Hysterectomies Are Associated with an Increased Risk of Depression: A Population-Based Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source
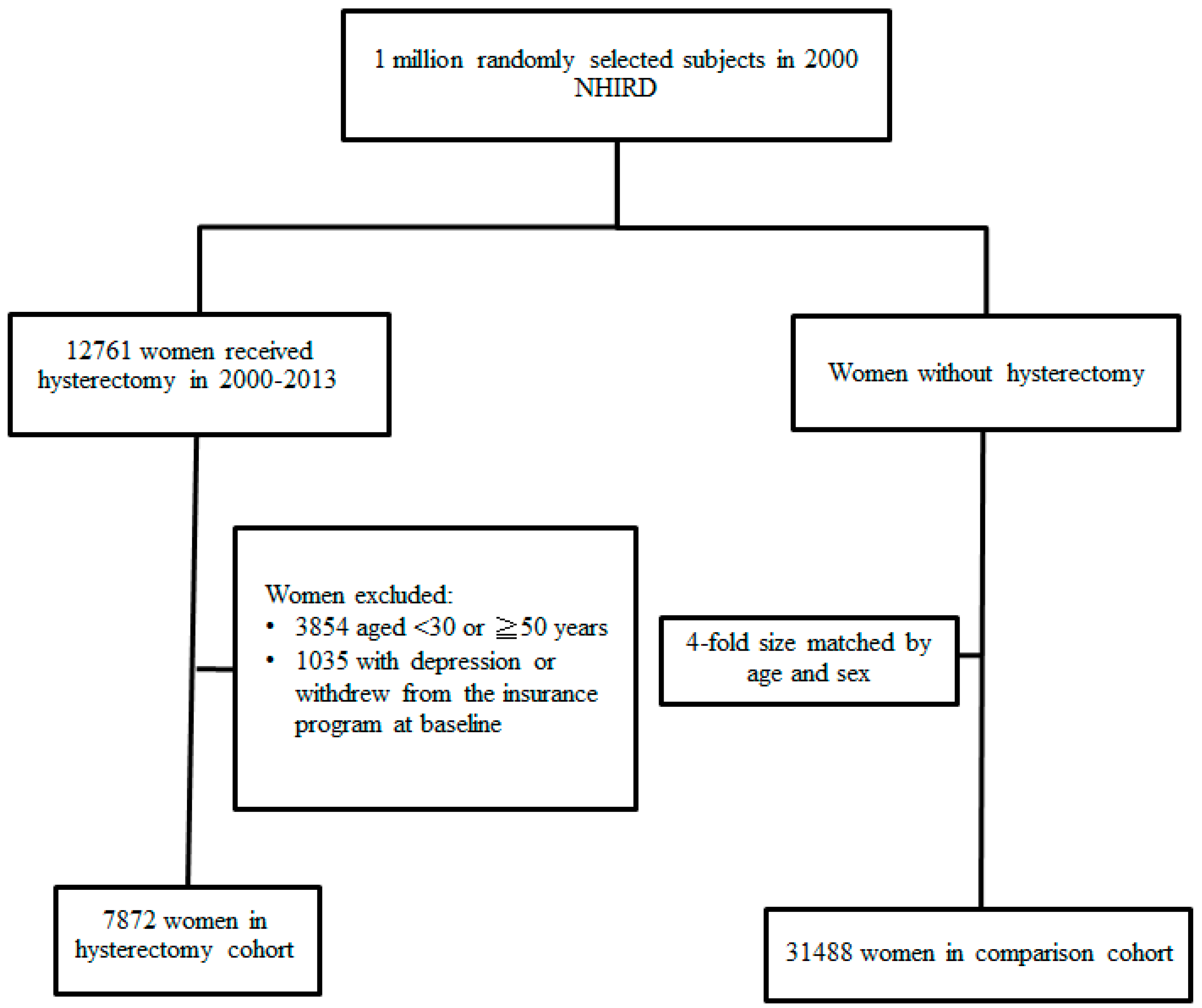
2.2. Sampled Participants
2.3. Outcomes, Relevant Variables, and Comorbidities
2.4. Statistical Analysis
3. Results
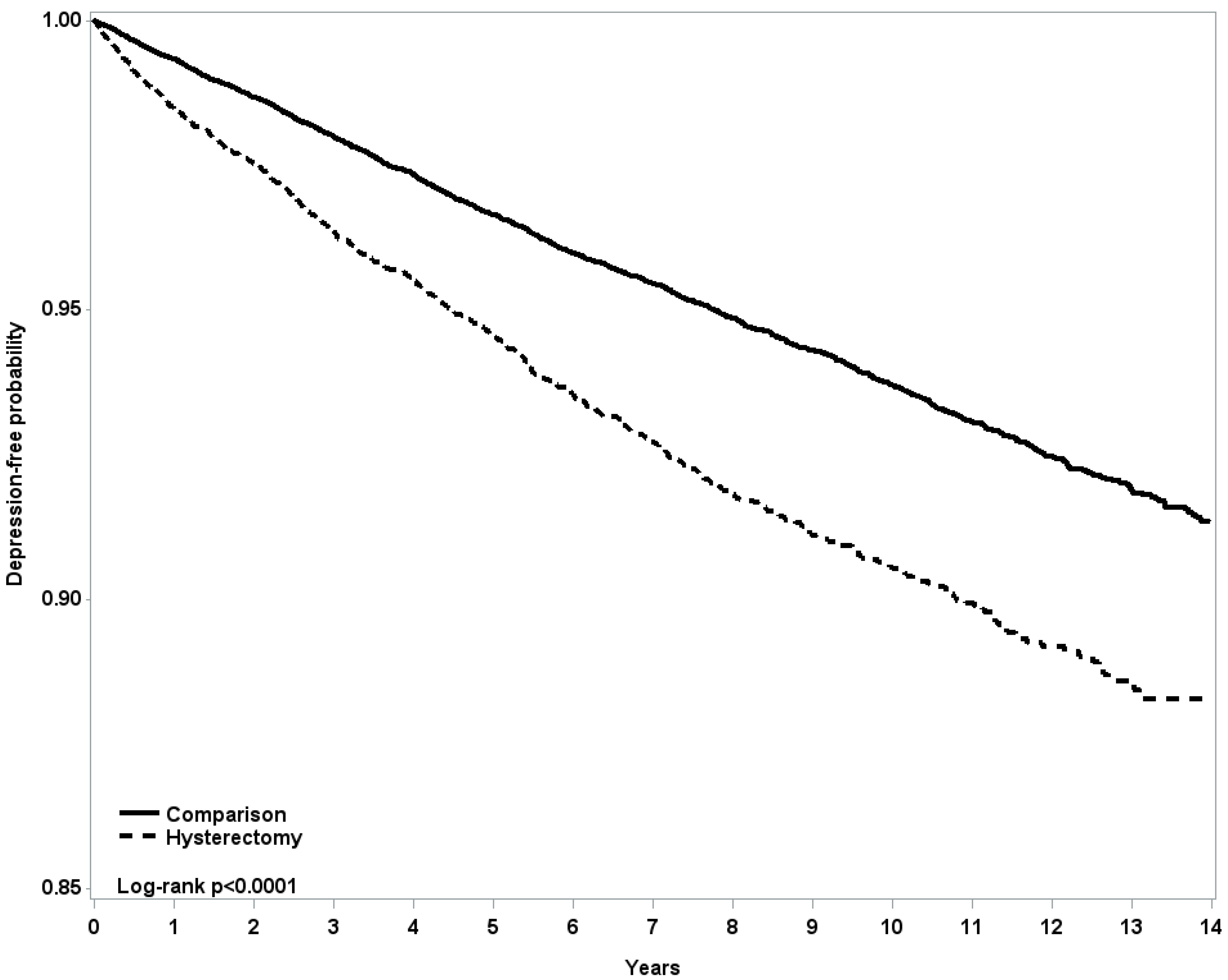
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ding, D.-C.; Chu, T.-Y.; Chang, Y.-H. Trend changes in the proportion of minimal invasive hysterectomies over a five-year period: A single-center experience. Tzu Chi Med. J. 2012, 24, 136–138. [Google Scholar] [CrossRef]
- Wu, M.-P.; Huang, K.-H.; Long, C.-Y.; Tsai, E.-M.; Tang, C.-H. Trends in various types of surgery for hysterectomy and distribution by patient age, surgeon age, and hospital accreditation: 10-year population-based study in Taiwan. J. Minim. Invasive Gynecol. 2010, 17, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Falcone, T.; Walters, M.D. Hysterectomy for benign disease. Obstet. Gynecol. 2008, 111, 753–767. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, C.M.; Steiner, C.A. Hysterectomy Rates in the United States 1990–1997. Obstet. Gynecol. 2002, 99, 229–234. [Google Scholar] [PubMed]
- Kjerulff, K.H.; Langenberg, P.W.; Rhodes, J.C.; Harvey, L.A.; Guzinski, G.M.; Stolley, P.D. Effectiveness of hysterectomy. Obstet. Gynecol. 2000, 95, 319–326. [Google Scholar] [PubMed]
- Darwish, M.; Atlantis, E.; Mohamed-Taysir, T. Psychological outcomes after hysterectomy for benign conditions: A systematic review and meta-analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 174, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Khastgir, G.; Studd, J.W.; Catalan, J. The psychological outcome of hysterectomy. Gynecol. Endocrinol. 2000, 14, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.; Bergquist, C.; Brännström, M.; Nordholm, L.; Silfverstolpe, G. Differences in aspects of personality and sexuality between perimenopausal women making different choices regarding prophylactic oophorectomy at elective hysterectomy. Acta Obstet. Gynecol. Scand. 2005, 84, 854–859. [Google Scholar] [CrossRef] [PubMed]
- Persson, P.; Brynhildsen, J.; Kjølhede, P.; Hysterectomy Multicentre Study Group in South-East Sweden. A 1-year follow up of psychological wellbeing after subtotal and total hysterectomy—A randomised study. BJOG 2010, 117, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Yen, J.-Y.; Chen, Y.-H.; Long, C.-Y.; Chang, Y.; Yen, C.-F.; Chen, C.-C.; Ko, C.-H. Risk factors for major depressive disorder and the psychological impact of hysterectomy: A prospective investigation. Psychosomatics 2008, 49, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Ewalds-Kvist, S.B.M.; Hirvonen, T.; Kvist, M.; Lertola, K.; Niemelä, P. Depression, anxiety, hostility and hysterectomy. J. Psychosom. Obstet. Gynaecol. 2005, 26, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.; Pandeya, N.; Byles, J.; Mishra, G. Hysterectomy and incidence of depressive symptoms in midlife women: The Australian Longitudinal Study on Women’s Health. Epidemiol. Psychiatr. Sci. 2017, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, K.E.; Ma, C.; Lamvu, G.M.; Langenberg, P.W.; Steege, J.F.; Kjerulff, K.H. Quality of life and sexual function after hysterectomy in women with preoperative pain and depression. Obstet. Gynecol. 2004, 104, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Helmy, Y.A.; Hassanin, I.M.A.; Elraheem, T.A.; Bedaiwy, A.A.; Peterson, R.S.; Bedaiwy, M.A. Psychiatric morbidity following hysterectomy in Egypt. Int. J. Gynaecol. Obstet. 2008, 102, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, C.M.; Sadler, L.; Stewart, A.W. A prospective study of outcomes five years after hysterectomy in premenopausal women. Aust. N. Z. J. Obstet. Gynaecol. 2008, 48, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.C.; Liao, C.C.; Chen, T.L. Perioperative medicine and Taiwan National Health Insurance Research Database. Acta Anaesthesiol. Taiwan 2016, 54, 93–96. [Google Scholar] [CrossRef] [PubMed]
- National Health Insurance Research Database. Available online: http://nhird.nhri.org.tw/en/index.html (accessed on 14 August 2018).
- Hamilton, K.J.; Arao, Y.; Korach, K.S. Estrogen hormone physiology: Reproductive findings from estrogen receptor mutant mice. Reprod. Biol. 2014, 14, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Kaump, G.R.; Spies, J.B. The impact of uterine artery embolization on ovarian function. J. Vasc. Interv. Radiol. 2013, 24, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Grambsch, P.; Young, E.A.; Meller, W.H. Pulsatile luteinizing hormone disruption in depression. Psychoneuroendocrinology 2004, 29, 825–829. [Google Scholar] [CrossRef]
- Benmansour, S.; Adeniji, O.S.; Privratsky, A.A.; Frazer, A. Effects of Long-Term Treatment with Estradiol and Estrogen Receptor Subtype Agonists on Serotonergic Function in Ovariectomized Rats. Neuroendocrinology 2016, 103, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Hernandez, O.T.; Martinez-Mota, L.; Herrera-Perez, J.J.; Jimenez-Rubio, G. Role of estradiol in the expression of genes involved in serotonin neurotransmission: Implications for female depression. Curr. Neuropharmacol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Torrealday, S.; Kodaman, P.; Pal, L. Premature ovarian insufficiency—An update on recent advances in understanding and management. F1000Research 2017, 6, 2069. [Google Scholar] [CrossRef] [PubMed]
- Bove, R.; Secor, E.; Chibnik, L.B.; Barnes, L.L.; Schneider, J.A.; Bennett, D.A.; De Jager, P.L. Age at surgical menopause influences cognitive decline and Alzheimer pathology in older women. Neurology 2014, 82, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Shu, C.-C.; Lin, J.-W.; Lin, Y.-F.; Hsu, N.-C.; Ko, W.-J. Evaluating the performance of a hospitalist system in Taiwan: A pioneer study for nationwide health insurance in Asia. J. Hosp. Med. 2011, 6, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Yeh, J.S.; Cheng, H.-M.; Hsu, P.-F.; Sung, S.-H.; Liu, W.-L.; Fang, H.-L.; Chuang, S.-Y. Hysterectomy in young women associates with higher risk of stroke: A nationwide cohort study. Int. J. Cardiol. 2013, 168, 2616–2621. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.C.; Lo, T.C.; Chang, J.H.; Kuo, H.W. Variations in aging, gender, menopause, and obesity and their effects on hypertension in Taiwan. Int. J. Hypertens. 2014, 2014, 515297. [Google Scholar] [CrossRef] [PubMed]
| Hysterectomy (n = 7872) | Comparison (n = 31,488) | p-Value | |
|---|---|---|---|
| N (%) | N (%) | ||
| Age, years | |||
| 30–39 | 1400 (17.8) | 5600 (17.8) | 1.0000 |
| 40–49 | 6472 (82.2) | 25,888 (82.2) | |
| Mean (SD) | 43.1 (4.0) | 43.1 (4.0) | 1.0000 |
| Follow-up duration (years) | |||
| Mean (SD) | 7.0 (4.1) | 7.6 (4.1) | <0.0001 |
| Oophorectomy | 1963 (24.9) | 1146 (3.6) | <0.0001 |
| Hormone use | 2170 (27.6) | 3920 (12.5) | <0.0001 |
| Comorbidity, n (%) | |||
| Anxiety | 1473 (18.7) | 4293 (13.6) | <0.0001 |
| Cancer | 280 (3.6) | 412 (1.3) | <0.0001 |
| Stroke | 282 (3.6) | 813 (2.6) | <0.0001 |
| CAD | 316 (4.0) | 780 (2.5) | <0.0001 |
| N | Event | PY | Rate | Crude | Adjusted * | |||
|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |||||
| All | ||||||||
| Comparison group | 31,488 | 1566 | 238,597 | 0.66 | 1 (reference) | 1 (reference) | ||
| Hysterectomy | 7872 | 566 | 55,234 | 1.02 | 1.56 (1.41–1.71) | <0.0001 | 1.35 (1.22–1.50) | <0.0001 |
| Age 30–39 | ||||||||
| Comparison group | 5600 | 275 | 46,844 | 0.59 | 1 (reference) | 1 (reference) | ||
| Hysterectomy | 1400 | 111 | 10,718 | 1.04 | 1.75 (1.41–2.19) | <0.0001 | 1.57 (1.24–1.99) | 0.0002 |
| Age 40–49 | ||||||||
| Comparison group | 25,888 | 1291 | 191,753 | 0.67 | 1 (reference) | 1 (reference) | ||
| Hysterectomy | 6472 | 455 | 44,517 | 1.02 | 1.51 (1.36–1.69) | <0.0001 | 1.30 (1.16–1.47) | <0.0001 |
| Hysterectomy | Oophorectomy | N | Event | PY | Rate | Crude | Adjusted * | ||
|---|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | ||||||
| No | No | 30,342 | 1509 | 230,667 | 0.65 | 1 (reference) | 1 (reference) | ||
| No | Yes | 1146 | 57 | 7931 | 0.72 | 1.09 (0.84–1.42) | 0.51 | 1.00 (0.77–1.30) | 0.99 |
| Yes | No | 5909 | 404 | 40,453 | 1.00 | 1.52 (1.36–1.69) | <0.0001 | 1.34 (1.20–1.50) | <0.0001 |
| Yes | Yes | 1963 | 162 | 14,781 | 1.10 | 1.67 (1.42–1.97) | <0.0001 | 1.46 (1.23–1.72) | <0.0001 |
| Hysterectomy | Use Hormone | N | Event | PY | Rate | Crude | Adjusted * | ||
|---|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | ||||||
| No | No | 27,568 | 1244 | 204,231 | 0.61 | 1 (reference) | 1 (reference) | ||
| No | Yes | 3920 | 322 | 34,366 | 0.94 | 1.56 (1.38–1.76) | <0.0001 | 1.34 (1.19–1.52) | <0.0001 |
| Yes | No | 5702 | 355 | 37,167 | 0.96 | 1.55 (1.38–1.75) | <0.0001 | 1.42 (1.26–1.60) | <0.0001 |
| Yes | Yes | 2170 | 211 | 18,068 | 1.17 | 1.93 (1.67–2.24) | <0.0001 | 1.63 (1.40–1.91) | <0.0001 |
| Hysterectomy | Oophorectomy | Use Hormone | N | Event | PY | Rate | Crude | Adjusted * | ||
|---|---|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |||||||
| No | No | No | 26,673 | 1207 | 198,240 | 0.61 | 1 (reference) | 1 (reference) | ||
| No | Yes | No | 895 | 37 | 5991 | 0.62 | 1.01 (0.73–1.40) | 0.96 | 0.97 (0.70–1.35) | 0.85 |
| No | No | Yes | 3669 | 302 | 32,427 | 0.93 | 1.55 (1.36–1.76) | <0.0001 | 1.34 (1.18–1.53) | <0.0001 |
| No | Yes | Yes | 251 | 20 | 1939 | 1.03 | 1.70 (1.09–2.64) | 0.02 | 1.40 (0.90–2.18) | 0.14 |
| Yes | No | No | 4566 | 284 | 29,500 | 0.96 | 1.57 (1.38–1.78) | <0.0001 | 1.45 (1.27–1.65) | <0.0001 |
| Yes | Yes | No | 1136 | 71 | 7667 | 0.93 | 1.51 (1.19–1.92) | 0.0008 | 1.37 (1.07–1.74) | 0.01 |
| Yes | No | Yes | 1343 | 120 | 10,953 | 1.10 | 1.81 (1.50–2.19) | <0.0001 | 1.48 (1.23–1.79) | <0.0001 |
| Yes | Yes | Yes | 827 | 91 | 7115 | 1.28 | 2.12 (1.72–2.63) | <0.0001 | 1.99 (1.61–2.47) | <0.0001 |
| Hysterectomy | Anxiety | Stroke | CAD | N | Event | PY | Rate | Crude | Adjusted * | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | ||||||||
| No | No | No | No | 26,306 | 1027 | 20,4752 | 0.50 | 1 (reference) | 1 (reference) | ||
| No | Yes | No | No | 3669 | 388 | 22,946 | 1.69 | 3.35 (2.98–3.77) | <0.0001 | 3.29 (2.92–3.70) | <0.0001 |
| No | No | Yes | No | 481 | 35 | 3753 | 0.93 | 1.86 (1.33–2.60) | 0.0003 | 1.81 (1.29–2.54) | 0.0005 |
| No | No | No | Yes | 376 | 26 | 2793 | 0.93 | 1.85 (1.26–2.73) | 0.002 | 1.84 (1.24–2.71) | 0.002 |
| No | Yes | Yes | No | 252 | 41 | 1799 | 2.28 | 4.53 (3.32–6.19) | <0.0001 | 4.32 (3.16–5.91) | <0.0001 |
| No | Yes | No | Yes | 324 | 39 | 1986 | 1.96 | 3.88 (2.82–5.34) | <0.0001 | 3.71 (2.69–5.12) | <0.0001 |
| No | No | Yes | Yes | 32 | 2 | 238 | 0.84 | 1.67 (0.42–6.69) | 0.47 | 1.69 (0.42–6.75) | 0.46 |
| No | Yes | Yes | Yes | 48 | 8 | 329 | 2.43 | 4.83 (2.41–9.68) | <0.0001 | 4.42 (2.20–8.87) | <0.0001 |
| Yes | No | No | No | 6085 | 344 | 44,119 | 0.78 | 1.55 (1.37–1.75) | <0.0001 | 1.46 (1.29–1.67) | <0.0001 |
| Yes | Yes | No | No | 1228 | 149 | 7426 | 2.01 | 3.97 (3.35–4.72) | <0.0001 | 3.68 (3.09–4.40) | <0.0001 |
| Yes | No | Yes | No | 155 | 18 | 1133 | 1.59 | 3.16 (1.98–5.04) | <0.0001 | 2.92 (1.83–4.67) | <0.0001 |
| Yes | No | No | Yes | 142 | 17 | 953 | 1.78 | 3.55 (2.20–5.74) | <0.0001 | 3.34 (2.07–5.41) | <0.0001 |
| Yes | Yes | Yes | No | 88 | 9 | 567 | 1.59 | 3.15 (1.63–6.07) | 0.0006 | 2.93 (1.52–5.65) | 0.001 |
| Yes | Yes | No | Yes | 135 | 18 | 789 | 2.28 | 4.50 (2.83–7.18) | <0.0001 | 4.19 (2.63–6.70) | <0.0001 |
| Yes | No | Yes | Yes | 17 | 6 | 119 | 5.04 | 10.0 (4.50–22.4) | <0.0001 | 9.78 (4.38–21.8) | <0.0001 |
| Yes | Yes | Yes | Yes | 22 | 5 | 129 | 3.88 | 7.65 (3.18–18.4) | <0.0001 | 7.25 (3.01–17.5) | <0.0001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harnod, T.; Chen, W.; Wang, J.-H.; Lin, S.-Z.; Ding, D.-C. Hysterectomies Are Associated with an Increased Risk of Depression: A Population-Based Cohort Study. J. Clin. Med. 2018, 7, 366. https://doi.org/10.3390/jcm7100366
Harnod T, Chen W, Wang J-H, Lin S-Z, Ding D-C. Hysterectomies Are Associated with an Increased Risk of Depression: A Population-Based Cohort Study. Journal of Clinical Medicine. 2018; 7(10):366. https://doi.org/10.3390/jcm7100366
Chicago/Turabian StyleHarnod, Tomor, Weishan Chen, Jen-Hung Wang, Shinn-Zong Lin, and Dah-Ching Ding. 2018. "Hysterectomies Are Associated with an Increased Risk of Depression: A Population-Based Cohort Study" Journal of Clinical Medicine 7, no. 10: 366. https://doi.org/10.3390/jcm7100366
APA StyleHarnod, T., Chen, W., Wang, J.-H., Lin, S.-Z., & Ding, D.-C. (2018). Hysterectomies Are Associated with an Increased Risk of Depression: A Population-Based Cohort Study. Journal of Clinical Medicine, 7(10), 366. https://doi.org/10.3390/jcm7100366






