Identification of Recurrence-Related microRNAs from Bone Marrow in Hepatocellular Carcinoma Patients
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Whole BM Fraction
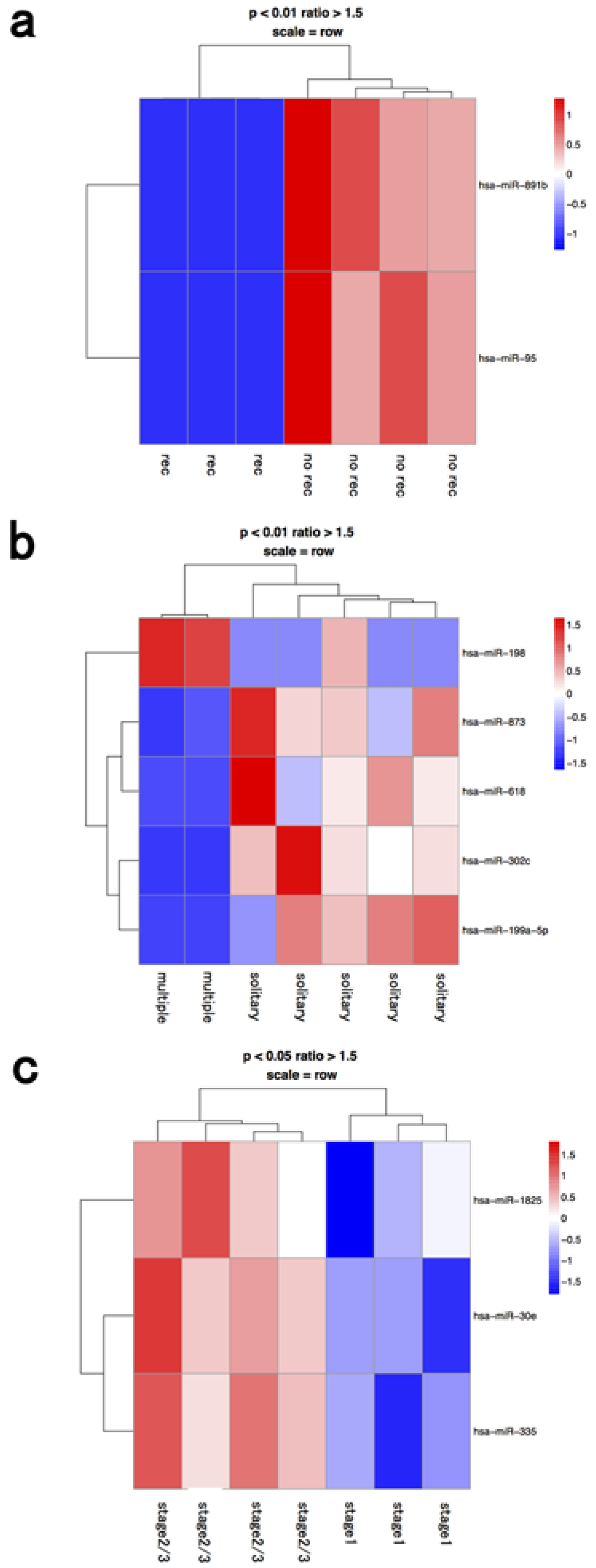
| BM Fraction | Clinical Significance | Upregulated microRNA | Downregulated microRNA |
|---|---|---|---|
| whole BM | recurrence | hsa-mir-891b | |
| recurrence | hsa-mir-95 | ||
| multiple | hsa-mir-198 | hsa-mir-873 | |
| multiple | hsa-mir-618 | ||
| multiple | hsa-mir-302c | ||
| multiple | hsa-mir-199a-5p | ||
| stage 2≥ | hsa-mir-1825 | ||
| stage 2≥ | hsa-mir-30e | ||
| stage 2≥ | hsa-mir-335 | ||
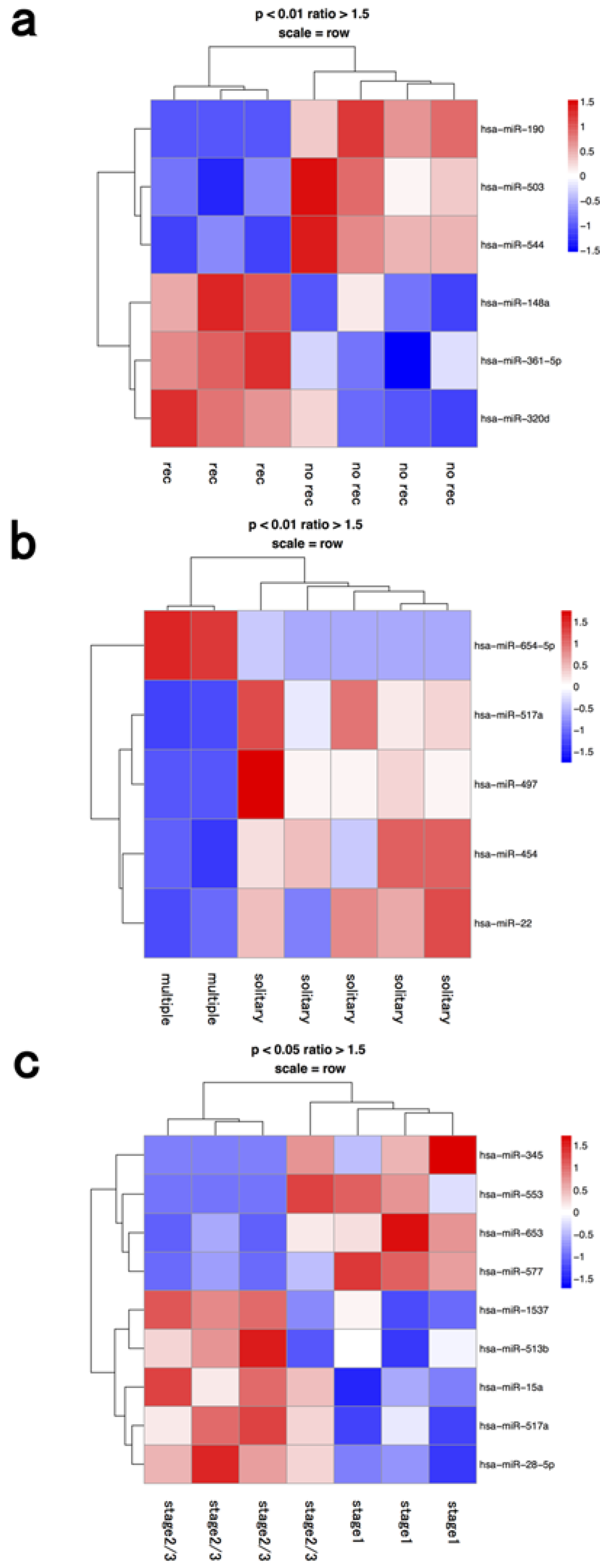
| lymphocyte | recurrence | hsa-mir-148a | hsa-mir-190 |
| recurrence | hsa-mir-361-5p | hsa-mir-503 | |
| recurrence | hsa-mir-320d | hsa-mir-544 | |
| multiple | hsa-mir-654-5p | hsa-mir-517a | |
| multiple | hsa-mir-497 | ||
| multiple | hsa-mir-454 | ||
| multiple | hsa-mir-22 | ||
| stage 2≥ | hsa-mir-1537 | hsa-mir-345 | |
| stage 2≥ | hsa-mir-513b | hsa-mir-553 | |
| stage 2≥ | hsa-mir-15a | hsa-mir-653 | |
| stage 2≥ | hsa-mir-517a | hsa-mir-577 | |
| stage 2≥ | hsa-mir-28-5p | ||
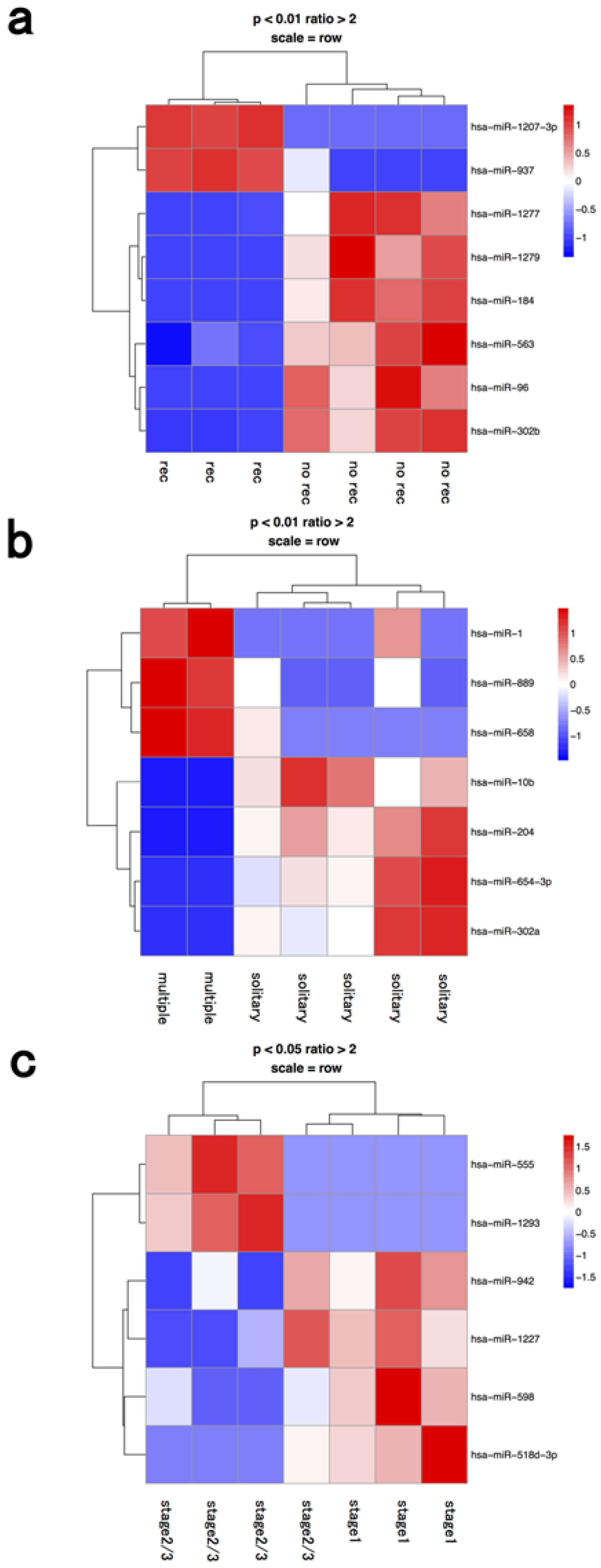
| macrophage | recurrence | hsa-mir-1207-3p | hsa-mir-1277 |
| recurrence | hsa-mir-937 | hsa-mir-1279 | |
| recurrence | hsa-mir-184 | ||
| recurrence | hsa-mir-563 | ||
| recurrence | hsa-mir-96 | ||
| recurrence | hsa-mir-302b | ||
| multiple | hsa-mir-1 | hsa-mir-10b | |
| multiple | hsa-mir-889 | hsa-mir-204 | |
| multiple | hsa-mir-658 | hsa-mir-654-3p | |
| multiple | hsa-mir-302a | ||
| stage 2≥ | hsa-mir-555 | hsa-mir-942 | |
| stage 2≥ | hsa-mir-1293 | hsa-mir-1227 | |
| stage 2≥ | hsa-mir-598 | ||
| stage 2≥ | hsa-mir-518d-3p |
3.2. Lymphocyte Fraction
3.3. Macrophage Fraction
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Okita, K. Clinical aspects of hepatocellular carcinoma in japan. Intern. Med. 2006, 45, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M. Updated treatment approach to hepatocellular carcinoma. J. Gastroenterol. 2005, 40, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Croce, C.M. Causes and consequences of microRNA dysregulation in cancer. Nat. Rev. Genet. 2009, 10, 704–714. [Google Scholar] [CrossRef] [PubMed]
- Morita, K.; Taketomi, A.; Shirabe, K.; Umeda, K.; Kayashima, H.; Ninomiya, M.; Uchiyama, H.; Soejima, Y.; Maehara, Y. Clinical significance and potential of hepatic microRNA-122 expression in hepatitis c. Liver Int. 2011, 31, 474–484. [Google Scholar] [CrossRef] [PubMed]
- Calin, G.A.; Croce, C.M. MicroRNA signatures in human cancers. Nat. Revi. Cancer 2006, 6, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Sugimachi, K.; Matsumura, T.; Hirata, H.; Uchi, R.; Ueda, M.; Ueo, H.; Shinden, Y.; Iguchi, T.; Eguchi, H.; Shirabe, K.; et al. Identification of a bona fide microRNA biomarker in serum exosomes that predicts hepatocellular carcinoma recurrence after liver transplantation. Br. J. Cancer 2015, 112, 532–538. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, T.; Sugimachi, K.; Iinuma, H.; Takahashi, Y.; Kurashige, J.; Sawada, G.; Ueda, M.; Uchi, R.; Ueo, H.; Takano, Y.; et al. Exosomal microRNA in serum is a novel biomarker of recurrence in human colorectal cancer. Br. J. Cancer 2015, 113, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.; Raposo, G. Exosomes—Vesicular carriers for intercellular communication. Curr. Opin. Cell Biol. 2009, 21, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Peinado, H.; Aleckovic, M.; Lavotshkin, S.; Matei, I.; Costa-Silva, B.; Moreno-Bueno, G.; Hergueta-Redondo, M.; Williams, C.; Garcia-Santos, G.; Ghajar, C.; et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through met. Nat. Med. 2012, 18, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Mathivanan, S.; Fahner, C.J.; Reid, G.E.; Simpson, R.J. Exocarta 2012: Database of exosomal proteins, RNA and lipids. Nucleic Acids Res. 2012, 40, D1241–D1244. [Google Scholar] [CrossRef] [PubMed]
- Valadi, H.; Ekstrom, K.; Bossios, A.; Sjostrand, M.; Lee, J.J.; Lotvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Hannafon, B.N.; Ding, W.Q. Intercellular communication by exosome-derived microRNAs in cancer. Int. J. Mol. Sci. 2013, 14, 14240–14269. [Google Scholar] [CrossRef] [PubMed]
- Cereghetti, D.M.; Lee, P.P. Tumor-derived exosomes contain microRNAs with immunological function: Implications for a novel immunosuppression mechanism. MicroRNA (Shariqah, United Arab Emirates) 2014, 2, 194–204. [Google Scholar] [CrossRef]
- Ono, M.; Kosaka, N.; Tominaga, N.; Yoshioka, Y.; Takeshita, F.; Takahashi, R.U.; Yoshida, M.; Tsuda, H.; Tamura, K.; Ochiya, T. Exosomes from bone marrow mesenchymal stem cells contain a microRNA that promotes dormancy in metastatic breast cancer cells. Sci. Signal. 2014, 7, ra63. [Google Scholar] [CrossRef] [PubMed]
- Mimori, K.; Fukagawa, T.; Kosaka, Y.; Ishikawa, K.; Iwatsuki, M.; Yokobori, T.; Hirasaki, S.; Takatsuno, Y.; Sakashita, H.; Ishii, H.; et al. A large-scale study of mt1-mmp as a marker for isolated tumor cells in peripheral blood and bone marrow in gastric cancer cases. Ann. Surg. Oncol. 2008, 15, 2934–2942. [Google Scholar] [CrossRef] [PubMed]
- Ota, D.; Mimori, K.; Yokobori, T.; Iwatsuki, M.; Kataoka, A.; Masuda, N.; Ishii, H.; Ohno, S.; Mori, M. Identification of recurrence-related microRNAs in the bone marrow of breast cancer patients. Int. J. Oncol. 2011, 38, 955–962. [Google Scholar] [PubMed]
- Akiyoshi, S.; Fukagawa, T.; Ueo, H.; Ishibashi, M.; Takahashi, Y.; Fabbri, M.; Sasako, M.; Maehara, Y.; Mimori, K.; Mori, M. Clinical significance of mir-144-zfx axis in disseminated tumour cells in bone marrow in gastric cancer cases. Br. J. Cancer 2012, 107, 1345–1353. [Google Scholar] [CrossRef] [PubMed]
- Takeyama, H.; Yamamoto, H.; Yamashita, S.; Wu, X.; Takahashi, H.; Nishimura, J.; Haraguchi, N.; Miyake, Y.; Suzuki, R.; Murata, K.; et al. Decreased mir-340 expression in bone marrow is associated with liver metastasis of colorectal cancer. Mol. Cancer Ther. 2014, 13, 976–985. [Google Scholar] [CrossRef] [PubMed]
- Shirabe, K.; Mano, Y.; Muto, J.; Matono, R.; Motomura, T.; Toshima, T.; Takeishi, K.; Uchiyama, H.; Yoshizumi, T.; Taketomi, A.; et al. Role of tumor-associated macrophages in the progression of hepatocellular carcinoma. Surg. Today 2012, 42, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mossanen, J.C.; Tacke, F. Role of lymphocytes in liver cancer. Oncoimmunology 2013, 2, e26468. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.F.; Xu, X.; Huang, J.; Fei, Q.L.; Chen, F.; Li, Y.D.; Han, Z.G. Down-regulation of mir-517a and mir-517c promotes proliferation of hepatocellular carcinoma cells via targeting pyk2. Cancer Lett. 2013, 329, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Yoshitomi, T.; Kawakami, K.; Enokida, H.; Chiyomaru, T.; Kagara, I.; Tatarano, S.; Yoshino, H.; Arimura, H.; Nishiyama, K.; Seki, N.; et al. Restoration of mir-517a expression induces cell apoptosis in bladder cancer cell lines. Oncol. Rep. 2011, 25, 1661–1668. [Google Scholar] [PubMed]
- Sun, C.K.; Man, K.; Ng, K.T.; Ho, J.W.; Lim, Z.X.; Cheng, Q.; Lo, C.M.; Poon, R.T.; Fan, S.T. Proline-rich tyrosine kinase 2 (pyk2) promotes proliferation and invasiveness of hepatocellular carcinoma cells through c-src/erk activation. Carcinogenesis 2008, 29, 2096–2105. [Google Scholar] [CrossRef] [PubMed]
- Furuta, M.; Kozaki, K.; Tanimoto, K.; Tanaka, S.; Arii, S.; Shimamura, T.; Niida, A.; Miyano, S.; Inazawa, J. The tumor-suppressive mir-497-195 cluster targets multiple cell-cycle regulators in hepatocellular carcinoma. PLoS ONE 2013, 8, e60155. [Google Scholar] [CrossRef] [PubMed]
- Creevey, L.; Ryan, J.; Harvey, H.; Bray, I.M.; Meehan, M.; Khan, A.R.; Stallings, R.L. MicroRNA-497 increases apoptosis in mycn amplified neuroblastoma cells by targeting the key cell cycle regulator wee1. Mol. Cancer 2013, 12, 23. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.T.; Jiang, C.C.; Wang, G.P.; Li, Y.P.; Wang, C.Y.; Guo, X.Y.; Yang, R.H.; Feng, Y.; Wang, F.H.; Tseng, H.Y.; et al. MicroRNA-497 targets insulin-like growth factor 1 receptor and has a tumour suppressive role in human colorectal cancer. Oncogene 2013, 32, 1910–1920. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.J.; Duan, L.J.; Qian, X.Q.; Xu, D.; Liu, H.L.; Zhu, Y.J.; Qi, J. Tumor-suppressive microRNA-497 targets ikkbeta to regulate NF-kappab signaling pathway in human prostate cancer cells. Am. J. Cancer Res. 2015, 5, 1795–1804. [Google Scholar] [PubMed]
- Shao, X.J.; Miao, M.H.; Xue, J.; Xue, J.; Ji, X.Q.; Zhu, H. The down-regulation of microRNA-497 contributes to cell growth and cisplatin resistance through pi3k/akt pathway in osteosarcoma. Cell Physiol. Biochem. 2015, 36, 2051–2062. [Google Scholar] [CrossRef] [PubMed]
- Phua, Y.W.; Nguyen, A.; Roden, D.L.; Elsworth, B.; Deng, N.; Nikolic, I.; Yang, J.; McFarland, A.; Russell, R.; Kaplan, W.; et al. MicroRNA profiling of the pubertal mouse mammary gland identifies mir-184 as a candidate breast tumour suppressor gene. Breast Cancer Res. 2015, 17, 83. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.S.; Liu, X.B.; Wong, B.Y.; Ng, R.W.; Yuen, A.P.; Wei, W.I. Mature mir-184 as potential oncogenic microRNA of squamous cell carcinoma of tongue. Clinical cancer research : an official journal of the American Association for Cancer Research 2008, 14, 2588–2592. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.C.; Lin, P.L.; Cheng, Y.W.; Wu, T.C.; Chou, M.C.; Chen, C.Y.; Lee, H. MicroRNA-184 deregulated by the microRNA-21 promotes tumor malignancy and poor outcomes in non-small cell lung cancer via targeting cdc25a and c-myc. Ann. Surg. Oncol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yao, J.; Zhang, X.; Guo, B.; Le, X.; Cubberly, M.; Li, Z.; Nan, K.; Song, T.; Huang, C. MiRNA-302b suppresses human hepatocellular carcinoma by targeting akt2. Mol. Cancer Res. 2014, 12, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhu, M.J.; Ren, A.M.; Wu, H.F.; Han, W.M.; Tan, R.Y.; Tu, R.Q. A ten-microRNA signature identified from a genome-wide microRNA expression profiling in human epithelial ovarian cancer. PLoS ONE 2014, 9, e96472. [Google Scholar] [CrossRef] [PubMed]
- Ujihira, T.; Ikeda, K.; Suzuki, T.; Yamaga, R.; Sato, W.; Horie-Inoue, K.; Shigekawa, T.; Osaki, A.; Saeki, T.; Okamoto, K.; et al. MicroRNA-574-3p, identified by microRNA library-based functional screening, modulates tamoxifen response in breast cancer. Sci. Rep. 2015, 5, 7641. [Google Scholar] [CrossRef] [PubMed]
- Shirabe, K.; Motomura, T.; Muto, J.; Toshima, T.; Matono, R.; Mano, Y.; Takeishi, K.; Ijichi, H.; Harada, N.; Uchiyama, H.; et al. Tumor-infiltrating lymphocytes and hepatocellular carcinoma: Pathology and clinical management. Int. J. Clin. Oncol. 2010, 15, 552–558. [Google Scholar] [CrossRef] [PubMed]
- De Visser, K.E.; Eichten, A.; Coussens, L.M. Paradoxical roles of the immune system during cancer development. Nat. Rev. Cancer 2006, 6, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Rolny, C.; Capparuccia, L.; Casazza, A.; Mazzone, M.; Vallario, A.; Cignetti, A.; Medico, E.; Carmeliet, P.; Comoglio, P.M.; Tamagnone, L. The tumor suppressor semaphorin 3b triggers a prometastatic program mediated by interleukin 8 and the tumor microenvironment. J. Exp. Med. 2008, 205, 1155–1171. [Google Scholar] [CrossRef] [PubMed]
- Van Zijl, F.; Mair, M.; Csiszar, A.; Schneller, D.; Zulehner, G.; Huber, H.; Eferl, R.; Beug, H.; Dolznig, H.; Mikulits, W. Hepatic tumor-stroma crosstalk guides epithelial to mesenchymal transition at the tumor edge. Oncogene 2009, 28, 4022–4033. [Google Scholar] [CrossRef] [PubMed]
- Aucher, A.; Rudnicka, D.; Davis, D.M. MicroRNAs transfer from human macrophages to hepato-carcinoma cells and inhibit proliferation. J. Immunol. (Baltimore, Md.: 1950) 2013, 191, 6250–6260. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sugimachi, K.; Sakimura, S.; Tomokuni, A.; Uchi, R.; Hirata, H.; Komatsu, H.; Shinden, Y.; Iguchi, T.; Eguchi, H.; Masuda, T.; et al. Identification of Recurrence-Related microRNAs from Bone Marrow in Hepatocellular Carcinoma Patients. J. Clin. Med. 2015, 4, 1600-1611. https://doi.org/10.3390/jcm4081600
Sugimachi K, Sakimura S, Tomokuni A, Uchi R, Hirata H, Komatsu H, Shinden Y, Iguchi T, Eguchi H, Masuda T, et al. Identification of Recurrence-Related microRNAs from Bone Marrow in Hepatocellular Carcinoma Patients. Journal of Clinical Medicine. 2015; 4(8):1600-1611. https://doi.org/10.3390/jcm4081600
Chicago/Turabian StyleSugimachi, Keishi, Shotaro Sakimura, Akira Tomokuni, Ryutaro Uchi, Hidenari Hirata, Hisateru Komatsu, Yoshiaki Shinden, Tomohiro Iguchi, Hidetoshi Eguchi, Takaaki Masuda, and et al. 2015. "Identification of Recurrence-Related microRNAs from Bone Marrow in Hepatocellular Carcinoma Patients" Journal of Clinical Medicine 4, no. 8: 1600-1611. https://doi.org/10.3390/jcm4081600
APA StyleSugimachi, K., Sakimura, S., Tomokuni, A., Uchi, R., Hirata, H., Komatsu, H., Shinden, Y., Iguchi, T., Eguchi, H., Masuda, T., Morita, K., Shirabe, K., Eguchi, H., Maehara, Y., Mori, M., & Mimori, K. (2015). Identification of Recurrence-Related microRNAs from Bone Marrow in Hepatocellular Carcinoma Patients. Journal of Clinical Medicine, 4(8), 1600-1611. https://doi.org/10.3390/jcm4081600





