Hyponatremia: A Risk Factor for Early Overt Encephalopathy after Transjugular Intrahepatic Portosystemic Shunt Creation
Abstract
:1. Introduction
2. Experimental Methods
2.1. Patient Selection
2.2. Patient Characteristics
2.3. Post-TIPS Outcomes
| Overall | EOE | No EOE | p | |
|---|---|---|---|---|
| (N = 81) | (N = 15) | (N = 66) | ||
| Demographics | ||||
| Male | 58 (72%) | 12 (80%) | 46 (70%) | 0.42 |
| Female | 23 (28%) | 3 (20%) | 20 (30%) | 0.42 |
| Mean age ± SD | 57 ± 8 | 60 ± 6 | 57 ± 8 | 0.10 |
| Etiology (N) * | ||||
| HCV/HBV | 50 (62%) | 10 (67%) | 40 (61%) | 0.67 |
| Alcohol | 16 (20%) | 4 (27%) | 12 (18%) | 0.45 |
| Other | 15 (19%) | 1 (7%) | 14 (21%) | 0.19 |
| Clinical Characteristics | ||||
| Refractory Ascites | 69 (85%) | 15 (100%) | 54 (81%) | 0.07 |
| TIPS Stenosis | 3 (4%) | 0 (0%) | 3 (5%) | 0.40 |
| Hydrothorax | 14 (17%) | 3 (20%) | 11 (17%) | 0.76 |
| Portal Vein Thrombosis | 7 (9%) | 1 (7%) | 6 (9%) | 0.76 |
| History of Overt HE | 24 (30%) | 2 (13%) | 22 (33%) | 0.13 |
| Pre-TIPS HE Prophylaxis | 55 (68%) | 11 (73%) | 44 (67%) | 0.62 |
| Laboratory Parameters (mean ± SD) | ||||
| Serum [Na+] (Normal = 133–146 mEq/L) ** | 132.7 ± 5.2 | 130.4 ± 4.0 | 133.3 ± 5.3 | 0.02 |
| Serum pre-TIPS [Na+] (133–146 mEq/L) ** | 132.6 ± 5.3 | 129.6 ± 5.0 | 133.3 ± 5.1 | 0.01 |
| Creatinine (0.6–1.3 mg/dL) | 1.3 ± 0.9 | 1.2 ± 0.6 | 1.3 ± 1.0 | 0.61 |
| Albumin (3.5–5.0 g/dL) | 2.6 ± 0.7 | 2.8 ± 0.7 | 2.5 ± 0.7 | 0.13 |
| AST (5–40 IU/L) | 80 ± 79 | 104 ± 159 | 75 ± 45 | 0.48 |
| ALT (7–56 IU/L) | 55 ± 49 | 64 ± 83 | 53 ± 38 | 0.62 |
| INR (0.8–1.2) | 1.6 ± 0.4 | 1.8 ± 0.5 | 1.6 ± 0.3 | 0.14 |
| Alkaline Phosphatase (40–140 IU/L) | 135 ± 65 | 154 ± 97 | 131 ± 55 | 0.38 |
| Total Bilirubin (0.1–1.2 mg/dL) | 3.1 ± 2.6 | 4.6 ± 4.5 | 2.7 ± 1.8 | 0.11 |
| MELD and Child-Pugh Scores | ||||
| MELD | 17 ± 4 | 19 ± 5 | 17 ± 4 | 0.15 |
| MELD-Na | 21 ± 5 | 24 ± 5 | 20 ± 5 | 0.01 |
| Child-Pugh Score | 11 ± 2 | 11 ± 2 | 10 ± 2 | 0.08 |
2.4. Statistical Analysis
3. Results and Discussion
3.1. Patient Characteristics
3.2. Post-TIPS Outcomes
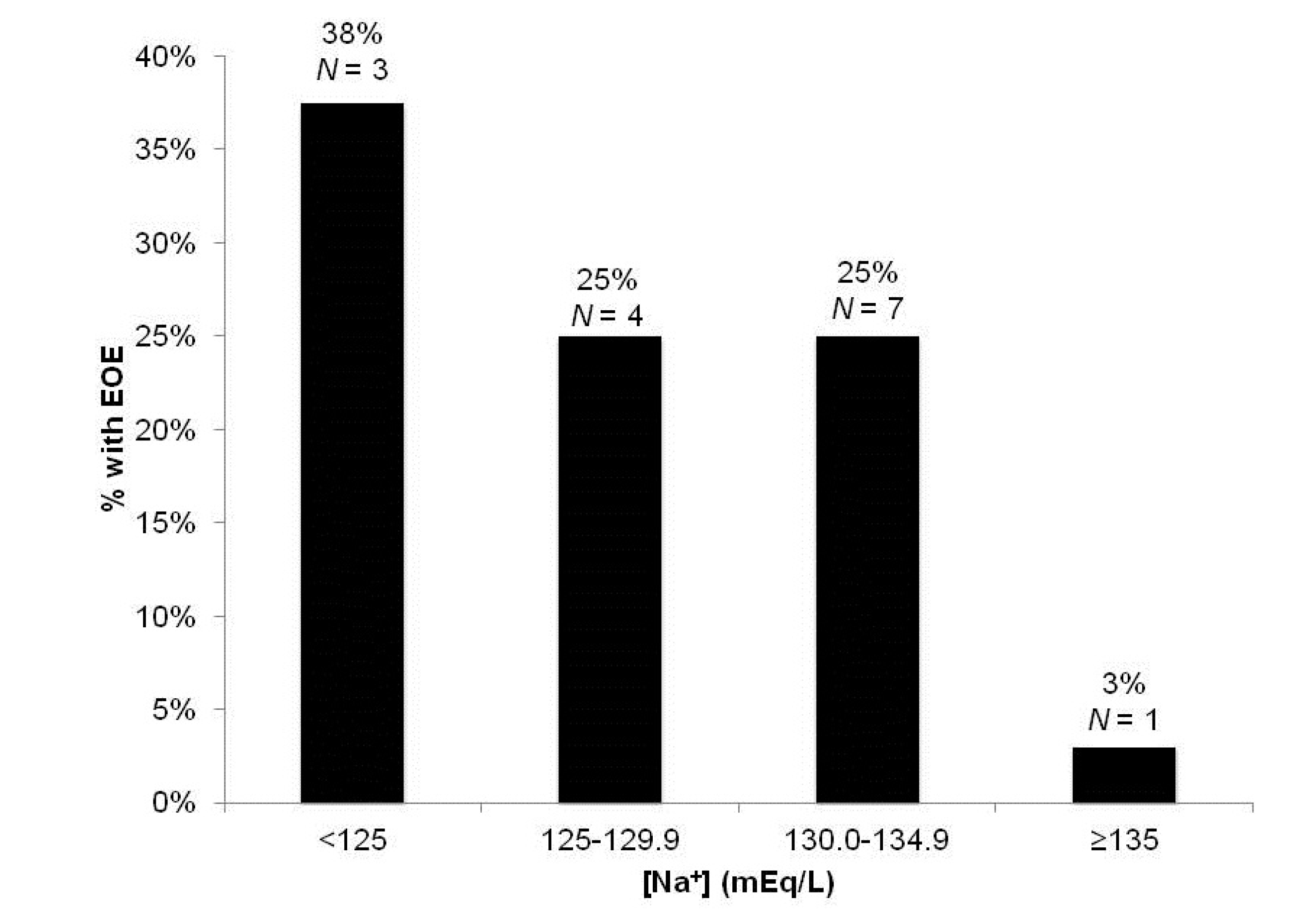
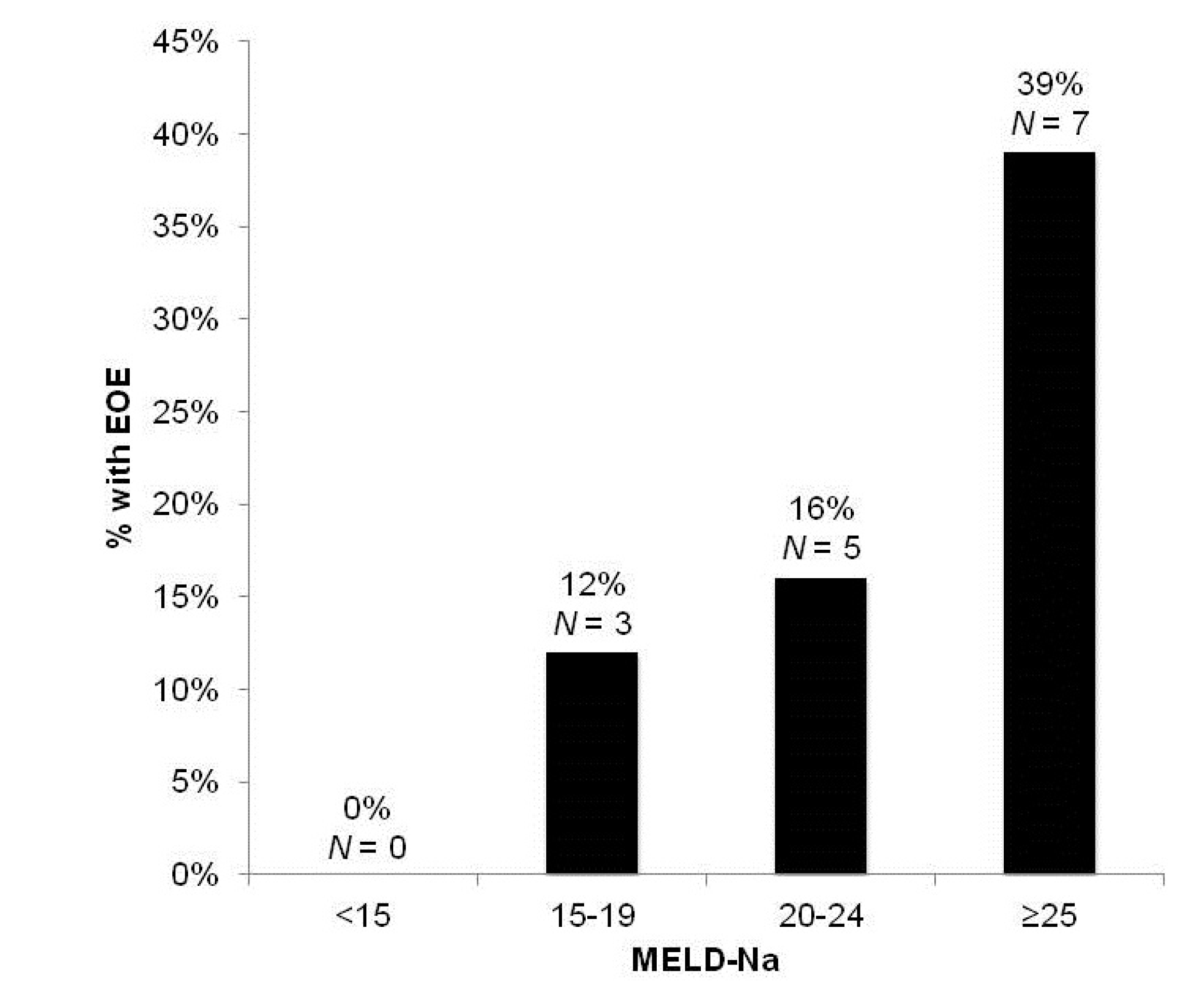
3.2.1. Development of Early Overt Encephalopathy (EOE)
| Variable | EOE (p) | LOS (p) |
|---|---|---|
| Age | 0.15 | 0.23 |
| Pre-TIPS [Na+] | 0.03 * | <0.01 * |
| Creatinine | 0.83 | 0.19 |
| Albumin | 0.12 | 0.04 * |
| AST | 0.25 | 0.25 |
| ALT | 0.43 | 0.17 |
| INR | 0.13 | 0.01 * |
| Total Bilirubin | 0.02 * | <0.01 * |
| MELD | 0.10 | <0.01 * |
| MELD-Na | <0.01 * | <0.01 * |
| Childs-Pugh Score | 0.19 | <0.01 * |
| Ascites | 0.24 | 0.31 |
| Portal Vein Thrombosis | 0.96 | 0.61 |
| History of HE | 0.90 | 0.78 |
| Diabetes mellitus (DM) | 0.55 | 0.81 |
| Insulin-Dependent DM | 0.42 | 0.05 * |
| Parameter | Odds-Ratio | Standard Error | Wald (χ2) |
|---|---|---|---|
| Intercept | −1.76 | 0.52 | 11.38 |
| Pre-TIPS Na | −2.16 | 1.07 | 4.03 |
| Total Bilirubin | 0.21 | 0.11 | 3.76 |
| Factor | Odds Ratio | 95% Confidence Interval |
|---|---|---|
| Pre-TIPS Na ≥135 (mEq/L) | 0.52 | 1.05–71.43 |
| Total Bilirubin | 1.07 | 1.00–1.51 |
3.2.2. Length of Stay after TIPS Insertion
3.2.3. In-Hospital Mortality
3.3. Discussion
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Riordan, S.M.; Williams, R. Treatment of hepatic encephalopathy. N. Engl. J. Med. 1997, 337, 473–479. [Google Scholar] [CrossRef]
- Atterbury, C.E.; Maddrey, W.C.; Conn, H.O. Neomycin-sorbitol and lactulose in the treatment of acute portal-systemic encephalopathy. A controlled, double-blind clinical trial. Am. J. Dig. Dis. 1978, 23, 398–406. [Google Scholar] [CrossRef]
- Blei, A.T. The pathophysiology of brain edema in acute liver failure. Neurochem. Int. 2005, 47, 71–77. [Google Scholar] [CrossRef]
- Dam, G.; Keiding, S.; Munk, O.L.; Ott, P.; Vilstrup, H.; Bak, L.K.; Waagepetersen, H.S.; Schousboe, A.; Sørensen, M. Hepatic encephalopathy is associated with decreased cerebral oxygen metabolism and blood flow not increased ammonia uptake. Hepatology 2013, 57, 258–265. [Google Scholar] [CrossRef]
- Iversen, P.; Sørensen, M.; Bak, L.K.; Waagepetersen, H.S.; Vafaee, M.S.; Borghammer, P.; Mouridsen, K.; Jensen, S.B.; Vilstrup, H.; Schousboe, A.; et al. Low cerebral oxygen consumption and blood flow in patients with cirrhosis and an acute episode of hepatic encephalopathy. Gastroenterology 2009, 136, 863–871. [Google Scholar] [CrossRef]
- Blei, A.T.; Olafsson, S.; Therrien, G.; Butterworth, R.F. Ammonia-induced brain edema and intracranial hypertension in rats after portacaval anastomosis. Hepatology 1994, 19, 1437–1444. [Google Scholar] [CrossRef]
- Cordoba, J.; Gottstein, J.; Blei, A.T. Hyponatremia exacerbates ammonia-induced brain edema in rats after portacaval anastomosis. J. Hepatol. 1998, 29, 589–594. [Google Scholar] [CrossRef]
- Guevara, M.; Baccaro, M.E.; Torre, A.; Gomez-Anson, B.; Rios, J.; Torres, F.; Rami, L.; Monte-Tubio, G.C.; Martin-Llahi, M.; Arroyo, V.; et al. Hyponatremia is a risk factor of hepatic encephalopathy in patients with cirrhosis: A prospective study with time-dependent analysis. Am. J. Gastroenterol. 2009, 104, 1382–1389. [Google Scholar] [CrossRef]
- Angeli, P.; Wong, F.; Watson, H.; Gines, P. Hyponatremia in cirrhosis: Results of a patient population survey. Hepatology 2006, 44, 1535–1542. [Google Scholar] [CrossRef]
- Rossle, M.; Haag, K.; Ochs, A.; Sellinge, M.; Noldge, G.; Perarnau, J.M.; Berger, E.; Blum, U.; Gabelmann, A.; Hauenstein, K.; et al. The transjugular intrahepatic portosystemic stent-shunt procedure for variceal haemorrhage. N. Engl. J. Med. 1994, 3, 165–172. [Google Scholar]
- LaBerge, J.M.; Somberg, K.A.; Lake, J.R.; Gordon, R.L.; Kerlan, R.K., Jr.; Ascher, N.L.; Roberts, J.P.; Simor, M.M.; Doherty, C.A.; Hahn, J.; et al. Two-year outcome following transjugular intrahepatic portosystemic stent-shunt for variceal bleeding: Results in 90 patients. Gastroenterology 1995, 108, 1143–1151. [Google Scholar] [CrossRef]
- Russo, M.W.; Sood, A.; Jacobson, I.M.; Brown, R.S., Jr. Transjugular intrahepatic portosystemic shunt for refractory ascites: An analysis of the literature on efficacy, morbidity, and mortality. Am. J. Gastroenterol. 2003, 98, 2521–2527. [Google Scholar]
- Riggio, O.; Angeloni, S.; Salvatori, F.M.; de Santis, A.; Cerini, F.; Farcomeni, A.; Attili, A.F.; Merli, M. Incidence, natural history and risk factors of hepatic encephalopathy after transjugular intrahepatic portosystemic with polytetrafluoroethylene-covered stent grafts. Am. J. Gastroenterol. 2008, 103, 2738–2746. [Google Scholar] [CrossRef]
- Jalan, R.; Elton, R.A.; Redhead, D.N.; Finlayson, N.D.; Hayes, P.C. Analysis of prognostic variables in the prediction of mortality, shunt failure, varicealrebleeding and encephalopathy following the transjugular intrahepatic portosystemic stent-shunt for variceal hemorrhage. J. Hepatol. 1995, 23, 123–128. [Google Scholar] [CrossRef]
- Bai, M.; Qi, X.; Yang, Z.; Fan, D.; Han, G. Predictors of hepatic encephalopathy after transjugular intrahepatic portosystemic shunt in cirrhotic patients: A systematic review. J. Gastroenterol. Hepatol. 2011, 26, 943–951. [Google Scholar] [CrossRef]
- Zuckerman, D.A.; Darcy, M.D.; Bocchini, T.P.; Hildebolt, C.F. Encephalopathy after transjugular intrahepatic portosystemic shunting: Analysis of incidence and potential risk factors. Am. J. Roentgenol. 1997, 169, 1727–1731. [Google Scholar] [CrossRef]
- Poordad, F.F. The burden of hepatic encephalopathy. Aliment. Pharmacol. Ther. 2007, 25, 3–9. [Google Scholar] [CrossRef]
- Leevy, C.B.; Phillips, J.A. Hospitalizations during the use of rifaximin vs. lactulose for the treatment of hepatic encephalopathy. Dig. Dis. Sci. 2007, 52, 737–741. [Google Scholar] [CrossRef]
- Swain, M.; Butterworth, R.F.; Blei, A.T. Ammonia and related amino acids in the pathogenesis of brain edema in acute ischemic liver failure in rats. Hepatology 1992, 15, 449–453. [Google Scholar] [CrossRef]
- Skowrońska, M.; Albrecht, J. Oxidative and nitrosative stress in ammonia neurotoxicity. Neurochem. Int. 2013, 62, 731–737. [Google Scholar] [CrossRef]
- Häussinger, D.; Görg, B. Interaction of oxidative stress, astrocyte swelling and cerebral ammonia toxicity. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 87–92. [Google Scholar] [CrossRef]
- Cordoba, J.; Alonso, J.; Rovira, A.; Jacas, C.; Sanpedro, F.; Castells, L.; Vargas, V.; Margarit, C.; Kulisewsky, J.; Esteban, R.; et al. The development of low-grade cerebral edema in cirrhosis is supported by the evaluation of 1H-magnetic resonance abnormalities after liver transplantation. J. Hepatol. 2001, 35, 598–604. [Google Scholar] [CrossRef]
- Lodi, R.; Tonon, C.; Stracciari, A.; Weiger, M.; Camaggi, V.; Iotti, S.; Donati, G.; Guarino, M.; Bolondi, L.; Barbiroli, B. Diffusion MRI showes increase water apparent diffucsion coefficient in the brains of cirrhotics. Neurology 2004, 62, 762–766. [Google Scholar]
- Haussinger, D. Low grade cerebeal edema and the pathogenesis of hepatic encephalopathy in cirrhosis. Hepatology 2006, 43, 1187–1190. [Google Scholar] [CrossRef]
- Decaux, G. Is asymptomatic hyponatremia really asymptomatic? Am. J. Med. 2006, 119, 79–82. [Google Scholar] [CrossRef]
- Schrier, R.W. Does “asymptomatic hyponatremia” exist? Nat. Rev. Nephrol. 2010, 6. [Google Scholar] [CrossRef]
- Kokko, J.P. Symptomatic hyponatremia with hypoxia is a medical emergency. Kidney Int. 2006, 69, 1291–1293. [Google Scholar] [CrossRef]
- Ayus, J.C.; Armstrong, D.; Arieff, A.I. Hyponatremia with hypoxia: Effects on brain adaptation, perfusion and histology in rodents. Kidney Int. 2006, 69, 1319–1325. [Google Scholar]
- Gines, P.; Berl, T.; Bernardi, M.; Bichet, D.G.; Harmon, G.; Jimenez, W.; Liard, J.F.; Martin, P.Y.; Schrier, R.W. Hyponatremia in cirrhosis: From pathogenesis to treatment. Hepatology 1998, 28, 851–864. [Google Scholar] [CrossRef]
- Bichet, D.; Szatalowicz, V.; Chaimovitz, C.; Schrier, R.W. Role of vasopressin in abnormal water excretion in cirrhotic patients. Ann. Intern. Med. 1982, 96, 423–427. [Google Scholar]
- Epstein, M. Derangement of renal water handling in liver disease. Gastroenterology 1985, 89, 1415–1425. [Google Scholar]
- Liamis, G.; Milionis, H.; Elisf, M. A review of drug-induced hyponatremia. Am. J. Kidney Dis. 2008, 52, 144–153. [Google Scholar] [CrossRef]
- Frederick, R.T. Current concepts in the pathophysiology and management of hepatic encephalopathy. J. Gastroenterol. Hepatol. 2011, 7, 222–233. [Google Scholar]
- Restuccia, T.; Gomez-Anson, B.; Guevara, M.; Alessandria, C.; Torre, A.; Alayrach, M.E.; Terra, C.; Martin, M.; Castellvi, M.; Rami, L.; et al. Effects of dilutionalhyponatremia on brain organic osmolytes and water content in patients with cirrhosis. Hepatology 2004, 39, 1613–1622. [Google Scholar] [CrossRef]
- Ahluwalia, V.; Wade, J.B.; Thacker, L.; Kraft, K.A.; Sterling, R.K.; Stravitz, R.T.; Fuchs, M.; Bouneva, I.; Puri, P.; Luketic, V.; et al. Differential impact of hyponatremia and hepatic encephalopathy on health-related quality of life and brain metabolite abnormalities in cirrhosis. J. Hepatol. 2013, 59, 467–473. [Google Scholar] [CrossRef]
- Amodio, P.; Del Piccolo, F.; Petteno, E.; Mapelli, D.; Angeli, P.; Iemmolo, R.; Muraca, M.; Musto, C.; Gerunda, G.; Rizzo, C.; et al. Prevalence and prognostic value of quantified electroencephalogram (EEG) alterations in cirrhotic patients. J. Hepatol. 2001, 35, 37–45. [Google Scholar] [CrossRef]
- Jalan, R.; Dabos, K.; Redhead, D.N.; Lee, A.; Hayes, P.C. Elevation of intracranial pressure following transjugular intrahepatic portosystemic stent-shunt for varicealhaemorrhage. J. Hepatol. 1997, 27, 928–933. [Google Scholar] [CrossRef]
- Somberg, K.A.; Riegler, J.L.; LaBerge, J.M.; Doherty-Simor, M.M.; Bachetti, P.; Roberts, J.P.; Lake, J.R. Hepatic encephalopathy after transjugular intrahepatic portosystemic shunts: Incidence and risk factors. Am. J. Gastroenterol. 1995, 90, 549–555. [Google Scholar]
- Rossle, M.; Deibert, P.; Haag, K.; Ochs, A.; Olschewski, M.; Siegerstetter, V.; Hauenstein, K.H.; Geiger, R.; Stiepak, C.; Keller, W.; et al. Randomised trial of transjugular-intrahepatic-portosystemic shunt vs. endoscopy plus propranolol for prevention of varicealrebleeding. Lancet 1997, 349, 1043–1049. [Google Scholar] [CrossRef]
- Sanyal, A.J.; Freedman, A.M.; Shiffman, M.L.; Purdum, P.P., III; Luketic, V.A.; Cheatham, A.K. Portosystemic encephalopathy after transjugular intrahepatic portosystemic shunt: Results of a prospective controlled study. Hepatology 1994, 20, 46–55. [Google Scholar]
- Nolte, W.; Wiltfang, J.; Schindler, C.; Munke, H.; Unterberg, K.; Zumhasch, U.; Figulla, H.R.; Werner, G.; Hartmann, H.; Ramadori, G. Portosystemic hepatic encephalopathy after transjugular intrahepatic portosystemic shunt in patients with cirrhosis: Clinical, laboratory, psychometric, and electroencephalographic investigations. Hepatology 1998, 28, 1215–1225. [Google Scholar] [CrossRef]
- Mamiya, Y.; Kanazawa, H.; Kimura, Y.; Narahara, Y.; Yamate, Y.; Nakatsuka, K.; Sakamoto, C. Hepatic encephalopathy after transjugular intrahepatic portosystemic shunt. Hepatol. Res. 2004, 30, 162–168. [Google Scholar] [CrossRef]
- Guevara, M.; Baccaro, M.E.; Ríos, J.; Martin-Llahi, M.; Uriz, J.; Ruiz del Arbol, L.; Planas, R.; Monescillo, A.; Guarner, C.; Crespo, J.; et al. Risk factors for hepatic encephalopathy in patients with cirrhosis and refractory ascites: Relevance of serum sodium concentration. Liver Int. 2010, 30, 1137–1142. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Merola, J.; Chaudhary, N.; Qian, M.; Jow, A.; Barboza, K.; Charles, H.; Teperman, L.; Sigal, S. Hyponatremia: A Risk Factor for Early Overt Encephalopathy after Transjugular Intrahepatic Portosystemic Shunt Creation. J. Clin. Med. 2014, 3, 359-372. https://doi.org/10.3390/jcm3020359
Merola J, Chaudhary N, Qian M, Jow A, Barboza K, Charles H, Teperman L, Sigal S. Hyponatremia: A Risk Factor for Early Overt Encephalopathy after Transjugular Intrahepatic Portosystemic Shunt Creation. Journal of Clinical Medicine. 2014; 3(2):359-372. https://doi.org/10.3390/jcm3020359
Chicago/Turabian StyleMerola, Jonathan, Noami Chaudhary, Meng Qian, Alexander Jow, Katherine Barboza, Hearns Charles, Lewis Teperman, and Samuel Sigal. 2014. "Hyponatremia: A Risk Factor for Early Overt Encephalopathy after Transjugular Intrahepatic Portosystemic Shunt Creation" Journal of Clinical Medicine 3, no. 2: 359-372. https://doi.org/10.3390/jcm3020359
APA StyleMerola, J., Chaudhary, N., Qian, M., Jow, A., Barboza, K., Charles, H., Teperman, L., & Sigal, S. (2014). Hyponatremia: A Risk Factor for Early Overt Encephalopathy after Transjugular Intrahepatic Portosystemic Shunt Creation. Journal of Clinical Medicine, 3(2), 359-372. https://doi.org/10.3390/jcm3020359




