Comparative Effectiveness of the Core Components of Cardiac Rehabilitation on Mortality and Morbidity: A Systematic Review and Network Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Information Sources and Search Strategy
2.2. Inclusion and Exclusion Criteria
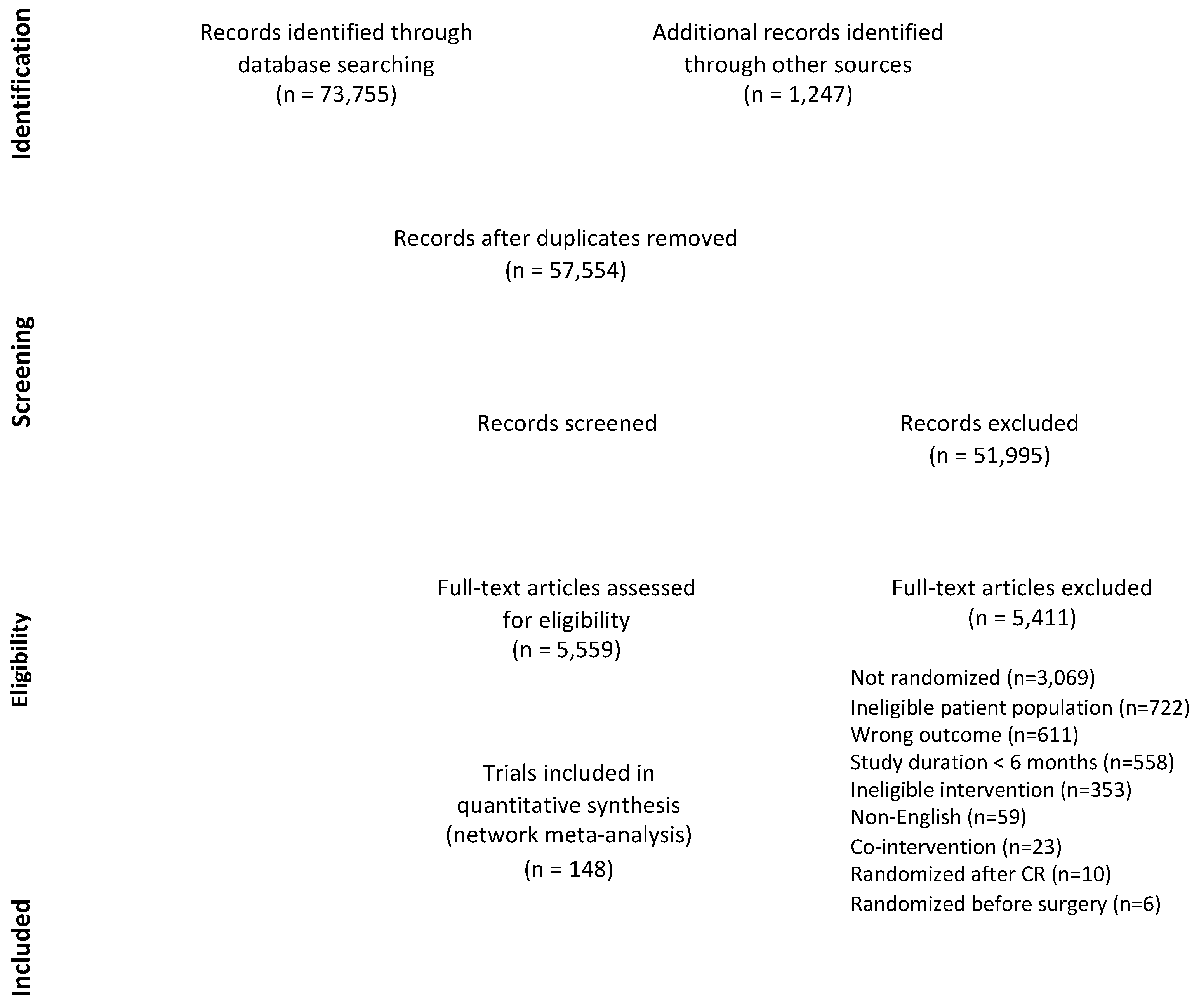
2.3. Study Selection
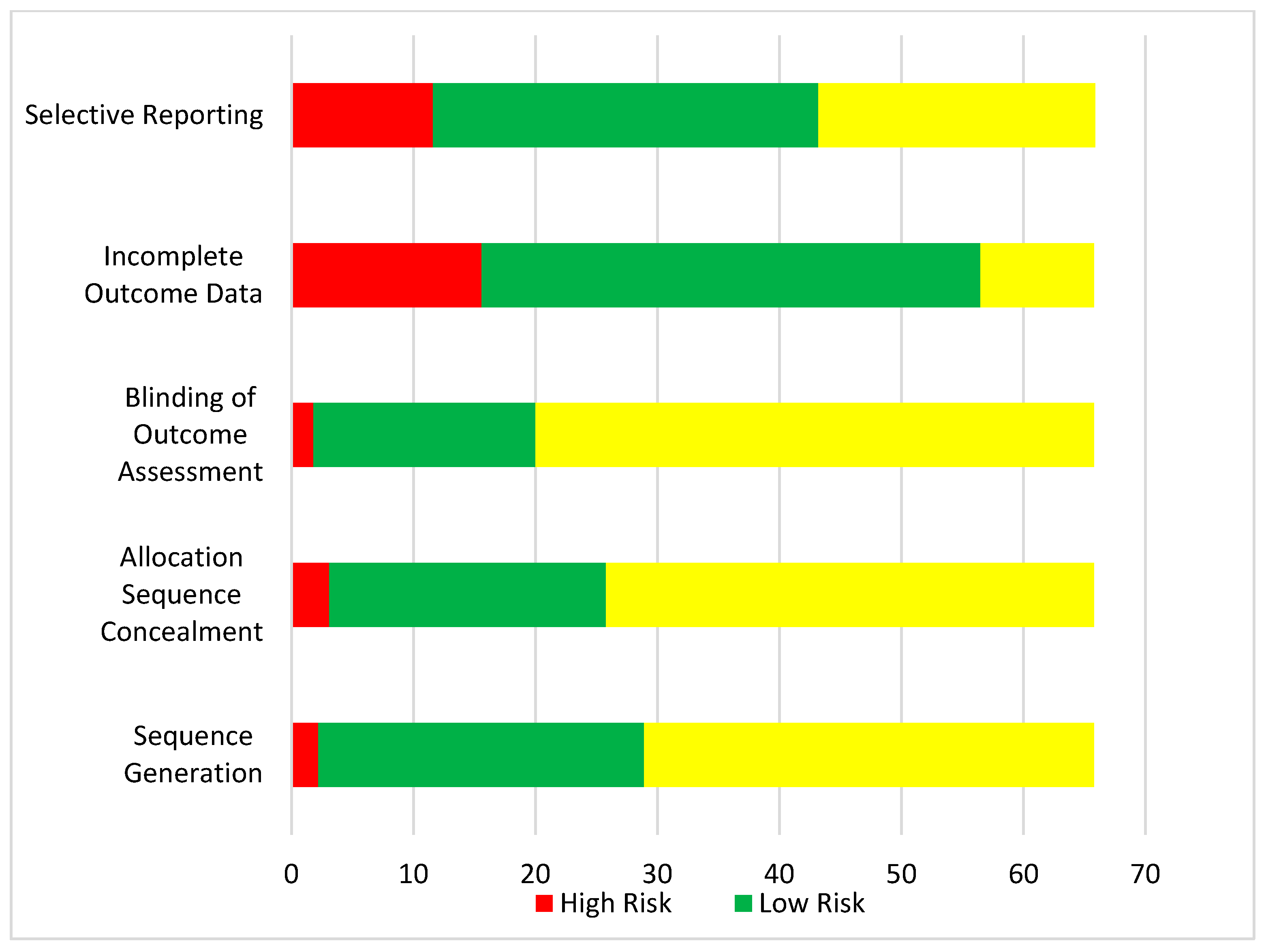
2.4. Data Extraction Process and Quality Assessment
2.5. Data Synthesis and Analysis
3. Results
Effects of Core Components
4. Discussion
4.1. Implications
4.2. Limitations
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Global Health Observatory Data Repository. Available online: http://apps.who.int/gho/data/?theme=main (accessed on 7 December 2014).
- Roth, G.A.; Johnson, C.; Abajobir, A.; Abd-Allah, F.; Abera, S.F.; Abyu, G.; Ahmed, M.; Aksut, B.; Alam, T.; Alam, K.; et al. Global, Regional, and National Burden of Cardiovascular Diseases for 10 Causes, 1990 to 2015. J. Am. Coll. Cardiol. 2017, 70, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Leon, A.S.; Franklin, B.A.; Costa, F.; Balady, G.J.; Berra, K.A.; Stewart, K.J.; Thompson, P.D.; Williams, M.A.; Lauer, M.S. Cardiac rehabilitation and secondary prevention of coronary heart disease: An American Heart Association scientific statement from the Council on Clinical Cardiology (Subcommittee on Exercise, Cardiac Rehabilitation, and Prevention) and the Council on Nutrition, Physical Activity, and Metabolism (Subcommittee on Physical Activity), in collaboration with the American association of Cardiovascular and Pulmonary Rehabilitation. Circulation 2005, 111, 369–376. [Google Scholar] [PubMed]
- Balady, G.J.; Williams, M.A.; Ades, P.A.; Bittner, V.; Comoss, P.; Foody, J.M.; Franklin, B.; Sanderson, B.; Southard, D. Core components of cardiac rehabilitation/secondary prevention programs: 2007 update: A scientific statement from the American Heart Association Exercise, Cardiac Rehabilitation, and Prevention Committee, the Council on Clinical Cardiology; the Councils on Cardiovascular Nursing, Epidemiology and Prevention, and Nutrition, Physical Activity, and Metabolism; and the American Association of Cardiovascular and Pulmonary Rehabilitation. Circulation 2007, 115, 2675–2682. [Google Scholar] [PubMed]
- Anderson, L.; Oldridge, N.; Thompson, D.R.; Zwisler, A.D.; Rees, K.; Martin, N.; Taylor, R.S. Exercise-Based Cardiac Rehabilitation for Coronary Heart Disease Cochrane Systematic Review and Meta-Analysis. J. Am. Coll. Cardiol. 2016, 67, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Piepoli, M.F.; Corrà, U.; Adamopoulos, S.; Benzer, W.; Bjarnason-Wehrens, B.; Cupples, M.; Dendale, P.; Doherty, P.; Gaita, D.; Höfer, S.; et al. Secondary prevention in the clinical management of patients with cardiovascular diseases. Core components, standards and outcome measures for referral and delivery: A policy statement from the cardiac rehabilitation section of the European Association for Cardiovascular Prevention & Rehabilitation. Endorsed by the Committee for Practice Guidelines of the European Society of Cardiology. Eur. J. Prev. Cardiol. 2014, 21, 664–681. [Google Scholar] [PubMed]
- Cardiac Rehabilitation and Cardiovascular Disease Prevention (CACR). Canadian Guidelines for Cardiac Rehabilitation and Cardiovascular Disease Prevention 2015. Available online: https://cacpr.ca/Guidelines (accessed on 5 July 2017).
- Woodruffe, S.; Neubeck, L.; Clark, R.A.; Gray, K.; Ferry, C.; Finan, J.; Sanderson, S.; Briffa, T.G. Australian Cardiovascular Health and Rehabilitation Association (ACRA) core components of cardiovascular disease secondary prevention and cardiac rehabilitation 2014. Heart Lung Circul. 2015, 24, 430–441. [Google Scholar] [CrossRef]
- Buckley, J.P.; Furze, G.; Doherty, P.; Speck, L.; Connolly, S.; Hinton, S.; Jones, J.L. BACPR scientific statement: British standards and core components for cardiovascular disease prevention and rehabilitation. Heart 2013, 99, 1069–1071. [Google Scholar] [CrossRef]
- Hamm, L.F.; Sanderson, B.K.; Ades, P.A.; Berra, K.; Kaminsky, L.A.; Roitman, J.L.; Williams, M.A. Core competencies for cardiac rehabilitation/secondary prevention professionals: 2010 update: Position statement of the American Association of Cardiovascular and Pulmonary Rehabilitation. J. Cardiopulm. Rehabil. Prev. 2011, 31, 2–10. [Google Scholar] [CrossRef]
- Van Halewijn, G.; Deckers, J.; Tay, H.Y.; van Domburg, R.; Kotseva, K.; Wood, D. Lessons from contemporary trials of cardiovascular prevention and rehabilitation: A systematic review and meta-analysis. Int. J. Cardiol. 2017, 232, 294–303. [Google Scholar] [CrossRef]
- Kachur, S.; Chongthammakun, V.; Lavie, C.J.; De Schutter, A.; Arena, R.; Milani, R.V.; Franklin, B.A. Impact of cardiac rehabilitation and exercise training programs in coronary heart disease. Prog. Cardiovasc. Dis. 2017, 60, 103–114. [Google Scholar] [CrossRef]
- Clark, A.M. What are the components of complex interventions in healthcare? Theorizing approaches to parts, powers and the whole intervention. Soc. Sci. Med. 2013, 93, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Altman, D.G.; Sterne, J.A.C. Chapter 8: Assessing Risk of Bias in Included Studies. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). Available online: http://handbook-5-1.cochrane.org (accessed on 14 June 2017).
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS. Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Hutton, B.; Salanti, G.; Caldwell, D.M.; Chaimani, A.; Schmid, C.H.; Cameron, C.; Ioannidis, J.P.; Straus, S.; Thorlund, K.; Jansen, J.P.; et al. The PRISMA extension statement for reporting of systematic reviews incorporating network metaanalyses of health care interventions: Checklist and explanations. Ann. Intern. Med. 2015, 162, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Balady, G.J.; Ades, P.A.; Comoss, P.; Limacher, M.; Pina, I.L.; Southard, D.; Williams, M.A.; Bazzarre, T. Core components of cardiac rehabilitation/secondary prevention programs: A statement for healthcare professionals from the American Heart Association and the American Association of Cardiovascular and Pulmonary Rehabilitation Writing Group. Circulation 2000, 102, 1069–1073. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Welton, N.J.; Caldwell, D.M.; Adamopoulos, E.; Vedhara, K. Mixed treatment comparison meta-analysis of complex interventions: Psychological interventions in coronary heart disease. Am. J. Epidemiol. 2009, 169, 1158–1165. [Google Scholar] [CrossRef] [PubMed]
- Franchini, A.J.; Dias, S.; Ades, A.E.; Jansen, J.P.; Welton, N.J. Accounting for correlation in network meta-analysis with multi-arm trials. Res. Synth. Methods 2012, 3, 142–160. [Google Scholar] [CrossRef] [PubMed]
- Dias, S.; Sutton, A.J.; Ades, A.E.; Welton, N.J. Evidence synthesis for decision making 2: A generalized linear modeling framework for pairwise and network meta-analysis of randomized controlled trials. Med. Decis. Mak. 2013, 33, 607–617. [Google Scholar] [CrossRef]
- Dias, S.; Sutton, A.J.; Welton, N.J.; Ades, A.E. Evidence synthesis for decision making 3: Heterogeneity—Subgroups, meta-regression, bias, and bias-adjustment. Med. Decis. Mak. 2013, 33, 618–640. [Google Scholar] [CrossRef]
- Dias, S.; Welton, N.J.; Sutton, A.J.; Caldwell, D.M.; Lu, G.; Ades, A.E. Evidence synthesis for decision making 4: Inconsistency in networks of evidence based on randomized controlled trials. Med. Decis. Mak. 2013, 33, 641–656. [Google Scholar] [CrossRef]
- Albus, C.; Theissen, P.; Hellmich, M.; Griebenow, R.; Wilhelm, B.; Aslim, D.; Schicha, H.; Köhle, K. Long-term effects of a multimodal behavioral intervention on myocardial perfusion-a randomized controlled trial. Int. J. Behav. Med. 2009, 16, 219–226. [Google Scholar] [CrossRef]
- Albus, C. A stepwise psychotherapy intervention to reduce risk in coronary artery disease (SPIRR-CAD)—Results of a randomised, controlled multicentre trial. J. Psychosom. Res. 2014, 76, 495. [Google Scholar] [CrossRef]
- Aldana, S.G.; Greenlaw, R.; Salberg, A.; Merrill, R.M.; Hager, R.; Jorgensen, R.B. The effects of an intensive lifestyle modification program on carotid artery intima-media thickness: A randomized trial. Am. J. Heal. Promot. 2007, 21, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Allison, T.G.; Farkouh, M.E.; Smars, P.A.; Evans, R.W.; Squires, R.W.; Gabriel, S.E.; Kopecky, S.L.; Gibbons, R.J.; Reeder, G.S. Management of coronary risk factors by registered nurses versus usual care in patients with unstable angina pectoris (a chest pain evaluation in the emergency room [CHEER] substudy). Am. J. Cardiol. 2000, 86, 133–138. [Google Scholar] [CrossRef]
- Andersson, A.; Sundel, K.L.; Undén, A.L.; Schenck-Gustafsson, K.; Eriksson, I. A five-year rehabilitation programme for younger women after a coronary event reduces the need for hospital care. Scand. J. Public Health 2010, 38, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Appels, A.; van Elderen, T.; Bär, F.; van der Pol, G.; Erdman, R.A.; Assman, M.; Trijsburg, W.; van Diest, R.; van Dixhoorn, J.; Pedersen, S.S. Effects of a behavioural intervention on quality of life and related variables in angioplasty patients. Results of the EXhaustion Intervention Trial. J. Psychosom. Res. 2006, 61, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Asbury, E.A.; Webb, C.M.; Collins, P. Group support to improve psychosocial well-being and primary-care demands among women with cardiac syndrome X. Climacteric 2011, 14, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Bambauer, K.Z.; Aupont, O.; Stone, P.H.; Locke, S.E.; Mullan, M.G.; Colagiovanni, J.; McLaughlin, T.J. The Effect of a Telephone Counseling Intervention on Self-Rated Health of Cardiac Patients. Psychosom. Med. 2005, 67, 539–545. [Google Scholar] [CrossRef]
- Barnason, S.; Zimmerman, L.; Nieveen, J.; Schulz, P.; Miller, C.; Hertzog, M.; Tu, C. Influence of a symptom management telehealth intervention on older adults’ early recovery outcomes after coronary artery bypass surgery. Heart Lung J. Acute Crit. Care 2009, 38, 364–376. [Google Scholar] [CrossRef]
- Beckie, T.M.; Beckstead, J.W. The effects of a cardiac rehabilitation program tailored for women on global quality of life: A randomized clinical trial. J. Womens Health 2010, 19, 1977–1985. [Google Scholar] [CrossRef]
- Belardinelli, R.B.; Paolini, P.; Cianci, G.C.G.; Piva, R.P.R.; Solenghi, M.S.M. The efficay of exercise training after coronary angioplasty: The ETICA trial. Eur. Heart. J. 2001, 22, 200. [Google Scholar]
- Belardinelli, R.; Paolini, I.; Cianci, G.; Piva, R.; Georgiou, D.; Purcaro, A. Exercise training intervention after coronary angioplasty: The ETICA trial. J. Am. Coll. Cardiol. 2001, 37, 1891–1900. [Google Scholar] [CrossRef]
- Bengtsson, K. Rehabilitation after myocardial infarction. A controlled study. Scand. J. Rehabil. Med. 1983, 15, 1–9. [Google Scholar] [CrossRef]
- Beresnevaite, M. Exploring the benefits of group psychotherapy in reducing alexithymia in coronary heart disease patients: A preliminary study. Psychother. Psychosom. 2000, 69, 117–122. [Google Scholar] [CrossRef]
- Berkman, L.F.; Blumenthal, J.; Burg, M.; Carney, R.M.; Catellier, D.; Cowan, M.J.; Czajkowski, S.M.; DeBusk, R.; Hosking, J.; Jaffe, A.; et al. Effects of treating depression and low perceived social support on clinical events after myocardial infarction the enhancing recovery in coronary heart disease patients (ENRICHD) randomized trial. JAMA 2003, 289, 3106–3116. [Google Scholar] [PubMed]
- Bertie, J.; King, A.; Reed, N.; Marshall, A.J.; Ricketts, C. Benefits and weaknesses of a cardiac rehabilitation programme. J. R. Coll. Physicians. Lond. 1992, 26, 147–151. [Google Scholar] [PubMed]
- Bethell, H.J.N.; Turner, S.C.; Mulleet, M.A. Cardiac rehabilitation in the community: 11-year follow-up after a randomized controlled trial. Coronary Health Care 1999, 3, 183–188. [Google Scholar] [CrossRef]
- Black, J.L.; Allison, T.G.; Williams, D.E.; Rummans, T.A.; Gau, G.T. Effect of intervention for psychological distress on rehospitalization rates in cardiac rehabilitation patients. Psychosomatics 1998, 39, 134–143. [Google Scholar] [CrossRef]
- Blasco, A.; Carmona, M.; Fernández-Lozano, I.; Salvador, C.H.; Pascual, M.; Sagredo, P.G.; Somolinos, R.; Muñoz, A.; García-López, F.; Escudier, J.M.; et al. Evaluation of a telemedicine service for the secondary prevention of coronary artery disease. J. Cardiopulm. Rehabil. Prev. 2012, 32, 25–31. [Google Scholar] [CrossRef]
- Blumenthal, J.A.; Jiang, W.; Babyak, M.A.; Krantz, D.S.; Frid, D.J.; Coleman, R.E.; Waugh, R.; Hanson, M.; Appelbaum, M.; O’Connor, C.; et al. Stress management and exercise training in cardiac patients with myocardial ischemia effects on prognosis and evaluation of mechanisms. Arch. Intern. Med. 1997, 157, 2213–2223. [Google Scholar] [CrossRef]
- Blumenthal, J.A.; Sherwood, A.; Smith, P.J.; Watkins, L.; Mabe, S.; Kraus, W.E.; Ingle, K.; Miller, P.; Hinderliter, A. Enhancing cardiac rehabilitation with stress management training: A randomized clinical efficacy trial. Circulation 2016, 133, 1341–1350. [Google Scholar] [CrossRef] [PubMed]
- Broadbent, E.; Ellis, C.J.; Thomas, J.; Gamble, G.; Petrie, K.J. Further development of an illness perception intervention for myocardial infarction patients: A randomized controlled trial. J. Psychosom. Res. 2009, 67, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Burgess, A.W.; Lerner, D.J.; D’Agostino, R.B.; Vokonas, P.S.; Hartman, C.R.; Gaccione, P. A randomized control trial of cardiac rehabilitation. Soc. Sci. Med. 1987, 24, 359–370. [Google Scholar] [CrossRef]
- Burr, M.L.; Ashfield-Watt, P.; Dunstan, F.; Fehily, A.M.; Breay, P.; Ashton, T.; Zotos, P.C.; Haboubi, N.A.; Elwood, P.C. Lack of benefit of dietary advice to men with angina: Results of a controlled trial. Eur. J. Clin. Nutr. 2003, 57, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Burell, G. Behaviour modification after coronary artery bypass graft surgery: Effects on cardiac morbidity and mortality. J. Rehabil. Sci. 1995, 8, 39–40. [Google Scholar]
- Byrkjeland, R.; Njerve, I.U.; Anderssen, S.; Arnesen, H.; Seljeflot, I.; Solheim, S. Effects of exercise training on HbA 1c and VO 2peak in patients with type 2 diabetes and coronary artery disease: A randomised clinical trial. Diabetes Vasc. Dis. Res. 2015, 12, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Carson Carson, P.; Phillips, R.; Lloyd, M.; Tucker, H.; Neophytou, M.; Buch, N.J.; Gelson, A.; Lawton, A.; Simpson, T. Exercise after myocardial infarction: A controlled trial. J. R. Coll. Physicians Lond. 1982, 16, 147–151. [Google Scholar] [PubMed]
- Chow, C.K.; Redfern, J.; Hillis, G.S.; Thakkar, J.; Santo, K.; Hackett, M.L.; Jan, S.; Graves, N.; de Keizer, L.; Barry, T.; et al. Effect of lifestyle-focused text messaging on risk factor modification in patients with coronary heart disease. JAMA 2015, 314, 1255–1263. [Google Scholar] [CrossRef] [PubMed]
- Claesson, M.; Birgander, L.S.; Jansson, J.H.; Lindahl, B.; Burell, G.; Asplund, K.; Mattsson, C. Cognitive-behavioural stress management does not improve biological cardiovascular risk indicators in women with ischaemic heart disease: A randomized-controlled trial. J. Intern. Med. 2006, 260, 320–331. [Google Scholar] [CrossRef] [PubMed]
- Clark, N.M.; Janz, N.K.; Dodge, J.A.; Schork, M.A.; Fingerlin, T.E.; Wheeler, J.R.C.; Liang, J.; Keteyian, S.J.; Santinga, J.T. Changes in functional health status of older women with heart disease: Evaluation of a program based on self-regulation. J. Gerontol. 2000, 55, S117–S126. [Google Scholar] [CrossRef]
- Cohen, A.; Assyag, P.; Boyer-Chatenet, L.; Cohen-Solal, A.; Perdrix, C.; Dalichampt, M.; Michel, P.L.; Montalescot, G.; Ravaud, P.; Steg, P.G.; et al. An Education Program for Risk Factor Management After an Acute Coronary Syndrome. JAMA Intern. Med. 2014, 174, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Costa e Silva, R.; Pellanda, L.; Portal, V.; Maciel, P.; Furquim, A.; Schaan, B. Transdiciplinary approach to the follow-up of patients after myocardial infarction. Clinics 2008, 63, 489–496. [Google Scholar] [PubMed]
- Cupples, M.E.; McKnigth, A. Five year follow up of patients at high cardiovascular risk who took part in randomised controlled trial of health promotion. BMJ 1999, 319, 687–688. [Google Scholar] [CrossRef] [PubMed]
- Dalal, H.M.; Evans, P.H.; Campbell, J.L.; Taylor, R.S.; Watt, A.; Read, K.L.; Mourant, A.J.; Wingham, J.; Thompson, D.R.; Pereira Gray, D.J. Home-based versus hospital-based rehabilitation after myocardial infarction: A randomized trial with preference arms—Cornwall Heart Attack Rehabilitation Management Study (CHARMS). Int. J. Cardiol. 2007, 119, 202–211. [Google Scholar] [CrossRef]
- Davidson, K.W.; Gidron, Y.; Mostofsky, E.; Trudeau, K.J. Hospitalization cost offset of a hostility intervention for coronary heart disease patients. J. Consult. Clin. Psychol. 2007, 75, 657–662. [Google Scholar] [CrossRef]
- Davidson, K.W.; Rieckmann, N.; Clemow, L.; Schwartz, J.E.; Shimbo, D.; Medina, V.; Albanese, G.; Kronish, I.; Hegel, M.; Burg, M.M. Enhanced depression care for patients with acute coronary syndrome and persistent depressive symptoms coronary psychosocial evaluation studies randomized controlled trial. Arch. Intern. Med. 2010, 170, 600–608. [Google Scholar] [CrossRef]
- De Lorgeril, M.; Salen, P.; Martin, J.-L.; Monjaud, I.; Delaye, J.; Mamelle, N. Mediterranean diet, traditional risk factors, and the rate of cardiovascular complications after myocardial infarction: Final report of the Lyon diet heart study. Circulation 1999, 99, 779–785. [Google Scholar] [CrossRef]
- DeBusk, R.F.; Miller, N.H.; Superko, H.R.; Dennis, C.A.; Thomas, R.J.; Lew, H.T.; Berger, W.E., III; Heller, R.S.; Rompf, J.; Gee, D.; et al. A Case-Management System for Coronary Risk Factor Modification after Acute Myocardial Infarction. Ann. Intern. Med. 1994, 120, 721–729. [Google Scholar] [CrossRef]
- Donohue, J.M.; Belnap, B.H.; Men, A.; He, F.; Roberts, M.S.; Schulberg, H.C.; Reynolds, C.F., III; Rollman, B.L. Twelve-month cost-effectiveness of telephone-delivered collaborative care for treating depression following cabg surgery: A randomized controlled. Gen. Hosp. Psychiatry 2014, 36, 453–459. [Google Scholar] [CrossRef]
- Du, L.; Dong, P.; Jia, J.; Li, Z.; Lai, L.; Yang, X.; Wang, S.; Yang, X.; Li, Z.; Shang, X.; et al. Impacts of intensive follow-up on the long-term prognosis of percutaneous coronary intervention in acute coronary syndrome patients—A single center prospective randomized controlled study in a Chinese population. Eur. J. Prev. Cardiol. 2016, 23, 1077–1085. [Google Scholar] [CrossRef]
- Dugmore, L.D.; Tipson, R.J.; Phillips, M.H.; Flint, E.J.; Stentiford, N.H.; Bone, M.F.; Littler, W.A. Changes in cardiorespiratory fitness, psychological wellbeing, quality of life, and vocational status following a 12 month cardiac exercise rehabilitation programme. Heart 1999, 81, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Edworthy, S.M.; Baptie, B.; Galvin, D.; Brant, R.F.; Churchill-Smith, T.; Manyari, D.; Belenkie, I. Effects of an enhanced secondary prevention program for patients with heart disease: A prospective randomized trial. Can. J. Cardiol. 2007, 23, 1066–1072. [Google Scholar] [CrossRef]
- Engblom, E.; Hletanent, E.K.; Hamalafnent, H.; Kallioj, V.; Lnberg, M.; Knutst, L.-R. Exercise habits and physical performance during comprehensive rehabilitation after coronary artery bypass surgery. Eur. Heart J. 1992, 13, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Erdman, R.A.M.; Duivenvoorden, H.J. Psychologic evaluation of a cardiac rehabilitation program: A randomized clinical trial in patients with myocardial infarction. J. Cardiac. Rehabil. 1983, 3, 696–704. [Google Scholar]
- Fihn, S.D.; Bucher, J.B.; McDonell, M.; Diehr, P.; Rumsfeld, J.S.; Doak, M.; Dougherty, C.; Gerrity, M.; Heidenreich, P.; Larsen, G.; et al. Collaborative care intervention for stable ischemic heart disease. Arch. Intern. Med. 2011, 171, 1471–1479. [Google Scholar] [CrossRef] [PubMed]
- Frasure-Smith, N.; Prince, R. The Ischemic heart disease life stress monitoring program: 18-month mortality results. Psychosom. Med. 1985, 47, 431–445. [Google Scholar] [CrossRef] [PubMed]
- Frasure-Smith, N.; Lespérance, F.; Prince, R.H.; Verrier, P.; Garber, R.A.; Juneau, M.; Wolfson, C.; Bourassa, M.G. Randomised trial of home-based psychosocial nursing intervention for patients recovering from myocardial infarction. Lancet 1997, 350, 473–479. [Google Scholar] [CrossRef]
- Fridlund, B.; Pihlgren, C.; Wannestig, L.B. A supportive–educative caring rehabilitation programme; improvements of physical health after myocardial infarction. J. Clin. Nurs. 1992, 1, 141–146. [Google Scholar] [CrossRef]
- Friedman, M.; Thoresen, C.E.; Gill, J.J.; Powell, L.H.; Ulmer, D.; Thompson, L.; Price, V.A.; Rabin, D.D.; Breall, W.S.; Dixon, T.; et al. of type A behavior and reduction in cardiac recurrences in postmyocardial infarction patients. Am. Heart J. 1984, 108, 237–248. [Google Scholar] [CrossRef]
- Froelicher, V.; Jensen, D.; Genter, F.; Sullivan, M.; McKirnan, M.D.; Witztum, K.; Scharf, J.; Strong, M.L.; Ashburn, W. A randomized trial of exercise training in patients with coronary heart disease. JAMA 1984, 252, 1291–1297. [Google Scholar] [CrossRef]
- Furber, S.; Butler, L.; Phongsavan, P.; Mark, A.; Bauman, A. randomised controlled trial of a pedometer-based telephone intervention to increase physical activity among cardiac patients not attending cardiac rehabilitation. Patient Educ. Couns. 2010, 80, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Furze, G.; Cox, H.; Morton, V.; Chuang, L.H.; Lewin, R.J.; Nelson, P.; Carty, R.; Norris, H.; Patel, N.; Elton, P. Randomized controlled trial of a lay-facilitated angina management programme. J. Adv. Nurs. 2012, 68, 2267–2279. [Google Scholar] [CrossRef] [PubMed]
- Ghroubi, S.; Elleuch, W.; Abid, L.; Kammoun, S.; Elleuch, M.-H. The effects of cardiovascular rehabilitation after coronary stenting. Ann. Phys. Rehabil. Med. 2012, 55, e309. [Google Scholar] [CrossRef]
- Giallauria, F.; Cirillo, P.; Lucci, R.; Pacileo, M.; Lorenzo, A.D.; D’Agostino, M.; Moschella, S.; Psaroudaki, M.; Del Forno, D.; Orio, F.; et al. Left ventricular remodelling in patients with moderate systolic dysfunction after myocardial infarction: Favourable effects of exercise training and predictive role of N-terminal pro-brain natriuretic peptide. Eur. J. Cardiovasc. Prev. Rehabil. 2008, 15, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Giannuzzi, P.; Tavazzi, L.; Temporelli, P.L.; Corrà, U.; Imparato, A.; Gattone, M.; Giordano, A.; Sala, L.; Schweiger, C.; Malinverni, C. Long-term physical training and left ventricular remodelling after anterior myocardial infraction: Results of the exercise in anterior myocardial infraction (EAMI) trial. J. Am. Coll. Cardiol. 1993, 22, 1821–1829. [Google Scholar] [CrossRef]
- Giannuzzi, P.; Temporelli, P.L.; Corra, U.; Gattone, M.; Giordano, A.; Tavazzi, L. Attenuation of unfavorable remodeling by exercise training in post infarction patients with left ventricular dysfunction: Results of the exercise in left ventricular dysfunction (ELVD) trial. Circulation 1997, 96, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Gortner, S.R.; Gilliss, C.L.; Shinn, J.A.; Sparacino, P.A.; Rankin, S.; Leavitt, M.; Price, M.; Hudes, M. Improving recovery following cardiac surgery: A randomized clinical trial. J. Adv. Nurs. 1988, 13, 649–661. [Google Scholar] [CrossRef]
- Gulliksson, M.; Burrel, G.; Vessby, B.; Lundin, L.; Toss, H.; Svardsudd, K. Randomized controlled trial of cognitive behavioral therapy vs standard treatment to prevent recurrent cardiovascular events in patients with coronary heart disease. secondary prevention in Uppsala primary health care project (SUPRIM). Arch. Inter. Med. 2011, 171, 134–140. [Google Scholar] [CrossRef]
- Gutschker, A.; Schaller, K.; Geissler, W. Results of physical conditioning in patients with acute myocardial infarction over 65 years of age. Cardiology 1977, 62, 109. [Google Scholar]
- Hämäiäinen, H.; Kallio, V.; Knuts, L.R.; Arstila, M.; Aaito-Setälä, L.; Härmälä, V.; Kaarsalo, E.; Laurila, S.; Pere, E. Community approach in rehabilitation and secondary prevention after acute myocardial infarction: Results of a randomized clinical trial. J. Cardiopulm. Rehabil. 1991, 11, 221–226. [Google Scholar] [CrossRef]
- Hambrecht, R.; Niebauer, J.; Marburger, C.; Grunze, M.; Kälberer, B.; Hauer, K.; Schlierf, G.; Kübler, W.; Schuler, G. Various intensities of leisure time physical activity in patients with coronary artery disease: Effects on cardiorespiratory fitness and progression of coronary atherosclerotic lesions. J. Am. Coll. Cardiol. 1993, 22, 468–477. [Google Scholar] [CrossRef]
- Hanssen, T.A.; Nordrehaug, J.E.; Eide, G.E.; Hanestad, B.R. Improving outcomes after myocardial infarction: A randomized controlled trial evaluating effects of a telephone follow-up intervention. Eur. J. Cardiovasc. Prev. Rehabil. 2007, 14, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Harald, T.; Reimer, O.; Tijssen, J.G.P.; Peters, R.J.G. Non-Cardiac Readmissions Are Reduced in Patients Attending a Nurse-Coordinated Prevention Program: Results from the RESPONSE Randomised Clinical Trial; EuroPrevent Congress of the European Association of Preventive Cardiology: Lisbon, Portugal, 2015. [Google Scholar]
- Haskell, W.L.; Alderman, E.L.; Fair, J.M.; Maron, D.J.; Mackey, S.F.; Superko, H.R.; Williams, P.T.; Johnstone, I.M.; Champagne, M.A.; Krauss, R.M.; et al. Effects of intensive multiple risk factor reduction on coronary atherosclerosis and clinical cardiac events in men and women with coronary artery disease. The Stanford Coronary Risk Intervention Project (SCRIP). Circulation 1994, 89, 975–990. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, A.L.; Patrao, T.A.; Atherton, J.; Ware, R.S.; Taylor, C.B.; O’Neil, A.; Foreman, R.; Oldenburg, B.F. Effect of a telephone-delivered coronary heart disease secondary prevention program (ProActive Heart) on quality of life and health behaviours: Primary outcomes of a randomised controlled trial. Int. J. Behav. Med. 2013, 20, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Heller, R.F.; Knapp, J.C.; Valenti, L.A.; Dobson, A.J. Secondary prevention after acute myocardial infarction. Am. J. Cardiol. 1993, 72, 759–762. [Google Scholar] [CrossRef]
- Higgins, H.C.; Hayes, R.L.; Mckenna, K.T. Rehabilitation outcomes following percutaneous coronary interventions (PCI). Patient Educ. Couns. 2001, 43, 219–230. [Google Scholar] [CrossRef]
- Hofman-Bang, C.; Lisspers, J.; Nordlander, R.; Nygren, A.; Sundin, O.; Ohman, A.; Rydén, L. Two-year results of a controlled study of residential rehabilitation for patients treated with percutaneous transluminal coronary angioplasty. A randomized study of a multifactorial programme. Eur. Heart J. 1999, 20, 1465–1474. [Google Scholar] [CrossRef]
- Jolly, K.; Taylor, R.S.; Lip, G.Y.H.; Singh, S. Reproducibility and safety of the incremental shuttle walking test for cardiac rehabilitation. Int. J. Cardiol. 2008, 125, 144–145. [Google Scholar] [CrossRef]
- Jones, D.A.; West, R.R. Psychological rehabilitation after myocardial infarction: Multicentre randomised controlled trial. BMJ 1996, 313, 1517–1521. [Google Scholar] [CrossRef]
- Kallio, V.; Hämäläinen, H.; Hakkila, J.; Luurila, O. Reduction in sudden deaths by a multifactorial intervention programme after acute myocardial infarction. Lancet 1979, 314, 1091–1094. [Google Scholar] [CrossRef]
- Karlsson, M.R.; Edström-Plüss, C.; Held, C.; Henriksson, P.; Billing, E.; Wallén, N.H. Effects of expanded cardiac rehabilitation on psychosocial status in coronary artery disease with focus on typed characteristics. J. Behav. Med. 2007, 30, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Koertge, J.; Janszky, I.; Sundin, Ö.; Blom, M.; Georgiades, A.; László, K.D.; Alinaghizadeh, H.; Ahnve, S. Effects of a stress management program on vital exhaustion and depression in women with coronary heart disease: A randomized controlled intervention study. J. Intern. Med. 2008, 263, 281–293. [Google Scholar] [CrossRef]
- Kure, C.E.; Ski, C.F.; Stewart, S.; Chan, Y.K.; Carrington, M.J.; Thompson, D.R. Potential impact of depression on health outcomes in a randomised control trial of multidisciplinary, nurse-led, home based intervention (HBI) to reduce secondary cardiac events. Glob. Heart 2014, 9, e29. [Google Scholar] [CrossRef]
- La Rovere, M.T.; Bersano, C.; Gnemmi, M.; Specchia, G.; Schwartz, P.J. Exercise-induced increase in baroreflex sensitivity predicts improved prognosis after myocardial infarction. Circulation 2002, 106, 945–949. [Google Scholar] [CrossRef] [PubMed]
- Leemrijse, C.J.; Peters, R.J.G.; von Birgelen, C.; van Dijk, L.; van Hal, J.M.; Kuijper, A.F.; Snaterse, M.; Veenhof, C. The telephone lifestyle intervention ‘Hartcoach’ has modest impact on coronary risk factors: A randomised multicentre trial. Eur. J. Prev. Cardiol. 2016, 23, 1658–1668. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, N.; Paul, A.; Moebus, S.; Budde, T.; Dobos, G.J.; Michalsen, A. Effects of lifestyle modification on coronary artery calcium progression and prognostic factors in coronary patients 3-Year results of the randomized SAFE-LIFE trial. Atherosclerosis 2011, 219, 630–636. [Google Scholar] [CrossRef] [PubMed]
- Leizorovicz, A.; Sait-Pierre, A.; Vasselon, C.; Boissel, J.P. Comparison of a rehabilitation programme, a counselling programme and usual care after an acute myocardial infarction: Results of a long-term randomized trial. Eur. Heart J. 1991, 12, 612–616. [Google Scholar] [CrossRef]
- Lewin, B.; Robertson, I.H.; Cay, E.L.; Irving, J.B.; Campbell, M. Effects of self-help post-myocardial-irfaction rehabilitation on psychological adjustment and use of health services. Lancet 1992, 339, 1036–1040. [Google Scholar] [CrossRef]
- Lewin, R.J.P.; Furze, G.; Robinson, J.; Griffith, K.; Wiseman, S.; Pye, M.; Boyle, R. A randomised controlled trial of a self-management plan for patients with newly diagnosed angina. Br. J. Gen. Pract. 2002, 52, 194–196. [Google Scholar]
- Lidell, E.; Fridlund, B. Long-term Effects of a Comprehensive Rehabilitation Programme after Myocardial Infarction. Scand. J. Caring Sci. 1996, 10, 67–74. [Google Scholar] [CrossRef]
- Lie, I.; Arnesen, H.; Sandvik, L.; Hamilton, G.H.; Bunch, E. Effects of a home-based intervention program on anxiety and depression 6 months after coronary artery bypass grafting: A randomized controlled trial. J. Psychosom. Res. 2007, 62, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Yaseri, M.; Pakpour, A.H.; Malm, D.; Broström, A.; Fridlund, B.; Burri, A.; Webb, T.L. Can a multifaceted intervention including motivational interviewing improve medication adherence, quality of life, and mortality rates in older patients undergoing coronary artery bypass surgery? A multicenter, randomized controlled trial with 18-month follow-up. Drugs Aging 2017, 34, 143–156. [Google Scholar] [PubMed]
- Manchanda, S.C.; Narang, R.; Reddy, K.S.; Sachdeva, U.; Prabhakaran, D.; Dharmanand, S.; Rajani, M.; Bijlani, R. Retardation of coronary atherosclerosis with yoga lifestyle intervention. J. Assoc. Physicians India 2000, 48, 687–694. [Google Scholar] [PubMed]
- Marcus, S.; Orha, I.; Cioranu, R.; Apetrei, E. Long-Term (25 Years) Efficacy of Comprehensive Post Myocardial Infarction Rehabilitation Program; XXI Congress of the European Society of Cardiology: Bologna, Spain, 1999. [Google Scholar]
- Maroto-Montero, J.M.; Ramírez, R.A.; Morales-Durán, M.D.; de Pablo Zarzosa, C.; Abraira, V. Cardiac rehabilitation in patients with myocardial infarction: A 10-year follow-up study. Rev. Esp. Cardiol. 2005, 58, 1181–1187. [Google Scholar] [CrossRef]
- Marra, S.; Paolillo, V.; Spadaccini, F.; Angelino, P.F. Long-term follow-up after a controlled randomized post-myocardial infarction rehabilitation programme: Effects on morbidity and mortality. Eur. Heart J. 1985, 6, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Mayou, R.A.; Thompson, D.R.; Clements, A.; Davies, C.H.; Goodwin, S.J.; Normington, K.; Hicks, N.; Price, J. Guideline-based early rehabilitation after myocardial infarction. J. Psychosom. Res. 2002, 52, 89–95. [Google Scholar] [CrossRef]
- Meisinger, C.; Stollenwerk, B.; Kirchberger, I.; Seidl, H.; Wende, R.; Kuch, B.; Holle, R. Effects of a nurse-based case management compared to usual care among aged patients with myocardial infarction: Results from the randomized controlled KORINNA study. BMC Geriatr. 2013, 13, 115. [Google Scholar] [CrossRef]
- Michalsen, A.; Grossman, P.; Lehmann, N.; Knoblauch, N.T.; Paul, A.; Moebus, S.; Budde, T.; Dobos, G.J. Psychological and quality-of-life outcomes from a comprehensive stress reduction and lifestyle program in patients with coronary artery disease: Results of a randomized trial. Psychother. Psychosom. 2005, 74, 344–352. [Google Scholar] [CrossRef]
- Miller, N.H.; Haskell, W.L.; Berra, K.; Debusk, R.F.; Iii, B.; Lew, H. Home versus group exercise training for increasing functional capacity after myocardial infarction. Circulation 1984, 70, 645–649. [Google Scholar] [CrossRef]
- Mittag, O.; China, C.; Hoberg, E.; Juers, E.; Kolenda, K.D.; Richardt, G.; Maurischat, C.; Raspe, H. Outcomes of cardiac rehabilitation with versus without a follow-up intervention rendered by telephone (Luebeck follow-up trial): Overall and gender-specific effects. Int. J. Rehabil. Res. 2006, 29, 295–302. [Google Scholar] [CrossRef]
- Mohiuddin, S.M.; Mooss, A.N.; Hunter, C.B.; Grollmes, T.L.; Cloutier, D.A.; Hilleman, D.E. Intensive smoking cessation intervention reduces mortality in high-risk smokers with cardiovascular disease. Chest 2007, 131, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Moholdt, T.; Bekken, V.M.; Grimsmo, J.; Slørdahl, S.A.; Wisløff, U. Home-Based aerobic interval training improves peak oxygen uptake rqual to residential cardiac rehabilitation: S randomized, controlled trial. PLoS. One 2012, 7, e41199. [Google Scholar] [CrossRef] [PubMed]
- Molino-Lova, R.; Pasquini, G.; Vannetti, F.; Paperini, A.; Forconi, T.; Polcaro, P.; Zipoli, R.; Cecchi, F.; Macchi, C. Effects of a structured physical activity intervention on measures of physical performance in frail elderly patients after cardiac rehabilitation: A pilot study with 1-year follow-up. Intern. Emerg. Med. 2013, 8, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Muñiz, J.; Gómez-Doblas, J.J.; Santiago-Pérez, M.I.; Lekuona-Goya, I.; Murga-Eizagaetxebarría, N.; de Teresa-Galván, S.S.E.; Cruz-Fernández, J.M.; Castro-Beiras, A.; CAM2 Project Working Group. The effect of post-discharge educational intervention on patients in achieving objectives in modifiable risk factors six months after discharge following an episode of acute coronary syndrome, (CAM-2 Project): A randomized controlled trial. Health Qual. Life Outcomes 2010, 8, 137. [Google Scholar]
- Munk, P.S.; Staal, E.M.; Butt, N.; Isaksen, K.; Larsen, A.I. High-intensity interval training may reduce in-stent restenosis following percutaneous coronary intervention with stent implantation. Am. Heart J. 2009, 158, 734–741. [Google Scholar] [CrossRef] [PubMed]
- Murphy, A.W.; Cupples, M.E.; Smith, S.M.; Byrne, M.; Byrne, M.C.; Newell, J. Effect of tailored practice and patient care plans on secondary prevention of heart disease in general practice: Cluster randomised controlled trial. BMJ 2009, 339, b4220. [Google Scholar] [CrossRef] [PubMed]
- Mutwalli, H.; Fallows, S.; Ashmeg, A.; Abukhudair, W.; Arnous, A.; Zamzami, M. A controlled trial of home-based cardiac rehabilitation versus usual hospital care in cardiac patients. J. Saudi Heart Assoc. 2012, 24, 286. [Google Scholar] [CrossRef]
- Myers, J.; Goebbels, U.; Dzeikan, G.; Froelicher, V.; Bremerich, J.; Mueller, P.; Buser, P.; Dubach, P. Exercise training and myocardial remodeling in patients with reduced ventricular function: One-year follow-up with magnetic resonance imaging. Am. Heart J. 2000, 139, 252–261. [Google Scholar] [CrossRef]
- Naughton, J.; Dorn, J.; Oberman, A.; Gorman, P.A.; Cleary, P. Maximal exercise systolic pressure, exercise training, and mortality in myocardial infarction patients. Am. J. Cardiol. 2000, 85, 416–420. [Google Scholar] [CrossRef]
- Neubeck, L.; Freedman, S.; Briffa, T.; Bauman, A.; Redfern, J. Four-year follow-up of the choice of health options in prevention of cardiovascular events randomized controlled trial. Eur J Cardiovasc Prev. Rehabil. 2011, 18, 278–286. [Google Scholar] [CrossRef]
- Oerkild, B.; Frederiksen, M.; Hansen, J.F.; Prescott, E. Home-based cardiac rehabilitation is an attractive alternative to no cardiac rehabilitation for elderly patients with coronary heart disease: Results from a randomised clinical trial. BMJ Open 2012, 2, 1820. [Google Scholar] [CrossRef] [PubMed]
- Oldenburg, B.; Perkins, R.J.; Andrews, G. Controlled trial of psychological intervention in myocardial infarction. J. Consul. Clin. Psychol. 1985, 53, 852–859. [Google Scholar] [CrossRef]
- Oldridge, N.; Guyatt, G.; Jones, N.; Crowe, J.; Singer, J.; Feeny, D.; McKelvie, R.; Runions, J.; Streiner, D.; Torrance, G. Effects on quality of life with comprehensive rehabilitation after acute myocardial infarction. Am. J. Cardiol. 1991, 67, 1084–1089. [Google Scholar] [CrossRef]
- Oranta, O.; Luutonen, S.; Salokangas, R.K.; Vahlberg, T.; Leino-Kilpi, H. The effects of interpersonal counselling on health-related quality of life after myocardial infarction. J. Clin. Nurs. 2011, 20, 3373–3382. [Google Scholar] [CrossRef]
- Ornish, D.; Brown, S.E.; Billings, J.H.; Billings, J.H.; Armstrong, W.T.; Ports, T.A.; McLanahan, S.M.; Kirkeeide, R.L.; Brand, R.J.; Gould, K.L. Can lifestyle changes reverse coronary heart disease? Lancet 1990, 336, 129–133. [Google Scholar] [CrossRef]
- Orth-Gomer, K.; Schneiderman, N.; Wang, H.X.; Walldin, C.; Blom, M.; Jernberg, T. Stress reduction prolongs life in women with coronary disease: The stockholm women’s intervention trial for coronary heart disease (SWITCHD). Circ. Cardiovasc. Qual. Outcomes 2009, 2, 25–32. [Google Scholar] [CrossRef]
- Otterstad, J.E. Influence on lifestyle measures and five-year coronary risk by a comprehensive lifestyle intervention programme in patients with coronary heart disease. Eur. J. Cardiovasc. Prev. Rehabil. 2003, 10, 429–437. [Google Scholar] [CrossRef]
- Park, J.H.; Tahk, S.J.; Bae, S.H.; Son, Y.J. Effects of a psychoeducational intervention for secondary prevention in Korean patients with coronary artery disease: A pilot study. Int. J. Nurs. Pract. 2013, 19, 295–305. [Google Scholar] [CrossRef]
- Raffo, J.A.; Luksic, I.Y.; Kappagoda, C.T.; Mary, D.A.S.G.; Whitaker, W.; Linden, R.J. Effects of physical training on myocardial ischaemia in patients with coronary artery disease. Br. Heart J. 1980, 43, 262–269. [Google Scholar] [CrossRef]
- Rahe, R.H.; O’Neil, T.; Hagan, A.; Arthur, R.J. Brief group therapy following myocardial infarction: Eighteen-month follow-up of a controlled trial. Int. J. Psychiatry Med. 1975, 6, 349–358. [Google Scholar] [CrossRef]
- Rakowska, J.M. Brief strategic therapy in first myocardial infarction patients with increased levels of stress: A randomized clinical trial. Anxiety Stress Coping 2015, 28, 687–705. [Google Scholar] [CrossRef] [PubMed]
- Redfern, J.; Briffa, T.; Ellis, E.; Freedman, S.B. Choice of secondary prevention improves risk factors after acute coronary syndrome: 1-year follow-up of the CHOICE (Choice of Health Options In prevention of Cardiovascular Events) randomised controlled trial. Heart 2009, 95, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Rollman, B.L.; Belnap, B.H.; LeMenager, M.S.; Mazumdar, S.; Houck, P.R.; Counihan, P.J.; Kapoor, W.N.; Schulberg, H.C.; Reynolds, C.F., III. Telephone-delivered collaborative care for treating post-cabg depression a randomized controlled trial. JAMA 2009, 302, 2095–2103. [Google Scholar] [CrossRef] [PubMed]
- Roman, O.; Gutierrez, M.; Luksic, I.; Chavez, E.; Camuzzi, A.L.; Villalón, E.; Klenner, C.; Cumsille, F. Cardiac rehabilitation after acute myocardial infarction: 9-year controlled follow-up study. Cardiology 1983, 70, 223–231. [Google Scholar] [PubMed]
- Roncella, A.; Pristipino, C.; Cianfrocca, C.; Scorza, S.; Pasceri, V.; Pelliccia, F.; Denollet, J.; Pedersen, S.S.; Speciale, G. One-year results of the randomized, controlled, short-term psychotherapy in acute myocardial infarction (STEP-IN-AMI) trial. Int. J. Cardiol. 2013, 170, 132–139. [Google Scholar] [CrossRef]
- Saffi, M.A.L.; Polanczyk, C.A.; Rabelo-Silva, E.R. Lifestyle interventions reduce cardiovascular risk in patients with coronary artery disease: A randomized clinical trial. Eur. J. Cardiovasc. Nurs. 2014, 13, 436–443. [Google Scholar] [CrossRef]
- Salminen, M.; Isoaho, R.; Vahlberg, T.; Ojanlatva, A.; Kivelä, S.-L. Effects of a health advocacy, counselling, and activation programme on depressive symptoms in older coronary heart disease patients. Int. J. Geriatr. Psychiatry 2005, 20, 552–558. [Google Scholar] [CrossRef]
- Santaularia, N.; Caminal, J.; Arnau, A.; Perramon, M.; Montesinos, J.; Abenoza Guardiola, M.; Jaarsma, T. The efficacy of a supervised exercise training programme on readmission rates in patients with myocardial ischemia: Results from a randomised controlled trial. Eur. J. Cardiovasc. Nurs. 2017, 16, 201–212. [Google Scholar] [CrossRef]
- Schneider, R.H.; Grim, C.E.; Rainforth, M.V.; Kotchen, T.; Nidich, S.I.; Gaylord-King, C.; Salerno, J.W.; Kotchen, J.M.; Alexander, C.N. Stress reduction in the secondary prevention of cardiovascular disease: Randomized, controlled trial of transcendental meditation and health education in blacks. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 750–758. [Google Scholar] [CrossRef]
- Schuler, G.; Hambrecht, R.; Schlierf, G.; Niebauer, J.; Hauer, K.; Neumann, J.; Hoberg, E.; Drinkmann, A.; Bacher, F.; Grunze, M.; et al. Regular physical exercise and low-fat diet. Effects on progression of coronary artery disease. Circulation 1992, 86, 1–11. [Google Scholar] [CrossRef]
- Sebregts, E.H.W.J.; Falger, P.R.J.; Appels, A.; Kester, A.D.M.; Bär, F.W.H.M. Psychological effects of a short behavior modification program in patients with acute myocardial infarction or coronary artery bypass grafting. A randomized controlled trial. J. Psychosom. Res. 2005, 58, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.W.; Oberman, A.; Barnes, G.; Eggert, D.; Barton, S.N.; Hellerstein, H.; Insua, J.; Yoran, C.; Fardy, P.; Franklin, B.A.; et al. Effects of a prescribed supervised exercise program on mortality and cardiovascular morbidity in patients after a myocardial infarction the national exercise and heart disease project. Am. J. Cardiol. 1981, 48, 39–46. [Google Scholar] [CrossRef]
- Singh, R.B.; Rastogi, S.S.; Verma, R.; Laxmi, B.; Singh, R.; Ghosh, S.; Niaz, M.A. Randomised controlled trial of cardioprotective diet in patients with recent acute myocardial infarction: Results of one year follow up. BMJ 1992, 304, 1015–1019. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.B.; Dubnov, G.; Niaz, M.A.; Ghosh, S.; Singh, R.; Rastogi, S.S.; Manor, O.; Pella, D.; Berry, E.M. Effect of an Indo-Mediterranean diet on progression of coronary artery disease in high risk patients (Indo-Mediterranean Diet Heart Study): A randomised single-blind trial. Lancet 2002, 360, 1455–1461. [Google Scholar] [CrossRef]
- Sivarajan, E.S.; Bruce, R.A.; Lindskog, B.D.; Almes, M.J.; Belanger, L.; Green, B. Treadmill test responses to an early exercise program after myocardial infarction: A randomized study. Circulation 1982, 65, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Specchia, G.; De Servi, S.; Scirè, A.; Assandri, J.; Berzuini, C.; Angoli, L.; La Rovere, M.T.; Cobelli, F. Interaction between exercise training and ejection fraction in predicting prognosis after a first myocardial infarction. Circulation 1996, 94, 978–982. [Google Scholar] [CrossRef]
- Stahle, A.; Mattsson, E.; Ryden, L.; Unden, A.L.; Nordlander, R. Improved physical fitness and quality of life following training of elderly patients after acute coronary events—A 1-year follow-up randomized controlled study. Eur. Heart J. 1999, 20, 1475–1484. [Google Scholar] [CrossRef]
- Stern, M.J.; Gorman, P.A.; Kaslow, L. The group counseling v exercise therapy study a controlled intervention with subjects following myocardial infarction. Arch. Intern. Med. 1983, 143, 1719–1725. [Google Scholar] [CrossRef]
- Strandberg, T.E.; Pitkala, K.H.; Berglind, S.; Nieminen, M.S.; Tilvis, R.S. Multifactorial intervention to prevent recurrent cardiovascular events in patients 75 years or older: The drugs and evidence-based medicine in the elderly (DEBATE) study: A randomized, controlled trial. Am. Heart J. 2006, 152, 585–592. [Google Scholar] [CrossRef]
- Todd, C.; Bradnam, M.S.; Cooke, M.B.; Ballantyne, D. Effects of daily high-intensity exercise on myocardial perfusion in angina pectoris. Am. J. Cardiol. 1991, 68, 1593–1599. [Google Scholar] [CrossRef]
- Toobert, D.J.; Glasgow, R.E.; Radcliffe, J.L. Physiologic and related behavioral outcomes from the women’s lifestyle heart trial. Ann. Behav. Med. 2000, 22, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Trzcieniecka-Green, A.; Steptoe, A. The effects of stress management on the quality of life of patients following acute myocardial infarction or coronary bypass surgery. Eur. Heart J. 1996, 17, 1663–1670. [Google Scholar] [CrossRef] [PubMed]
- Turner, A.; Hambridge, J.; Baker, A.; Bowman, J.; McElduff, P. Randomised controlled trial of group cognitive behaviour therapy versus brief intervention for depression in cardiac patients. Aust. N. Z. J. Psychiatry 2013, 47, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Vahedian-Azimi, A.; Miller, A.C.; Hajiesmaieli, M.; Kangasniemi, M.; Alhani, F.; Jelvehmoghaddam, H.; Fathi, M.; Farzanegan, B.; Ardehali, S.H.; Hatamian, S.; et al. Cardiac rehabilitation using the Family-Centered Empowerment Model versus home-based cardiac rehabilitation in patients with myocardial infarction: A randomised controlled trial. Open Heart 2016, 3, e000349. [Google Scholar] [CrossRef]
- Vale, M.J.; Jelinek, M.V.; Best, J.D.; Dart, A.M.; Grigg, L.E.; Hare, D.L.; Ho, B.P.; Newman, R.W.; McNeil, J.J.; COACH Study Group. Coaching patients On Achieving Cardiovascular Health (COACH): A multicenter randomized trial in patients with coronary heart disease. Arch. Intern. Med. 2003, 163, 2775–2783. [Google Scholar] [CrossRef]
- Van Dlxhoorn, J.; Duivenvoordenf, H.J.; Staal, J.A.; Poolf, J.; Verhagef, F. Cardiac events after myocardial infarction: Possible effect of relaxation therapy. Eur. Heart J. 1987, 8, 1210–1214. [Google Scholar] [CrossRef]
- Vermeulen, A.; Lie, K.I.; Durrer, D. Effects of cardiac rehabilitation after myocardial infarction: Changes in coronary risk factors and long-term prognosis. Am. Heart J. 1983, 105, 798–801. [Google Scholar] [CrossRef]
- Vona, M.; Chapuis, L.; Iannino, T.; Ferrari, E.; Von Segesser, L.K. Efficacy of two long-term intervention strategies to promote long-term adherence to lifestyle changes and to reduce cardiovascular events in patients with coronary artery disease. Eur. Heart J. 2009, 30, 474. [Google Scholar]
- Vona, M.; Iannino, T.; De Pascalis, S.; Gallardo, C.; Cook, S.; Vermeulen, B. Impact of endothelial dysfunction improvement after exercise training on prognosis in patients with recent myocardial infarction. Circulation 2010, 122, A13764. [Google Scholar]
- West, R.R.; Jones, D.A.; Henderson, A.H. Rehabilitation after myocardial infarction trial (RAMIT): Multi-centre randomised controlled trial of comprehensive cardiac rehabilitation in patients following acute myocardial infarction. Heart 2012, 98, 637–644. [Google Scholar] [CrossRef]
- Wheeler, J.R.; Janz, N.K.; Dodge, J.A. Can a disease self-management program reduce health care costs? The case of older women with heart disease. Med. Care 2003, 41, 706–715. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmsen, L.; Sanne, H.; Elmfeldt, D.; Grimby, G.; Tibblin, G.; Wedel, H. A controlled trial of physical training after myocardial infarction effects on risk factors, nonfatal reinfarction, and death. Prev. Med. 1975, 4, 491–508. [Google Scholar] [CrossRef]
- Xavier, D.; Gupta, R.; Kamath, D.; Sigamani, A.; Devereaux, P.J.; George, N.; Joshi, R.; Pogue, J.; Pais, P.; Yusuf, S. Community health worker-based intervention for adherence to drugs and lifestyle change after acute coronary syndrome: A multicentre, open, randomised controlled trial. Lancet Diabetes Endocrinol. 2016, 4, 244–253. [Google Scholar] [CrossRef]
- Yu, C.M.; Lau, C.P.; Chau, J.; McGhee, S.; Kong, S.L.; Cheung, B.M.; Li, L.S. A short course of cardiac rehabilitation program is highly cost effective in improving long-term quality of life in patients with recent myocardial infarction or percutaneous coronary intervention. Arch. Phys. Med. Rehabil. 2004, 85, 1915–1922. [Google Scholar] [CrossRef] [PubMed]
- Zetta, S.; Smith, K.; Jones, M.; Allcoat, P.; Sullivan, F. Evaluating the angina plan in patients admitted to hospital with angina: A randomized controlled trial. Cardiovasc. Ther. 2011, 29, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.X.; Ho, S.C.; Sit, J.W.H.; He, H.G. The effects of a transtheoretical model-based exercise stage-matched intervention on exercise behavior in patients with coronary heart disease: A randomized controlled trial. Patient Educ. Couns. 2014, 95, 384–392. [Google Scholar] [CrossRef]
- Abell, B.; Glasziou, P.; Hoffmann, T. The contribution of individual exercise training components to clinical outcomes in randomised controlled trials of cardiac rehabilitation: A systematic review and meta-regression. Sports Med. Open 2017, 3, 19. [Google Scholar] [CrossRef]
- Anderson, L.; Sharp, G.A.; Norton, R.J.; Dalal, H.; Dean, S.G.; Jolly, K.; Cowie, A.; Zawada, A.; Taylor, R.S. Home-based versus centre-based cardiac rehabilitation. Cochrane Database Syst. Rev. 2017, 6, CD007130. [Google Scholar] [CrossRef]
- Kraal, J.J.; Vromen, T.; Spee, R.; Kemps, H.M.C.; Peek, N. The influence of training characteristics on the effect of exercise training in patients with coronary artery disease: Systematic review and meta-regression analysis. Int. J. Cardiol. 2017, 245, 52–58. [Google Scholar] [CrossRef]
- Rauch, B.; Davos, C.H.; Doherty, P.; Saure, D.; Metzendorf, M.I.; Salzwedel, A.; Völler, H.; Jensen, K.; Schmid, J.P. The prognostic effect of cardiac rehabilitation in the era of acute revascularisation and statin therapy: A systematic review and meta-analysis of randomized and non-randomized studies—The Cardiac Rehabilitation Outcome Study (CROS). Eur. J. Prev. Cardiol. 2016, 23, 1914–1939. [Google Scholar] [CrossRef]
- Powell, R.; Mcgregor, G.; Ennis, S.; Kimani, P.K.; Underwood, M. Is exercise-based cardiac rehabilitation effective? A systematic review and meta-analysis to re-examine the evidence. BMJ Open 2018, 8, e019656. [Google Scholar]
- Clark, A.M.; Harding, L.; Vandermeer, B.; McAlister, F.A. Meta-analysis: Secondary prevention programs for patients with coronary artery disease. Ann. Intern. Med. 2005, 143, 659–672. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, G.T.; Buring, J.E.; Yusuf, S.; Goldhaber, S.Z.; Olmstead, E.M.; Paffenbarger, R.S., Jr.; Hennekens, C.H. An overview of randomized trials of rehabilitation with exercise after myocardial infarction. Circulation 1989, 80, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Oldridge, N.B.; Guyatt, G.H.; Fischer, M.E.; Rimm, A.A. Cardiac rehabilitation after myocardial infarction. Combined experience of randomized clinical trials. JAMA 1988, 260, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, J.A.; Rees, K.; Taylor, R.S.; Thompson, D.; Oldridge, N.; Ebrahim, S. Exercise-based rehabilitation for coronary heart disease. Cochrane Database Syst. Rev. 2001, CD001800. [Google Scholar] [CrossRef]
- Heran, B.S.; Chen, J.M.; Ebrahim, S.; Moxham, T.; Oldridge, N.; Rees, K.; Thompson, D.R.; Taylor, R.S. Exercise-based cardiac rehabilitation for coronary heart disease. Cochrane Database Syst. Rev. 2011, CD001800. [Google Scholar] [CrossRef]
- Oldridge, N. Exercise-based cardiac rehabilitation in patients with coronary heart disease: Meta-analysis outcomes revisited. Future Cardiol. 2012, 8, 729–751. [Google Scholar] [CrossRef]
- Whalley, B.; Thompson, D.R.; Taylor, R.S. Psychological interventions for coronary heart disease: Cochrane systematic review and meta-analysis. Int. J. Behav. Med. 2014, 21, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.; Brown, J.P.; Clark, A.M.; Dalal, H.; Rossau, H.K.; Bridges, C.; Taylor, R.S. Patient education in the management of coronary heart disease. Cochrane Database Syst. Rev. 2017, 6, CD008895. [Google Scholar] [CrossRef]
- Chaves, G.S.S.; Ghisi, G.L.M.; Grace, S.L.; Oh, P.; Ribeiro, A.L.; Britto, R.R. Effects of comprehensive cardiac rehabilitation on functional capacity in a middle-income country: A randomised controlled trial. Heart 2018, 1–8. [Google Scholar] [CrossRef]
- Pesah, E.; Turk-Adawi, K.; Supervia, M.; Lopez Jimenez, F.; Bogdanska, N.; Grace, S.L. Cardiac rehabilitation delivery in low- and middle-income countries: Availability, characteristics and barriers. World NCDs Congress. Chandigarh, India. November. Int. J. Non-Commun. Dis. 2017, 2, s57–s58. [Google Scholar]
- Supervia, M.; Turk-Adawi, K.; Lopez Jimenez, F.; Pesah, E.; Rongjing, D.; Britto, R.; Bjarnason-Wehrens, B.; Derman, W.; Abreu, A.; Babu, A.; et al. Nature of cardiac rehabilitation services around the globe. eClinicalMedicine 2018. under review. [Google Scholar]
- Turk-Adawi, K.; Sarrafzadegan, N.; Grace, S.L. Global availability of cardiac rehabilitation. Nat. Rev. Cardiol. 2014, 11, 586–596. [Google Scholar] [CrossRef]
- Turk-Adawi, K.I.; Terzic, C.; Bjarnason-Wehrens, B.; Grace, S.L. Cardiac rehabilitation in Canada and Arab countries: Comparing availability and program characteristics. BMC Health Serv. Res. 2015, 15, 521. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomised trials. BMC Med. 2010, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, T.C.; Glasziou, P.P.; Boutron, I.; Milne, R.; Perera, R.; Moher, D.; Altman, D.G.; Barbour, V.; Macdonald, H.; Johnston, M.; et al. Better reporting of interventions: Template for intervention description and replication (TIDieR) checklist and guide. BMJ 2014, 348, 1687. [Google Scholar] [CrossRef]
| # | N | Indication (Post-MI %) | Age (Mean Years, SD) | Sex (% male) | Follow-Up (Mean Months, SD) | |
|---|---|---|---|---|---|---|
| All Endpoints | 143 | 69,910 | 55 (37.2%) | 58.7 (6.4) | 83.1 | 36.5 (38.3) |
| Primary Endpoints | ||||||
| All-Cause Mortality | 118 | 44,462 | 50 (90.9%) | 58.9 (6.4) | 85.6 | 37.2 (35.9) |
| CV Mortality | 42 | 16,770 | 21 (38.2%) | 56.9 (6.5) | 92.9 | 47.9 (48.7) |
| Secondary Endpoints | ||||||
| Any MI | 52 | 16,690 | 28 (50.9%) | 55.6 (5.4) | 86.5 | 42.3 (43.7) |
| Fatal MI | 16 | 4261 | 9 (16.4%) | 53.7 (4.5) | 100.0 | 47.7 (51.8) |
| Non-Fatal MI | 31 | 11,919 | 18 (32.7%) | 54.5 (4.3) | 83.9 | 46.2 (47.3) |
| Any Revascularization | 49 | 16,947 | 25 (45.5%) | 56.8 (4.9) | 83.3 | 34.9 (38.9) |
| CABG | 33 | 7391 | 18 (32.7%) | 56.3 (4.9) | 84.4 | 33.0 (38.9) |
| PCI | 23 | 8859 | 10 (18.2%) | 56.8 (4.8) | 87.0 | 27.0 (16.0) |
| Any Hospitalization | 45 | 14,440 | 16 (29.1%) | 59.0 (5.8) | 82.6 | 31.5 (33.5) |
| CV Hospitalization | 24 | 7925 | 10 (18.2%) | 57.6 (4.1) | 88.0 | 28.5 (30.1) |
| Outcome | Model | ||
|---|---|---|---|
| Main Effects | Two-Way Interaction | Full-Interaction | |
| All-Cause Mortality | 1147.7 | 1147.5 | 1148.7 |
| CV Mortality | 415.8 | 415.8 | 417.2 |
| Total MI | 536.4 | 536.8 | 538.6 |
| Fatal MI | 143.5 | 141.4 | 142.0 |
| Non-Fatal MI | 318.4 | 319.3 | 317.9 |
| Revascularization † | 545.8 | 545.7 | 537.0 |
| CABG † | 319.0 | 319.2 | 314.9 |
| PCI † | 239.8 | 237.6 | 236.2 |
| All-Cause Hospitalization | 549.9 | 549.9 | 551.4 |
| CV Hospitalization | 275.0 | 277.0 | 279.3 |
| Outcome | Component | ||||
|---|---|---|---|---|---|
| Nutritional Counseling | Risk Factor Modification | Psychosocial Management | Patient Education | Exercise Training | |
| All-Cause Mortality | |||||
| Estimate & 95% Credible Interval | 1.07 (0.78–1.46) | 0.87 (0.66–1.15) | 0.68 (0.54–0.85) | 0.98 (0.78–1.20) | 0.74 (0.60–0.92) |
| Probability Best | 0.01 | 0.04 | 0.67 | 0.01 | 0.28 |
| CV Mortality | |||||
| Estimate & 95% Credible Interval | 1.11 (0.68–1.74) | 0.72 (0.43–1.22) | 0.76 (0.53–1.11) | 0.95 (0.62–1.39) | 0.75 (0.53–1.05) |
| Probability Best | 0.03 | 0.40 | 0.24 | 0.06 | 0.28 |
| Total MI | |||||
| Estimate & 95% Credible Interval | 0.94 (0.56–1.55) | 0.86 (0.54–1.38) | 0.76 (0.57–0.99) | 0.68 (0.47–0.99) | 0.75 (0.56–0.99) |
| Probability Best | 0.08 | 0.10 | 0.17 | 0.45 | 0.20 |
| Fatal MI | |||||
| Estimate & 95% Credible Interval | 1.99 (0.57–6.86) | 0.54 (0.13–2.34) | 0.50 (0.21–1.13) | 0.58 (0.25–1.13) | 0.54 (0.31–0.87) |
| Probability Best | 0.01 | 0.34 | 0.29 | 0.15 | 0.21 |
| Non-Fatal MI | |||||
| Estimate & 95% Credible Interval | 0.93 (0.37–2.47) | 1.05 (0.37–2.68) | 0.86 (0.51–1.40) | 0.83 (0.42–1.47) | 0.78 (0.45–1.28) |
| Probability Best | 0.23 | 0.16 | 0.16 | 0.21 | 0.25 |
| All-Cause Hospitalization | |||||
| Estimate & 95% Credible Interval | 1.19 (0.70–1.94) | 0.97 (0.67–1.38) | 0.76 (0.58–0.96) | 0.87 (0.63–1.18) | 0.83 (0.60–1.13) |
| Probability Best | 0.04 | 0.06 | 0.49 | 0.16 | 0.25 |
| CV Hospitalization | |||||
| Estimate & 95% Credible Interval | 0.37 (0.09–1.45) | 0.70 (0.44–1.14) | 0.78 (0.55–1.00) | 1.03 (0.73–1.41) | 0.75 (0.39–1.12) |
| Probability Best | 0.74 | 0.11 | 0.04 | 0.00 | 0.11 |
| Components | Arms (n) | Odds Ratio (Mean) | Odds Ratio (Median) | Credible Intervals | p (OR < 1) | |
|---|---|---|---|---|---|---|
| 2.5% | 97.5% | |||||
| NC | 2 | 0.83 | 0.79 | 0.47 | 1.38 | 81.4 |
| RFM | 4 | 0.29 | 0.26 | 1.12 | 0.59 | 100.0 |
| PM | 5 | 0.93 | 0.93 | 0.66 | 1.24 | 70.2 |
| NC, PM | 2 | 1.26 | 1.11 | 0.40 | 2.96 | 41.5 |
| ET | 14 | 0.78 | 0.77 | 0.54 | 1.08 | 93.5 |
| RFM, ET | 2 | 1.92 | 1.65 | 0.61 | 4.82 | 16.2 |
| PM, ET | 2 | 0.72 | 0.65 | 0.25 | 1.67 | 83.4 |
| NC, PM, ET | 1 | 0.16 | 0.13 | 0.03 | 0.50 | 99.8 |
| NC, RFM, PM, ET | 1 | 2.49 | 1.76 | 0.36 | 8.89 | 23.4 |
| PE | 12 | 0.91 | 0.89 | 0.56 | 1.43 | 69.8 |
| NC, PE | 1 | 0.43 | 0.36 | 0.13 | 1.11 | 96.3 |
| NC, RFM, PE | 3 | 0.66 | 0.64 | 0.40 | 1.06 | 96.3 |
| PM, PE | 7 | 0.93 | 0.92 | 0.66 | 1.26 | 74.0 |
| RFM, PM, PE | 2 | 1.09 | 0.98 | 0.45 | 2.36 | 51.5 |
| ET, PE | 1 | 0.92 | 0.85 | 0.41 | 1.85 | 68.8 |
| NC, ET, PE | 2 | 1.38 | 1.07 | 0.29 | 4.03 | 45.9 |
| RFM, ET, PE | 1 | 1.44 | 1.29 | 0.48 | 3.26 | 30.9 |
| NC, FFM, ET, PE | 1 | 1.55 | 1.18 | 0.26 | 5.16 | 41.9 |
| PM, ET, PE | 3 | 0.80 | 0.75 | 0.36 | 1.50 | 78.8 |
| NC, PM, ET, PE | 1 | 1.07 | 0.80 | 0.20 | 3.45 | 61.8 |
| RFM, PM, ET, PE | 1 | 1.65 | 1.34 | 0.41 | 4.62 | 31.7 |
| NC, RFM, PM, ET, PE | 3 | 0.34 | 0.31 | 0.15 | 0.69 | 99.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kabboul, N.N.; Tomlinson, G.; Francis, T.A.; Grace, S.L.; Chaves, G.; Rac, V.; Daou-Kabboul, T.; Bielecki, J.M.; Alter, D.A.; Krahn, M. Comparative Effectiveness of the Core Components of Cardiac Rehabilitation on Mortality and Morbidity: A Systematic Review and Network Meta-Analysis. J. Clin. Med. 2018, 7, 514. https://doi.org/10.3390/jcm7120514
Kabboul NN, Tomlinson G, Francis TA, Grace SL, Chaves G, Rac V, Daou-Kabboul T, Bielecki JM, Alter DA, Krahn M. Comparative Effectiveness of the Core Components of Cardiac Rehabilitation on Mortality and Morbidity: A Systematic Review and Network Meta-Analysis. Journal of Clinical Medicine. 2018; 7(12):514. https://doi.org/10.3390/jcm7120514
Chicago/Turabian StyleKabboul, Nader N., George Tomlinson, Troy A. Francis, Sherry L. Grace, Gabriela Chaves, Valeria Rac, Tamara Daou-Kabboul, Joanna M. Bielecki, David A. Alter, and Murray Krahn. 2018. "Comparative Effectiveness of the Core Components of Cardiac Rehabilitation on Mortality and Morbidity: A Systematic Review and Network Meta-Analysis" Journal of Clinical Medicine 7, no. 12: 514. https://doi.org/10.3390/jcm7120514
APA StyleKabboul, N. N., Tomlinson, G., Francis, T. A., Grace, S. L., Chaves, G., Rac, V., Daou-Kabboul, T., Bielecki, J. M., Alter, D. A., & Krahn, M. (2018). Comparative Effectiveness of the Core Components of Cardiac Rehabilitation on Mortality and Morbidity: A Systematic Review and Network Meta-Analysis. Journal of Clinical Medicine, 7(12), 514. https://doi.org/10.3390/jcm7120514






