5.3%) | 20 (11.0%) | 0.166 |
| Congestive heart failure | 0.683 | 0.780 | |||||
| 0 | 67 (26.1%) | 33 (27.0%) | 34 (25.2%) | 19 (25.0%) | 48 (26.5%) | ||
| I | 100 (38.9%) | 43 (35.2%) | 57 (42.2%) | 28 (36.8%) | 72 (39.8%) | ||
| II | 51 (19.8%) | 24 (19.7%) | 27 (20.0%) | 14 (18.4%) | 37 (20.4%) | ||
| III | 31 (12.1%) | 17 (13.9%) | 14 (10.4%) | 12 (15.8%) | 19 (10.5%) | ||
| Laboratory data at ICU admission | |||||||
| BUN (mg/dL) | 48.0 ± 33.5 | 58.1 ± 34.5 | 38.9 ± 29.9 | <0.001 | 48.9 ± 36.6 | 47.7 ± 32.2 | 0.783 |
| pH | 7.4 ± 0.1 | 7.4 ± 0.8 | 7.4 ± 0.1 | 0.659 | 7.4 ± 0.1 | 7.4 ± 0.1 | 0.612 |
| FiO2 | 0.5 ± 0.2 | 0.5 ± 0.2 | 0.5 ± 0.2 | 0.916 | 5 ± 0.2 | 0.5 ± 0.2 | 0.218 |
| SBP (mmHg) | 121.0 ± 28.4 | 129.8 ± 28.8 | 113.0 ± 25.6 | <0.001 | 126.0 ± 25.6 | 118.8 ± 29.3 | 0.063 |
| GCS | 11.9 ± 4.2 | 12.3 ± 4.0 | 11.6 ± 4.4 | 0.164 | 11.9 ± 4.1 | 11.9 ± 4.3 | 0.948 |
| SOFA | 8.9 ± 3.5 | 8.3 ± 3.1 | 9.5 ± 3.7 | 0.008 | 8.7 ± 3.4 | 9.1 ± 3.6 | 0.410 |
| APACHE II | 16.3 ± 6.2 | 15.6 ± 6.0 | 9.5 ± 3.8 | 0.094 | 15.0 ± 6.4 | 16.9 ± 6.0 | 0.025 |
| MODS | 5.9 ± 3.7 | 5.5 ± 3.4 | 6.4 ± 3.8 | 0.040 | 5.7 ± 3.3 | 6.0 ± 3.8 | 0.507 |
| Etiology of AKI | |||||||
| Shock | 150 (58.4%) | 56 (5.9%) | 94 (69.6) | <0.001 | 40 (52.6%) | 110 (60.8%) | 0.268 |
| Sepsis | 98 (38.1%) | 26 (23.8%) | 69 (51.1%) | <0.001 | 22 (28.9%) | 76 (42.0%) | 0.067 |
| Drug-induced | 3 (1.2%) | 0 (0%) | 3 (2.2%) | 0.249 | 0 (0%) | 3 (1.7%) | 0.557 |
| Rhabdomyolysis | 9 (3.5%) | 5 (4.1%) | 4 (3.0%) | 0.740 | 4 (5.3%) | 5 (2.8%) | 0.457 |
| Pigmentation | 6 (2.3%) | 4 (3.3%) | 2 (1.5%) | 0.427 | 4 (5.3%) | 2 (1.1%) | 0.065 |
| Contrast | 37 (14.4%) | 22 (18.0%) | 15 (11.1%) | 0.154 | 13 (17.1%) | 24 (13.3%) | 0.440 |
| Other | 26 (10.1%) | 16 (13.1%) | 10 (7.4%) | 0.150 | 7 (9.2%) | 19 (10.5%) | 0.825 |
| At initiating dialysis | |||||||
| Admission to dialysis (days) | 40.3 ± 27.1 | 42.0 ± 31.8 | 37.1 ± 47.5 | 0.335 | 45.8 ± 33.9 | 36.8 ± 43.1 | 0.106 |
| Mechanical Ventilation | 185 (72.0%) | 74 (60.7%) | 111 (82.2%) | <0.001 | 49 (64.5%) | 136 (75.1%) | 0.095 |
| Emergency Surgery | 100 (38.9%) | 49 (40.2%) | 51 (37.8%) | 0.703 | 33 (43.4%) | 67 (37.0%) | 0.400 |
| IABP | 27 (10.5%) | 10 (8.2%) | 17 (12.6%) | 0.310 | 7 (9.2%) | 20 (11.0%) | 0.824 |
| Urine output (mL/24 h) | 591.7 ± 790.3 | 750.3 ± 1013.0 | 448.3 ± 472.1 | 0.002 | 869.7 ± 1188.7 | 474.9 ± 503.1 | <0.001 |
| AKI risk prediction score | 22.6 ± 6.9 | 20.2 ± 6.5 | 24.9 ± 6.5 | <0.001 | 20.8 ± 6.4 | 23.4 ± 7.0 | 0.004 |
| Body weight (kg) | 66.8 ± 14.3 | 68.6 ± 15.9 | 67.8 ± 16.9 | 0.055 | 70.0 ± 15.9 | 65.5 ± 13.4 | 0.021 |
| IE | 8.2 ± 15.0 | 4.7 ± 8.3 | 11.3 ± 18.7 | <0.001 | 5.24 ± 9.32 | 9.43 ± 16.75 | 0.041 |
| SOFA | 10.9 ± 3.9 | 9.1 ± 3.2 | 12.6 ± 3.8 | <0.001 | 9.4 ± 3.3 | 11.6 ± 4.0 | <0.001 |
| APACHE II | 17.8 ± 6.4 | 15.6 ± 5.4 | 19.8 ± 6.7 | <0.001 | 15.5 ± 5.7 | 18.7 ± 6.5 | <0.001 |
| MODS | 8.1 ± 4.1 | 6.5 ± 3.7 | 9.5 ± 3.9 | <0.001 | 7.0 ± 3.6 | 8.6 ± 4.2 | 0.005 |
| Phosphate (mg/dL) | 4.5 ± 1.7 | 4.8 ± 1.6 | 4.3 ± 1.8 | 0.085 | 4.8 ± 1.5 | 4.4 ± 1.8 | 0.333 |
| 25 OH Vit D, ng/mL | 11.7 ± 5.6 | 10.8 ± 5.5 | 12.9 ± 5.9 | 0.471 | 11.2 ± 7.1 | 12.0 ± 5.2 | 0.812 |
| 1,25 diOH Vit D, pg/mL | 27.3 ± 6.5 | 25.5 ± 6.4 | 29.7 ± 6.4 | 0.545 | 28.9 ± 6.8 | 26.6 ± 6.6 | 0.545 |
| Kidney function marker | |||||||
| BUN (mg/dL) | 82.4 ± 47.2 | 82.7 ± 51.5 | 82.5 ± 45.4 | 0.922 | 82.3 ± 51.5 | 82.5 ± 45.4 | 0.978 |
| Creatinine (mg/dL) | 2.0 ± 1.6 | 4.1 ± 2.2 | 4.2 ± 2.4 | 0.745 | 4.1 ± 2.2 | 4.2 ± 2.4 | 0.745 |
| Urine NGAL (ng/mL) | 197.5 ± 85.3 | 191.0 ± 93.3 | 203.5 ± 77.1 | 0.254 | 189.2 ± 97.6 | 201.0 ± 79.7 | 0.330 |
| Urine NGAL/Cre | 6.9 ± 11.1 | 6.8 ± 12.5 | 6.9 ± 9.7 | 0.912 | 5.0 ± 6.9 | 7.7 ± 12.4 | 0.085 |
| Urine KIM1 (ng/mL) | 6.0 ± 5.8 | 5.8 ± 5.8 | 6.2 ± 5.8 | 0.529 | 5.9 ± 6.5 | 5.7 ± 5.4 | 0.139 |
| Urine KIM1/Cre | 0.1 ± 0.2 | 0.1 ± 0.2 | 0.1 ± 0.1 | 0.993 | 0.1 ± 0.1 | 0.1 ± 0.2 | 0.699 |
| Urine cFGF-23/Cre | 877.4 ± 994.3 | 671.4 ± 924.9 | 1063.5 ± 1021.2 | <0.001 | 699.1 ± 1015.0 | 952.2 ± 978.6 | 0.062 |
| Plasma iFGF-23 (pg/mL) | 304.2 ± 468.0 | 395.1 ± 635.6 | 269.0 ± 385.2 | 0.265 | 320.4 ± 551.8 | 300.2 ± 449.5 | 0.875 |
| Plasma cFGF-23 (RU/mL) | 2630.1 ± 2259.5 | 1926.7 ± 1745.4 | 3265.9 ± 2479.0 | <0.001 | 1925.3 ± 1917.3 | 2926.1 ± 2330.0 | 0.001 |
| Indication for dialysis | |||||||
| Azotemia | 123 (47.9%) | 58 (47.5%) | 65 (48.1%) | 0.999 | 32 (42.1%) | 91 (50.3%) | 0.274 |
| Fluid overload | 111 (43.2%) | 51 (41.8%) | 60 (44.4%) | 0.706 | 30 (39.5%) | 81 (44.8%) | 0.491 |
| Electrolyte disorders | 18 (7.0%) | 10 (8.2%) | 18 (5.9%) | 0.626 | 7 (9.2%) | 11 (6.1%) | 0.423 |
| Metabolic acidosis | 46 (17.9%) | 17 (13.9%) | 29 (21.5%) | 0.143 | 11 (14.5%) | 35 (19.3%) | 0.380 |
| Oliguria | 166 (64.6%) | 69 (56.6%) | 97 (71.9%) | 0.013 | 46 (56.6%) | 123 (68.0%) | 0.088 |
| Uremic encephalopathy | 12 (4.7%) | 9 (7.4%) | 3 (2.2%) | 0.074 | 6 (7.9%) | 6 (3.3%) | 0.191 |
| Dialysis modality | |||||||
| CVVH | 62 (21.1%) | 16 (13.1) | 46 (34.1%) | <0.001 | 15 (19.7%) | 47 (26.0%) | 0.296 |
| IHD | 62 (29.2%) | 47 (38.5%) | 28 (20.7%) | 27 (35.5%) | 48 (26.5%) | ||
| SLED | 120 (46.7%) | 59 (48.5%) | 61 (45.2%) | 34 (44.7%) | 86 (47.5%) | ||
| Relevant outcome parameters | |||||||
| Hospital length of stay (days) | 54.7 ± 50.4 | 52.3 ± 41.1 | 56.9 ± 57.6 | 0.459 | 59.0 ± 46.3 | 52.9 ± 52.0 | 0.383 |
| Duration of hospital dialysis (days) | 82.4 ± 60.7 | 42.0 ± 31.8 | 37.1 ± 47.5 | 0.335 | 45.8 ± 33.9 | 36.8 ± 43.1 | 0.745 |
| Serum cFGF23 Categories | Low cFGF-23 | High cFGF-23 | p |
|---|---|---|---|
| (n = 116) | (n = 141) | ||
| Patient characteristics | |||
| Age (years) | 65.8 ± 16.0 | 65.7 ± 17.2 | 0.973 |
| Gender (male) | 77 (66.4%) | 90 (63.8%) | 0.695 |
| Baseline creatinine (mg/dL) | 2.2 ± 1.9 | 1.8 ± 1.3 | 0.039 |
| eGFR (MDRD) (mL/min/1.73 m2) | 54.4 ± 42.5 | 56.5 ± 39.8 | 0.690 |
| Comorbidities | |||
| Diabetes mellitus | 33 (43.4%) | 82 (45.3%) | 0.891 |
| Cirrhosis | 1 (0.9%) | 8 (5.7%) | 0.044 |
| COPD | 7 (6.0%) | 8 (5.7%) | 0.999 |
| CAD | 27 (23.3%) | 27 (19.1%) | 0.445 |
| CVA | 12 (10.3%) | 12 (8.5%) | 0.670 |
| Congestive heart failure | 0.265 | ||
| 0 | 32 (27.6%) | 35 (24.8%) | |
| I | 37 (31.9%) | 63 (44.7%) | |
| II | 27 (23.3%) | 24 (17.0%) | |
| III | 15 (12.9%) | 16 (11.3%) | |
| IV | 0 (0%) | 8 (5.5%) | |
| Laboratory data at ICU admission | |||
| BUN (mg/dL) | 48.3 ± 36.1 | 47.8 ± 31.4 | 0.897 |
| pH | 7.4 ± 0.1 | 7.4 ± 0.1 | 0.354 |
| FiO2 | 0.5 ± 0.2 | 0.5 ± 0.2 | 0.609 |
| SBP | 126.1 ± 29.2 | 116.7 ± 27.0 | 0.008 |
| GCS | 11.9 ± 4.3 | 11.9 ± 4.2 | 0.984 |
| SOFA | 8.2 ± 3.6 | 9.6 ± 3.3 | 0.001 |
| APACHE II | 15.9 ± 6.0 | 16.6 ± 6.4 | 0.405 |
| MODS | 5.7 ± 3.5 | 6.1 ± 3.8 | 0.328 |
| Etiology of AKI | |||
| Shock | 66 (56.9%) | 84 (59.6%) | 0.704 |
| Sepsis | 40 (34.5%) | 58 (41.1%) | 0.303 |
| Rhabdomyolysis | 7 (6.0%) | 2 (1.4%) | 0.083 |
| Drug-induced | 2 (1.7%) | 1 (0.7%) | 0.591 |
| Pigmentation | 5 (4.3%) | 1 (0.7%) | 0.094 |
| Contrast | 17 (14.7%) | 20 (14.2%) | 0.999 |
| Others | 12 (10.3%) | 14 (9.9%) | 0.999 |
| At initiating dialysis | |||
| Admission to dialysis (days) | 35.5 ± 34.1 | 42.6 ± 45.4 | 0.163 |
| Mechanical ventilation | 78 (67.2%) | 107 (75.9%) | 0.128 |
| Emergency Surgery | 45 (38.8%) | 55 (39.0%) | 0.999 |
| IABP | 13 (11.2%) | 14 (9.9%) | 0.839 |
| Urine output (mL/24 h) | 650.9 ± 642.9 | 542.9 ± 892.8 | 0.277 |
| AKI risk prediction score | 21.5 ± 6.7 | 23.5 ± 6.9 | 0.021 |
| Body weight (kg) | 67.2 ± 15.4 | 66.5 ± 13.3 | 0.728 |
| IE | 7.14 ± 11.5 | 9.1 ± 17.4 | 0.310 |
| SOFA | 10.6 ± 4.3 | 11.2 ± 3.6 | 0.218 |
| APACHE II | 17.8 ± 6.7 | 11.8 ± 6.2 | 0.980 |
| MODS | 7.8 ± 4.4 | 8.4 ± 3.8 | 0.222 |
| Phosphate, mg/dL | 4.1 ± 1.7 | 4.9 ± 1.7 | 0.021 |
| 25 OH Vit D, ng/mL | 11.0 ± 5.8 | 12.5 ± 5.6 | 0.617 |
| 1,25 diOH Vit D, pg/mL | 29.7 ± 6.9 | 25.0 ± 5.5 | 0.149 |
| Kidney function marker | |||
| BUN (mg/dL) | 81.2 ± 45.8 | 83.4 ± 48.4 | 0.714 |
| Creatinine (mg/dL) | 4.2 ± 2.4 | 4.1 ± 2.3 | 0.677 |
| Urine KIM1 (ng/mL) | 5.9 ± 5.9 | 6.1 ± 5.7 | 0.800 |
| Urine KIM1/Cre | 0.13 ± 0.18 | 0.14 ± 0.14 | 0.715 |
| Urine NGAL (ng/mL) | 196.5 ± 86.1 | 198.2 ± 85.0 | 0.916 |
| Urine NGAL/Cre | 7.0 12.9 | 6.8 ± 9.4 | 0.877 |
| Urine cFGF-23/Cre | 523.4 ± 747.2 | 1173.3 ± 1077.6 | <0.001 |
| Plasma iFGF-23 (pg/mL) | 257.6 ± 243.0 | 325.50 ± 542.3 | 0.536 |
| Indication for dialysis | |||
| Azotemia | 56 (48.3%) | 67 (47.5%) | 0.999 |
| Fluid overload | 48 (41.4%) | 63 (44.7%) | 0.615 |
| Electrolyte disorders | 7 (60%) | 11 (7.8%) | 0.631 |
| Metabolic acidosis | 22 (19.0%) | 24 (17.0%) | 0.745 |
| Oliguria | 73 (62.9%) | 93 (66.0%) | 0.694 |
| Uremic complication | 7 (6.0%) | 5 (3.5%) | 0.386 |
| Dialysis modality | 0.011 | ||
| CVVH | 44 (37.9%) | 31 (22.0%) | |
| IHD | 21 (18.1%) | 41 (29.1%) | |
| SLED | 51 (44.0%) | 69 (48.9%) | |
| Outcomes of interest | |||
| Hospital length of stay (days) | 49.0 ± 43.0 | 59.4 ± 55.5 | 0.101 |
| Duration of hospital dialysis (days) | 39.9 ± 34.4 | 39.0 ± 45.5 | 0.862 |
| Hospital mortality | 42 (36.2%) | 82 (58.2%) | <0.001 |
| Composite outcome at discharge | 69 (59.5%) | 104 (73.8%) | <0.001 |
| 90-day mortality | 45 (38.8%) | 90 (63.8%) | <0.001 |
| 90-day weaning from dialysis | 47 (40.5%) | 29 (20.6%) | <0.001 |
| 90-day composite outcome | 69 (59.5%) | 112 (79.4%) | <0.001 |
| Independent Variables | Hospital Mortality | Composite Outcome at Discharge | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p | OR | 95% CI | p | |
| Age (per year) | 1.03 | 1.01–1.04 | 0.007 | 1.03 | 1.01–1.04 | 0.004 |
| SOFA (per score) | 1.26 | 1.15–1.39 | <0.001 | 1.12 | 1.03–1.22 | 0.011 |
| High cFGF-23 | 1.80 | 1.01–3.24 | 0.043 | 1.80 | 1.01–3.19 | 0.045 |
| 90-Day Mortality | 90-Day Composite Outcome | |||||
| Age (per year) | 1.03 | 1.01–1.05 | 0.001 | 1.03 | 1.01–1.05 | 0.001 |
| SOFA (per score) | 1.30 | 1.17–1.44 | 0.037 | 1.17 | 1.07–1.27 | <0.001 |
| High cFGF-23 | 2.19 | 1.20–4.00 | 0.011 | 2.39 | 1.31–4.35 | 0.005 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, V.-C.; Shiao, C.-C.; Chi, N.-H.; Wang, C.-H.; Chueh, S.-C.J.; Liou, H.-H.; Spapen, H.D.; Honore, P.M.; Chu, T.-S. Outcome Prediction of Acute Kidney Injury Biomarkers at Initiation of Dialysis in Critical Units. J. Clin. Med. 2018, 7, 202. https://doi.org/10.3390/jcm7080202
Wu V-C, Shiao C-C, Chi N-H, Wang C-H, Chueh S-CJ, Liou H-H, Spapen HD, Honore PM, Chu T-S. Outcome Prediction of Acute Kidney Injury Biomarkers at Initiation of Dialysis in Critical Units. Journal of Clinical Medicine. 2018; 7(8):202. https://doi.org/10.3390/jcm7080202
Chicago/Turabian StyleWu, Vin-Cent, Chih-Chung Shiao, Nai-Hsin Chi, Chih-Hsien Wang, Shih-Chieh Jeff Chueh, Hung-Hsiang Liou, Herbert D. Spapen, Patrick M. Honore, and Tzong-Shinn Chu. 2018. "Outcome Prediction of Acute Kidney Injury Biomarkers at Initiation of Dialysis in Critical Units" Journal of Clinical Medicine 7, no. 8: 202. https://doi.org/10.3390/jcm7080202
APA StyleWu, V.-C., Shiao, C.-C., Chi, N.-H., Wang, C.-H., Chueh, S.-C. J., Liou, H.-H., Spapen, H. D., Honore, P. M., & Chu, T.-S. (2018). Outcome Prediction of Acute Kidney Injury Biomarkers at Initiation of Dialysis in Critical Units. Journal of Clinical Medicine, 7(8), 202. https://doi.org/10.3390/jcm7080202






