Effect of Acute Exercise Mode on Serum Brain-Derived Neurotrophic Factor (BDNF) and Task Switching Performance
Abstract
1. Introduction
2. Method
2.1. Participants
2.2. Acute Exercise Intensity Manipulation Check
2.3. Blood Sampling and Analysis
2.4. Cognitive Task
2.5. Procedure
2.6. Statistical Analysis
3. Results
3.1. Exercise Intensity Manipulation
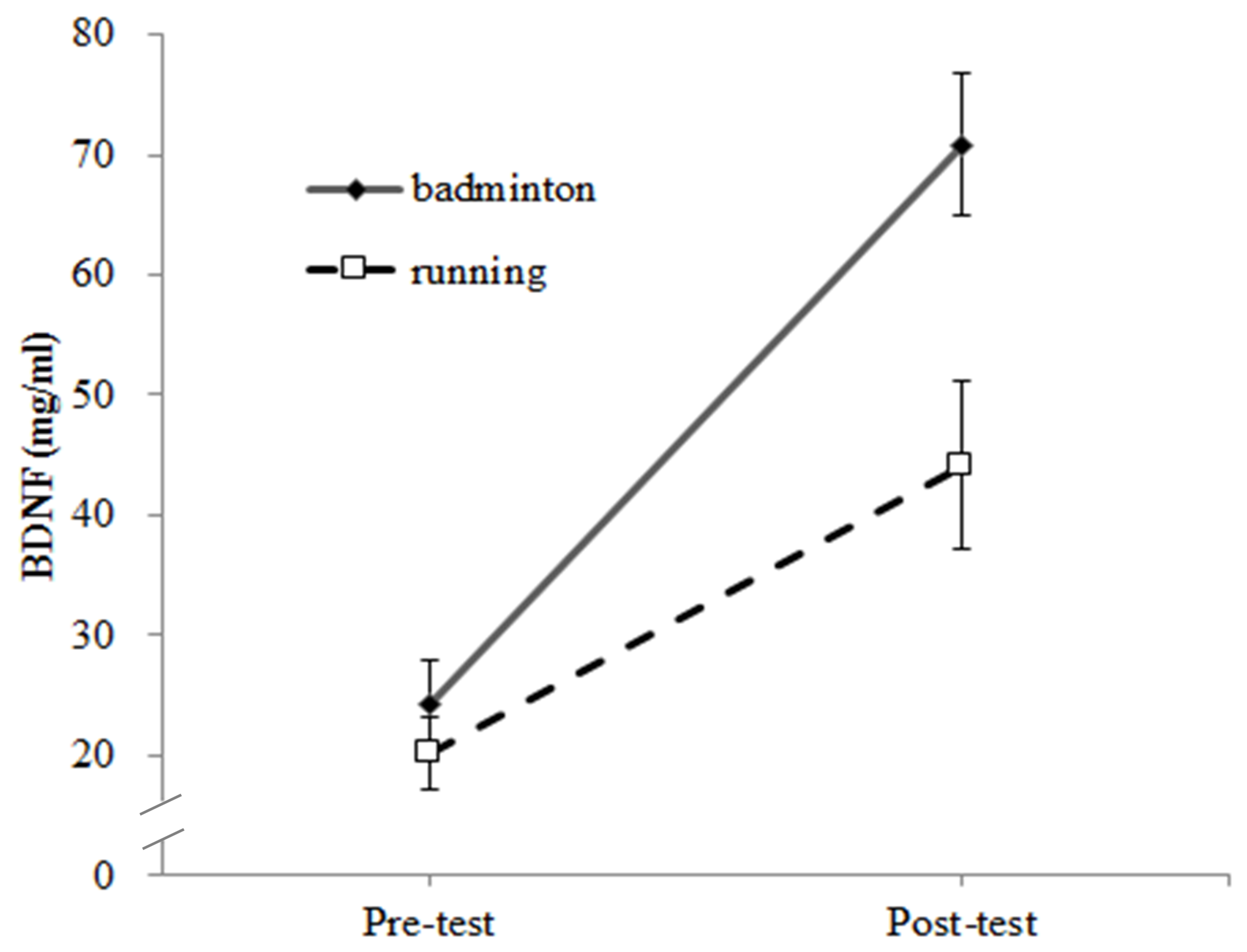
3.2. BDNF Response
3.3. Behavioral Data
3.3.1. Global Switch Effect
3.3.2. Local Switch Effect
3.3.3. Switch Cost Effect
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Colcombe, S.; Kramer, A.F. Fitness effects on the cognitive function of older adults: A meta-analytic study. Psychol. Sci. 2003, 14, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Buchman, A.S.; Wilson, R.S.; Bennett, D.A. Total daily activity is associated with cognition in older persons. Am. J. Geriatr. Psychiatry 2008, 16, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Pesce, C.; Audiffren, M. Does acute exercise switch off switch costs? A study with younger and older athletes. J. Sport Exerc. Psychol. 2011, 33, 609–626. [Google Scholar] [CrossRef] [PubMed]
- Pesce, C.; Cereatti, L.; Forte, R.; Crova, C.; Casella, R. Acute and chronic exercise effects on attentional control in older road cyclists. Gerontology 2011, 57, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.K.; Ku, P.W.; Tomporowski, P.D.; Chen, F.T.; Huang, C.C. Effects of acute resistance exercise on late-middle-age adults’ goal planning. Med. Sci. Sports Exerc. 2012, 44, 1773–1779. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.K.; Tsai, C.L.; Huang, C.C.; Wang, C.C.; Chu, I.H. Effects of acute resistance exercise on cognition in late middle-aged adults: General or specific cognitive improvement? J. Sci. Med. Sport 2014, 17, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Audiffren, M.; Tomporowski, P.D.; Zagrodnik, J. Acute aerobic exercise and information processing: Modulation of executive control in a random number generation task. Acta Psychol. 2009, 132, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Lambourne, K.; Audiffren, M.; Tomporowski, P.D. Effects of acute exercise on sensory and executive processing tasks. Med. Sci. Sports Exerc. 2010, 42, 1396–1402. [Google Scholar] [CrossRef] [PubMed]
- Drollette, E.S.; Scudder, M.R.; Raine, L.B.; Moore, R.D.; Saliba, B.J.; Pontifex, M.B.; Hillman, C.H. Acute exercise facilitates brain function and cognition in children who need it most: An ERP study of individual differences in inhibitory control capacity. Dev. Cogn. Neurosci. 2014, 7, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.K.; Liu, S.; Yu, H.H.; Lee, Y.H. Effect of acute exercise on executive function in children with attention deficit hyperactivity disorder. Arch. Clin. Neuropsychol. 2012, 27, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.L.; Huang, C.J.; Tsai, Y.J.; Chang, Y.K.; Hung, T.M. Neuroelectric and behavioral effects of acute exercise on task switching in children with attention-deficit/hyperactivity disorder. Front. Psychol. 2016, 7, 1589. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, S.S.; Chang, Y.K.; Hung, T.-M.; Fang, C.L. The effects of acute resistance exercise on young and older males’ working memory. Psychol. Sport. Exerc. 2016, 22, 286–293. [Google Scholar] [CrossRef]
- Hsieh, S.S.; Chang, Y.K.; Fang, C.L.; Hung, T.M. Acute resistance exercise facilitates attention control in adult males without an age-moderating effect. J. Sport Exerc. Psychol. 2016, 38, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Pontifex, M.B.; Hillman, C.H.; Fernhall, B.; Thompson, K.M.; Valentini, T.A. The effect of acute aerobic and resistance exercise on working memory. Med. Sci. Sports Exerc. 2009, 41, 927–934. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.A.; Wrisberg, C.A. Motor Learning and Performance: A Situation-Based Learning Approach, 4th ed.; Human Kinetics: Champaign, IL, USA, 2008. [Google Scholar]
- Huang, C.J.; Lin, P.C.; Hung, C.L.; Chang, Y.K.; Hung, T.M. Type of physical exercise and inhibitory function in older adults: An event-related potential study. Psychol. Sport. Exerc. 2014, 15, 205–211. [Google Scholar] [CrossRef]
- Dai, C.T.; Chang, Y.K.; Huang, C.J.; Hung, T.M. Exercise mode and executive function in older adults: An ERP study of task-switching. Brain Cogn. 2013, 83, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.L.; Wang, W.L. Exercise-mode-related changes in task-switching performance in the elderly. Front. Behav. Neurosci. 2015, 9, 56. [Google Scholar] [CrossRef] [PubMed]
- Tyler, W.J.; Alonso, M.; Bramham, C.R.; Pozzo-Miller, L.D. From acquisition to consolidation: On the role of brain-derived neurotrophic factor signaling in hippocampal-dependent learning. Learn. Mem. 2002, 9, 224–237. [Google Scholar] [CrossRef] [PubMed]
- McAllister, A.K.; Katz, L.C.; Lo, D.C. Neurotrophins and synaptic plasticity. Annu. Rev. Neurosci. 1999, 22, 295–318. [Google Scholar] [CrossRef] [PubMed]
- Ferris, L.T.; Williams, J.S.; Shen, C.L. The effect of acute exercise on serum brain-derived neurotrophic factor levels and cognitive function. Med. Sci. Sports Exerc. 2007, 39, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Griffin, E.W.; Mullally, S.; Foley, C.; Warmington, S.A.; O’Mara, S.M.; Kelly, A.M. Aerobic exercise improves hippocampal function and increases BDNF in the serum of young adult males. Physiol. Behav. 2011, 104, 934–941. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Liu, X.; Lin, E.J.; Wang, C.; Choi, E.Y.; Riban, V.; Lin, B.; During, M.J. Environmental and genetic activation of a brain-adipocyte BDNF/leptin axis causes cancer remission and inhibition. Cell 2010, 142, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Hillman, C.H.; Pontifex, M.B.; Castelli, D.M.; Khan, N.A.; Raine, L.B.; Scudder, M.R.; Drollette, E.S.; Moore, R.D.; Wu, C.-T.; Kamijo, K. Effects of the FITKids randomized controlled trial on executive control and brain function. Pediatrics 2014, 134, e1063–e1071. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A. Executive functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [PubMed]
- Monsell, S. Task switching. Trends Cogn. Sci. 2003, 7, 134–140. [Google Scholar] [CrossRef]
- Kray, J.; Lindenberger, U. Adult age differences in task switching. Psychol. Aging 2000, 15, 126. [Google Scholar] [CrossRef] [PubMed]
- Winter, B.; Breitenstein, C.; Mooren, F.C.; Voelker, K.; Fobker, M.; Lechtermann, A.; Krueger, K.; Fromme, A.; Korsukewitz, C.; Floel, A.; et al. High impact running improves learning. Neurobiol. Learn. Mem. 2007, 87, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Karvonen, M.J.; Kentala, E.; Mustala, O. The effects of training heart rate: A longitudinal study. Ann. Med. Exp. Biol. Fenn. 1957, 35, 307–315. [Google Scholar] [PubMed]
- Robergs, R.A.; Landwehr, R. The surprising history of the “HRmax = 220-age” equation. J. Exercise Physl. 2002, 5, 1–10. [Google Scholar]
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics: Champaign, IL, USA, 1998. [Google Scholar]
- Rogers, R.D.; Monsell, S. Costs of a predictible switch between simple cognitive tasks. J. Exp. Psychol. Gen. 1995, 124, 207–231. [Google Scholar] [CrossRef]
- Hillman, C.H.; Snook, E.M.; Jerome, G.J. Acute cardiovascular exercise and executive control function. Int. J. Psychophysiol. 2003, 48, 307–314. [Google Scholar] [CrossRef]
- Kempermann, G.; Fabel, K.; Ehninger, D.; Babu, H.; Leal-Galicia, P.; Garthe, A.; Wolf, S. Why and how physical activity promotes experience-induced brain plasticity. Front. Neurosci. 2010, 4, 189. [Google Scholar] [CrossRef] [PubMed]
- Coles, K.; Tomporowski, P.D. Effects of acute exercise on executive processing, short-term and long-term memory. J. Sports Sci. 2008, 26, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Salthouse, T.A.; Davis, H.P. Organization of cognitive abilities and neuropsychological variables across the lifespan. Dev. Rev. 2006, 26, 31–54. [Google Scholar] [CrossRef]
- Kamijo, K.; Takeda, Y. Regular physical activity improves executive function during task switching in young adults. Int. J. Psychophysiol. 2010, 75, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Themanson, J.R.; Hillman, C.H.; Curtin, J.J. Age and physical activity influences on action monitoring during task switching. Neurobiol. Aging 2006, 27, 1335–1345. [Google Scholar] [CrossRef] [PubMed]
- Anderson-Hanley, C.; Arciero, P.J.; Brickman, A.M.; Nimon, J.P.; Okuma, N.; Westen, S.C.; Merz, M.E.; Pence, B.D.; Woods, J.A.; Kramer, A.F.; et al. Exergaming and older adult cognition: A cluster randomized clinical trial. Am. J. Prev. Med. 2012, 42, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Vandierendonck, A. Executive functions and task switching. Psychol. Belg. 2000, 40, 211–226. [Google Scholar]
- Voss, M.W.; Kramer, A.F.; Basak, C.; Prakash, R.S.; Roberts, B. Are expert athletes ‘expert’ in the cognitive laboratory? A meta-analytic review of cognition and sport expertise. Appl. Cogn. Psychol. 2010, 24, 812–826. [Google Scholar] [CrossRef]
- Scisco, J.L.; Leynes, P.A.; Kang, J. Cardiovascular fitness and executive control during task-switching: An ERP study. Int. J. Psychophysiol. 2008, 69, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Allport, A.; Styles, E.A.; Hsieh, S. Shifting Intentional Set: Exploring the Dynamic Control of Tasks. In Attention and Performance XV; Umiltà, C., Moscovitch, M., Eds.; The MIT Press: Cambridge, UK, 1994; pp. 421–452. [Google Scholar]
- Torres-Aleman, I. Toward a comprehensive neurobiology of IGF-I. Dev. Neurobiol. 2010, 70, 384–396. [Google Scholar] [CrossRef] [PubMed]
- Arwert, L.; Deijen, J.; Drent, M. The relation between insulin-like growth factor I levels and cognition in healthy elderly: A meta-analysis. Growth Horm. IGF Res. 2005, 15, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Cubeddu, A.; Bucci, F.; Giannini, A.; Russo, M.; Daino, D.; Russo, N.; Merlini, S.; Pluchino, N.; Valentino, V.; Casarosa, E.; et al. Brain-derived neurotrophic factor plasma variation during the different phases of the menstrual cycle in women with premenstrual syndrome. Psychoneuroendocrinology 2011, 36, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Nougier, V.; Stein, J.-F.; Bonnel, A.-M. Information processing in sport and “orienting of attention”. Int. J. Sport Psychol. 1991, 22, 307–327. [Google Scholar]
- Nagamatsu, L.S.; Handy, T.C.; Hsu, C.L.; Voss, M.; Liu-Ambrose, T. Resistance training promotes cognitive and functional brain plasticity in seniors with probable mild cognitive impairment. Arch. Intern. Med. 2012, 172, 666–668. [Google Scholar] [CrossRef] [PubMed]
| Variable | Total |
|---|---|
| Sample size | 20 |
| Age (years) | 23.15 ± 2.48 |
| Height (m) | 1.75 ± 0.07 |
| Weight (kg) | 68.98 ± 8.86 |
| BMI (kg/m2) | 22.49 ± 2.10 |
| HR-rest (bpm) | 60.75 ± 8.94 |
| HR-max (bpm) | 189.95 ± 1.67 |
| 60% HRR (bpm) | 138.30 ± 3.64 |
| Variable | Pre-Test | |||
|---|---|---|---|---|
| Open-Skill | Closed-Skill | t | p | |
| Global switch RT (ms) | ||||
| Pure | 475.625 ± 56.02 | 485.12 ± 81.07 | −0.66 | 0.52 |
| Mixed | 727.13 ± 151.03 | 766.21 ± 151.61 | −0.98 | 0.34 |
| Global switch cost | 251.51 ± 123.6 | 281.09 ± 108.37 | ||
| Local switch RT (ms) | ||||
| Non-switch | 477.54 ± 78.51 | 740.3 ± 142.89 | −0.86 | 0.4 |
| Switch | 656.47 ± 124.58 | 792.12 ± 164.94 | −1.09 | 0.29 |
| Local switch cost | 41.23 ± 65.46 | 51.83 ± 57.46 | ||
| Global switch accuracy (%) | ||||
| Pure | 96.2 ± 3.17 | 93.35 ± 11.42 | 1.09 | 0.29 |
| Mixed | 92.3 ± 5.54 | 89.02 ± 11.43 | 1.26 | 0.22 |
| Local switch accuracy (%) | ||||
| Non-switch | 93.2 ± 6.72 | 90.85 ± 12.08 | 0.94 | 0.36 |
| Switch | 91.5 ± 5.66 | 87.85 ± 11.05 | 1.4 | 0.18 |
| Variable | Post-Test | Sessions p-Value | Interactions p-Value | |||||
|---|---|---|---|---|---|---|---|---|
| Conditions | Open-Skill | Closed-Skill | Conditions | Open-Skill | Closed-Skill | |||
| Global switch RT (ms) | Pure | 477.54 ± 78.51 | 485.12 ± 81.07 | Mixed | 656.47 ± 124.58 | 766.21 ± 151.61 | 0.74 | 0.05 |
| Local switch RT (ms) | Non-switch | 639.64 ± 115.31 | 663.69 ± 120.1 | Switch | 673.29 ± 136.77 | 694.49 ± 120.54 | 0.3 | 0.78 |
| Global switch accuracy (%) | Pure | 95.55 ± 3.83 | 96.45 ± 3.24 | Mixed | 91.5 ± 7.21 | 93.35 ± 3.69 | 0.19 | 0.44 |
| Local switch accuracy (%) | Non-switch | 94.5 ± 4.25 | 94.45 ± 4.21 | Switch | 88.5 ± 13.18 | 92.35± 3.91 | 0.22 | 0.16 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hung, C.-L.; Tseng, J.-W.; Chao, H.-H.; Hung, T.-M.; Wang, H.-S. Effect of Acute Exercise Mode on Serum Brain-Derived Neurotrophic Factor (BDNF) and Task Switching Performance. J. Clin. Med. 2018, 7, 301. https://doi.org/10.3390/jcm7100301
Hung C-L, Tseng J-W, Chao H-H, Hung T-M, Wang H-S. Effect of Acute Exercise Mode on Serum Brain-Derived Neurotrophic Factor (BDNF) and Task Switching Performance. Journal of Clinical Medicine. 2018; 7(10):301. https://doi.org/10.3390/jcm7100301
Chicago/Turabian StyleHung, Chiao-Ling, Jun-Wei Tseng, Hsiao-Han Chao, Tsung-Min Hung, and Ho-Seng Wang. 2018. "Effect of Acute Exercise Mode on Serum Brain-Derived Neurotrophic Factor (BDNF) and Task Switching Performance" Journal of Clinical Medicine 7, no. 10: 301. https://doi.org/10.3390/jcm7100301
APA StyleHung, C.-L., Tseng, J.-W., Chao, H.-H., Hung, T.-M., & Wang, H.-S. (2018). Effect of Acute Exercise Mode on Serum Brain-Derived Neurotrophic Factor (BDNF) and Task Switching Performance. Journal of Clinical Medicine, 7(10), 301. https://doi.org/10.3390/jcm7100301






