Endovascular Interventions for the Morbidly Adherent Placenta
Abstract
1. Introduction
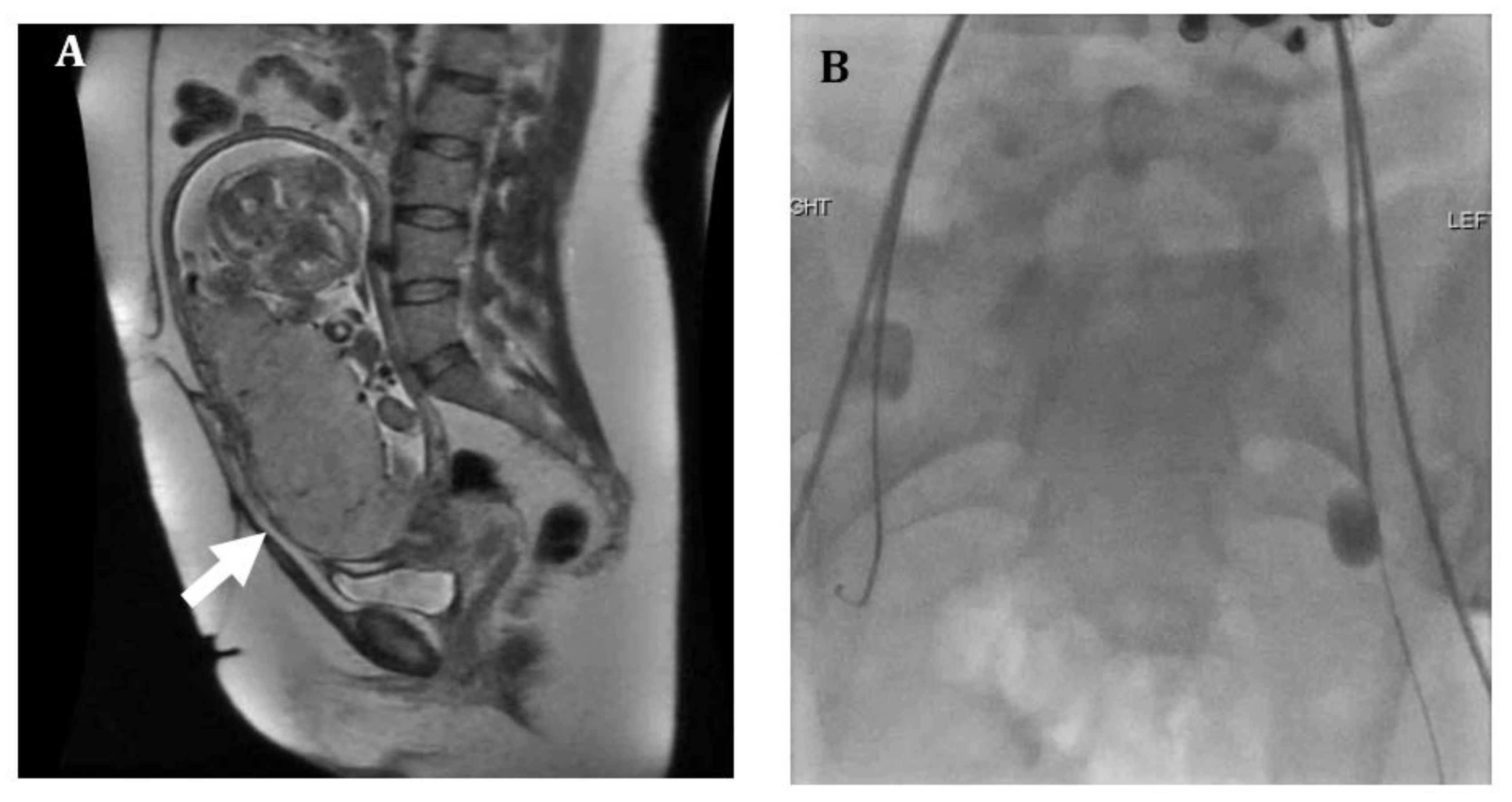
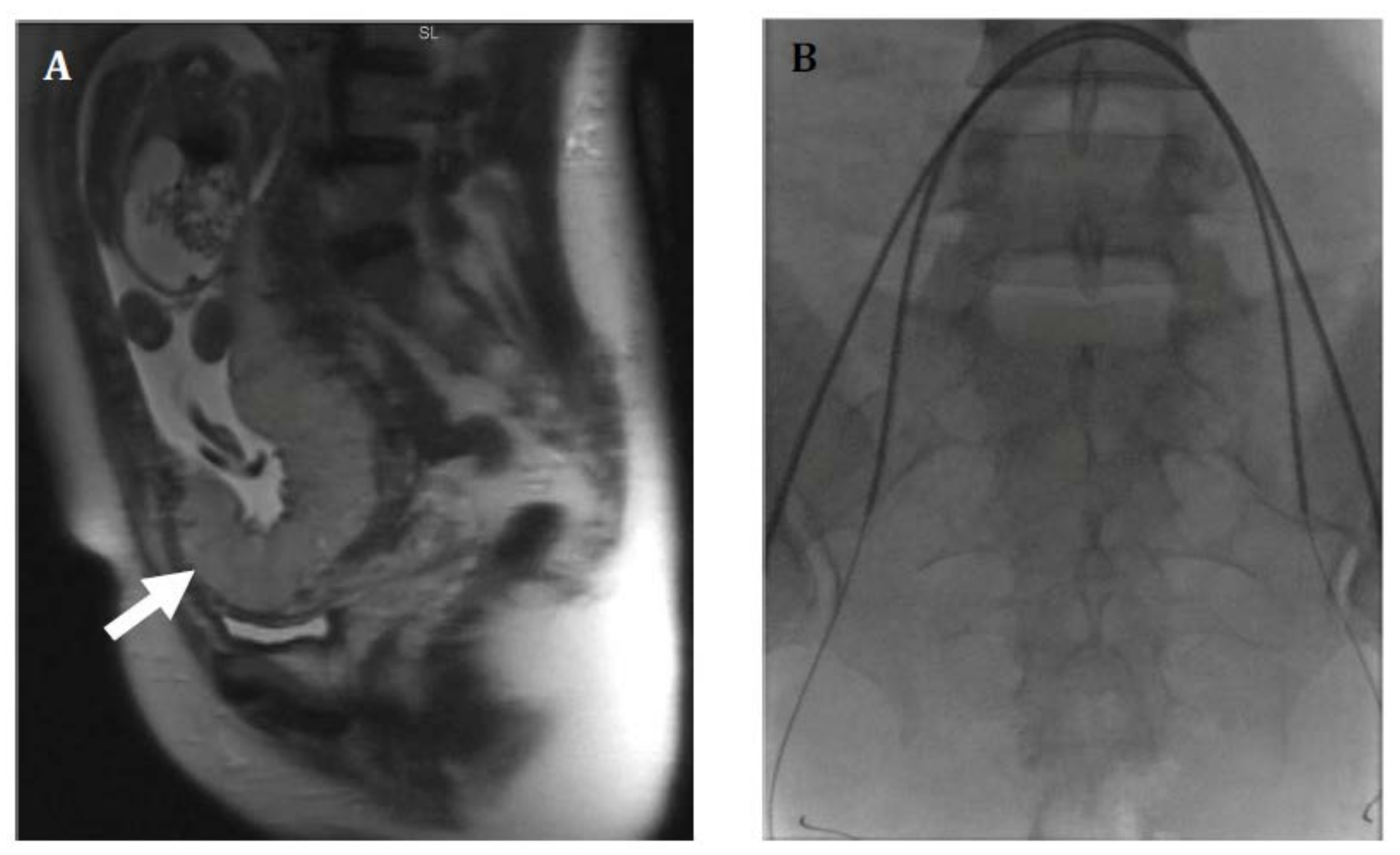
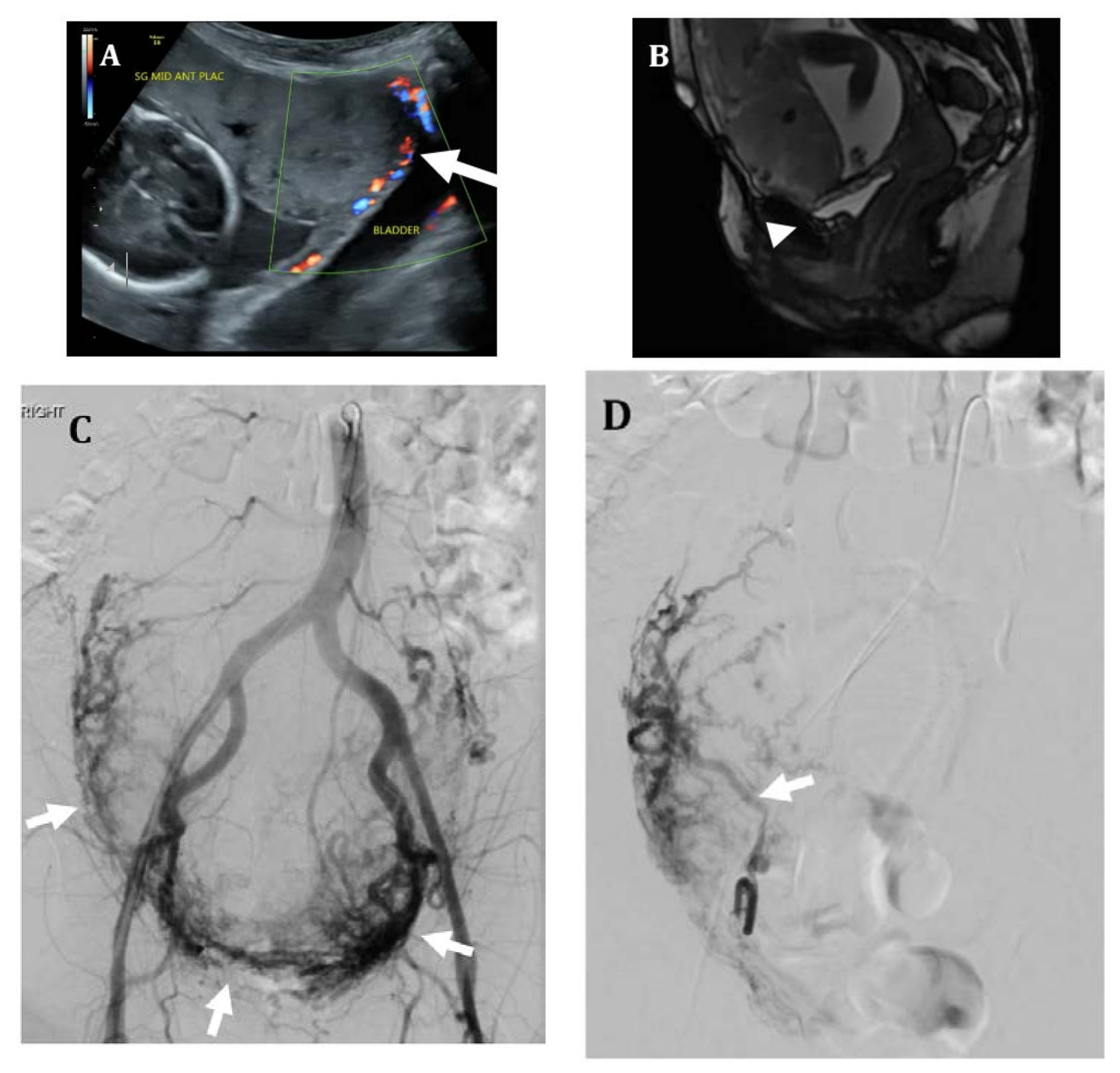
2. Imaging
3. Surgical Intervention
4. Endovascular Interventions
4.1. Internal Iliac Balloon Catheter Placement
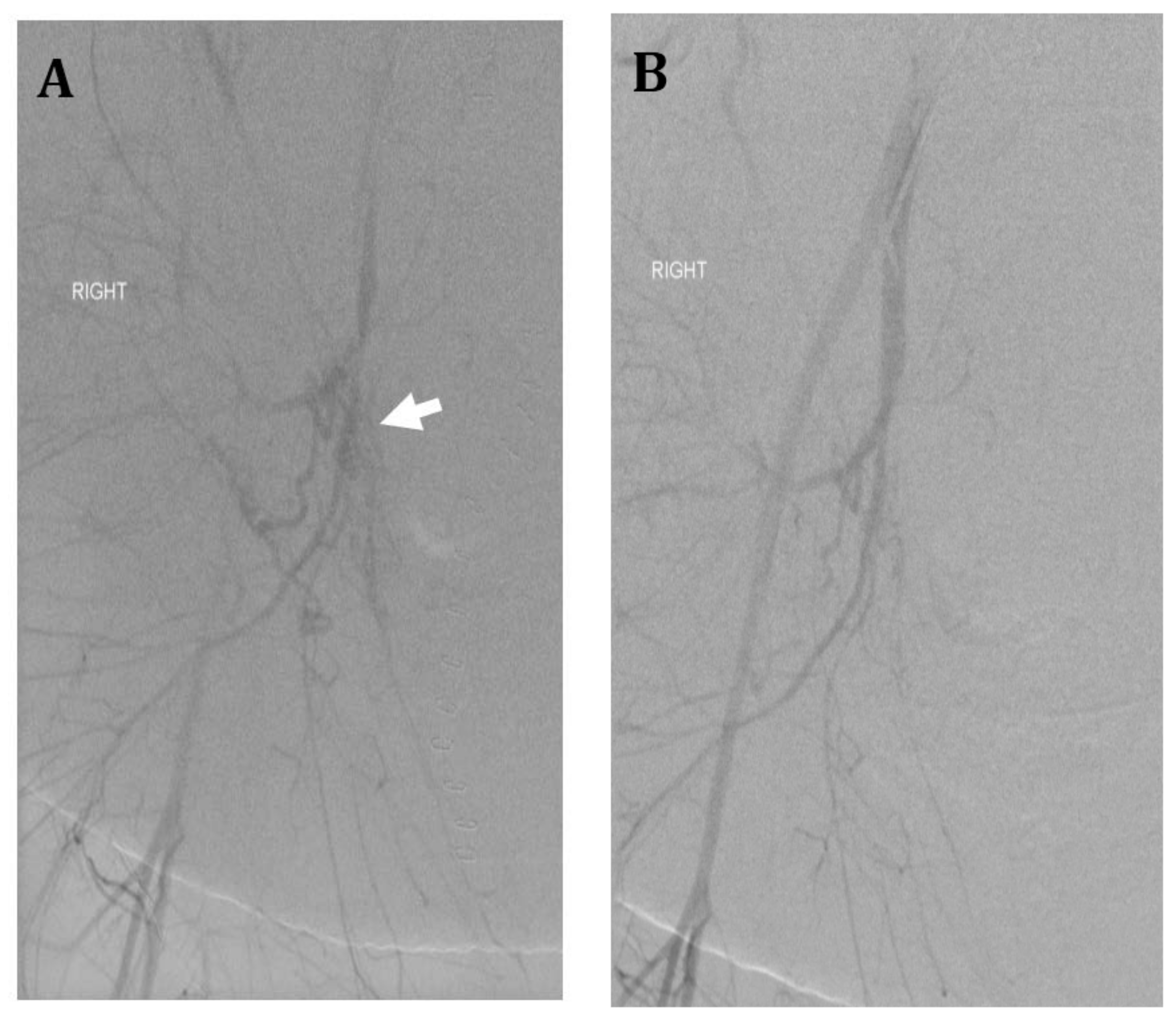
4.2. Infrarenal Aortic Balloon Catheter
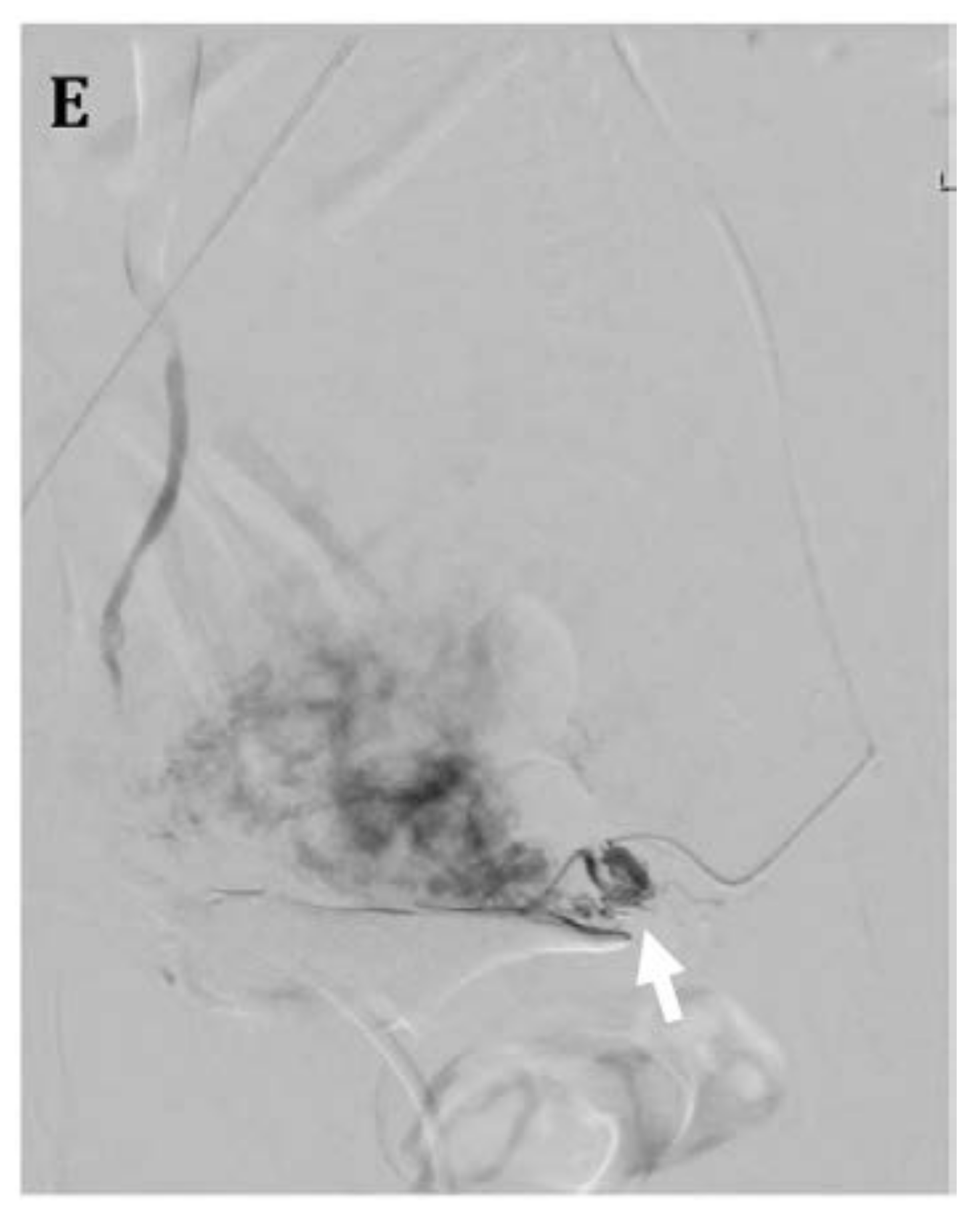
4.3. Uterine Artery Embolization
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kilcoyne, A.; Shenoy-Bhangle, A.S.; Roberts, D.J.; Sisodia, R.C.; Gervais, D.A.; Lee, S.I. MRI of Placenta Accreta, Placenta Increta, and Placenta Percreta: Pearls and Pitfalls. AJR Am. J. Roentgenol. 2017, 208, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Picel, A.C.; Wolford, B.; Cochran, R.L.; Ramos, G.A.; Roberts, A.C. Prophylactic Internal Iliac Artery Occlusion Balloon Placement to Reduce Operative Blood Loss in Patients with Invasive Placenta. J. Vasc. Interv. Radiol. 2018, 29, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Guleria, K.; Gupta, B.; Agarwal, S.; Suneja, A.; Vaid, N.; Jain, S. Abnormally invasive placenta: Changing trends in diagnosis and management. Acta Obstet. Gynecol. Scand. 2013, 92, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Mazouni, C.; Gorincour, G.; Juhan, V.; Bretelle, F. Placenta accreta: A review of current advances in prenatal diagnosis. Placenta 2007, 28, 599–603. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, D.L.; Kingdom, J.C.; Amsalem, H.; Beecroft, J.R.; Windrim, R.C.; Kachura, J.R. Conservative management of invasive placenta using combined prophylactic internal iliac artery balloon occlusion and immediate postoperative uterine artery embolization. Can. Assoc. Radiol. J. 2015, 66, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Oyelese, Y.; Smulian, J.C. Placenta previa, placenta accreta, and vasa previa. Obstet. Gynecol. 2006, 107, 927–941. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.A.; Chollet, J.A.; Goodwin, T.M. Clinical risk factors for placenta previa-placenta accreta. Am. J. Obstet. Gynecol. 1997, 177, 210–214. [Google Scholar] [CrossRef]
- Wu, S.; Kocherginsky, M.; Hibbard, J.U. Abnormal placentation: Twenty-year analysis. Am. J. Obstet. Gynecol. 2005, 192, 1458–1461. [Google Scholar] [CrossRef]
- Mc, K.R.; D’Errico, E. Placenta accreta: Clinical manifestations and conservative management. N. Engl. J. Med. 1951, 245, 159–165. [Google Scholar]
- Bowman, Z.S.; Eller, A.G.; Bardsley, T.R.; Greene, T.; Varner, M.W.; Silver, R.M. Risk factors for placenta accreta: A large prospective cohort. Am. J. Perinatol. 2014, 31, 799–804. [Google Scholar] [PubMed]
- Silver, R.M. Abnormal Placentation: Placenta Previa, Vasa Previa, and Placenta Accreta. Obstet. Gynecol. 2015, 126, 654–668. [Google Scholar] [CrossRef] [PubMed]
- Usta, I.M.; Hobeika, E.M.; Musa, A.A.; Gabriel, G.E.; Nassar, A.H. Placenta previa-accreta: Risk factors and complications. Am. J. Obstet. Gynecol. 2005, 193, 1045–1049. [Google Scholar] [CrossRef] [PubMed]
- Grobman, W.A.; Gersnoviez, R.; Landon, M.B.; Spong, C.Y.; Leveno, K.J.; Rouse, D.J.; Varner, M.W.; Moawad, A.H.; Caritis, S.N.; Harper, M.; et al. Pregnancy outcomes for women with placenta previa in relation to the number of prior cesarean deliveries. Obstet. Gynecol. 2007, 110, 1249–1255. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, K.E.; Sellers, S.; Spark, P.; Kurinczuk, J.J.; Brocklehurst, P.; Knight, M. Incidence and risk factors for placenta accreta/increta/percreta in the UK: A national case-control study. PLoS ONE 2012, 7, e52893. [Google Scholar] [CrossRef] [PubMed]
- Fox, K.A.; Shamshirsaz, A.A.; Carusi, D.; Secord, A.A.; Lee, P.; Turan, O.M.; Huls, C.; Abuhamad, A.; Simhan, H.; Barton, J.; et al. Conservative management of morbidly adherent placenta: Expert review. Am. J. Obstet. Gynecol. 2015, 213, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Society for Maternal-Fetal Medicine; Belfort, M.A. Placenta accreta. Am. J. Obstet. Gynecol. 2010, 203, 430–439. [Google Scholar]
- Baughman, W.C.; Corteville, J.E.; Shah, R.R. Placenta accreta: Spectrum of US and MR imaging findings. Radiographics 2008, 28, 1905–1916. [Google Scholar] [CrossRef] [PubMed]
- Dashe, J.S.; McIntire, D.D.; Ramus, R.M.; Santos-Ramos, R.; Twickler, D.M. Persistence of placenta previa according to gestational age at ultrasound detection. Obstet. Gynecol. 2002, 99, 692–697. [Google Scholar] [PubMed]
- Masselli, G.; Gualdi, G. MR imaging of the placenta: What a radiologist should know. Abdom. Imaging 2013, 38, 573–587. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-A.; Narra, V.R. Magnetic resonance imaging with true fast imaging with steady-state precession and half-fourier acquisition single-shot turbo spin-echo sequences in cases of suspected placenta accreta. Acta Radiol. 2004, 45, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Leyendecker, J.R.; DuBose, M.; Hosseinzadeh, K.; Stone, R.; Gianini, J.; Childs, D.D.; Snow, A.N.; Mertz, H. MRI of pregnancy-related issues: Abnormal placentation. AJR Am. J. Roentgenol. 2012, 198, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Masselli, G.; Brunelli, R.; Casciani, E.; Polettini, E.; Piccioni, M.G.; Anceschi, M.; Gualdi, G. Magnetic resonance imaging in the evaluation of placental adhesive disorders: Correlation with color Doppler ultrasound. Eur. Radiol. 2008, 18, 1292–1299. [Google Scholar] [CrossRef] [PubMed]
- Committee on Obstetric Practice. Committee opinion no. 529: Placenta accreta. Obstet. Gynecol. 2012, 120, 207–211. [Google Scholar]
- Eller, A.G.; Bennett, M.A.; Sharshiner, M.; Masheter, C.; Soisson, A.P.; Dodson, M.; Silver, R.M. Maternal morbidity in cases of placenta accreta managed by a multidisciplinary care team compared with standard obstetric care. Obstet. Gynecol. 2011, 117, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Shamshirsaz, A.A.; Fox, K.A.; Salmanian, B.; Diaz-Arrastia, C.R.; Lee, W.; Baker, B.W.; Ballas, J.; Chen, Q.; Van Veen, T.R.; Javadian, P.; et al. Maternal morbidity in patients with morbidly adherent placenta treated with and without a standardized multidisciplinary approach. Am. J. Obstet. Gynecol. 2015, 212, 218. [Google Scholar] [CrossRef] [PubMed]
- Belfort, M.A.; Shamshiraz, A.A.; Fox, K. Minimizing blood loss at cesarean-hysterectomy for placenta previa percreta. Am. J. Obstet. Gynecol. 2017, 216, 78. [Google Scholar] [CrossRef] [PubMed]
- Sentilhes, L.; Ambroselli, C.; Kayem, G.; Provansal, M.; Fernandez, H.; Perrotin, F.; Winer, N.; Pierre, F.; Benachi, A.; Dreyfus, M. Maternal outcome after conservative treatment of placenta accreta. Obstet. Gynecol. 2010, 115, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Ballas, J.; Hull, A.D.; Saenz, C.; Warshak, C.R.; Roberts, A.C.; Resnik, R.R.; Moore, T.R.; Ramos, G.A. Preoperative intravascular balloon catheters and surgical outcomes in pregnancies complicated by placenta accreta: A management paradox. Am. J. Obstet. Gynecol. 2012, 207, 216. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.H.; Tay, K.H.; Sheah, K.; Kwek, K.; Wong, K.; Tan, H.K.; Tan, B.S. Perioperative endovascular internal iliac artery occlusion balloon placement in management of placenta accreta. AJR Am. J. Roentgenol. 2007, 189, 1158–1163. [Google Scholar] [CrossRef] [PubMed]
- Salim, R.; Chulski, A.; Romano, S.; Garmi, G.; Rudin, M.; Shalev, E. Precesarean prophylactic balloon catheters for suspected placenta accreta. Obstet. Gynecol. 2015, 126, 1022–1028. [Google Scholar] [CrossRef] [PubMed]
- Cali, G.; Forlani, F.; Giambanco, L.; Amico, M.L.; Vallone, M.; Puccio, G.; Luigi, A. Prophylactic use of intravascular balloon catheters in women with placenta accreta, increta and percreta. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 179, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Liu, Z.; Zhao, X.; Liu, C.; Wang, Y.; Chu, Q.; Wang, X.; Chen, Z. Outcome of pregnancies after balloon occlusion of the infrarenal abdominal aorta during caesarean in 230 patients with placenta praevia accreta. Cardiovasc. Interv. Radiol. 2016, 39, 1573–1579. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.L.; Duan, X.H.; Han, X.W.; Wang, L.; Zhao, X.L.; Chen, Z.M.; Chu, Q.J.; Zhang, W. Comparison of temporary abdominal aortic occlusion with internal iliac artery occlusion for patients with placenta accrete—A non-randomised prospective study. Vasa 2017, 46, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.-C.; Ou, H.-Y.; Tsang, L.L.-C.; Kung, F.-T.; Hsu, T.-Y.; Cheng, Y.-F. Prophylactic intraoperative uterine artery embolization to control hemorrhage in abnormal placentation during late gestation. Fertil. Steril. 2009, 91, 1951–1955. [Google Scholar] [CrossRef] [PubMed]
- Izbizky, G.; Meller, C.; Grasso, M.; Velazco, A.; Peralta, O.; Otaño, L.; Garcia-Monaco, R. Feasibility and safety of prophylactic uterine artery catheterization and embolization in the management of placenta accreta. J. Vasc. Interv. Radiol. 2015, 26, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Yang, Z.Q.; Mohammed, W.; Feng, Y.L.; Shi, H.B.; Zhou, X. Prophylactic uterine artery embolization assisted cesarean section for the prevention of intrapartum hemorrhage in high-risk patients. Cardiovasc. Interv. Radiol. 2014, 37, 1458–1463. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pan, Y.; Zhou, X.; Yang, Z.; Cui, S.; De, W.; Sun, L. Retrospective cohort study of prophylactic intraoperative uterine artery embolization for abnormally invasive placenta. Int. J. Gynecol. Obstet. 2017, 137, 45–50. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaufman, C.; Tadros, A. Endovascular Interventions for the Morbidly Adherent Placenta. J. Clin. Med. 2018, 7, 92. https://doi.org/10.3390/jcm7050092
Kaufman C, Tadros A. Endovascular Interventions for the Morbidly Adherent Placenta. Journal of Clinical Medicine. 2018; 7(5):92. https://doi.org/10.3390/jcm7050092
Chicago/Turabian StyleKaufman, Claire, and Anthony Tadros. 2018. "Endovascular Interventions for the Morbidly Adherent Placenta" Journal of Clinical Medicine 7, no. 5: 92. https://doi.org/10.3390/jcm7050092
APA StyleKaufman, C., & Tadros, A. (2018). Endovascular Interventions for the Morbidly Adherent Placenta. Journal of Clinical Medicine, 7(5), 92. https://doi.org/10.3390/jcm7050092



