The Impact and Mechanism of Methylated Metabotropic Glutamate Receptors 1 and 5 in the Hippocampus on Depression-Like Behavior in Prenatal Stress Offspring Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Animal Model Establishment
2.3. Drug Treatment
2.4. Sucrose Preference Test, SPT
2.5. Open Field Test, OFT
2.6. Forced Swimming Test, FST
2.7. Separation of the Hippocampus
2.8. Quantitative Real-Time RT-PCR
2.9. Western Blotting
2.10. Methylation Analysis by Bisulfite Sequencing PCR
2.11. Statistics
3. Result
3.1. 5-azaD Treatment Group Showed Significant Antidepressant Effect
3.2. mGluR1 and mGluR5 mRNA Expression in the Hippocampus
3.3. mGluR1 and mGluR5 Protein Expression in the Hippocampus
3.4. Promoter Methylation of mGluR1 Gene in the Hippocampus of Offspring
3.5. Promoter Methylation of mGluR5 Gene in the Hippocampus of Offspring
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cuijpers, P.; van Straten, A.; Andersson, G. Internet-administered cognitive behavior therapy for health problems: A systematic review. J. Behav. Med. 2008, 31, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Murakumi, J. Gender and depression Explaining the Different Rates of depression Between Men and Women. Available online: https://pdfs.semanticscholar.org/9dc8/ea74361dec8a28be2bbdc2f756d765a3f32f.pdf#page=28 (accessed on 17 May 2018).
- Ardoino, G.I.; Queirolo, E.I.; Barg, G.; Ciccariello, D.A.; Kordas, K. The relationship among depression, parenting stress, and partner support in low-income women from Montevideo, Uruguay. Health Care Women Int. 2015, 36, 392–408. [Google Scholar] [CrossRef] [PubMed]
- Markham, J.A.; Koenig, J.I. Prenatal stress: Role in psychotic and depressive diseases. Psychopharmacology 2011, 214, 89–106. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ma, Y.; Hu, J.; Cheng, W.; Jiang, H.; Zhang, X.; Li, M.; Ren, J.; Li, X. Prenatal chronic mild stress induces depression-like behavior and sex-specific changes in regional glutamate receptor expression patterns in adult rats. Neuroscience 2015, 301, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.A.; Dhami, P.; Feber, A.; Cortázar, D.; Suzuki, Y.; Schulz, R.; Schär, P.; Beck, S. Resources for methylome analysis suitable for gene knockout studies of potential epigenome modifiers. GigaScience 2012, 1, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Levine, B.; Robertson, I.H.; Clare, L.; Carter, G.; Hong, J.; Wilson, B.A.; Duncan, J.; Stuss, D.T. Rehabilitation of executive functioning: An experimental–clinical validation of Goal Management Training. J. Int. Neuropsychol. Soc. 2000, 6, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Musazzi, L.; Treccani, G.; Mallei, A.; Popoli, M. The action of antidepressants on the glutamate system: Regulation of glutamate release and glutamate receptors. Biol. Psychiatry 2013, 73, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Pitsikas, N. The metabotropic glutamate receptors: Potential drug targets for the treatment of anxiety disorders. Eur. J. Pharmacol. 2014, 723, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Harvey, B.H.; Shahid, M. Metabotropic and ionotropic glutamate receptors as neurobiological targets in anxiety and stress-related disorders: Focus on pharmacology and preclinical translational models. Pharmacol. Biochem. Behav. 2012, 100, 775–800. [Google Scholar] [CrossRef] [PubMed]
- Nicoletti, F.; Bockaert, J.; Collingridge, G.L.; Conn, P.J.; Ferraguti, F.; Schoepp, D.D.; Wroblewski, J.T.; Pin, J.P. Metabotropic glutamate receptors: From the workbench to the bedside. Neuropharmacology 2011, 60, 1017–1041. [Google Scholar] [CrossRef] [PubMed]
- Amalric, M. Targeting metabotropic glutamate receptors (mGluRs) in Parkinson’s disease. Curr. Opin. Pharmacol. 2015, 20, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Matrisciano, F.; Tueting, P.; Maccari, S.; Nicoletti, F.; Guidotti, A. Pharmacological activation of group-II metabotropic glutamate receptors corrects a schizophrenia-like phenotype induced by prenatal stress in mice. Neuropsychopharmacology 2012, 37, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Tavassoli, E.; Saboory, E.; Teshfam, M.; Rasmi, Y.; Roshan-Milani, S.; Ilkhanizadeh, B.; Hesari, A.K. Effect of prenatal stress on density of NMDA receptors in rat brain. Int. J. Dev. Neurosci. 2013, 31, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.M.; Huang, C.C.; Hsu, K.S. Prenatal stress alters hippocampal synaptic plasticity in young rat offspring through preventing the proteolytic conversion of pro-brain-derived neurotrophic factor (BDNF) to mature BDNF. J. Physiol. 2012, 590, 991–1010. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Sawa, A.; Iyoet, M.S. Increased levels ofglutamate in brains from patients with mood disorders. Biol. Psychiatry 2007, 62, 1310–1316. [Google Scholar] [CrossRef] [PubMed]
- Said, N.; Lakehayli, S.; Battas, O.; Hakkou, F.; Tazi, A. Effects of prenatal stress on anxiety-like behavior and nociceptive response in rats. J. Integr. Neurosci. 2015, 14, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Frye, C.A.; Wawrzycki, J. Effect of prenatal stress and gonadal hormone condition on depressive behaviors of female and male rats. Horm. Behav. 2003, 44, 319–326. [Google Scholar] [CrossRef]
- Bekku, N.; Yoshimura, H. Animal model of menopausal depressive-like state in female mice: Prolongation of immobility time in the forced swimming test following ovariectomy. Psychopharmacology 2005, 183, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Sun, X.L.; Wu, F.F.; Su, C.J.; Ding, J.H.; Hu, G. Female early adult depression results in detrimental impacts on the behavioral performance and brain development in offspring. CNS Neurosci. Ther. 2012, 18, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Biala, Y.N.; Bogoch, Y.; Bejar, C.; Linial, M.; Weinstock, M. Prenatal stress diminishes gender differences in behavior and in expression of hippocampal synaptic genes and proteins in rats. Hippocampus 2011, 21, 1114–1125. [Google Scholar] [CrossRef] [PubMed]
- Fumagalli, F.; Pasini, M.; Frasca, A.; Drago, F.; Racagni, G.; Riva, M.A. Prenatal stress alters glutamatergic system responsiveness in adult rat prefrontal cortex. J. Neurochem. 2009, 109, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Zuena, A.R.; Mairesse, J.; Casolini, P.; Cinque, C.; Alema, G.S.; Morley-Fletcher, S.; Chiodi, V.; Spagnoli, L.G.; Gradini, R.; Catalani, A.; et al. Prenatal restraint stress generates two distinct behavioral and neurochemical profiles in male and female rats. PLoS ONE 2008, 3, e2170. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.-Y.; Feng, S.; Joo, J.W.J.; Jacobsen, S.E.; Pellegrini, M. A comparative analysis of DNA methylation across human embryonic stem cell lines. Genome Biol. 2011, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Mav, D.; Grimm, S.A.; Jothi, R.; Shah, R.; Wade, P.A. Fine-tuning of epigenetic regulation with respect to promoter CpG content in a cell type-specific manner. Epigenetics 2014, 9, 747–759. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Useche, I.; Ke, J.; Tian, Y.; Shim, D.; Howell, S.C.; Qiu, X.; Yuan, C. DNA Methylation Regulated Nucleosome Dynamics. Sci. Rep. 2013, 3, 21. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.A. Functions of DNA methylation: Islands, start sites, gene bodies and beyond. Nat. Rev. Genet. 2012, 13, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.A.; Schade, R.; Terry, A.V., Jr. Variable prenatal stress results in impairments of sustained attention and inhibitory response control in a 5-choice serial reaction time task in rats. Neuroscience 2012, 218, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Strathdee, G.; Brown, R. Aberrant DNA methylation in cancer: Potential clinical interventions. Mol. Med. Rep. 2002, 4, 1–17. [Google Scholar] [CrossRef]
- Kahl, K.G.; Georgi, K.; Bleich, S.; Muschler, M.; Hillemacher, T.; Hilfiker-Kleinert, D.; Schweiger, U.; Ding, X.; Kotsiari, A.; Frieling, H. Altered DNA methylation of glucose transporter 1 and glucose transporter 4 in patients with major depressive disorder. J. Psychiatr. Res. 2016, 76, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Momparler, R.L. Epigenetic therapy of cancer with 5-aza-2′-deoxycytidine (decitabine). Semin. Oncol. 2005, 32, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Karahoca, M.; Momparler, R.L. Pharmacokinetic and pharmacodynamic analysis of 5-aza-2′-deoxycytidine (decitabine) in the design of its dose-schedule for cancer therapy. Clin. Epigenet. 2013, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Klengel, T.; Pape, J.; Binder, E.B.; Mehta, D. The role of DNA methylation in stress-related psychiatric disorders. Neuropharmacology 2014, 80, 115–132. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; Markou, A.; Lucki, I. Assessing antidepressant activity in rodents: Recent developments and future needs. Trends Pharmacol. Sci. 2002, 23, 238–245. [Google Scholar] [CrossRef]
- Morley-Fletcher, S.; Mairesse, J.; Maccari, S. Behavioural and Neuroendocrine Consequences of Prenatal Stress in Rat. Adapt. Maladapt. Asp. Dev. Stress 2012, 3, 175–193. [Google Scholar]
- Koehl, M.; Darnaudéry, M.; Dulluc, J.; Van Reeth, O.; Le Moal Maccari, M. Prenatal stress alters circadian activity of hypothalamo-pituitary-adrenal axis and hippocampal corticosteroid receptors in adult rats of both gender. Dev. Neurobiol. 2015, 40, 302–315. [Google Scholar] [CrossRef]
- Maccari, S.; Darnaudery, M.; Morley-Fletcher, S.; Zuena, A.R.; Cinque, C.; Van Reeth, O. Prenatal stress and long-term consequences: Implications of glucocorticoid hormones. Neurosci. Biobehav. Rev. 2003, 27, 119–127. [Google Scholar] [CrossRef]
- Scopinho, A.A.; Scopinho, M.; Lisboa, S.F.; Correa, F.M.; Guimarães, F.S.; Joca, S.R. Acute reversible inactivation of the ventral medial prefrontal cortex induces antidepressant-like effects in rats. Behav. Brain Res. 2010, 214, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates—The New Coronal Set; Elsevier: New York, NY, USA, 2004. [Google Scholar]
- Garcia, L.S.; Comim, C.M.; Valvassori, S.S.; Réus, G.Z.; Andreazza, A.C.; Stertz, L.; Fries, G.R.; Gavioli, E.C.; Kapczinski, F.; Quevedo, J. Chronic administration of ketamine elicits antidepressant-like effects in rats without affecting hippocampal brain-derived neurotrophic factor protein levels. Basic Clin. Pharmacol. Toxicol. 2008, 103, 502–506. [Google Scholar] [CrossRef] [PubMed]
- Dalla, C.; Antoniou, K.; Drossopoulou, G.; Xagoraris, M.; Kokras, N.; Sfikakis, A.; Papadopoulou-Daifoti, Z. Chronic mild stress impact: Are females more vulnerable. Neuroscience 2005, 135, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Grønli, J.; Murison, R.; Fiske, E.; Bjorvatn, B.; Sørensen, E.; Portas, C.M.; Ursin, R. Effects of chronic mild stress on sexual behavior, locomotor activity and consumption of sucrose and saccharine solutions. Physiol. Behav. 2005, 84, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Guan, L.; Zhu, Z.; Li, H. Reduced levels of NR1 and NR2A with depression-like behavior in different brain regions in prenatally stressed juvenile offspring. PLoS ONE 2013, 8, e81775. [Google Scholar] [CrossRef] [PubMed]
- Guan, L.; Jia, N.; Zhao, X.; Zhang, X.; Tang, G.; Yang, L.; Sun, H.; Wang, D.; Su, Q.; Song, Q.; et al. The involvement of ERK/CREB/Bcl-2 in depression-like behavior in prenatally stressed OR. Brain Res. Bull. 2013, 99, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sales, A.J.; Biojone, C.; Terceti, M.S.; Guimaraes, F.S.; Gomes, M.V.; Joca, S.R. Antidepressant-like effect induced by systemic and intra-hippocampal administration of DNA methylation inhibitors. Br. J. Pharmacol. 2011, 164, 1711–1721. [Google Scholar] [CrossRef] [PubMed]
- Smolders, I.; Clinckers, R.; Meurs, A.; De Bundel, D.; Portelli, J.; Ebinger, G.; Michotte, Y. Direct enhancement of hippocampal dopamine or serotonin levels as a pharmacodynamic measure of combined antidepressant-anticonvulsant action. Neuropharmacology 2008, 54, 1017–1028. [Google Scholar] [CrossRef] [PubMed]
- Pomierny-Chamio, L.; Poleszak, E.; Pilc, A.; Nowak, G. NMDA but not AMPA glutamatergic receptors are involved in the antidepressant-like activity of MTEP during the forecd swim test in mice. Pharmaol. Rep. 2010, 62, 1186–1190. [Google Scholar] [CrossRef]
- Chaki, S.; Ago, Y.; Palucha-Paniewiera, A.; Matrisciano, F.; Pilc, A. MGluR2/3 and mGluR5 receptors: Potential targets fornovel antidepressants. Neuropharmacology 2013, 66, 40–52. [Google Scholar] [CrossRef] [PubMed]
- Natsubori, T.; Inoue, H.; Abe, O.; Takano, Y.; Iwashiro, N.; Aoki, Y.; Koike, S.; Yahata, N.; Katsura, M.; Gonoi, W.; et al. Reduced frontal glutamate + glutamine and N-acetylaspartate levels in patients with chronic schizophrenia but not in those at clinical high risk for psychosis or with first-episode schizophrenia. Schizophr. Bull. 2014, 40, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Wierońska, J.M.; Brański, P.; Szewczyk, B.; Pałucha, A.; Papp, M.; Gruca, P.; Moryl, E.; Pilc, A. Changes in the expression of metabotropic glutamate receptor 5 (mGluR5) in the rat hippocampus in an animal model of depression. Pol. J. Pharmacol. 2001, 53, 659–662. [Google Scholar] [PubMed]
- Poncer, J.C. Shinozaki, H.; Miles, R. Dual modulation of synaptic inhibition by distinct metabotropic glutamate receptors in the rat hippocampus. J. Physiol. 1995, 485, 121–134. [Google Scholar] [CrossRef] [PubMed]
- Yuen, E.Y.; Liu, W.H.; Karatsoreos, I.N.; Feng, J.; McEwen, B.S.; Yan, Z. Acute stress enhances glutamatergic transmission in prefrontal cortex and facilitates working memory. Proc. Natl. Acad. Sci. USA 2009, 106, 14075–14079. [Google Scholar] [CrossRef] [PubMed]
- Qiu, D.; Knöpfel, T. Presynaptically expressed long-term depression at cerebellar parallel fiber synapses. Eur. J. Physiol. 2009, 457, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Morley-Fletcher, S.; Mairesse, J.; Soumier, A.; Banasr, M.; Fagioli, F.; Gabriel, C.; Mocaer, E.; Daszuta, A.; McEwen, B.; Nicoletti, F.; et al. agomelatine treatment corrects behavioral, cellular, and biochemical ab normalities induced by prenatal stress in rats. Psychopharmacology 2011, 217, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Attucci, S.; Carla, V.; Mannaioni, G.; Moroni, F. Activation of type 5 metabotropic glutamate receptors enhances NMDA responses in mice cortical wedges. Br. J. Pharmacol. 2011, 132, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.T.; Huang, F.L.; Hu, Z.L.; Zhang, W.J.; Qiao, X.Q.; Huang, Y.Q.; Dai, R.P.; Li, F.; Li, C.Q. Early-Life Social Isolation-Induced Depressive-Like Behavior in Rats Results in Microglial Activation and Neuronal Histone Methylation that Are Mitigated by Minocycline. Neurotox. Res. 2017, 31, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Nuyt, A.M.; Szyf, M. Developmental programming through epigenetic changes. Circ. Res. 2007, 100, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Van den Hove, D.L.; Kenis, G.; Brass, A.; Opstelten, R.; Rutten, B.P.; Bruschettini, M.; Blanco, C.E.; Lesch, K.P. Vulnerability versus resilience to prenatal stress in male and female rats; implications from gene expression profiles in the hippocampus and frontal cortex. Eur. Neuropsychopharmacol. 2013, 23, 1226–1246. [Google Scholar] [CrossRef] [PubMed]
- Mueller, B.R.; Bale, T.L. Sex-Specific Programming of Offspring Emotionality Following Stress Early in Pregnancy. J. Neurosci. 2008, 28, 9055–9065. [Google Scholar] [CrossRef] [PubMed]
- Rivera, R.M.; Stein, P.; Weaver, J.R.; Mager, J.; Schultz, R.M.; Bartolomei, M.S. Manipulations of mouse embryos prior to implantation result in aberrant expression of imprinted genes on day 9.5 of development. Hum. Mol. Genet. 2008, 17, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ogawa, M.; Wood, J.R.; Bartolomei, M.S.; Sammel, M.D.; Kusanovic, J.P.; Walsh, S.W.; Romero, R.; Strauss, J.F. Genetic and epigenetic mechanisms combine to control MMP1 expression and its association with preterm premature rupture of membranes. Hum. Mol. Genet. 2008, 17, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Maya Vetencourt, J.F.; Tiraboschi, E.; Spolidoro, M.; Castrén, E.; Maffei, L. Serotonin triggers a transient epigenetic mechanism that reinstates adult visual cortex plasticity in rats. Eur. J. Neurosci. 2011, 33, 49–57. [Google Scholar] [CrossRef] [PubMed]
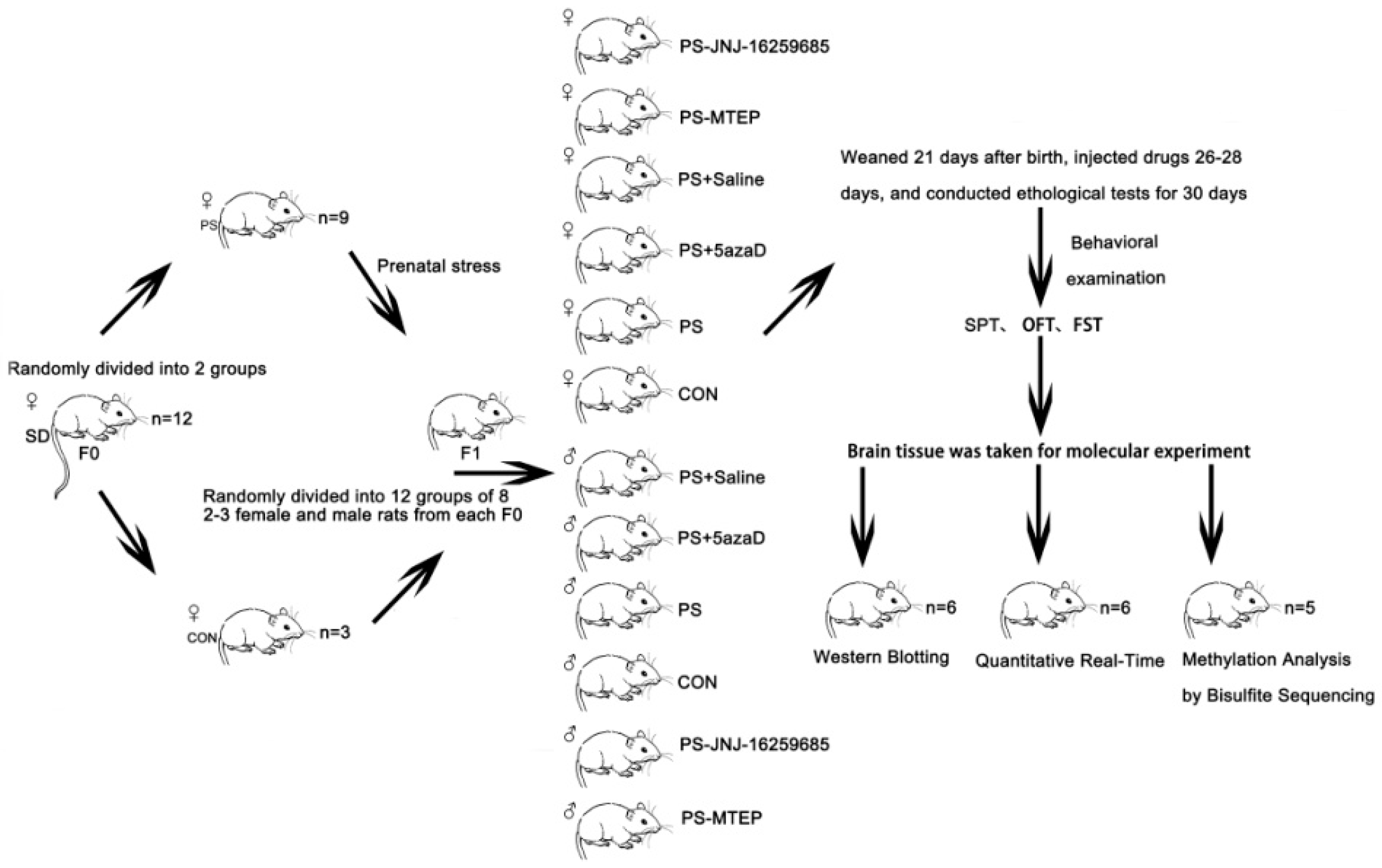
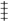 p < 0.001,
p < 0.001, 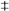 p < 0. 05 compare with PS.
p < 0. 05 compare with PS.
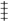 p < 0.001,
p < 0.001, 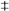 p < 0. 05 compare with PS.
p < 0. 05 compare with PS.
 p < 0.001 compare with PS.
p < 0.001 compare with PS.
 p < 0.001 compare with PS.
p < 0.001 compare with PS.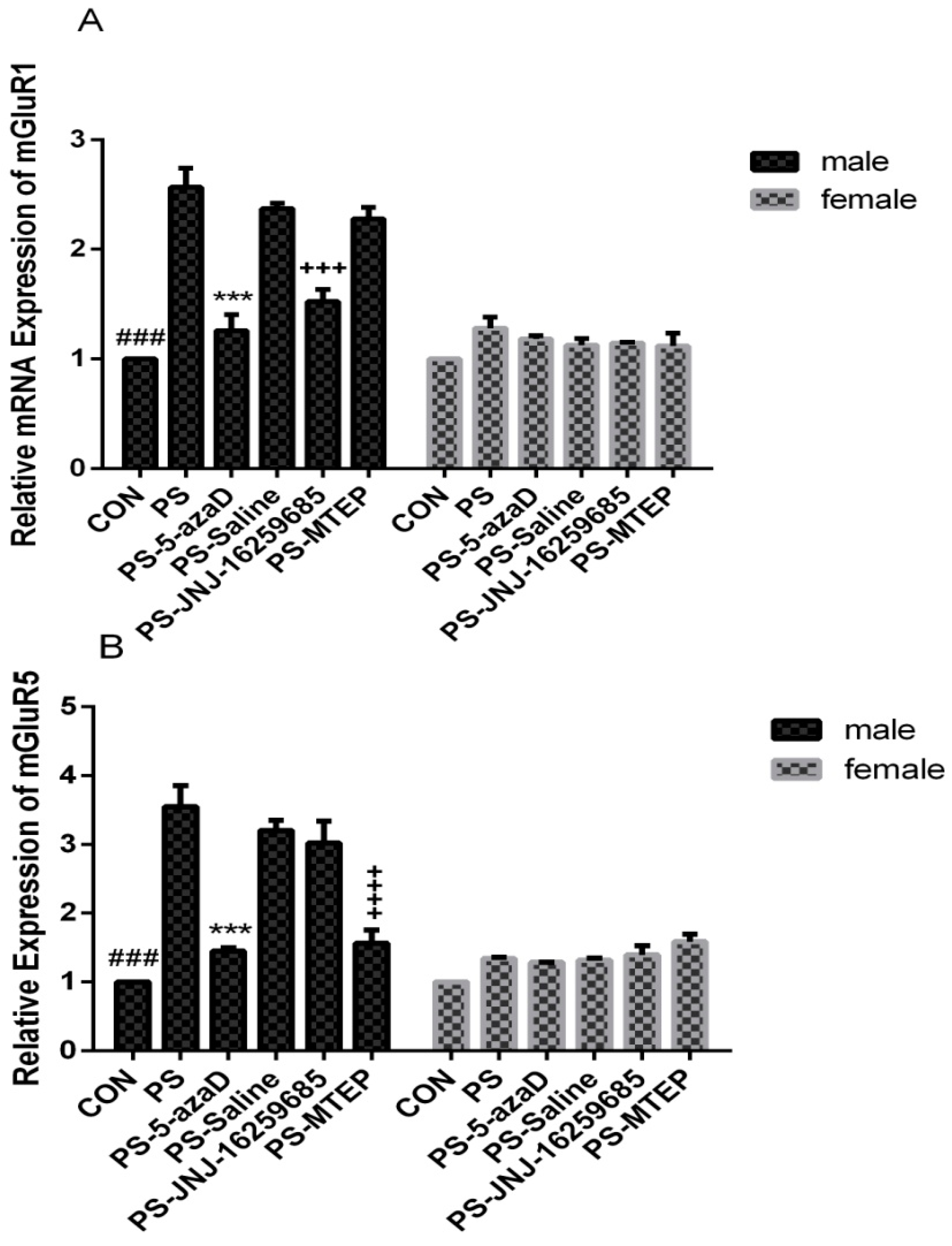
 p < 0.05 compare with PS.
p < 0.05 compare with PS.
 p < 0.05 compare with PS.
p < 0.05 compare with PS.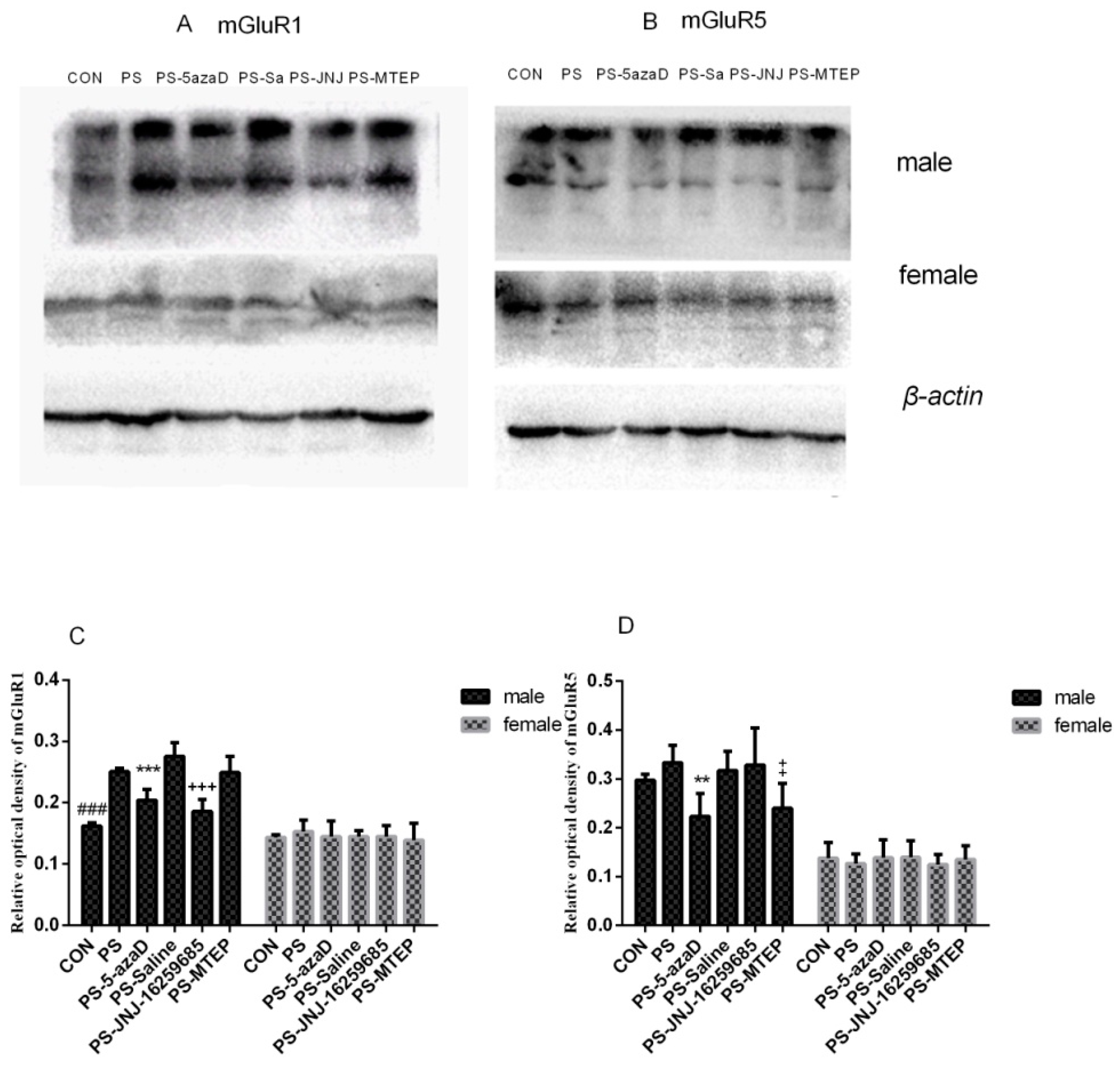
 p < 0.001 compare with PS.
p < 0.001 compare with PS.
 p < 0.001 compare with PS.
p < 0.001 compare with PS.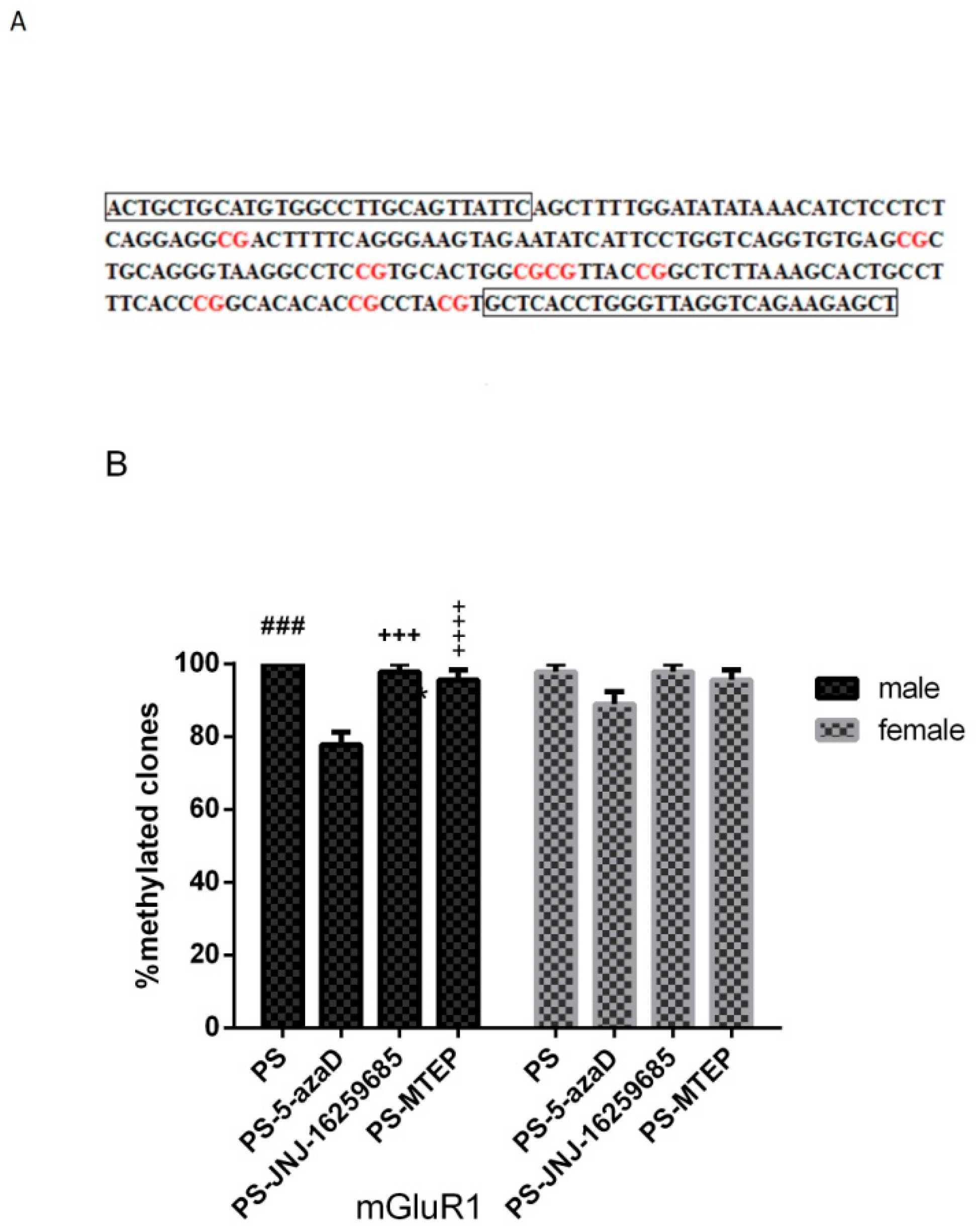
 p < 0.01 compare with PS.
p < 0.01 compare with PS.
 p < 0.01 compare with PS.
p < 0.01 compare with PS.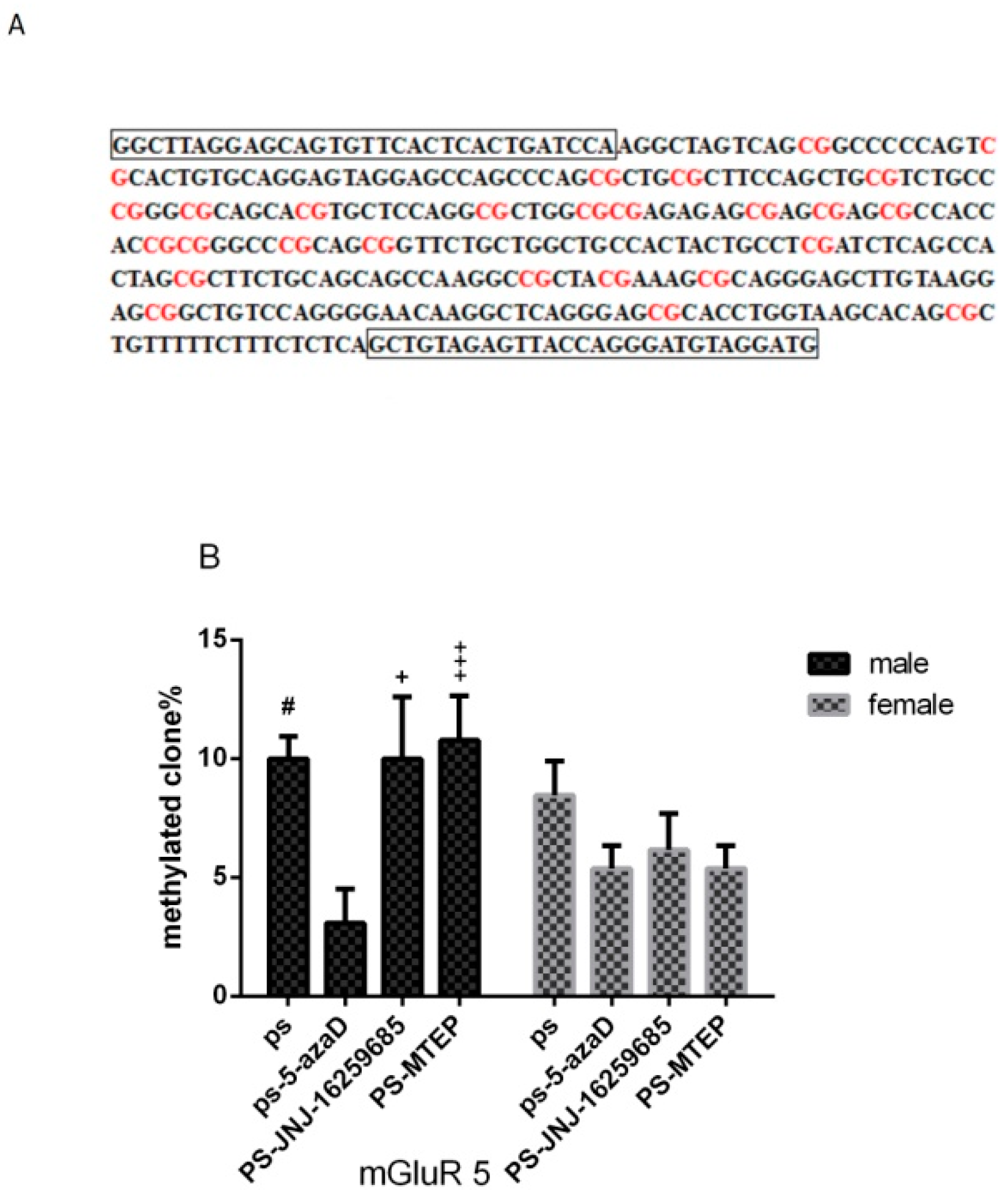
| mRNA | Primer(5′-3′) | Denatring °C (s) | Annealing °C (s) | Elongation °C (s) | Cycles |
|---|---|---|---|---|---|
| mGluR1 | 94 (30) | 56 (30) | 72 (60) | 39 | |
| F:GGTCCCTTCTGACACTTTGC | |||||
| R:AAGCATCCATTCCACTCTCG | |||||
| mGluR5 | 94 (30) | 56 (30) | 72 (60) | 39 | |
| F:TGAGAGGAAGTGTGGTGCAG | |||||
| R:CAAGTGTGATGTTGGGCAAG | |||||
| mRNA | Primer (5′-3′) | Denatring °C (s) | Annealing °C (s) | Elongation °C (s) | Cycles |
|---|---|---|---|---|---|
| mGluR1 | 94 (45) | 66 (45) | 72 (60) | 20 | |
| F:ATTGTTGTATGTGGTTTTGTAGTTATTT | |||||
| R:AACTCTTCTAACCTAACCCAAATAAAC | |||||
| mGluR5 | 94 (45) | 56 (45) | 72 (60) | 20 | |
| F:GGTTTAGGAGTAGTGTTTATTTATTGATTTA | |||||
| R:CATCCTACATCCCTAATAACTCTACAAC | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, T.; Dang, S.; Su, Q.; Zhang, H.; Zhang, J.; Zhang, L.; Zhang, X.; Lu, Y.; Li, H.; Zhu, Z. The Impact and Mechanism of Methylated Metabotropic Glutamate Receptors 1 and 5 in the Hippocampus on Depression-Like Behavior in Prenatal Stress Offspring Rats. J. Clin. Med. 2018, 7, 117. https://doi.org/10.3390/jcm7060117
Lin T, Dang S, Su Q, Zhang H, Zhang J, Zhang L, Zhang X, Lu Y, Li H, Zhu Z. The Impact and Mechanism of Methylated Metabotropic Glutamate Receptors 1 and 5 in the Hippocampus on Depression-Like Behavior in Prenatal Stress Offspring Rats. Journal of Clinical Medicine. 2018; 7(6):117. https://doi.org/10.3390/jcm7060117
Chicago/Turabian StyleLin, Tianwei, Shaokang Dang, Qian Su, Huiping Zhang, Junli Zhang, Lin Zhang, Xiaoxiao Zhang, Yong Lu, Hui Li, and Zhongliang Zhu. 2018. "The Impact and Mechanism of Methylated Metabotropic Glutamate Receptors 1 and 5 in the Hippocampus on Depression-Like Behavior in Prenatal Stress Offspring Rats" Journal of Clinical Medicine 7, no. 6: 117. https://doi.org/10.3390/jcm7060117
APA StyleLin, T., Dang, S., Su, Q., Zhang, H., Zhang, J., Zhang, L., Zhang, X., Lu, Y., Li, H., & Zhu, Z. (2018). The Impact and Mechanism of Methylated Metabotropic Glutamate Receptors 1 and 5 in the Hippocampus on Depression-Like Behavior in Prenatal Stress Offspring Rats. Journal of Clinical Medicine, 7(6), 117. https://doi.org/10.3390/jcm7060117




