Bullying at Workplace and Brain-Imaging Correlates
Abstract
:1. Introduction
2. Subjects and Methods
2.1. Volumetric Analysis with Manual Segmentation of the Hippocampus
2.2. Semiautomatic Volumetric Analysis with SIENAX
2.3. Automatic Volumetric Analysis with VBM
2.4. Statistics
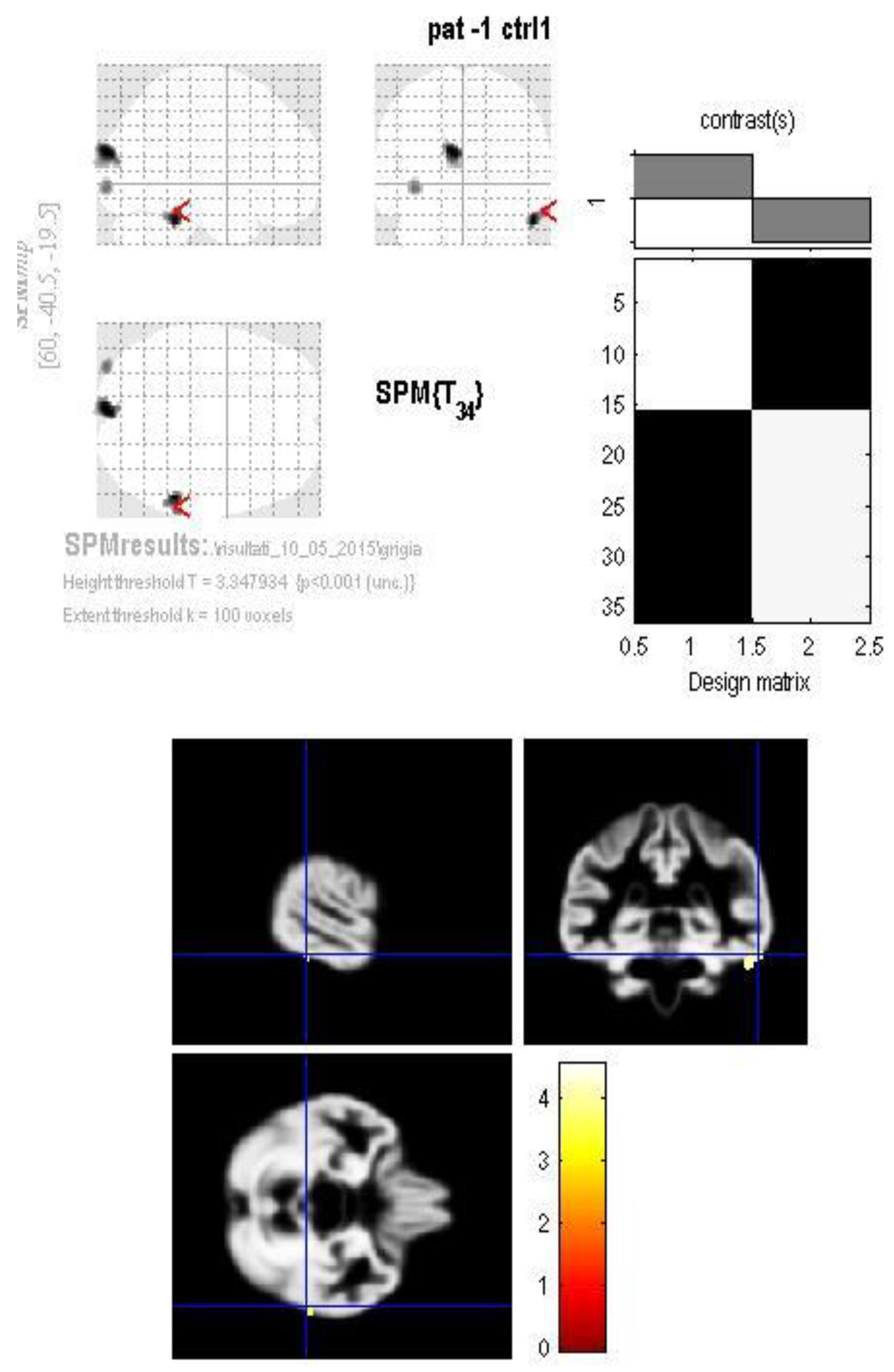
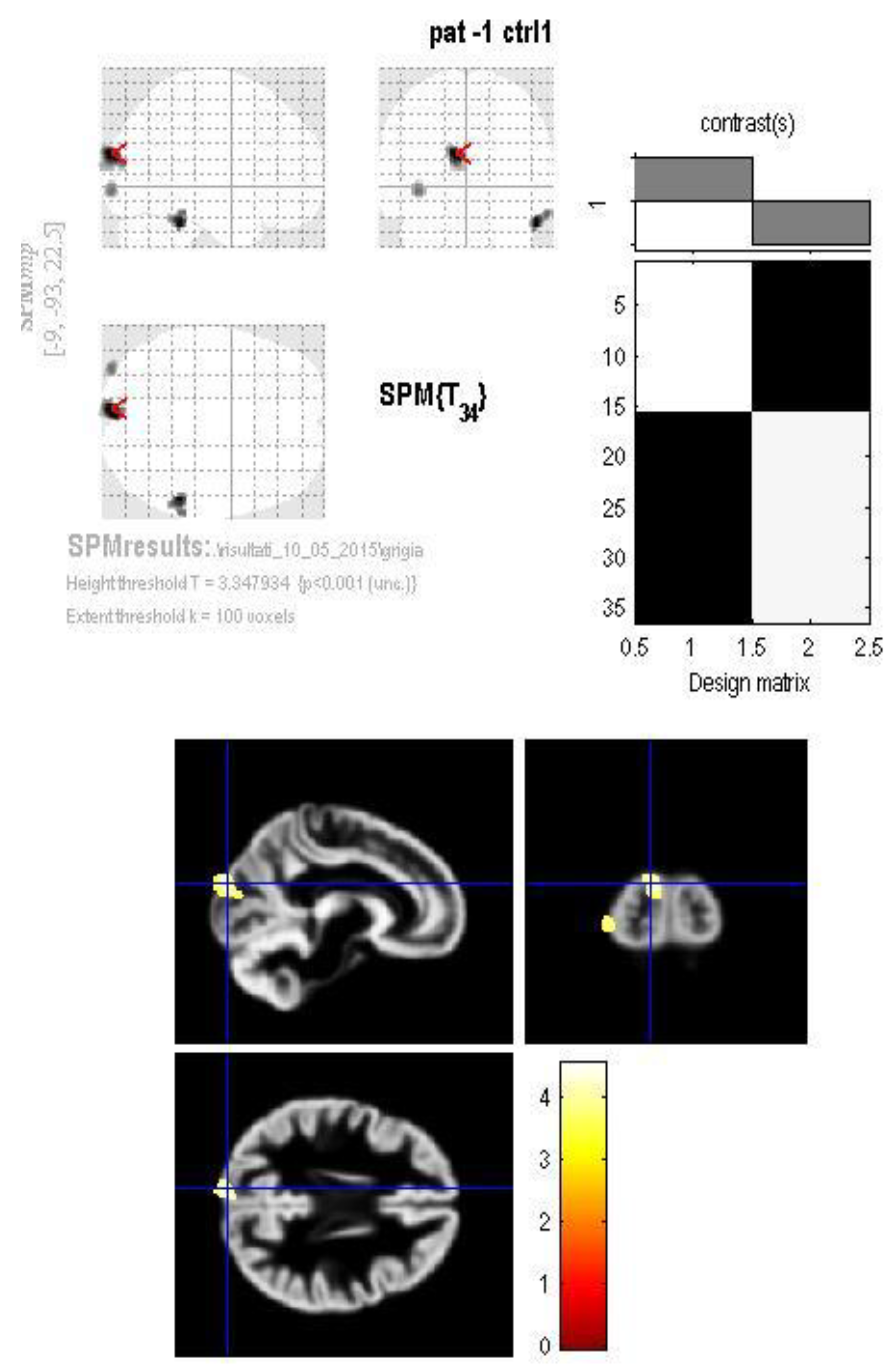
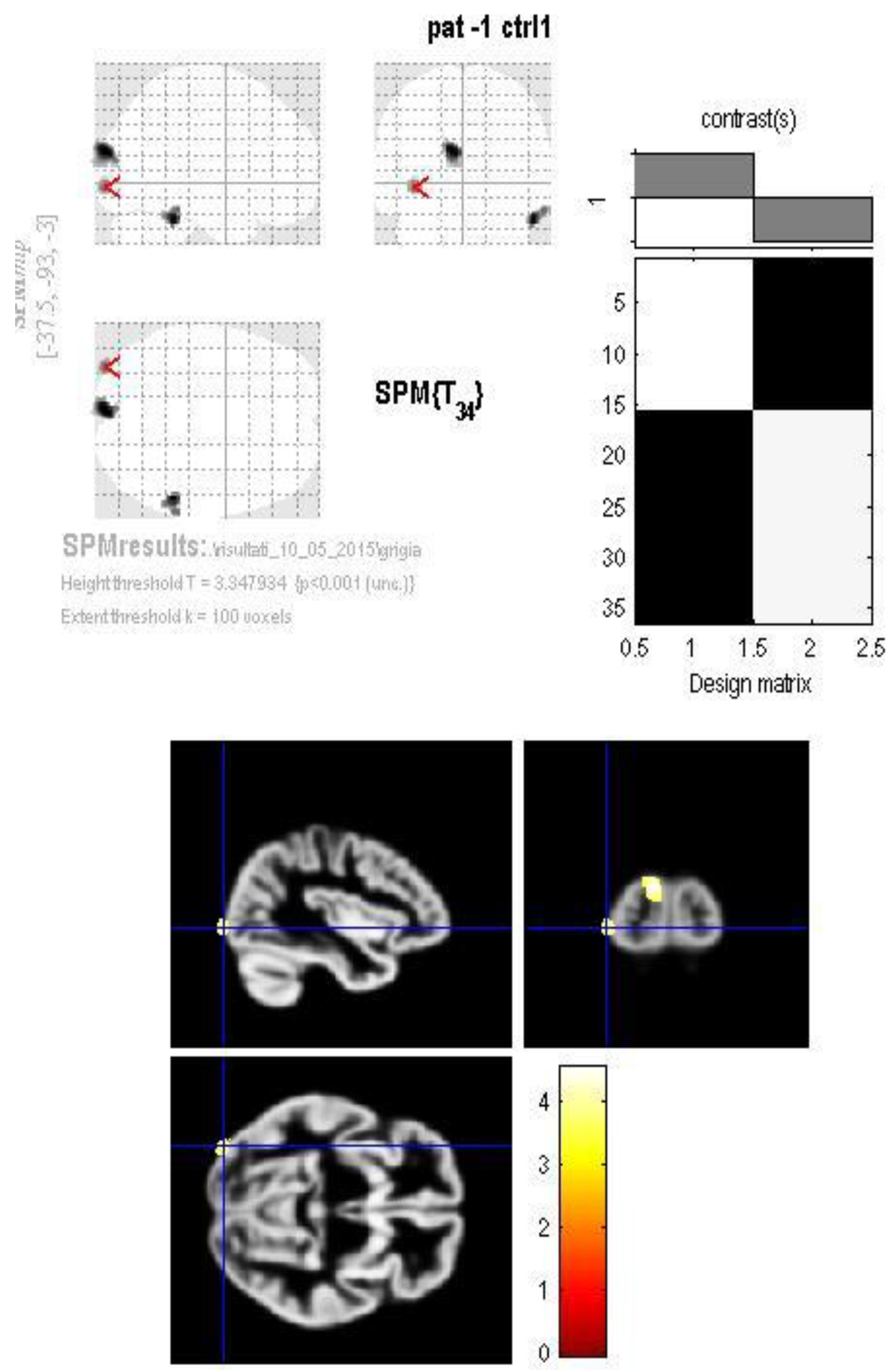
3. Results
4. Discussion
5. Limitations
- (1)
- The rather small sample of the subjects investigated is a significant limitation of the study. Therefore, even though the sensitivity was enhanced by the use of a homogenous study group, is difficult to generalize the results.
- (2)
- The cross-sectional method of this study makes difficult to demonstrate conclusively whether this brain characteristics are cause o consequence of work-related bullying.
- (3)
- Patients did not undergo neuropsychological assessment since cognitive impairment was in the exclusion criteria; furthermore, although some patients referred subjective cognitive complaints, these were neither confirmed by informants nor supported by impaired performances at screening tools. However, we strongly believe that further studies could use neuropsychological evaluations to better substantiate the hypothesis of work-related abnormalities of the semantic memory. Follow-up studies are also needed to evaluate if the nature of these brain-imaging correlates is reversible or not.
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Stansfeld, S.; Candy, B. Psychosocial work environment and mental health—A meta-analytic review. Scand. J. Work Environ. Health 2006, 32, 443–462. [Google Scholar] [CrossRef] [PubMed]
- Savić, C.; Belkić, K. Why are job stressors relevant for psychiatry? Br. J. Psychiatry 2014, 205, 425–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrie, J.E.; Head, J.; Shipley, M.J.; Vahtera, J.; Marmot, M.G.; Kivimaki, M. Injustice at work and incidence of psychiatric morbidity: The Whitehall II study. Occup. Environ. Med. 2006, 63, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Nolfe, G.; Petrella, C.; Blasi, F.; Zontini, G.; Nolfe, G. Psychopathological dimensions of harassment in the workplace (mobbing). Int. J. Ment. Health 2008, 36, 70–88. [Google Scholar] [CrossRef]
- Bamberger, S.G.; Vinding, A.L.; Larsen, A.; Nielsen, P.; Fonager, K.; Nielsen, R.N.; Ryom, P.; Omland, Ø. Impact of organisational change on mental health: A systematic review. Occup. Environ. Med. 2012, 69, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Nolfe, G.; Mancini, P.; Mancusi, R.; Zontini, G.; Nolfe, G. Work-related psychopathology: Rates in different work activities and relationship between subjective perception of work distress and psychiatric disturbances. Work 2014, 47, 501–508. [Google Scholar] [PubMed]
- Wang, H.X.; Wahlberg, M.; Karp, A.; Winblad, B.; Fratiglioni, L. Psychosocial stress at work is associated with increased dementia risk in late life. Alzheimers Dement. 2012, 8, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Andel, R.; Crowe, M.; Hahn, E.A. Work-Related Stress May Increase the Risk of Vascular Dementia. J. Am. Geriatr. Soc. 2012, 60, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Blix, E.; Perski, A.; Berglund, H.; Savic, I. Long Term Occupational Stress Is Associated with Regional Reductions in Brain Tissue Volumes. PLoS ONE 2013, 8, e64065. [Google Scholar] [CrossRef] [PubMed]
- Monteleone, P.; Nolfe, G.; Serritella, C.; Milano, V.; Di Cerbo, A.; Blasi, F.; Petrella, C.; Maj, M. Hypoactivity of the hypothalamo-pituitary-adrenal axis in victims of mobbing: Role of subjects’ temperament and chronicity of the work-related psychological distress. Psychother. Psychosom. 2009, 78, 381–383. [Google Scholar] [CrossRef] [PubMed]
- Wahlberg, K.; Ghatan, P.H.; Modell, S.; Nygren, A.; Inqvar, M.; Asberg, M.; Heilig, M. Suppressed neuroendocrine stress response in depressed women on job-stress-related long-term sick leave: A stable marker potentially suggestive of preexisting vulnerability. Biol. Psychiatry 2009, 65, 742–747. [Google Scholar] [CrossRef] [PubMed]
- Heim, C.; Ehlert, U.; Hellhammer, D.H. The potential role of hypocortisolism in the pathophysiology of stress-related bodily disorders. Psychoneuroendocrinology 2000, 25, 1–35. [Google Scholar] [CrossRef]
- Dedovic, K.; D’Aguiar, C.; Pruessner, J.C. What stress does to your brain: A review of neuroimaging studies. Can. J. Psychiatry 2009, 54, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.M.; Jerram, M.; Abbs, B.; Whitfield-Gabrieli, S.; Makris, N. Sex differences in stress response circuitry activation dependent on female hormonal cycle. J. Neurosci. 2010, 30, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, M.L.; Brachman, R.A.; Martinowich, K.; Schloesser, R.J.; Herkenham, M. Glucocorticoids orchestrate divergent effects on mood through adult neurogenesis. J. Neurosci. 2013, 33, 2961–2972. [Google Scholar] [CrossRef] [PubMed]
- Anacher, C. Adult hippocampal neurogenesis in depression: Behavioral implications and regulation by the stress system. Curr. Top. Behav. Neurosci. 2014, 18, 25–43. [Google Scholar]
- Jovanovic, H.; Perski, A.; Berglund, H.; Savic, I. Chronic stress is linked to 5-HT (1A) receptor changes and functional disintegration of the limbic networks. Neuroimage 2011, 55, 1178–1188. [Google Scholar] [CrossRef] [PubMed]
- Golkar, A.; Johansson, E.; Kasahara, M.; Osika, W.; Perski, A.; Savic, I. The influence of work-related stress on the regulation of emotion and on functional connectivity in the brain. PLoS ONE 2014, 9, e104550. [Google Scholar] [CrossRef] [PubMed]
- Cohen, R.A.; Grieve, S.; Hoth, K.F.; Paul, R.H.; Sweet, L.; Tate, D.; Gunstad, J.; Stroud, L.; McCaffery, J.; Hitsman, B.; et al. Early life stress and morphometry of the adult anterior cingulate cortex and caudate nuclei. Biol. Psychiatry 2006, 59, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J.D. Neuroimaging in posttraumatic stress disorder and other stress-related disorders. Neuroimaging Clin. N. Am. 2007, 17, 523–538. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J.D.; Elzinga, B.; Schmahl, C.; Vermetten, E. Structural and functional plasticity of the human brain in posttraumatic stress disorder. Prog. Brain Res. 2008, 167, 171–186. [Google Scholar] [PubMed]
- Papagni, S.A.; Benetti, S.; Arulanantham, S.; McCrory, E.; McGuire, P.; Mechelli, A. Effects of stressful life events on human brain structure: A longitudinal voxelbased morphometry study. Stress 2011, 14, 227–232. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, M.C.; Yusel, K.; Nazarov, A.; MacQueen, G.M. A meta-analysis examining clinical predictors of hippocampal volume in patients with major depressive disorder. J. Psychiatry Neurosci. 2009, 31, 41–54. [Google Scholar]
- Leyman, H. Mobbing and psychological terror at workplace. Violence Vict. 1990, 5, 119–126. [Google Scholar]
- First, M.B.; Gibbon, M.; Spitzer, R.L.; Williams, J.B.W. Structural Clinical Interview for Axis I DSM-IV Disorders-Non-Patients; SCID-I /NP, Version 2.0; New York State Psychiatric Institute: New York, NY, USA, 2002. [Google Scholar]
- Hamilton, M. Diagnosis and rating of anxiety. Br. J. Psychiatry 1969, 3, 76–79. [Google Scholar]
- Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 1960, 23, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Holmes, T.H.; Rahe, R.H. The Social Readjustment Rating Scale. J. Psychosom. Res. 1967, 11, 213–218. [Google Scholar] [CrossRef]
- Nolfe, G.; Triassi, M.; Cappuccio, A.; Carbone, U.; Nolfe, G. The Naples-Questionnaire of Work Distress (nQ-WD): Bullying at workplace, organizational dysfunctions and biopsycho-social effects. Open J. Psychiatry 2012, 2, 77–81. [Google Scholar] [CrossRef]
- Frisoni, G.B.; Jack, C.R.; Bocchetta, M.; Bauer, C.; Frederiksen, K.S.; Liu, Y.; Preboske, G.; Swihart, T.; Blair, M.; Cavedo, E.; et al. The EADC-ADNI harmonized protocol for manual hippocampal segmentation on magnetic resonance: Evidence of validity. Alzheimers Dement. 2015, 11, 111–125. [Google Scholar] [CrossRef] [PubMed]
- De Stefano, N.; Iannucci, G.; Sormani, M.P.; Guidi, L.; Bortolazzi, M.L.; Comi, G.; Federico, A.; Filippi, M. MR correlates of cerebral atrophy in patients with multiple sclerosis. J. Neurol. 2002, 249, 1072–1077. [Google Scholar] [CrossRef] [PubMed]
- Ashburner, J.; Friston, K.J. Voxel-based morphometry—The methods. Neuroimage 2000, 11, 805–821. [Google Scholar] [CrossRef] [PubMed]
- Mazziotta, J.; Toga, A.; Evans, A.; Lancaster, J.; Zilles, K.; Woods, R.; Paus, T.; Simpson, G.; Pike, B.; Holmes, C.; et al. A probabilistic atlas and reference system for the human brain: International Consortium for Brain Mapping (ICBM). Philos. Trans. R. Soc. B Biol. Sci. 2001, 356, 1293–1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Acheson, D.T.; Gresack, J.E.; Risbrough, V.B. Hippocampal dysfunction effects on context memory: Possible etiology for post-traumatic stress disorder. Neuropharmacology 2012, 62, 674–685. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Heninger, G.R.; Nestler, E.J. A molecular and cellular theory of depression. Arch. Gen. Psychiatry 1997, 54, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Mohanakrishnan Menon, P.; Nasrallah, H.A.; Lyons, J.A.; Scott, M.F.; Liberto, V. Single-voxel proton MR spectroscopy of right versus left hippocampi in PTSD. Psychiatry Res. 2003, 123, 101–108. [Google Scholar] [CrossRef]
- O’Doherty, D.C.; Chitty, K.M.; Saddiqui, S.; Lagopoulos, J. A systematic review and meta-analysis of magnetic resonance measurement of structural volumes in posttraumatic stress disorder. Psychiatry Res. 2015, 232, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Sorrels, S.F.; Paredes, M.F.; Cebrian-Silla, A.; Sandoval, K.; Qi, D.; Kelley, K.W.; James, D.; Mayer, S.; Chang, J.; Auguste, K.I. Human hippocampal neurogenesis drops sharply in children to undetectable levels in adults. Nature 2018, 555, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Desimone, R.; Ungerieider, L.G. Neural mechanisms of visual processing in monkeys. In Handbook of Neuropsychology; Goodglass, H., Damasio, A.R., Eds.; Elsevier: Amsterdam, The Netherlands, 1989; pp. 267–300. [Google Scholar]
- Sheldon, S.; Farb, N.; Palombo, D.J.; Levine, B. Intrinsic medial temporal lobe connectivity relates to individual differences in episodic autobiographical remembering. Cortex 2016, 74, 206–216. [Google Scholar] [CrossRef] [PubMed]
| Brain Sienax (Patients) | Brain Sienax (Controls) | Right Hippocampus (Patients) | Right Hippocampus (Controls) | Left Hippocampus (Patients) | Left Hippocampus (Controls) | |
|---|---|---|---|---|---|---|
| 1,487,785.08 ± 70445.2 | 1,441,922.35 ± 75173.7 | 3441.5 ± 3193.9 | 5948.8 ± 3193.0 | 3202.9 ± 2795.9 | 6137.5 ± 3079.9 | |
| f value | 3.582 | 5.731 | 9.052 | |||
| p value | p = 0.0667 | p = 0.0222 | p = 0.0048 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nolfe, G.; Cirillo, M.; Iavarone, A.; Negro, A.; Garofalo, E.; Cotena, A.; Lazazzara, M.; Zontini, G.; Cirillo, S. Bullying at Workplace and Brain-Imaging Correlates. J. Clin. Med. 2018, 7, 200. https://doi.org/10.3390/jcm7080200
Nolfe G, Cirillo M, Iavarone A, Negro A, Garofalo E, Cotena A, Lazazzara M, Zontini G, Cirillo S. Bullying at Workplace and Brain-Imaging Correlates. Journal of Clinical Medicine. 2018; 7(8):200. https://doi.org/10.3390/jcm7080200
Chicago/Turabian StyleNolfe, Giovanni, Mario Cirillo, Alessandro Iavarone, Alberto Negro, Elisabetta Garofalo, Annisa Cotena, Massimo Lazazzara, Gemma Zontini, and Sossio Cirillo. 2018. "Bullying at Workplace and Brain-Imaging Correlates" Journal of Clinical Medicine 7, no. 8: 200. https://doi.org/10.3390/jcm7080200
APA StyleNolfe, G., Cirillo, M., Iavarone, A., Negro, A., Garofalo, E., Cotena, A., Lazazzara, M., Zontini, G., & Cirillo, S. (2018). Bullying at Workplace and Brain-Imaging Correlates. Journal of Clinical Medicine, 7(8), 200. https://doi.org/10.3390/jcm7080200






