Amiloride Promotes Oligodendrocyte Survival and Remyelination after Spinal Cord Injury in Rats
Abstract
1. Introduction
2. Experimental Section
2.1. SCI Model
2.2. Administration of Amiloride
2.3. Evaluation of Hindlimb Function
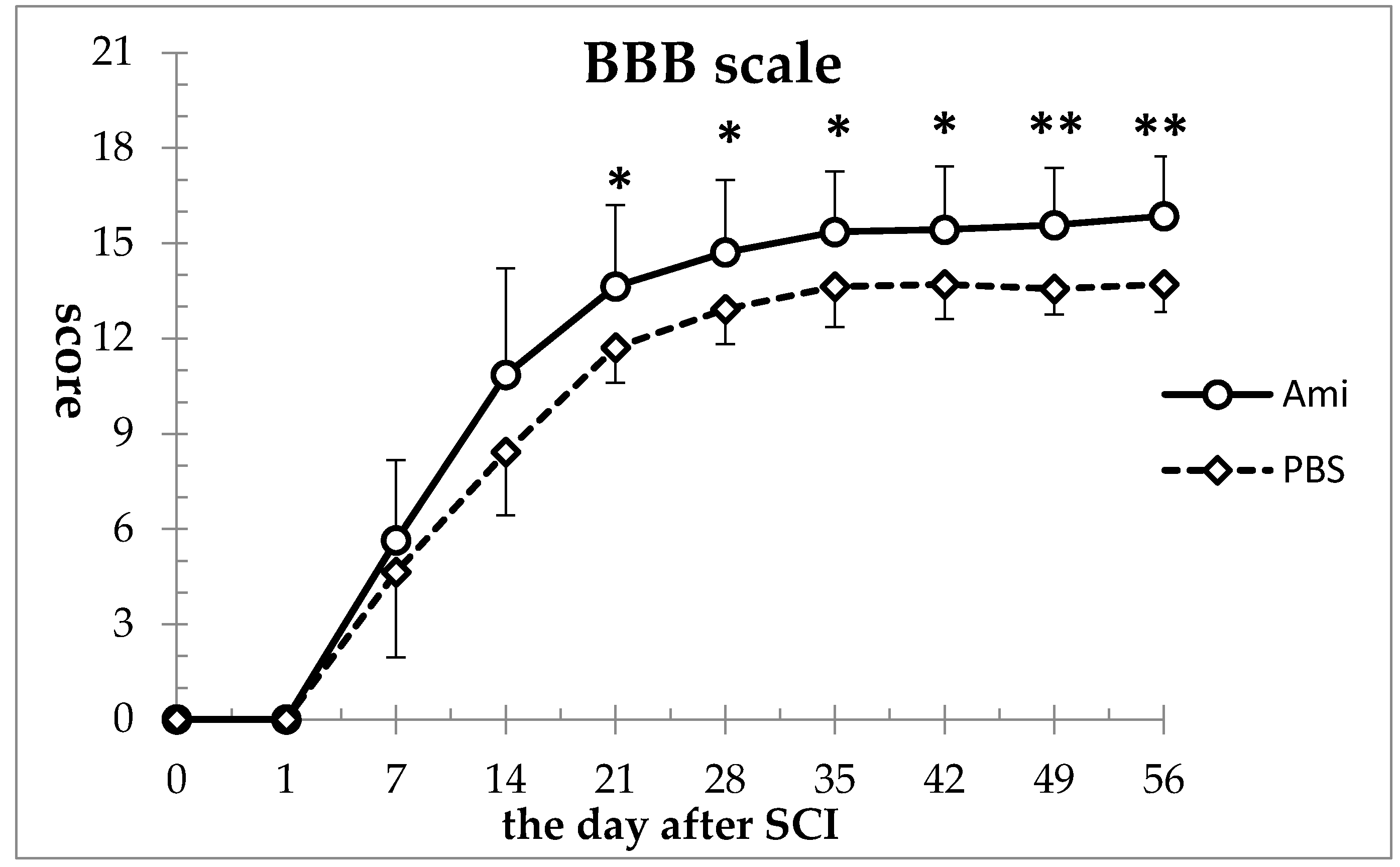
2.3.1. Basso, Beattie, Breanahan Locomotor Rating Scale (BBB) Scores
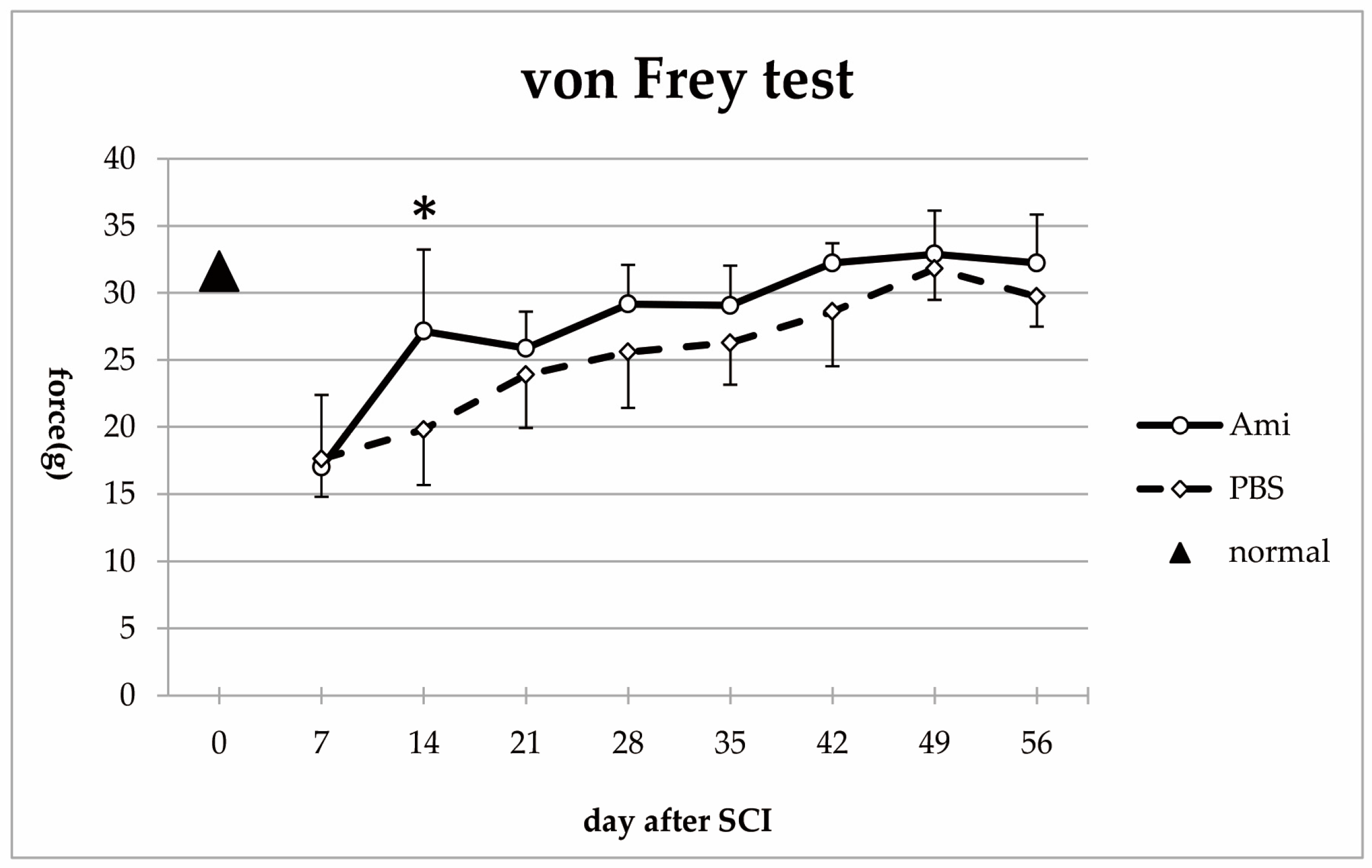
2.3.2. Dynamic Plantar Esthesiometer
2.4. Immunohistochemistry
2.5. Western Blot
2.6. Toluidine Blue Staining and Electron Microscopy
2.7. Statistical Analysis
3. Results
3.1. Hindlimb Motor Function
3.2. Hindlimb Sensory Function
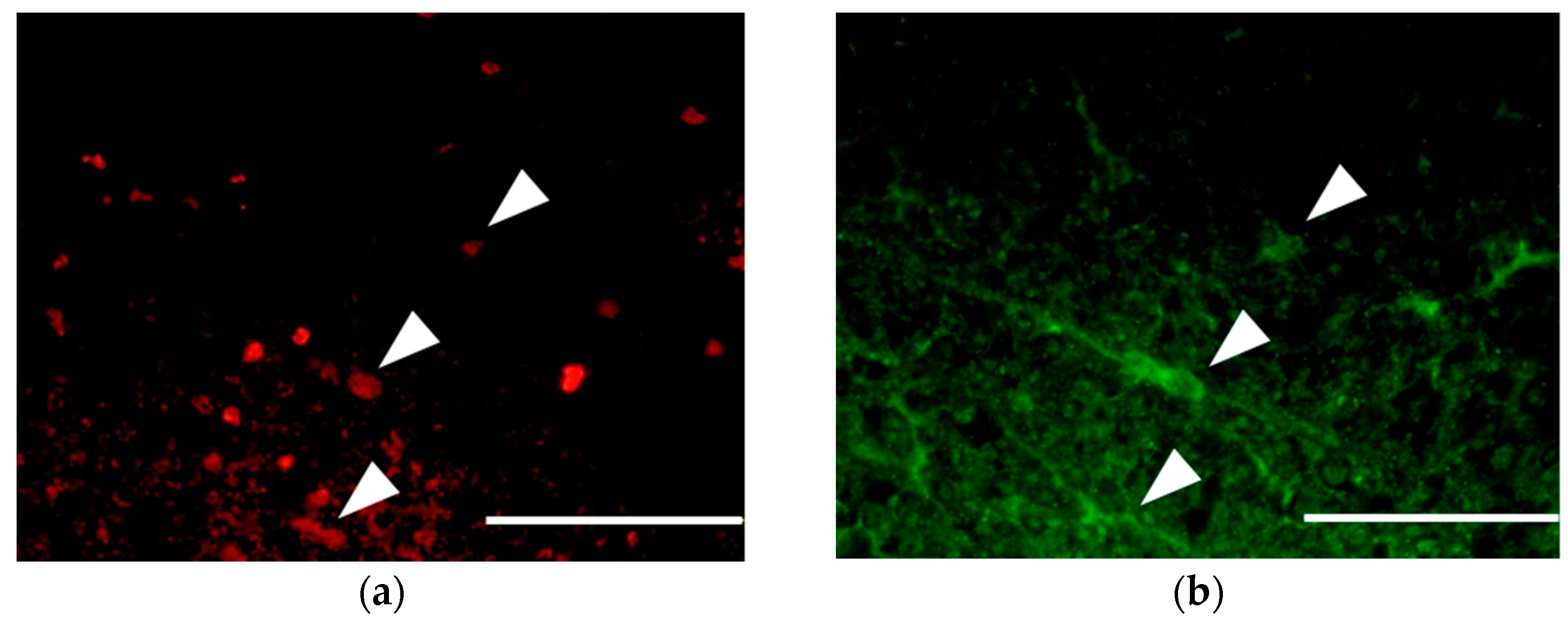
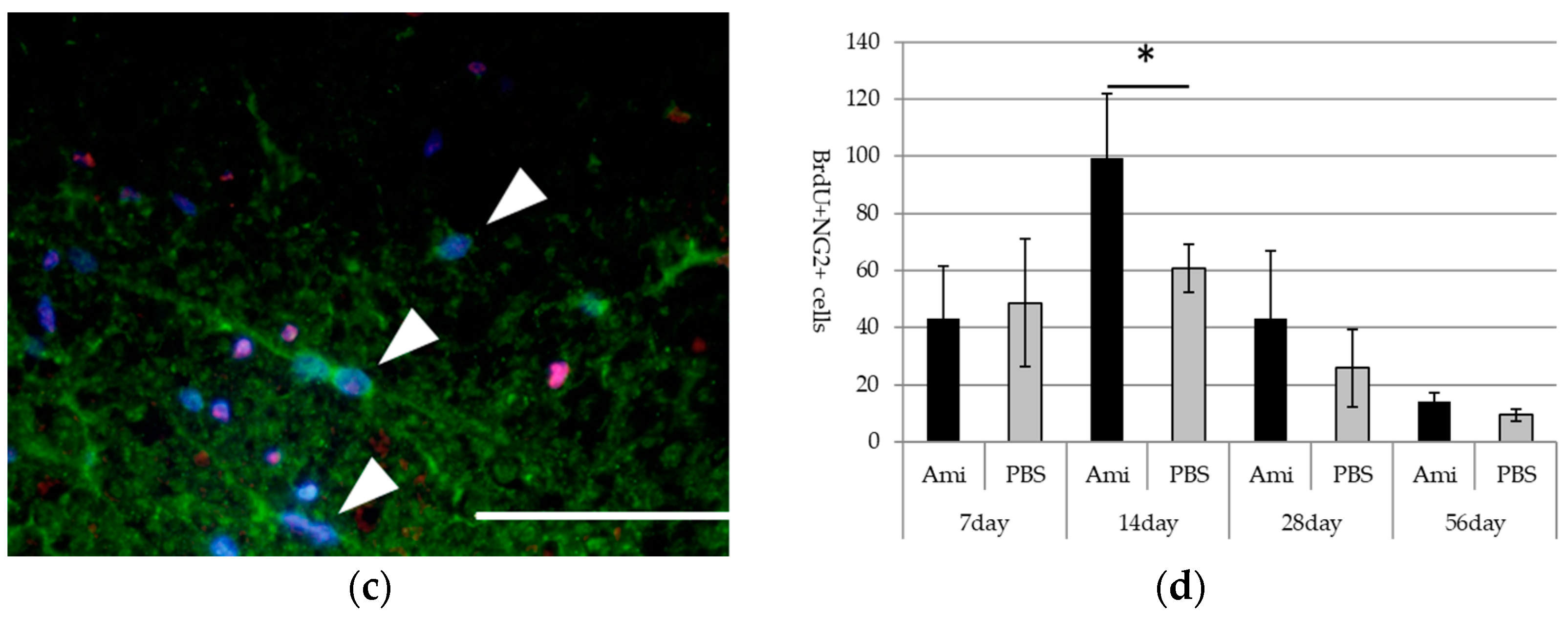
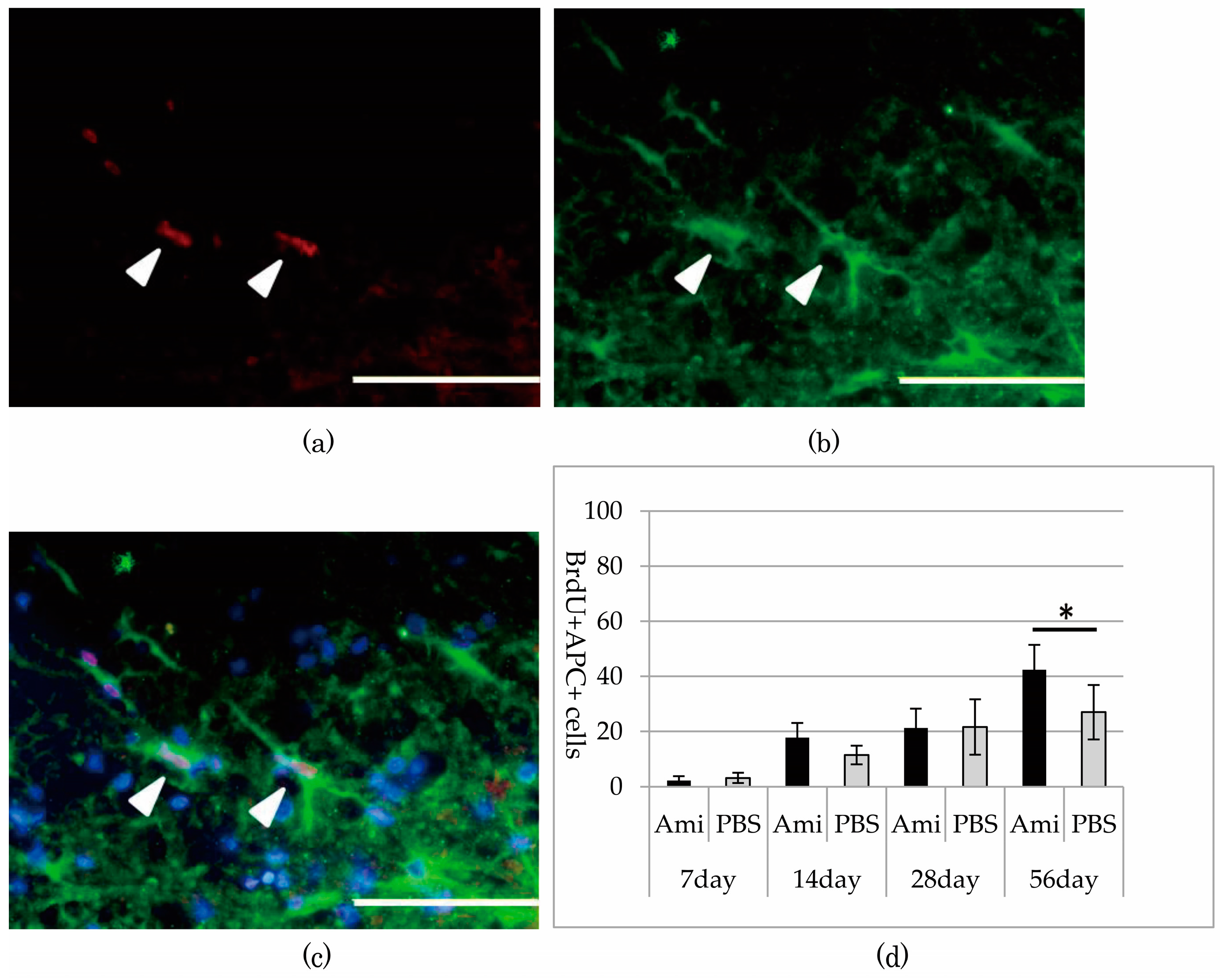
3.3. Survival and Proliferation of Oligodendrocyte Precursor Cells (OPCs)
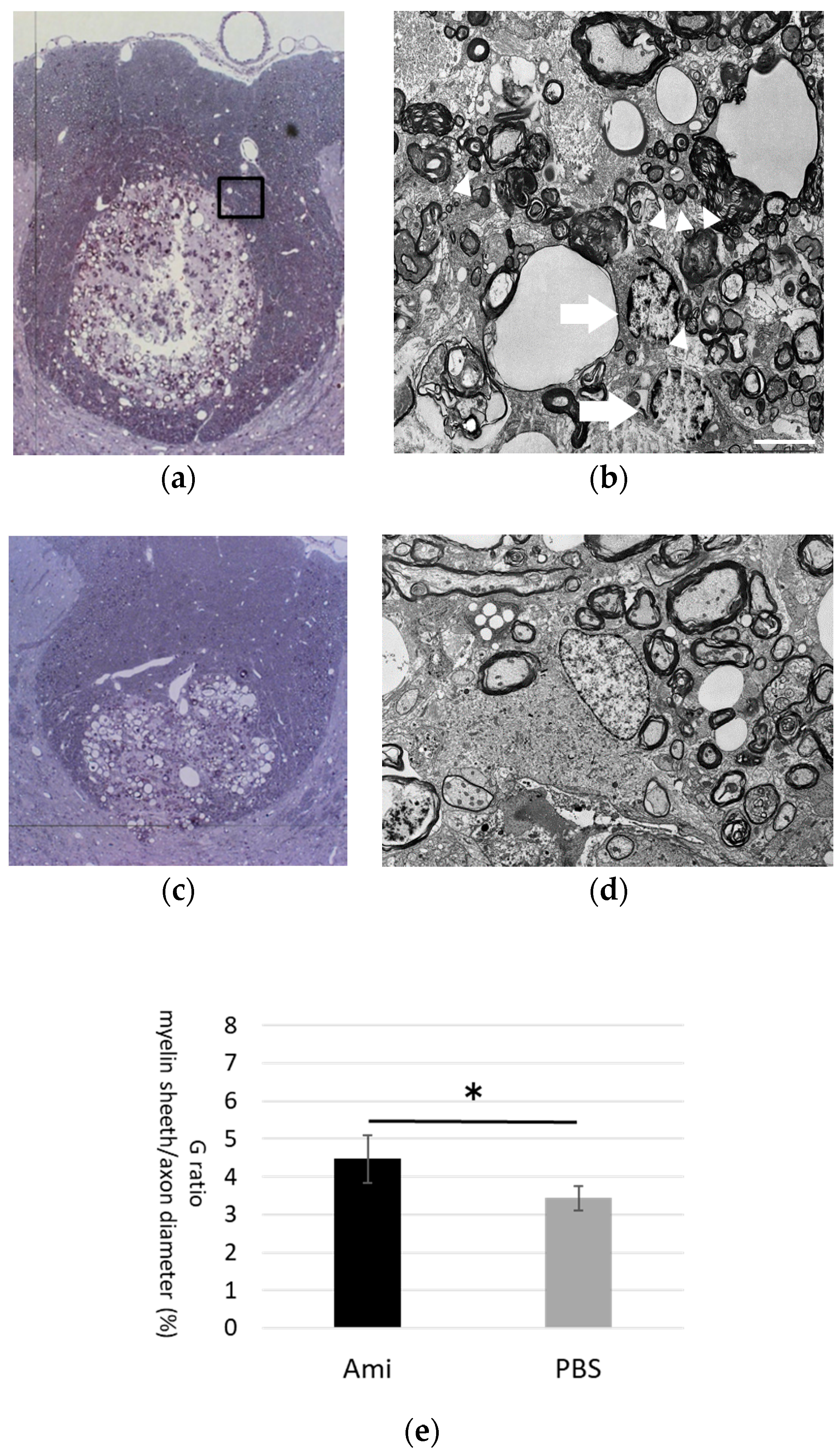
3.4. Remyelination
4. Discussion
4.1. The Apoptosis of Neural Cells during the Secondary Injury Phase of SCI
4.2. ER-Stress-Induced Apoptosis
4.3. Enhancing the ER Stress Response with Amiloride
4.4. The Survival and Differentiation of OPCs
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Thuret, S.; Moon, L.D.; Gage, F.H. Therapeutic interventions after spinal cord injury. Nat. Rev. Neurosci. 2006, 7, 628–643. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Z.; Xu, X.M.; Hu, R.; Du, C.; Zhang, S.X.; McDonald, J.W.; Dong, H.X.; Wu, Y.J.; Fan, G.S.; Jacquin, M.F.; et al. Neuronal and glial apoptosis after traumatic spinal cord injury. J. Neurosci. 1997, 17, 5395–5406. [Google Scholar] [PubMed]
- Beattie, M.S.; Farooqui, A.A.; Bresnahan, J.C. Review of current evidence for apoptosis after spinal cord injury. J. Neurotrauma 2000, 17, 915–925. [Google Scholar] [CrossRef] [PubMed]
- Casha, S.; Yu, W.R.; Fehlings, M.G. Oligodendroglial apoptosis occurs along degenerating axons and is associated with FAS and p75 expression following spinal cord injury in the rat. Neuroscience 2001, 103, 203–218. [Google Scholar] [CrossRef]
- McTigue, D.M. Potential Therapeutic Targets for PPARgamma after Spinal Cord Injury. PPAR. Res. 2008, 2008, 517162. [Google Scholar] [CrossRef] [PubMed]
- Oyinbo, C.A. Secondary injury mechanisms in traumatic spinal cord injury: A nugget of this multiply cascade. Acta. Neurobiol. Exp. 2011, 71, 281–299. [Google Scholar]
- Ishii, K.; Toda, M.; Nakai, Y.; Asou, H.; Watanabe, M.; Nakamura, M.; Yato, Y.; Fujimura, Y.; Kawakami, Y.; Toyama, Y.; et al. Increase of oligodendrocyte progenitor cells after spinal cord injury. J. Neurosci. Res. 2001, 65, 500–507. [Google Scholar] [CrossRef] [PubMed]
- McTigue, D.M.; Wei, P.; Stokes, B.T. Proliferation of NG2-positive cells and altered oligodendrocyte numbers in the contused rat spinal cord. J. Neurosci. 2001, 21, 3392–3400. [Google Scholar] [PubMed]
- Kuroiwa, M.; Watanabe, M.; Katoh, H.; Suyama, K.; Matsuyama, D.; Imai, T.; Mochida, J. Effect of amiloride on endoplasmic reticulum stress response in the injured spinal cord of rats. Eur. J. Neurosci. 2014, 40, 3120–3127. [Google Scholar] [CrossRef] [PubMed]
- Penas, C.; Guzmán, M.S.; Verdú, E.; Forés, J.; Navarro, X.; Casas, C. Spinal cord injury induces endoplasmic reticulum stress with different cell-type dependent response. J. Neurochem. 2007, 102, 1242–1255. [Google Scholar] [CrossRef] [PubMed]
- Ohri, S.S.; Maddie, M.A.; Zhao, Y.; Qiu, M.S.; Hetman, M.; Whittemore, S.R. Attenuating the endoplasmic reticulum stress response improves functional recovery after spinal cord injury. Glia 2011, 59, 1489–1502. [Google Scholar] [CrossRef] [PubMed]
- Durham-Lee, J.C.; Mokkapati, V.U.; Johnson, K.M.; Nesic, O. Amiloride improves locomotor recovery after spinal cord injury. J. Neurotrauma 2011, 28, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Friese, M.A.; Craner, M.J.; Etzensperger, R.; Vergo, S.; Wemmie, J.A.; Welsh, M.J.; Vincent, A.; Fugger, L. Acid-sensing ion channel-1 contributes to axonal degeneration in autoimmune inflammation of the central nervous system. Nat. Med. 2007, 13, 1483–1489. [Google Scholar] [CrossRef] [PubMed]
- Basso, D.M.; Beattie, M.S.; Bresnahan, J.C. A sensitive and reliable locomotor rating scale for open field testing in rats. J. Neurotrauma 1995, 12, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Tator, C.H. Biology of neurological recovery and functional restoration after spinal cord injury. Neurosurgery 1998, 42, 696–708. [Google Scholar] [CrossRef] [PubMed]
- Ramer, L.M.; Ramer, M.S.; Steeves, J.D. Setting the stage for functional repair of spinal cord injuries: A cast of thousands. Spinal Cord 2005, 43, 134–161. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Rabchevsky, A.G.; Hall, E.D. Role of peroxynitrite in secondary oxidative damage after spinal cord injury. J. Neurochem. 2007, 100, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.K.; Hogan, E.L.; Banik, N.L. Calpain in the pathophysiology of spinal cord injury: Neuroprotection with calpain inhibitors. Brain Res. Rev. 2003, 42, 169–185. [Google Scholar] [CrossRef]
- Dumont, R.J.; Okonkwo, D.O.; Verma, S.; Hurlbert, R.J.; Boulos, P.T.; Ellegala, D.B.; Dumont, A.S. Acute spinal cord injury, part I: Pathophysiologic mechanisms. Clin. Neuropharmacol. 2001, 24, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Amemiya, S.; Kamiya, T.; Nito, C.; Inaba, T.; Kato, K.; Ueda, M.; Shimazaki, K.; Katayama, Y. Anti-apoptotic and neuroprotective effects of edaravone following transient focal ischemia in rats. Eur. J. Pharmacol. 2005, 516, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Arias, R.L.; Sung, M.L.; Vasylyev, D.; Zhang, M.Y.; Albinson, K.; Kubek, K.; Kagan, N.; Beyer, C.; Lin, Q.; Dwyer, J.M.; et al. Amiloride is neuroprotective in an MPTP model of Parkinson’s disease. Neurobiol. Dis. 2008, 31, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Suyama, K.; Watanabe, M.; Sakabe, K.; Okada, Y.; Matsuyama, D.; Kuroiwa, M.; Mochida, J. Overexpression of GRP78 protects glial cells from endoplasmic reticulum stress. Neurosci. Lett. 2011, 504, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, D.; Watanabe, M.; Suyama, K.; Kuroiwa, M.; Mochida, J. Endoplasmic reticulum stress response in the rat contusive spinal cord injury model-susceptibility in specific cell types. Spinal Cord 2014, 52, 9–16. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xiong, Z.G.; Pignataro, G.; Li, M.; Chang, S.Y.; Simon, R.P. Acid-sensing ion channels (ASICs) as pharmacological targets for neurodegenerative diseases. Curr. Opin. Pharmacol. 2008, 8, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Hosoi, T.; Kume, A.; Otani, K.; Oba, T.; Ozawa, K. A unique modulator of endoplasmic reticulum stress-signalling pathways: The novel pharmacological properties of amiloride in glial cells. Br. J. Pharmacol. 2010, 159, 428–437. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xiong, Z.G.; Zhu, X.M.; Chu, X.P.; Minami, M.; Hey, J.; Wei, W.L.; MacDonald, J.F.; Wemmie, J.A.; Price, M.P.; Welsh, M.J.; et al. Neuroprotection in ischemia: Blocking calcium-permeable acid-sensing ion channels. Cell 2004, 118, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Toyama, Y.; Nishiyama, A. Differentiation of proliferated NG2-positive glial progenitor cells in a remyelinating lesion. J. Neurosci. Res. 2002, 69, 826–836. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imai, T.; Katoh, H.; Suyama, K.; Kuroiwa, M.; Yanagisawa, S.; Watanabe, M. Amiloride Promotes Oligodendrocyte Survival and Remyelination after Spinal Cord Injury in Rats. J. Clin. Med. 2018, 7, 46. https://doi.org/10.3390/jcm7030046
Imai T, Katoh H, Suyama K, Kuroiwa M, Yanagisawa S, Watanabe M. Amiloride Promotes Oligodendrocyte Survival and Remyelination after Spinal Cord Injury in Rats. Journal of Clinical Medicine. 2018; 7(3):46. https://doi.org/10.3390/jcm7030046
Chicago/Turabian StyleImai, Takeshi, Hiroyuki Katoh, Kaori Suyama, Masahiro Kuroiwa, Sho Yanagisawa, and Masahiko Watanabe. 2018. "Amiloride Promotes Oligodendrocyte Survival and Remyelination after Spinal Cord Injury in Rats" Journal of Clinical Medicine 7, no. 3: 46. https://doi.org/10.3390/jcm7030046
APA StyleImai, T., Katoh, H., Suyama, K., Kuroiwa, M., Yanagisawa, S., & Watanabe, M. (2018). Amiloride Promotes Oligodendrocyte Survival and Remyelination after Spinal Cord Injury in Rats. Journal of Clinical Medicine, 7(3), 46. https://doi.org/10.3390/jcm7030046





