Incidence, Etiology and Outcomes of Hyponatremia after Transsphenoidal Surgery: Experience with 344 Consecutive Patients at a Single Tertiary Center
Abstract
:1. Introduction
2. Methods
3. Results
3.1. Patient Characteristics
| Number of patients | 344 |
|---|---|
| Mean age | 48 ± 16 years |
| Gender (N (%)) | |
| Male | 153 (44.5) |
| Female | 191 (55.5) |
| Comorbidities (N (%)) | |
| Hypertension | 127 (36.9) |
| Diabetes mellitus | 56 (16.3) |
| Hypothyroidism | 42 (12.2) |
| Hyperlipidemia | 21 (6.1) |
| Coronary artery disease | 17 (4.9) |
| Atrial fibrillation | 4 (1.2) |
| GERD | 4 (1.2) |
| Asthma | 4 (1.2) |
| Cerebro-occlusive disease | 4 (1.2) |
| Breast cancer | 4 (1.2) |
| Depression | 4 (1.2) |
| Anxiety | 3 (0.9) |
| Migraines | 3 (0.9) |
| Obstructive sleep apnea | 3 (0.9) |
| PCOS | 3 (0.9) |
| BPH | 2 (0.6) |
| Renal cell carcinoma | 2 (0.6) |
| Sickle cell trait | 2 (0.6) |
| Rheumatoid arthritis | 2 (0.6) |
3.2. Tumor Characteristics
| Mean Tumor Size | |
|---|---|
| Preoperative (N = 344) | 6.37 ± 10.4 cm3 |
| Postoperative (N = 286) | 1.03 ± 4.55 cm3 |
| Completeness of Resection | |
| Gross total resection (N (%)) | 182 (63.6) |
| Mean absolute tumor resection (cm3) | 5.75 |
| Mean relative tumor resection (%) | 90.7 |
| Tumor Pathologies (N (%)) | |
| Null cell adenoma | 228 (66.3) |
| Functional adenoma | 55 (16.0) |
| Prolactinoma (PRL) | 23 (6.8) |
| Somatotroph adenoma (GH) | 10 (2.9) |
| LH/FSH | 5 (1.5) |
| LH | 4 (1.2) |
| Corticotroph adenoma (ACTH) | 4 (1.2) |
| FSH | 2 (0.6) |
| PRL/FSH | 1 (0.3) |
| PRL/LH | 1 (0.3) |
| PRL/ACTH | 1 (0.3) |
| PRL/ACTH/GH | 1 (0.3) |
| ACTH/FSH | 1 (0.3) |
| ACTH/GH | 1 (0.3) |
| ACTH/LH/FSH | 1 (0.3) |
| Rathke’s cleft cyst | 48 (14.0) |
| Chordoma | 3 (0.9) |
| Lymphocytic adenohypophysitis | 2 (0.6) |
| Null cell adenoma + Rathke’s cleft cyst | 2 (0.6) |
| Somatotroph adenoma (GH) + Rathke’s cleft cyst | 1 (0.3) |
| Craniopharyngioma | 1 (0.3) |
| Germinoma | 1 (0.3) |
| Chondrosarcoma | 1 (0.3) |
| Fibrous dysplasia | 1 (0.3) |
3.3. Preoperative Anterior Pituitary Function
3.4. Surgical Procedure
3.5. Hospital Course
3.6. Incidence of Postoperative Hyponatremia
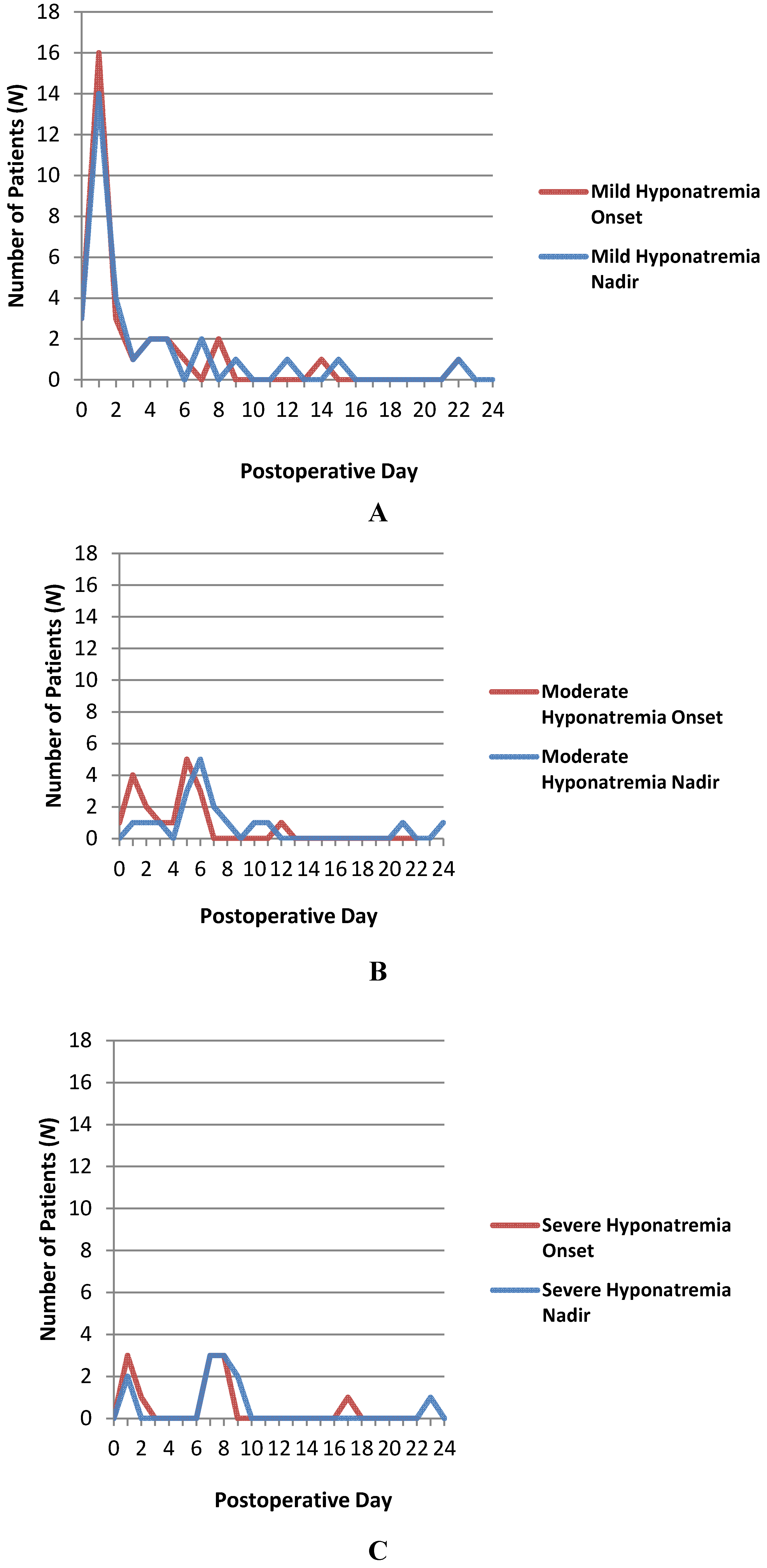
3.7. Clinical Manifestations of Hyponatremia
| Incidence of Post-operative Hyponatremia (N (%)) | 62 (18.0) |
|---|---|
| Hyponatremia Severity (N (%)) | |
| Mild (131–134 mEq/L) | 32 (51.6) |
| Moderate (125–130 mEq/L) | 19 (30.6) |
| Severe (≤124 mEq/L) | 11 (17.7) |
| Clinical Symptoms (N (%)) | |
| None | 43 (69.4) |
| Nausea/Vomiting | 10 (16.1) |
| Headache | 4 (6.5) |
| Malaise | 3 (4.8) |
| Agitation | 1 (1.6) |
| Confusion | 1 (1.6) |
| Balance difficulty | 1 (1.6) |
| Abdominal pain | 1 (1.6) |
| Primary Etiology (N (%)) | |
| Syndrome of inappropriate ADH secretion (SIADH) | 44 (71) |
| Cerebral salt wasting (CSW) | 15 (24.2) |
| Desmopressin acetate overdose | 3 (4.8) |
| Secondary Etiology (N (%)) | |
| Hypoadrenal state | 4 (6.5) |
| Desmopressin acetate overdose | 3 (4.8) |
| Ischemic cardiomyopathy | 1 (1.6) |
| Excessive oral intake of hypotonic fluids | 1 (1.6) |
3.8. Etiology of Hyponatremia
3.9. Treatment of Hyponatremia
3.10. Outcomes
| Hyponatremia Treatment Strategies (N (%)) | |
|---|---|
| None | 30 (48.4) |
| Fluid restriction | 27 (43.5) |
| Diuretics (e.g., furosemide) | 19 (30.6) |
| Hypertonic saline | 9 (14.5) |
| Normal saline | 7 (11.3) |
| Sodium chloride tablets | 3 (4.8) |
| Demeclocycline | 2 (3.2) |
| Corticosteroids | 2 (3.2) |
| Conivaptan | 1 (1.6) |
| Time to Resolution of Hyponatremia (days) | |
| All 62 patients with hyponatremia | 3.5 ± 4.1 |
| By Severity | |
| Mild hyponatremia (N = 32) | 2.0 ± 2.8 |
| Moderate hyponatremia (N = 19) | 5.1 ± 5.1 |
| Severe hyponatremia (N = 11) | 5.3 ± 3.6 |
| By Etiology | |
| Hyponatremia due to desmopressin acetate overdose (N = 3) | 1.5 ± 0.9 |
| Hyponatremia due to SIADH (N = 44) | 3.7 ± 4.0 |
| Hyponatremia due to CSW (N = 15) | 4.3 ± 4.1 |
| By Treatment | |
| Conivaptan (N = 1) | 1 |
| Normal saline (N = 7) | 4 ± 2.4 |
| Corticosteroids (N = 2) | 4 ± 2.8 |
| Sodium chloride tabs (N = 3) | 4.7 ± 1.2 |
| Hypertonic saline (N = 9) | 4.8 ± 3.9 |
| Fluid restriction (N = 27) | 5.7 ± 4.8 |
| Demeclocycline (N = 2) | 6 ± 1.4 |
| Diuretics (e.g., furosemide) (N = 19) | 6.3 ± 5.1 |
| Clinical Outcomes of Hyponatremia (N (%)) | |
| Complete resolution of symptoms | 62 (100) |
| Persistent neurological deficit | 0 (0) |
| Death | 0 (0) |
4. Discussion
4.1. Etiology of Post-transsphenoidal Hyponatremia
4.2. Incidence of Post-Transsphenoidal Hyponatremia
4.3. Onset and Clinical Manifestations of Post-Transsphenoidal Hyponatremia
4.4. Treatment of Post-Transsphenoidal Hyponatremia
4.5. Study Weaknesses
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Boscoe, A.; Paramore, C.; Verbalis, J.G. Cost of illness of hyponatremia in the United States. Cost Eff. Resour. Alloc. 2006, 4. [Google Scholar] [CrossRef]
- Fraser, J.F.; Stieg, P.E. Hyponatremia in the neurosurgical patient: Epidemiology, pathophysiology, diagnosis, and management. Neurosurgery 2006, 59, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.I.; Suri, M.F.; Sung, G.Y.; Straw, R.N.; Yahia, A.M.; Saad, M.; Guterman, L.R.; Hopkins, L.N. Prognostic significance of hypernatremia and hyponatremia among patients with aneurysmal subarachnoid hemorrhage. Neurosurgery 2002, 50, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Harrigan, M.R. Cerebral salt wasting syndrome: A review. Neurosurgery 1996, 38, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Hasan, D.; Wijdicks, E.F.; Vermeulen, M. Hyponatremia is associated with cerebral ischemia in patients with aneurysmal subarachnoid hemorrhage. Ann. Neurol. 1990, 27, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Doczi, T.; Bende, J.; Huszka, E.; Kiss, J. Syndrome of inappropriate secretion of antidiuretic hormone after subarachnoid hemorrhage. Neurosurgery 1981, 9, 394–397. [Google Scholar] [CrossRef] [PubMed]
- Moro, N.; Katayama, Y.; Igarashi, T.; Mori, T.; Kawamata, T.; Kojima, J. Hyponatremia in patients with traumatic brain injury: Incidence, mechanism, and response to sodium supplementation or retention therapy with hydrocortisone. Surg. Neurol. 2007, 68, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Atchison, J.W.; Wachendorf, J.; Haddock, D.; Mysiw, W.J.; Gribble, M.; Corrigan, J.D. Hyponatremia-associated cognitive impairment in traumatic brain injury. Brain Inj. 1993, 7, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Donati-Genet, P.C.; Dubuis, J.M.; Girardin, E.; Rimensberger, P.C. Acute symptomatic hyponatremia and cerebral salt wasting after head injury: An important clinical entity. J. Pediatr. Surg. 2001, 36, 1094–1097. [Google Scholar] [CrossRef] [PubMed]
- Doczi, T.; Tarjanyi, J.; Huszka, E.; Kiss, J. Syndrome of inappropriate secretion of antidiuretic hormone (SIADH) after head injury. Neurosurgery 1982, 10, 685–688. [Google Scholar] [CrossRef] [PubMed]
- Furlan, J.C.; Fehlings, M.G. Hyponatremia in the acute stage after traumatic cervical spinal cord injury: Clinical and neuroanatomic evidence for autonomic dysfunction. Spine 2009, 34, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Peruzzi, W.T.; Shapiro, B.A.; Meyer, P.R., Jr.; Krumlovsky, F.; Seo, B.W. Hyponatremia in acute spinal cord injury. Crit. Care Med. 1994, 22, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Patwari, A.K.; Singh, B.S.; Manorama, D.E. Inappropriate secretion of antidiuretic hormone in acute bacterial meningitis. Ann. Trop. Paediatr. 1995, 15, 179–183. [Google Scholar] [PubMed]
- Van de Beek, D.; de Gans, J.; Spanjaard, L.; Sela, S.; Vermeulen, M.; Dankert, J. Group a streptococcal meningitis in adults: Report of 41 cases and a review of the literature. Clin. Infect. Dis. 2002, 34. [Google Scholar] [CrossRef]
- Brouwer, M.C.; van de Beek, D.; Heckenberg, S.G.; Spanjaard, L.; de Gans, J. Hyponatraemia in adults with community-acquired bacterial meningitis. QJM 2007, 100, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Roca-Ribas, F.; Ninno, J.E.; Gasperin, A.; Lucas, M.; Llubia, C. Cerebral salt wasting syndrome as a postoperative complication after surgical resection of acoustic neuroma. Otol. Neurotol. 2002, 23, 992–995. [Google Scholar] [CrossRef] [PubMed]
- Staiger, R.D.; Sarnthein, J.; Wiesli, P.; Schmid, C.; Bernays, R.L. Prognostic factors for impaired plasma sodium homeostasis after transsphenoidal surgery. Br. J. Neurosurg. 2013, 27, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Sigounas, D.G.; Sharpless, J.L.; Cheng, D.M.; Johnson, T.G.; Senior, B.A.; Ewend, M.G. Predictors and incidence of central diabetes insipidus after endoscopic pituitary surgery. Neurosurgery 2008, 62, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Zada, G.; Liu, C.Y.; Fishback, D.; Singer, P.A.; Weiss, M.H. Recognition and management of delayed hyponatremia following transsphenoidal pituitary surgery. J. Neurosurg. 2007, 106, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.; Zuyuan, R.; Changbao, S.; Renzhi, W.; Yi, Y.; Wenbin, M. Hyponatremia after transspheniodal surgery of pituitary adenoma. Chin. Med. Sci. J. 2003, 18, 120–123. [Google Scholar] [PubMed]
- Olson, B.R.; Gumowski, J.; Rubino, D.; Oldfield, E.H. Pathophysiology of hyponatremia after transsphenoidal pituitary surgery. J. Neurosurg. 1997, 87, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Beukhof, C.M.; Hoorn, E.J.; Lindemans, J.; Zietse, R. Novel risk factors for hospital-acquired hyponatraemia: A matched case-control study. Clin. Endocrinol. 2007, 66, 367–372. [Google Scholar] [CrossRef]
- Gill, G.; Huda, B.; Boyd, A.; Skagen, K.; Wile, D.; Watson, I.; van Heyningen, C. Characteristics and mortality of severe hyponatraemia—A hospital-based study. Clin. Endocrinol. 2006, 65, 246–249. [Google Scholar] [CrossRef]
- Rahman, M.; Friedman, W.A. Hyponatremia in neurosurgical patients: Clinical guidelines development. Neurosurgery 2009, 65, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Clayton, J.A.; Le Jeune, I.R.; Hall, I.P. Severe hyponatraemia in medical in-patients: Aetiology, assessment and outcome. QJM 2006, 99, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Huda, M.S.; Boyd, A.; Skagen, K.; Wile, D.; van Heyningen, C.; Watson, I.; Wong, S.; Gill, G. Investigation and management of severe hyponatraemia in a hospital setting. Postgrad. Med. J. 2006, 82, 216–219. [Google Scholar] [CrossRef] [PubMed]
- McGee, S.; Abernethy, W.B., III; Simel, D.L. The rational clinical examination. Is this patient hypovolemic? JAMA 1999, 281, 1022–1029. [Google Scholar]
- Saeed, B.O.; Beaumont, D.; Handley, G.H.; Weaver, J.U. Severe hyponatraemia: Investigation and management in a district general hospital. J. Clin. Pathol. 2002, 55, 893–896. [Google Scholar] [CrossRef] [PubMed]
- Cole, C.D.; Gottfried, O.N.; Liu, J.K.; Couldwell, W.T. Hyponatremia in the neurosurgical patient: Diagnosis and management. Neurosurg. Focus 2004, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Adrogue, H.J.; Madias, N.E. Hyponatremia. N. Engl. J. Med. 2000, 342, 1581–1589. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C. The effect of interruption of the supraoptic-hypophyseal tracts on the antidiuretic, pressor and oxytocic activity of the posterior lobe of the hypophysis. Endocrinology 1936, 20, 762–768. [Google Scholar] [CrossRef]
- Gagel, O.; Klaes, H. Hypothalamo-hypophyseal regulation of water metabolism. Klin. Wochenschr. 1950, 28, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, S.J.; Meanock, C.I.; Turner, G.F.; Smythe, P.J.; Pickard, J.D.; Noble, A.R.; Walker, V. Fluid balance and secretion of antidiuretic hormone following transsphenoidal pituitary surgery. A preliminary series. J. Neurosurg. 1985, 63, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.F.; Laws, E.R., Jr.; Fossett, D. Delayed hyponatremia after transsphenoidal surgery for pituitary adenoma. Report of nine cases. J. Neurosurg. 1995, 83, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.L.; Tyrrell, J.B.; Wilson, C.B. Delayed onset of hyponatremia after transsphenoidal surgery for pituitary adenomas. Neurosurgery 1995, 37, 649–653. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.I.; Cho, W.H.; Choi, B.K.; Cha, S.H.; Song, G.S.; Choi, C.H. Delayed hyponatremia following transsphenoidal surgery for pituitary adenoma. Neurol. Med. Chir. 2008, 48, 489–492. [Google Scholar] [CrossRef]
- Hensen, J.; Henig, A.; Fahlbusch, R.; Meyer, M.; Boehnert, M.; Buchfelder, M. Prevalence, predictors and patterns of postoperative polyuria and hyponatraemia in the immediate course after transsphenoidal surgery for pituitary adenomas. Clin. Endocrinol. 1999, 50, 431–439. [Google Scholar] [CrossRef]
- Andrews, B.T.; Fitzgerald, P.A.; Tyrell, J.B.; Wilson, C.B. Cerebral salt wasting after pituitary exploration and biopsy: Case report. Neurosurgery 1986, 18, 469–471. [Google Scholar] [CrossRef] [PubMed]
- Diringer, M.; Ladenson, P.W.; Borel, C.; Hart, G.K.; Kirsch, J.R.; Hanley, D.F. Sodium and water regulation in a patient with cerebral salt wasting. Arch. Neurol. 1989, 46, 928–930. [Google Scholar] [CrossRef] [PubMed]
- Harrigan, M.R. Cerebral salt wasting syndrome. Crit. Care Clin. 2001, 17, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.F. Hyponatremia in patients with central nervous system disease: SIADH versus CSW. Trends Endocrinol. Metab. 2003, 14, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Berendes, E.; Walter, M.; Cullen, P.; Prien, T.; Van Aken, H.; Horsthemke, J.; Schulte, M.; von Wild, K.; Scherer, R. Secretion of brain natriuretic peptide in patients with aneurysmal subarachnoid haemorrhage. Lancet 1997, 349, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Tomida, M.; Muraki, M.; Uemura, K.; Yamasaki, K. Plasma concentrations of brain natriuretic peptide in patients with subarachnoid hemorrhage. Stroke 1998, 29, 1584–1587. [Google Scholar] [CrossRef] [PubMed]
- De Zeeuw, D.; Janssen, W.M.; de Jong, P.E. Atrial natriuretic factor: Its (patho)physiological significance in humans. Kidney Int. 1992, 41, 1115–1133. [Google Scholar]
- Weidmann, P.; Hasler, L.; Gnadinger, M.P.; Lang, R.E.; Uehlinger, D.E.; Shaw, S.; Rascher, W.; Reubi, F.C. Blood levels and renal effects of atrial natriuretic peptide in normal man. J. Clin. Invest. 1986, 77, 734–742. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Goto, A.; Nagoshi, H.; Hui, C.; Omata, M. Role of brain ouabainlike compound in central nervous system-mediated natriuresis in rats. Hypertension 1994, 23, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Diederich, S.; Franzen, N.F.; Bahr, V.; Oelkers, W. Severe hyponatremia due to hypopituitarism with adrenal insufficiency: Report on 28 cases. Eur. J. Endocrinol. 2003, 148, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Kroll, M.; Juhler, M.; Lindholm, J. Hyponatraemia in acute brain disease. J. Intern. Med. 1992, 232, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Erkut, Z.A.; Pool, C.; Swaab, D.F. Glucocorticoids suppress corticotropin-releasing hormone and vasopressin expression in human hypothalamic neurons. J. Clin. Endocrinol. Metab. 1998, 83, 2066–2073. [Google Scholar] [PubMed]
- Raff, H. Glucocorticoid inhibition of neurohypophysial vasopressin secretion. Am. J. Physiol. 1987, 252, R635–R644. [Google Scholar] [PubMed]
- Tobias, J.D. Cerebrospinal fluid losses through ventricular catheters leading to hyponatremia in two children. South. Med. J. 1991, 84, 279–280. [Google Scholar] [CrossRef] [PubMed]
- Ngo, Q.N.; Ranger, A.; Singh, R.N.; Kornecki, A.; Seabrook, J.A.; Fraser, D.D. External ventricular drains in pediatric patients. Pediatr. Crit. Care Med. 2009, 10, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Macmohan, P.; Cooke, R.W. Hyponatremia caused by repeated cerebrospinal fluid drainage in post haemorrhagic hydrocephalus. Arch. Dis. Child. 1983, 58, 385–386. [Google Scholar] [CrossRef] [PubMed]
- Norlela, S.; Azmi, K.N.; Khalid, B.A. Syndrome of inappropriate antidiuretic hormone caused by continuous lumbar spinal fluid drainage after transphenoidal surgery. Singap. Med. J. 2006, 47, 75–76. [Google Scholar]
- Adrogue, H.M.; Madias, N.E. Hyponatremia. NEJM 2013, 342, 1581–1589. [Google Scholar] [CrossRef]
- Jahangiri, A.; Wagner, J.; Tran, M.T.; Miller, L.M.; Tom, M.W.; Kunwar, S.; Blevins, L., Jr.; Aghi, M.K. Factors predicting postoperative hyponatremia and efficacy of hyponatremia management strategies after more than 1000 pituitary operations. J. Neurosurg. 2013, 119, 1478–1483. [Google Scholar] [CrossRef] [PubMed]
- Sata, A.; Hizuka, N.; Kawamata, T.; Hori, T.; Takano, K. Hyponatremia after transsphenoidal surgery for hypothalamo-pituitary tumors. Neuroendocrinology 2006, 83, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Kristof, R.R.M.; Neuloh, G.; Klingmuller, D. Incidence, clinical manifestations, and course of water and electrolyte metabolism disturbances following transsphenoidal pituitary adenoma surgery: A prospective observational study. J. Neurosurg. 2009, 111, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.X.; Crofton, J.T.; Miller, J.; Sigman, C.J.; Liu, H.; Huber, J.M.; Brooks, D.P.; Share, L. Sex difference in urinary concentrating ability of rats with water deprivation. Am. J. Physiol. 1996, 270, R550–R555. [Google Scholar] [PubMed]
- Merkelbach, U.; Czernichow, P.; Gaillard, R.C.; Vallotton, M.B. Radioimmunoassay of (8-arginine)-vasopressin. Ii. Application to determination of antidiuretic hormone in urine. Acta Endocrinol. 1975, 80, 453–464. [Google Scholar] [PubMed]
- Grikiniene, J.; Volbekas, V.; Stakisaitis, D. Gender differences of sodium metabolism and hyponatremia as an adverse drug effect. Medicina 2004, 40, 935–942. [Google Scholar] [PubMed]
- Walker, V. Fluid balance disturbances in neurosurgical patients: Physiological basis and definitions. Acta Neurochir. Suppl. 1990, 47, 95–101. [Google Scholar] [PubMed]
- Randall, R.V.; Clark, E.C.; Dodge, H.W., Jr.; Love, J.G. Polyuria after operation for tumors in the region of the hypophysis and hypothalamus. J. Clin. Endocrinol. Metab. 1960, 20, 1614–1621. [Google Scholar] [CrossRef] [PubMed]
- Hollinshead, W.H. The interphase of diabetes insipidus. Mayo Clin. Proc. 1964, 39, 92–100. [Google Scholar] [PubMed]
- Olson, B.R.; Rubino, D.; Gumowski, J.; Oldfield, E.H. Isolated hyponatremia after transsphenoidal pituitary surgery. J. Clin. Endocrinol. Metab. 1995, 80, 85–91. [Google Scholar] [PubMed]
- Singer, P.A.; Sevilla, L.J. Postoperative endocrine management of pituitary tumors. Neurosurg. Clin. N. Am. 2003, 14, 123–138. [Google Scholar] [CrossRef] [PubMed]
- Berkenbosch, J.W.; Lentz, C.W.; Jimenez, D.F.; Tobias, J.D. Cerebral salt wasting syndrome following brain injury in three pediatric patients: Suggestions for rapid diagnosis and therapy. Pediatr. Neurosurg. 2002, 36, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Suarez, J.I.; Qureshi, A.I.; Parekh, P.D.; Razumovsky, A.; Tamargo, R.J.; Bhardwaj, A.; Ulatowski, J.A. Administration of hypertonic (3%) sodium chloride/acetate in hyponatremic patients with symptomatic vasospasm following subarachnoid hemorrhage. J. Neurosurg. Anesthesiol. 1999, 11, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Katayama, Y.; Kawamata, T.; Hirayama, T. Improved efficiency of hypervolemic therapy with inhibition of natriuresis by fludrocortisone in patients with aneurysmal subarachnoid hemorrhage. J. Neurosurg. 1999, 91, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Moro, N.; Katayama, Y.; Kojima, J.; Mori, T.; Kawamata, T. Prophylactic management of excessive natriuresis with hydrocortisone for efficient hypervolemic therapy after subarachnoid hemorrhage. Stroke 2003, 34, 2807–2811. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barber, S.M.; Liebelt, B.D.; Baskin, D.S. Incidence, Etiology and Outcomes of Hyponatremia after Transsphenoidal Surgery: Experience with 344 Consecutive Patients at a Single Tertiary Center. J. Clin. Med. 2014, 3, 1199-1219. https://doi.org/10.3390/jcm3041199
Barber SM, Liebelt BD, Baskin DS. Incidence, Etiology and Outcomes of Hyponatremia after Transsphenoidal Surgery: Experience with 344 Consecutive Patients at a Single Tertiary Center. Journal of Clinical Medicine. 2014; 3(4):1199-1219. https://doi.org/10.3390/jcm3041199
Chicago/Turabian StyleBarber, Sean M., Brandon D. Liebelt, and David S. Baskin. 2014. "Incidence, Etiology and Outcomes of Hyponatremia after Transsphenoidal Surgery: Experience with 344 Consecutive Patients at a Single Tertiary Center" Journal of Clinical Medicine 3, no. 4: 1199-1219. https://doi.org/10.3390/jcm3041199
APA StyleBarber, S. M., Liebelt, B. D., & Baskin, D. S. (2014). Incidence, Etiology and Outcomes of Hyponatremia after Transsphenoidal Surgery: Experience with 344 Consecutive Patients at a Single Tertiary Center. Journal of Clinical Medicine, 3(4), 1199-1219. https://doi.org/10.3390/jcm3041199




