Airway Mucus and Asthma: The Role of MUC5AC and MUC5B
Abstract
1. Asthma
2. Mucociliary Clearance
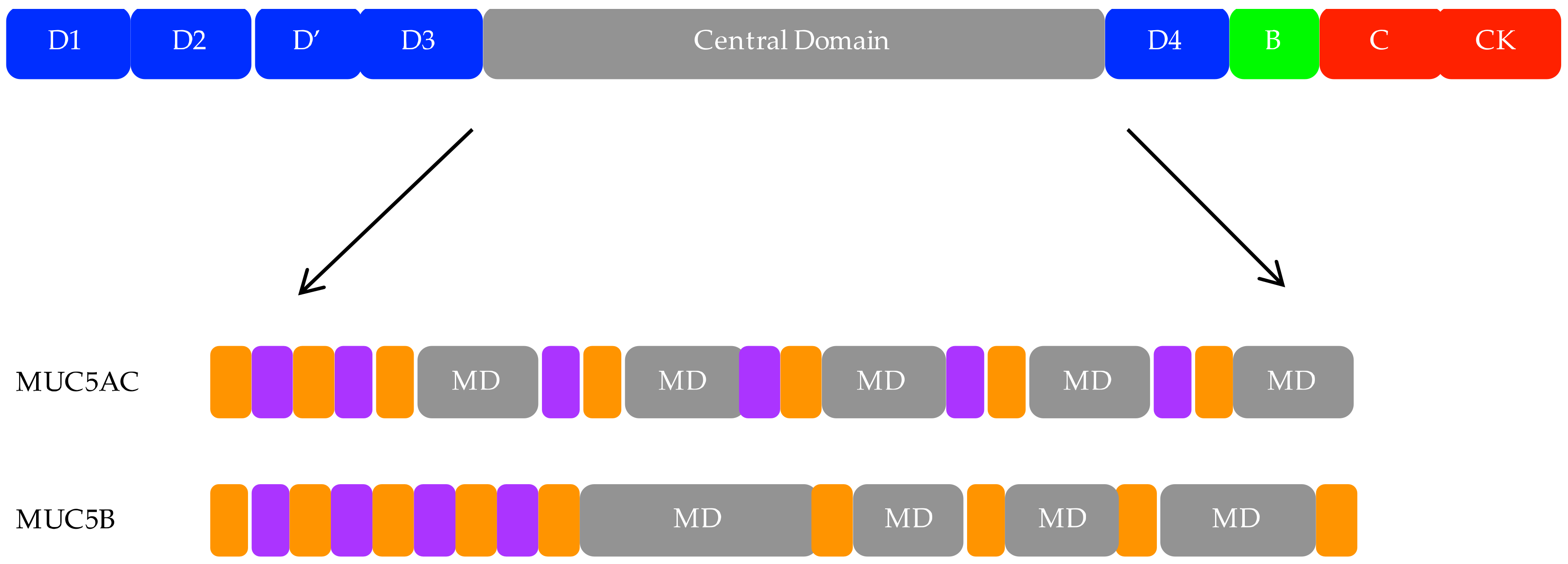
3. Polymeric Mucins
4. MUC5AC and MUC5B
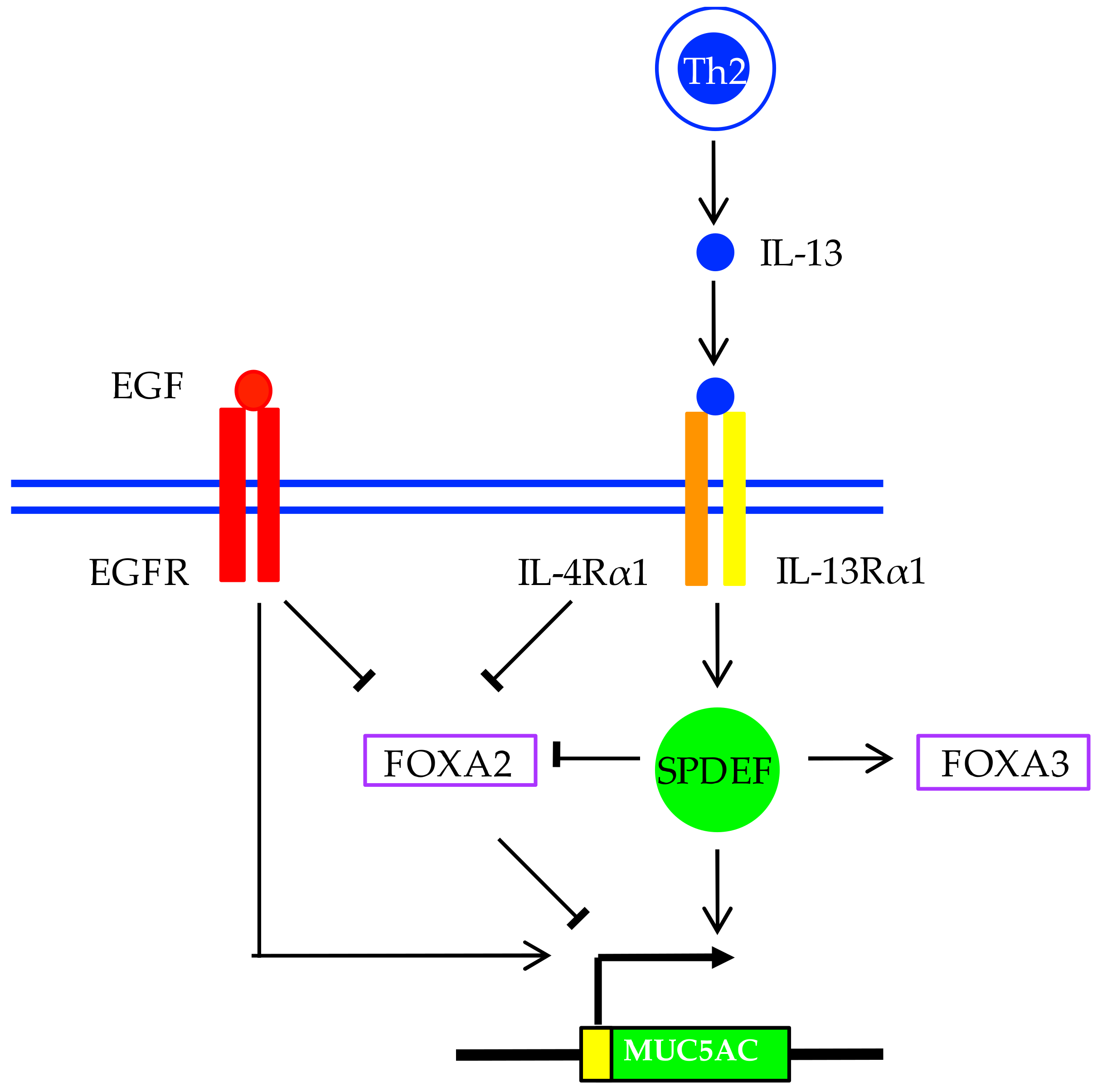
5. Regulation of MUC5AC and MUC5B Expression in Asthma
6. Goblet Cell Fate in Asthma
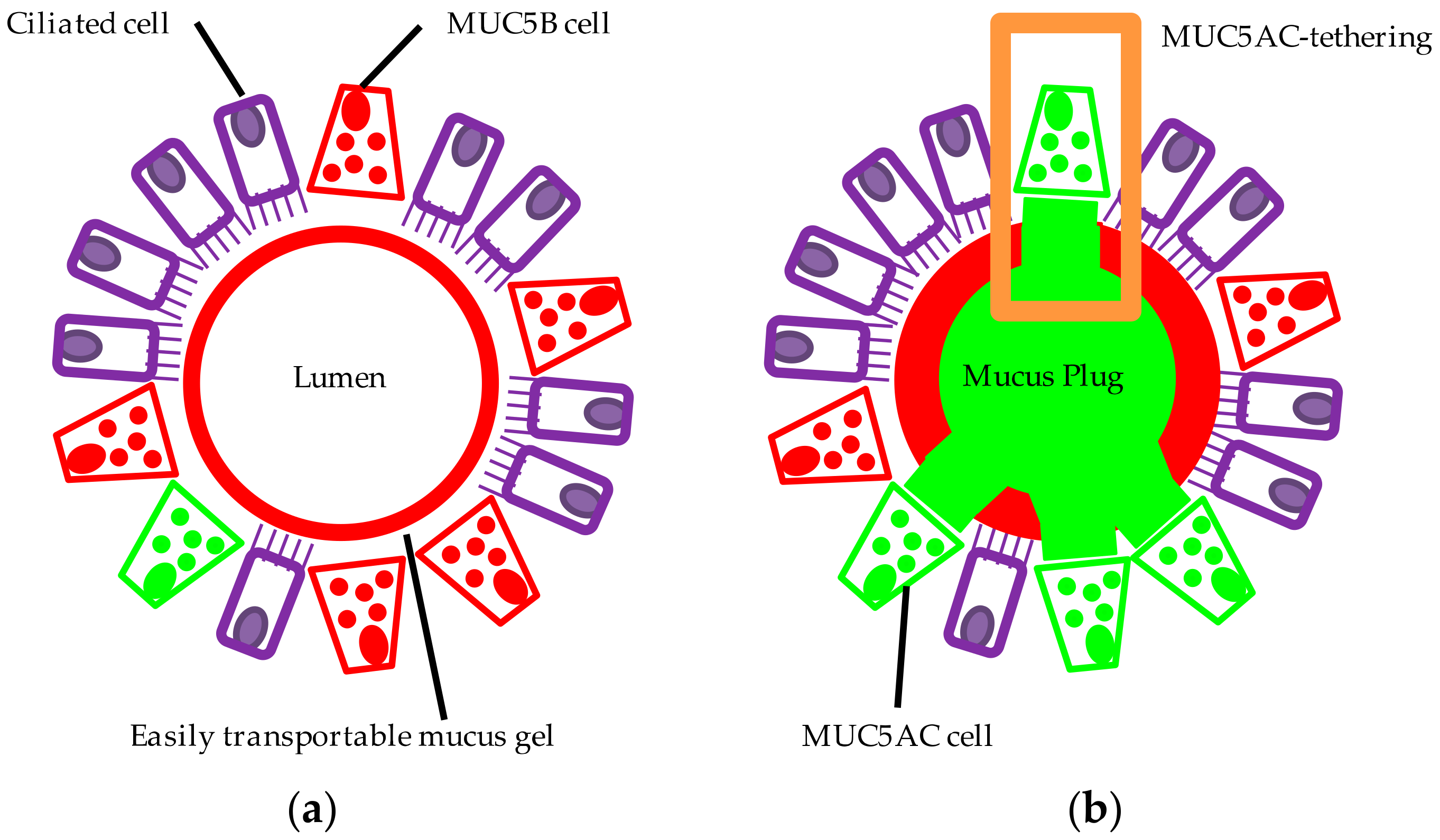
7. Altered MUC5AC and MUC5B Properties
8. Therapeutics
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Masoli, M.; Fabian, D.; Holt, S.; Beasley, R. Global Initiative for Asthma (GINA) Program The global burden of asthma: Executive summary of the GINA Dissemination Committee Report. Allergy 2004, 59, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Pearce, N.; Aït-Khaled, N.; Beasley, R.; Mallol, J.; Keil, U.; Mitchell, E.; Robertson, C. Worldwide trends in the prevalence of asthma symptoms: Phase III of the International Study of Asthma and Allergies in Childhood (ISAAC). Thorax 2007, 62, 758–766. [Google Scholar] [CrossRef] [PubMed]
- Fuhlbrigge, A.L.; Adams, R.J.; Guilbert, T.W.; Grant, E.; Lozano, P.; Janson, S.L.; Martinez, F.; Weiss, K.B.; Weiss, S.T. The Burden of Asthma in the United States. Am. J. Respir. Crit. Care Med. 2002, 166, 1044–1049. [Google Scholar] [CrossRef] [PubMed]
- Ferkol, T.; Schraufnagel, D. The Global Burden of Respiratory Disease. Ann. Am. Thorac. Soc. 2014, 11, 404–406. [Google Scholar] [CrossRef] [PubMed]
- Fanta, C. H. Asthma. N. Engl. J. Med. 2009, 360, 1002–1014. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, P.K.; Li, D. Airway mucosa: Secretory cells, mucus and mucin genes. Eur. Respir. J. 1997, 10, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Fahy, J.V.; Dickey, B.F. Airway Mucus Function and Dysfunction. N. Engl. J. Med. 2010, 363, 2233–2247. [Google Scholar] [CrossRef] [PubMed]
- Hackett, N.R.; Shaykhiev, R.; Walters, M.S.; Wang, R.; Zwick, R.K.; Ferris, B.; Witover, B.; Salit, J.; Crystal, R.G. The human airway epithelial basal cell transcriptome. PLoS ONE 2011, 6, e18378. [Google Scholar] [CrossRef] [PubMed]
- Thornton, D.J.; Sheehan, J.K. From mucins to mucus: Toward a more coherent understanding of this essential barrier. Proc. Am. Thorac. Soc. 2004, 1, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Erle, D.J.; Sheppard, D. The cell biology of asthma. J. Cell Biol. 2014, 205, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Button, B.; Cai, L.-H.; Ehre, C.; Kesimer, M.; Hill, D.B.; Sheehan, J.K.; Boucher, R.C.; Rubinstein, M. A Periciliary Brush Promotes the Lung Health by Separating the Mucus Layer from Airway Epithelia. Science 2012, 337, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.D. Lung mucus: A clinician’s view. Eur. Respir. J. 1997, 10, 1914–1917. [Google Scholar] [CrossRef] [PubMed]
- Dunnill, M.S. The pathology of asthma, with special reference to changes in the bronchial mucosa. J. Clin. Pathol. 1960, 13, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Messer, J.W.; Peters, G.A.; Bennett, W.A. Causes of Death and Pathologic Findings in 304 Cases of Bronchial Asthma. Dis. Chest 1960, 38, 616–624. [Google Scholar] [CrossRef] [PubMed]
- Kuyper, L.M.; Paré, P.D.; Hogg, J.C.; Lambert, R.K.; Ionescu, D.; Woods, R.; Bai, T.R. Characterization of airway plugging in fatal asthma. Am. J. Med. 2003, 115, 6–11. [Google Scholar] [CrossRef]
- Evans, C.M.; Kim, K.; Tuvim, M.J.; Dickey, B.F. Mucus hypersecretion in asthma: Causes and effects. Curr. Opin. Pulm. Med. 2009, 15, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Bateman, J.R.; Pavia, D.; Sheahan, N.F.; Agnew, J.E.; Clarke, S.W. Impaired tracheobronchial clearance in patients with mild stable asthma. Thorax 1983, 38, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Agnew, J.E.; Bateman, J.R.; Pavia, D.; Clarke, S.W. Radionuclide demonstration of ventilatory abnormalities in mild asthma. Clin. Sci. Lond. Engl. 1984, 66, 525–531. [Google Scholar] [CrossRef]
- O’Riordan, T.G.; Zwang, J.; Smaldone, G.C. Mucociliary Clearance in Adult Asthma. Am. Rev. Respir. Dis. 1992, 146, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Messina, M.S.; O’riordan, T.G.; Smaldone, G.C. Changes in Mucociliary Clearance during Acute Exacerbations of Asthma. Am. Rev. Respir. Dis. 1991, 143, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Thornton, D.J.; Rousseau, K.; McGuckin, M.A. Structure and function of the polymeric mucins in airways mucus. Annu. Rev. Physiol. 2008, 70, 459–486. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.M.; Koo, J.S. Airway mucus: The good, the bad, the sticky. Pharmacol. Ther. 2009, 121, 332–348. [Google Scholar] [CrossRef] [PubMed]
- Lang, T.; Hansson, G.C.; Samuelsson, T. Gel-forming mucins appeared early in metazoan evolution. Proc. Natl. Acad. Sci. USA 2007, 104, 16209–16214. [Google Scholar] [CrossRef] [PubMed]
- Rose, M.C.; Voynow, J.A. Respiratory Tract Mucin Genes and Mucin Glycoproteins in Health and Disease. Physiol. Rev. 2006, 86, 245–278. [Google Scholar] [CrossRef] [PubMed]
- Perez-Vilar, J.; Hill, R.L. The Structure and Assembly of Secreted Mucins. J. Biol. Chem. 1999, 274, 31751–31754. [Google Scholar] [CrossRef] [PubMed]
- Raviv, U.; Giasson, S.; Kampf, N.; Gohy, J.-F.; Jérôme, R.; Klein, J. Lubrication by charged polymers. Nature 2003, 425, 163–165. [Google Scholar] [CrossRef] [PubMed]
- Asker, N.; Axelsson, M.A.; Olofsson, S.O.; Hansson, G.C. Human MUC5AC mucin dimerizes in the rough endoplasmic reticulum, similarly to the MUC2 mucin. Biochem. J. 1998, 335, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Perez-Vilar, J.; Eckhardt, A.E.; DeLuca, A.; Hill, R.L. Porcine submaxillary mucin forms disulfide-linked multimers through its amino-terminal D-domains. J. Biol. Chem. 1998, 273, 14442–14449. [Google Scholar] [CrossRef] [PubMed]
- Perez-Vilar, J.; Eckhardt, A.E.; Hill, R.L. Porcine Submaxillary Mucin Forms Disulfide-bonded Dimers between Its Carboxyl-terminal Domains. J. Biol. Chem. 1996, 271, 9845–9850. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, J.K.; Kirkham, S.; Howard, M.; Woodman, P.; Kutay, S.; Brazeau, C.; Buckley, J.; Thornton, D.J. Identification of Molecular Intermediates in the Assembly Pathway of the MUC5AC Mucin. J. Biol. Chem. 2004, 279, 15698–15705. [Google Scholar] [CrossRef] [PubMed]
- Verdugo, P.; Deyrup-Olsen, I.; Aitken, M.; Villalon, M.; Johnson, D. Molecular Mechanism of Mucin Secretion: I. The Role of Intragranular Charge Shielding. J. Dent. Res. 1987, 66, 506–508. [Google Scholar] [CrossRef] [PubMed]
- Forstner, G. Signal Transduction Packaging and Secretion of Mucins. Annu. Rev. Physiol. 1995, 57, 585–605. [Google Scholar] [CrossRef] [PubMed]
- Ridley, C.; Kouvatsos, N.; Raynal, B.D.; Howard, M.; Collins, R.F.; Desseyn, J.-L.; Jowitt, T.A.; Baldock, C.; Davis, C.W.; Hardingham, T.E.; et al. Assembly of the Respiratory Mucin MUC5B: A new model for a gel-forming mucin. J. Biol. Chem. 2014, 289, 16409–16420. [Google Scholar] [CrossRef] [PubMed]
- Kesimer, M.; Makhov, A.M.; Griffith, J.D.; Verdugo, P.; Sheehan, J.K. Unpacking a gel-forming mucin: A view of MUC5B organization after granular release. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2010, 298, L15–L22. [Google Scholar] [CrossRef] [PubMed]
- Verdugo, P. Mucin exocytosis. Am. Rev. Respir. Dis. 1991, 144, S33–S37. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.Y.T.; Yang, N.; Quinton, P.M.; Chin, W.-C. A new role for bicarbonate in mucus formation. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2010, 299, L542–L549. [Google Scholar] [CrossRef] [PubMed]
- Audie, J.P.; Janin, A.; Porchet, N.; Copin, M.C.; Gosselin, B.; Aubert, J.P. Expression of human mucin genes in respiratory, digestive, and reproductive tracts ascertained by in situ hybridization. J. Histochem. Cytochem. Off. J. Histochem. Soc. 1993, 41, 1479–1485. [Google Scholar] [CrossRef]
- Porchet, N.; Pigny, P.; Buisine, M.-P.; Debailleul, V.; Degand, P.; Laine, A.; Aubert, J.-P. Human mucin genes: Genomic organization and expression of MUC4, MUC5AC and MUC5B. Biochem. Soc. Trans. 1995, 23, 800–805. [Google Scholar] [CrossRef] [PubMed]
- Reid, C.J.; Gould, S.; Harris, A. Developmental Expression of Mucin Genes in the Human Respiratory Tract. Am. J. Respir. Cell Mol. Biol. 1997, 17, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Buisine, M.-P.; Devisme, L.; Copin, M.-C.; Durand-Réville, M.; Gosselin, B.; Aubert, J.-P.; Porchet, N. Developmental Mucin Gene Expression in the Human Respiratory Tract. Am. J. Respir. Cell Mol. Biol. 1999, 20, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhao, Y.H.; Kalaslavadi, T.B.; Hamati, E.; Nehrke, K.; Le, A.D.; Ann, D.K.; Wu, R. Genome-Wide Search and Identification of a Novel Gel-Forming Mucin MUC19/Muc19 in Glandular Tissues. Am. J. Respir. Cell Mol. Biol. 2004, 30, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Hovenberg, H.W.; Davies, J.R.; Herrmann, A.; Lindén, C.J.; Carlstedt, I. MUC5AC, but not MUC2, is a prominent mucin in respiratory secretions. Glycoconj. J. 1996, 13, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Wickström, C.; Davies, J.R.; Eriksen, G.V.; Veerman, E.C.; Carlstedt, I. MUC5B is a major gel-forming, oligomeric mucin from human salivary gland, respiratory tract and endocervix: Identification of glycoforms and C-terminal cleavage. Biochem. J. 1998, 334, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, S.; Sheehan, J.K.; Knight, D.; Richardson, P.S.; Thornton, D.J. Heterogeneity of airways mucus: Variations in the amounts and glycoforms of the major oligomeric mucins MUC5AC and MUC5B. Biochem. J. 2002, 361, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Thornton, D.J.; Howard, M.; Khan, N.; Sheehan, J.K. Identification of two glycoforms of the MUC5B mucin in human respiratory mucus. Evidence for a cysteine-rich sequence repeated within the molecule. J. Biol. Chem. 1997, 272, 9561–9566. [Google Scholar] [CrossRef] [PubMed]
- Kesimer, M.; Kirkham, S.; Pickles, R.J.; Henderson, A.G.; Alexis, N.E.; Demaria, G.; Knight, D.; Thornton, D.J.; Sheehan, J.K. Tracheobronchial air-liquid interface cell culture: A model for innate mucosal defense of the upper airways? Am. J. Physiol.-Lung Cell. Mol. Physiol. 2009, 296, L92–L100. [Google Scholar] [CrossRef] [PubMed]
- Groneberg, D.A.; Eynott, P.R.; Lim, S.; Oates, T.; Wu, R.; Carlstedt, I.; Roberts, P.; McCann, B.; Nicholson, A.G.; Harrison, B.D.; et al. Expression of respiratory mucins in fatal status asthmaticus and mild asthma. Histopathology 2002, 40, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Groneberg, D.A.; Eynott, P.R.; Oates, T.; Lim, S.; Wu, R.; Carlstedt, I.; Nicholson, A.G.; Chung, K.F. Expression of MUC5AC and MUC5B mucins in normal and cystic fibrosis lung. Respir. Med. 2002, 96, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Hovenberg, H.W.; Davies, J.R.; Carlstedt, I. Different mucins are produced by the surface epithelium and the submucosa in human trachea: Identification of MUC5AC as a major mucin from the goblet cells. Biochem. J. 1996, 318, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Seibold, M.A.; Smith, R.W.; Urbanek, C.; Groshong, S.D.; Cosgrove, G.P.; Brown, K.K.; Schwarz, M.I.; Schwartz, D.A.; Reynolds, S.D. The Idiopathic Pulmonary Fibrosis Honeycomb Cyst Contains A Mucocilary Pseudostratified Epithelium. PLoS ONE 2013, 8, e58658. [Google Scholar] [CrossRef] [PubMed]
- Seibold, M.A.; Wise, A.L.; Speer, M.C.; Steele, M.P.; Brown, K.K.; Loyd, J.E.; Fingerlin, T.E.; Zhang, W.; Gudmundsson, G.; Groshong, S.D.; et al. A common MUC5B promoter polymorphism and pulmonary fibrosis. N. Engl. J. Med. 2011, 364, 1503–1512. [Google Scholar] [CrossRef] [PubMed]
- Bonser, L.R.; Zlock, L.; Finkbeiner, W.; Erle, D.J. Epithelial tethering of MUC5AC-rich mucus impairs mucociliary transport in asthma. J. Clin. Investig. 2016, 126, 2367–2371. [Google Scholar] [CrossRef] [PubMed]
- Lachowicz-Scroggins, M.E.; Yuan, S.; Kerr, S.C.; Dunican, E.M.; Yu, M.; Carrington, S.D.; Fahy, J.V. Abnormalities in MUC5AC and MUC5B Protein in Airway Mucus in Asthma. Am. J. Respir. Crit. Care Med. 2016, 194, 1296–1299. [Google Scholar] [CrossRef] [PubMed]
- Ermund, A.; Meiss, L.N.; Rodriguez-Pineiro, A.M.; Bähr, A.; Nilsson, H.E.; Trillo-Muyo, S.; Ridley, C.; Thornton, D.J.; Wine, J.J.; Hebert, H.; et al. The normal trachea is cleaned by MUC5B mucin bundles from the submucosal glands coated with the MUC5AC mucin. Biochem. Biophys. Res. Commun. 2017, 492, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Ostedgaard, L.S.; Moninger, T.O.; McMenimen, J.D.; Sawin, N.M.; Parker, C.P.; Thornell, I.M.; Powers, L.S.; Gansemer, N.D.; Bouzek, D.C.; Cook, D.P.; et al. Gel-forming mucins form distinct morphologic structures in airways. Proc. Natl. Acad. Sci. USA 2017, 114, 6842–6847. [Google Scholar] [CrossRef] [PubMed]
- Thornton, D.J.; Davies, J.R.; Kraayenbrink, M.; Richardson, P.S.; Sheehan, J.K.; Carlstedt, I. Mucus glycoproteins from ‘normal’ human tracheobronchial secretion. Biochem. J. 1990, 265, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, J.K.; Brazeau, C.; Kutay, S.; Pigeon, H.; Kirkham, S.; Howard, M.; Thornton, D.J. Physical characterization of the MUC5AC mucin: A highly oligomeric glycoprotein whether isolated from cell culture or in vivo from respiratory mucous secretions. Biochem. J. 2000, 347, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Raclawska, D.S.; Ttofali, F.; Fletcher, A.A.; Harper, D.N.; Bochner, B.S.; Janssen, W.J.; Evans, C.M. Mucins and Their Sugars. Critical Mediators of Hyperreactivity and Inflammation. Ann. Am. Thorac. Soc. 2016, 13 (Suppl. 1), S98–S99. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Ehre, C.; Abdullah, L.H.; Sheehan, J.K.; Roy, M.; Evans, C.M.; Dickey, B.F.; Davis, C.W. Munc13-2−/− baseline secretion defect reveals source of oligomeric mucins in mouse airways. J. Physiol. 2008, 586, 1977–1992. [Google Scholar] [CrossRef] [PubMed]
- Roy, M.G.; Livraghi-Butrico, A.; Fletcher, A.A.; McElwee, M.M.; Evans, S.E.; Boerner, R.M.; Alexander, S.N.; Bellinghausen, L.K.; Song, A.S.; Petrova, Y.M.; et al. Muc5b is required for airway defence. Nature 2014, 505, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Livraghi-Butrico, A.; Grubb, B.R.; Wilkinson, K.J.; Volmer, A.S.; Burns, K.A.; Evans, C.M.; O’Neal, W.K.; Boucher, R.C. Contribution of mucus concentration and secreted mucins Muc5ac and Muc5b to the pathogenesis of muco-obstructive lung disease. Mucosal Immunol. 2017, 10, 395–407. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.M.; Raclawska, D.S.; Ttofali, F.; Liptzin, D.R.; Fletcher, A.A.; Harper, D.N.; McGing, M.A.; McElwee, M.M.; Williams, O.W.; Sanchez, E.; et al. The polymeric mucin Muc5ac is required for allergic airway hyperreactivity. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Ehre, C.; Worthington, E.N.; Liesman, R.M.; Grubb, B.R.; Barbier, D.; O’Neal, W.K.; Sallenave, J.-M.; Pickles, R.J.; Boucher, R.C. Overexpressing mouse model demonstrates the protective role of Muc5ac in the lungs. Proc. Natl. Acad. Sci. USA 2012, 109, 16528–16533. [Google Scholar] [CrossRef] [PubMed]
- Koeppen, M.; McNamee, E.N.; Brodsky, K.S.; Aherne, C.M.; Faigle, M.; Downey, G.P.; Colgan, S.P.; Evans, C.M.; Schwartz, D.A.; Eltzschig, H.K. Detrimental role of the airway mucin Muc5ac during ventilator-induced lung injury. Mucosal Immunol. 2013, 6, 762–775. [Google Scholar] [CrossRef] [PubMed]
- Rock, J.R.; Randell, S.H.; Hogan, B.L.M. Airway basal stem cells: A perspective on their roles in epithelial homeostasis and remodeling. Dis. Models Mech. 2010, 3, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Curran, D.R.; Cohn, L. Advances in Mucous Cell Metaplasia. Am. J. Respir. Cell Mol. Biol. 2010, 42, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, P.G.; Modrek, B.; Choy, D.F.; Jia, G.; Abbas, A.R.; Ellwanger, A.; Arron, J.R.; Koth, L.L.; Fahy, J.V. T-helper Type 2–driven Inflammation Defines Major Subphenotypes of Asthma. Am. J. Respir. Crit. Care Med. 2009, 180, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Kuperman, D.A.; Lewis, C.C.; Woodruff, P.G.; Rodriguez, M.W.; Yang, Y.H.; Dolganov, G.M.; Fahy, J.V.; Erle, D.J. Dissecting asthma using focused transgenic modeling and functional genomics. J. Allergy Clin. Immunol. 2005, 116, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, P.G.; Boushey, H.A.; Dolganov, G.M.; Barker, C.S.; Yang, Y.H.; Donnelly, S.; Ellwanger, A.; Sidhu, S.S.; Dao-Pick, T.P.; Pantoja, C.; et al. Genome-wide profiling identifies epithelial cell genes associated with asthma and with treatment response to corticosteroids. Proc. Natl. Acad. Sci. USA 2007, 104, 15858–15863. [Google Scholar] [CrossRef] [PubMed]
- Zhen, G.; Park, S.W.; Nguyenvu, L.T.; Rodriguez, M.W.; Barbeau, R.; Paquet, A.C.; Erle, D.J. IL-13 and Epidermal Growth Factor Receptor Have Critical but Distinct Roles in Epithelial Cell Mucin Production. Am. J. Respir. Cell Mol. Biol. 2007, 36, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Solberg, O.D.; Ostrin, E.J.; Love, M.I.; Peng, J.C.; Bhakta, N.R.; Hou, L.; Nguyen, C.; Solon, M.; Nguyen, C.; Barczak, A.J.; et al. Airway Epithelial miRNA Expression Is Altered in Asthma. Am. J. Respir. Crit. Care Med. 2012, 186, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Lachowicz-Scroggins, M.E.; Finkbeiner, W.E.; Gordon, E.D.; Yuan, S.; Zlock, L.; Bhakta, N.R.; Woodruff, P.G.; Fahy, J.V.; Boushey, H.A. Corticosteroid and long-acting ß-agonist therapy reduces epithelial goblet cell metaplasia. Clin. Exp. Allergy 2017. [Google Scholar] [CrossRef] [PubMed]
- Kuperman, D.A.; Huang, X.; Koth, L.L.; Chang, G.H.; Dolganov, G.M.; Zhu, Z.; Elias, J.A.; Sheppard, D.; Erle, D.J. Direct effects of interleukin-13 on epithelial cells cause airway hyperreactivity and mucus overproduction in asthma. Nat. Med. 2002, 8, 885–889. [Google Scholar] [CrossRef] [PubMed]
- Laoukili, J.; Perret, E.; Willems, T.; Minty, A.; Parthoens, E.; Houcine, O.; Coste, A.; Jorissen, M.; Marano, F.; Caput, D.; et al. IL-13 alters mucociliary differentiation and ciliary beating of human respiratory epithelial cells. J. Clin. Investig. 2001, 108, 1817–1824. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.L.; Ling, M.F.; Adams, D.C.; Faustino, L.; Islam, S.A.; Afshar, R.; Griffith, J.W.; Harris, R.S.; Ng, A.; Radicioni, G.; et al. Allergic asthma is distinguished by sensitivity of allergen-specific CD4+ T cells and airway structural cells to type 2 inflammation. Sci. Transl. Med. 2016, 8, 359ra132. [Google Scholar] [CrossRef] [PubMed]
- Takeyama, K.; Dabbagh, K.; Lee, H.-M.; Agustí, C.; Lausier, J.A.; Ueki, I.F.; Grattan, K.M.; Nadel, J.A. Epidermal growth factor system regulates mucin production in airways. Proc. Natl. Acad. Sci. USA 1999, 96, 3081–3086. [Google Scholar] [CrossRef] [PubMed]
- Cras, T.D.L.; Acciani, T.H.; Mushaben, E.M.; Kramer, E.L.; Pastura, P.A.; Hardie, W.D.; Korfhagen, T.R.; Sivaprasad, U.; et al. Epithelial EGF receptor signaling mediates airway hyperreactivity and remodeling in a mouse model of chronic asthma. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2011, 300, L414–L421. [Google Scholar] [CrossRef] [PubMed]
- Takeyama, K.; Fahy, J.V.; Nadel, J.A. Relationship of epidermal growth factor receptors to goblet cell production in human bronchi. Am. J. Respir. Crit. Care Med. 2001, 163, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Donoghue, L.J.; Livraghi-Butrico, A.; McFadden, K.M.; Thomas, J.M.; Chen, G.; Grubb, B.R.; O’Neal, W.K.; Boucher, R.C.; Kelada, S.N.P. Identification of trans Protein QTL for Secreted Airway Mucins in Mice and a Causal Role for Bpifb1. Genetics 2017, 207, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Aikawa, T.; Shimura, S.; Sasaki, H.; Ebina, M.; Takishima, T. Marked goblet cell hyperplasia with mucus accumulation in the airways of patients who died of severe acute asthma attack. Chest 1992, 101, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Ordoñez, C.L.; Khashayar, R.; Wong, H.H.; Ferrando, R.; Wu, R.; Hyde, D.M.; Hotchkiss, J.A.; Zhang, Y.; Novikov, A.; Dolganov, G.; et al. Mild and moderate asthma is associated with airway goblet cell hyperplasia and abnormalities in mucin gene expression. Am. J. Respir. Crit. Care Med. 2001, 163, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Crystal, R.G. Airway Basal Cells. The “Smoking Gun” of Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2014, 190, 1355–1362. [Google Scholar] [CrossRef] [PubMed]
- Boucherat, O.; Morissette, M.C.; Provencher, S.; Bonnet, S.; Maltais, F. Bridging Lung Development with Chronic Obstructive Pulmonary Disease. Relevance of Developmental Pathways in Chronic Obstructive Pulmonary Disease Pathogenesis. Am. J. Respir. Crit. Care Med. 2015, 193, 362–375. [Google Scholar] [CrossRef] [PubMed]
- Danahay, H.; Pessotti, A.D.; Coote, J.; Montgomery, B.E.; Xia, D.; Wilson, A.; Yang, H.; Wang, Z.; Bevan, L.; Thomas, C.; et al. Notch2 Is Required for Inflammatory Cytokine-Driven Goblet Cell Metaplasia in the Lung. Cell Rep. 2015, 10, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Lafkas, D.; Shelton, A.; Chiu, C.; de Leon Boenig, G.; Chen, Y.; Stawicki, S.S.; Siltanen, C.; Reichelt, M.; Zhou, M.; Wu, X.; et al. Therapeutic antibodies reveal Notch control of transdifferentiation in the adult lung. Nature 2015, 528, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Rajavelu, P.; Chen, G.; Xu, Y.; Kitzmiller, J.A.; Korfhagen, T.R.; Whitsett, J.A. Airway epithelial SPDEF integrates goblet cell differentiation and pulmonary Th2 inflammation. J. Clin. Investig. 2015, 125, 2021–2031. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Korfhagen, T.R.; Karp, C.L.; Impey, S.; Xu, Y.; Randell, S.H.; Kitzmiller, J.; Maeda, Y.; Haitchi, H.M.; Sridharan, A.; et al. Foxa3 Induces Goblet Cell Metaplasia and Inhibits Innate Antiviral Immunity. Am. J. Respir. Crit. Care Med. 2014, 189, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Park, K.-S.; Korfhagen, T.R.; Bruno, M.D.; Kitzmiller, J.A.; Wan, H.; Wert, S.E.; Khurana Hershey, G.K.; Chen, G.; Whitsett, J.A. SPDEF regulates goblet cell hyperplasia in the airway epithelium. J. Clin. Investig. 2007, 117, 978–988. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Heijink, I.H.; Kistemaker, L.E.M.; Reinders-Luinge, M.; Kooistra, W.; Noordhoek, J.A.; Gosens, R.; Brandsma, C.A.; Timens, W.; Hiemstra, P.S.; et al. Aberrant DNA methylation and expression of SPDEF and FOXA2 in airway epithelium of patients with COPD. Clin. Epigenet. 2017, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Cano-Rodriquez, D.; Winkle, M.; Gjaltema, R.A.F.; Goubert, D.; Jurkowski, T.P.; Heijink, I.H.; Rots, M.G.; Hylkema, M.N. Targeted epigenetic editing of SPDEF reduces mucus production in lung epithelial cells. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2017, 312, L334–L347. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Korfhagen, T.R.; Xu, Y.; Kitzmiller, J.; Wert, S.E.; Maeda, Y.; Gregorieff, A.; Clevers, H.; Whitsett, J.A. SPDEF is required for mouse pulmonary goblet cell differentiation and regulates a network of genes associated with mucus production. J. Clin. Investig. 2009, 119, 2914–2924. [Google Scholar] [CrossRef] [PubMed]
- Wan, H.; Kaestner, K.H.; Ang, S.-L.; Ikegami, M.; Finkelman, F.D.; Stahlman, M.T.; Fulkerson, P.C.; Rothenberg, M.E.; Whitsett, J.A. Foxa2 regulates alveolarization and goblet cell hyperplasia. Development 2004, 131, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Wan, H. Luo, F.; Zhang, L.; Xu, Y.; Lewkowich, I.; Wills-Karp, M.; Whitsett, J.A. Foxa2 Programs Th2 Cell-Mediated Innate Immunity in the Developing Lung. J. Immunol. 2010, 184, 6133–6141. [Google Scholar] [CrossRef] [PubMed]
- Young, H.W.J.; Williams, O.W.; Chandra, D.; Bellinghausen, L.K.; Pérez, G.; Suárez, A.; Tuvim, M.J.; Roy, M.G.; Alexander, S.N.; Moghaddam, S.J.; et al. Central Role of Muc5ac Expression in Mucous Metaplasia and Its Regulation by Conserved 5′ Elements. Am. J. Respir. Cell Mol. Biol. 2007, 37, 273–290. [Google Scholar] [CrossRef] [PubMed]
- Welsh, K.G.; Rousseau, K.; Fisher, G.; Bonser, L.R.; Bradding, P.; Brightling, C.E.; Thornton, D.J.; Gaillard, E.A. MUC5AC and a glycosylated variant of MUC5B alter mucin composition in children with acute asthma. Chest 2017. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, J.K.; Richardson, P.S.; Fung, D.C.; Howard, M.; Thornton, D.J. Analysis of respiratory mucus glycoproteins in asthma: A detailed study from a patient who died in status asthmaticus. Am. J. Respir. Cell Mol. Biol. 1995, 13, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Innes, A.L.; McGrath, K.W.; Dougherty, R.H.; McCulloch, C.E.; Woodruff, P.G.; Seibold, M.A.; Okamoto, K.S.; Ingmundson, K.J.; Solon, M.C.; Carrington, S.D.; et al. The H Antigen at Epithelial Surfaces Is Associated with Susceptibility to Asthma Exacerbation. Am. J. Respir. Crit. Care Med. 2011, 183, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Vidriero, M.T.; Reid, L. Chemical markers of mucous and serum glycoproteins and their relation to viscosity in mucoid and purulent sputum from various hypersecretory diseases. Am. Rev. Respir. Dis. 1978, 117, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Innes, A.L.; Carrington, S.D.; Thornton, D.J.; Kirkham, S.; Rousseau, K.; Dougherty, R.H.; Raymond, W.W.; Caughey, G.H.; Muller, S.J.; Fahy, J.V. Ex Vivo Sputum Analysis Reveals Impairment of Protease-dependent Mucus Degradation by Plasma Proteins in Acute Asthma. Am. J. Respir. Crit. Care Med. 2009, 180, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Hollinger, M.; Lachowicz-Scroggins, M.E.; Kerr, S.C.; Dunican, E.M.; Daniel, B.M.; Ghosh, S.; Erzurum, S.C.; Willard, B.; Hazen, S.L.; et al. Oxidation increases mucin polymer cross-links to stiffen airway mucus gels. Sci. Transl. Med. 2015, 7, 276ra27. [Google Scholar] [CrossRef] [PubMed]
- Kesic, M.J.; Hernandez, M.; Jaspers, I. Airway protease/antiprotease imbalance in atopic asthmatics contributes to increased Influenza A virus cleavage and replication. Respir. Res. 2012, 13, 82. [Google Scholar] [CrossRef] [PubMed]
- Shimura, S.; Andoh, Y.; Haraguchi, M.; Shirato, K. Continuity of airway goblet cells and intraluminal mucus in the airways of patients with bronchial asthma. Eur. Respir. J. 1996, 9, 1395–1401. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, L.H.; Evans, J.R.; Wang, T.T.; Ford, A.A.; Makhov, A.M.; Nguyen, K.; Coakley, R.D.; Griffith, J.D.; Davis, C.W.; Ballard, S.T.; et al. Defective postsecretory maturation of MUC5B mucin in cystic fibrosis airways. JCI Insight 2017, 2, e89752. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.X.; Ostedgaard, L.S.; Hoegger, M.J.; Moninger, T.O.; Karp, P.H.; McMenimen, J.D.; Choudhury, B.; Varki, A.; Stoltz, D.A.; Welsh, M.J. Acidic pH increases airway surface liquid viscosity in cystic fibrosis. J. Clin. Investig. 2016, 126, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Shimura, S.; Sasaki, T.; Sasaki, H.; Takishima, T. Chemical properties of bronchorrhea sputum in bronchial asthma. Chest 1988, 94, 1211–1215. [Google Scholar] [CrossRef] [PubMed]
- Kerr, S.C.; Carrington, S.D.; Oscarson, S.; Gallagher, M.E.; Solon, M.; Yuan, S.; Ahn, J.N.; Dougherty, R.H.; Finkbeiner, W.E.; Peters, M.C.; et al. Intelectin-1 Is a Prominent Protein Constituent of Pathologic Mucus Associated with Eosinophilic Airway Inflammation in Asthma. Am. J. Respir. Crit. Care Med. 2014, 189, 1005–1007. [Google Scholar] [CrossRef] [PubMed]
- Capri, S.; Gallus, G.; Grassi, C.; Huchon, G.J.; Irvine, S.H.; Lopez-Vidriero, M.T.; Luisetti, M.; Lungarella, G.; Marriott, C.; Marsac, J.; et al. Recommendations for guidelines on clinical trials of mucoactive drugs in chronic bronchitis and chronic obstructive pulmonary disease. Task Group on Mucoactive Drugs. Chest 1994, 106, 1532–1537. [Google Scholar]
- Rogers, D.F.; Barnes, P.J. Treatment of airway mucus hypersecretion. Ann. Med. 2006, 38, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Sadowska, A.M. N-Acetylcysteine mucolysis in the management of chronic obstructive pulmonary disease. Ther. Adv. Respir. Dis. 2012, 6, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Daviskas, E.; Anderson, S.D.; Gonda, I.; Eberl, S.; Meikle, S.; Seale, J.P.; Bautovich, G. Inhalation of hypertonic saline aerosol enhances mucociliary clearance in asthmatic and healthy subjects. Eur. Respir. J. 1996, 9, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Cotgreave, I.A.; Eklund, A.; Larsson, K.; Moldéus, P.W. No penetration of orally administered N-acetylcysteine into bronchoalveolar lavage fluid. Eur. J. Respir. Dis. 1987, 70, 73–77. [Google Scholar] [PubMed]
- Shen, Y.; Cai, W.; Lei, S.; Zhang, Z. Effect of high/low dose N-acetylcysteine on chronic obstructive pulmonary disease: A systematic review and meta-analysis. COPD 2014, 11, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Dano, G. Bronchospasm caused by acetylcysteine in children with bronchial asthma. Acta Allergol. 1971, 26, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Gillissen, A.; Jaworska, M.; Orth, M.; Coffiner, M.; Maes, P.; App, E.M.; Cantin, A.M.; Schultze-Werninghaus, G. Nacystelyn, a novel lysine salt of N-acetylcysteine, to augment cellular antioxidant defence in vitro. Respir. Med. 1997, 91, 159–168. [Google Scholar] [CrossRef]
- Suk, J.S.; Boylan, N.J.; Trehan, K.; Tang, B.C.; Schneider, C.S.; Lin, J.-M.G.; Boyle, M.P.; Zeitlin, P.L.; Lai, S.K.; Cooper, M.J.; et al. N-acetylcysteine enhances cystic fibrosis sputum penetration and airway gene transfer by highly compacted DNA nanoparticles. Mol. Ther. J. Am. Soc. Gene Ther. 2011, 19, 1981–1989. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Wilson, D.B.; Brooks, R.C.; Sproule, B.J. Acute effects of nebulization of N-acetylcysteine on pulmonary mechanics and gas exchange. Am. Rev. Respir. Dis. 1970, 102, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, H.J.; Borowitz, D.S.; Christiansen, D.H.; Morris, E.M.; Nash, M.L.; Ramsey, B.W.; Rosenstein, B.J.; Smith, A.L.; Wohl, M.E. Effect of aerosolized recombinant human DNase on exacerbations of respiratory symptoms and on pulmonary function in patients with cystic fibrosis. The Pulmozyme Study Group. N. Engl. J. Med. 1994, 331, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Laube, B.L.; Auci, R.M.; Shields, D.E.; Christiansen, D.H.; Lucas, M.K.; Fuchs, H.J.; Rosenstein, B.J. Effect of rhDNase on airflow obstruction and mucociliary clearance in cystic fibrosis. Am. J. Respir. Crit. Care Med. 1996, 153, 752–760. [Google Scholar] [CrossRef] [PubMed]
- Katial, R.K.; Bernstein, D.; Prazma, C.M.; Lincourt, W.R.; Stempel, D.A. Long-term treatment with fluticasone propionate/salmeterol via Diskus improves asthma control versus fluticasone propionate alone. Allergy Asthma Proc. 2011, 32, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.S.; Custovic, A.; Lowe, L.A.; Aldington, S.; Williams, M.; Beasley, R.; Woodcock, A. Effect of addition of salmeterol versus doubling the dose of fluticasone propionate on specific airway resistance in children with asthma. Allergy Asthma Proc. 2010, 31, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Kitano, M.; Ishinaga, H.; Shimizu, T.; Takeuchi, K.; Majima, Y. Effects of clarithromycin and dexamethasone on mucus production in isografted rat trachea. Pharmacology 2011, 87, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Southam, D.S.; Ellis, R.; Wattie, J.; Glass, W.; Inman, M.D. Goblet cell rebound and airway dysfunction with corticosteroid withdrawal in a mouse model of asthma. Am. J. Respir. Crit. Care Med. 2008, 178, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Riesenfeld, E.P.; Sullivan, M.J.; Thompson-Figueroa, J.A.; Haverkamp, H.C.; Lundblad, L.K.; Bates, J.H.T.; Irvin, C.G. Inhaled salmeterol and/or fluticasone alters structure/function in a murine model of allergic airways disease. Respir. Res. 2010, 11, 22. [Google Scholar] [CrossRef] [PubMed]
- Fahy, J.V.; Fleming, H.E.; Wong, H.H.; Liu, J.T.; Su, J.Q.; Reimann, J.; Fick, R.B.; Boushey, H.A. The effect of an anti-IgE monoclonal antibody on the early- and late-phase responses to allergen inhalation in asthmatic subjects. Am. J. Respir. Crit. Care Med. 1997, 155, 1828–1834. [Google Scholar] [CrossRef] [PubMed]
- Corren, J.; Lemanske, R.F.J.; Hanania, N.A.; Korenblat, P.E.; Parsey, M.V.; Arron, J.R.; Harris, J.M.; Scheerens, H.; Wu, L.C.; Su, Z.; et al. Lebrikizumab Treatment in Adults with Asthma. N. Engl. J. Med. 2011, 365, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Hanania, N.A.; Korenblat, P.; Chapman, K.R.; Bateman, E.D.; Kopecky, P.; Paggiaro, P.; Yokoyama, A.; Olsson, J.; Gray, S.; Holweg, C.T.J.; et al. Efficacy and safety of lebrikizumab in patients with uncontrolled asthma (LAVOLTA I and LAVOLTA II): Replicate, phase 3, randomised, double-blind, placebo-controlled trials. Lancet Respir. Med. 2016, 4, 781–796. [Google Scholar] [CrossRef]
- Wenzel, S.; Ford, L.; Pearlman, D.; Spector, S.; Sher, L.; Skobieranda, F.; Wang, L.; Kirkesseli, S.; Rocklin, R.; Bock, B.; et al. Dupilumab in Persistent Asthma with Elevated Eosinophil Levels. N. Engl. J. Med. 2013, 368, 2455–2466. [Google Scholar] [CrossRef] [PubMed]
- Corren, J.; Parnes, J.R.; Wang, L.; Mo, M.; Roseti, S.L.; Griffiths, J.M.; van der Merwe, R. Tezepelumab in Adults with Uncontrolled Asthma. N. Engl. J. Med. 2017, 377, 936–946. [Google Scholar] [CrossRef] [PubMed]
- Rogers, D.F. Mucoactive drugs for asthma and COPD: Any place in therapy? Expert Opin. Investig. Drugs 2002, 11, 15–35. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonser, L.R.; Erle, D.J. Airway Mucus and Asthma: The Role of MUC5AC and MUC5B. J. Clin. Med. 2017, 6, 112. https://doi.org/10.3390/jcm6120112
Bonser LR, Erle DJ. Airway Mucus and Asthma: The Role of MUC5AC and MUC5B. Journal of Clinical Medicine. 2017; 6(12):112. https://doi.org/10.3390/jcm6120112
Chicago/Turabian StyleBonser, Luke R., and David J. Erle. 2017. "Airway Mucus and Asthma: The Role of MUC5AC and MUC5B" Journal of Clinical Medicine 6, no. 12: 112. https://doi.org/10.3390/jcm6120112
APA StyleBonser, L. R., & Erle, D. J. (2017). Airway Mucus and Asthma: The Role of MUC5AC and MUC5B. Journal of Clinical Medicine, 6(12), 112. https://doi.org/10.3390/jcm6120112




