Towards a Treatment of Stress Urinary Incontinence: Application of Mesenchymal Stromal Cells for Regeneration of the Sphincter Muscle
Abstract
:1. Introduction
2. Results and Discussion
2.1. Autologous Progenitor Cells
2.1.1. Mesenchymal Stromal Cells
| Stem cell source | Bone marrow | Adipose tissue | Urine | Striated muscle |
|---|---|---|---|---|
| Cell Type | MSC | ADSC | USC | satellite cell |
| Key Surface | CD73, CD90 | CD34, CD73 | CD44, CD73 | α7β1 integrin |
| Inclusion Marker(s) | CD105, CD146 | CD90, CD105 | CD90, CD105 | |
| Key Surface | CD11b, CD14 | CD11b, CD14 | CD31, CD34 | ø |
| Exclusion Marker(s) | CD34, CD45 | CD45 | CD45 | |
| Key Intracellular | vimentin, αSMA | STRO-1 | unknown | Pax7 |
| Marker(s) | STRO-1 | |||
| Cell Availability | abundant | abundant | abundant | limited |
| Isolation/Preparation | simple | feasible | very simple | complex |
| Differentiation | osteo, chondro | osteo, chondro | osteo, chondro | myoblast |
| Capacities | adipo, SMC * | adipo, SMC * | adipo, SMC * | myotube |
| endothelial | ||||
| urothelial | ||||
| Smooth Muscle Differentiation | established | established | established | ø |
| Striated Muscle Differentiation | complex | questionable | published | published |
| but not confirmed | ||||
| Mode of Action | paracrine/trophic | paracrine/trophic | paracrine/trophic | generation of |
| SMC generation | SMC generation | SMC generation | striated muscle cells | |
| Target in SUI | lissosphincter | lissosphincter | lissosphincter | rhabdosphincter |
| Key References | [15,33,55] | [34,56] | [57,58] | [59,60] |
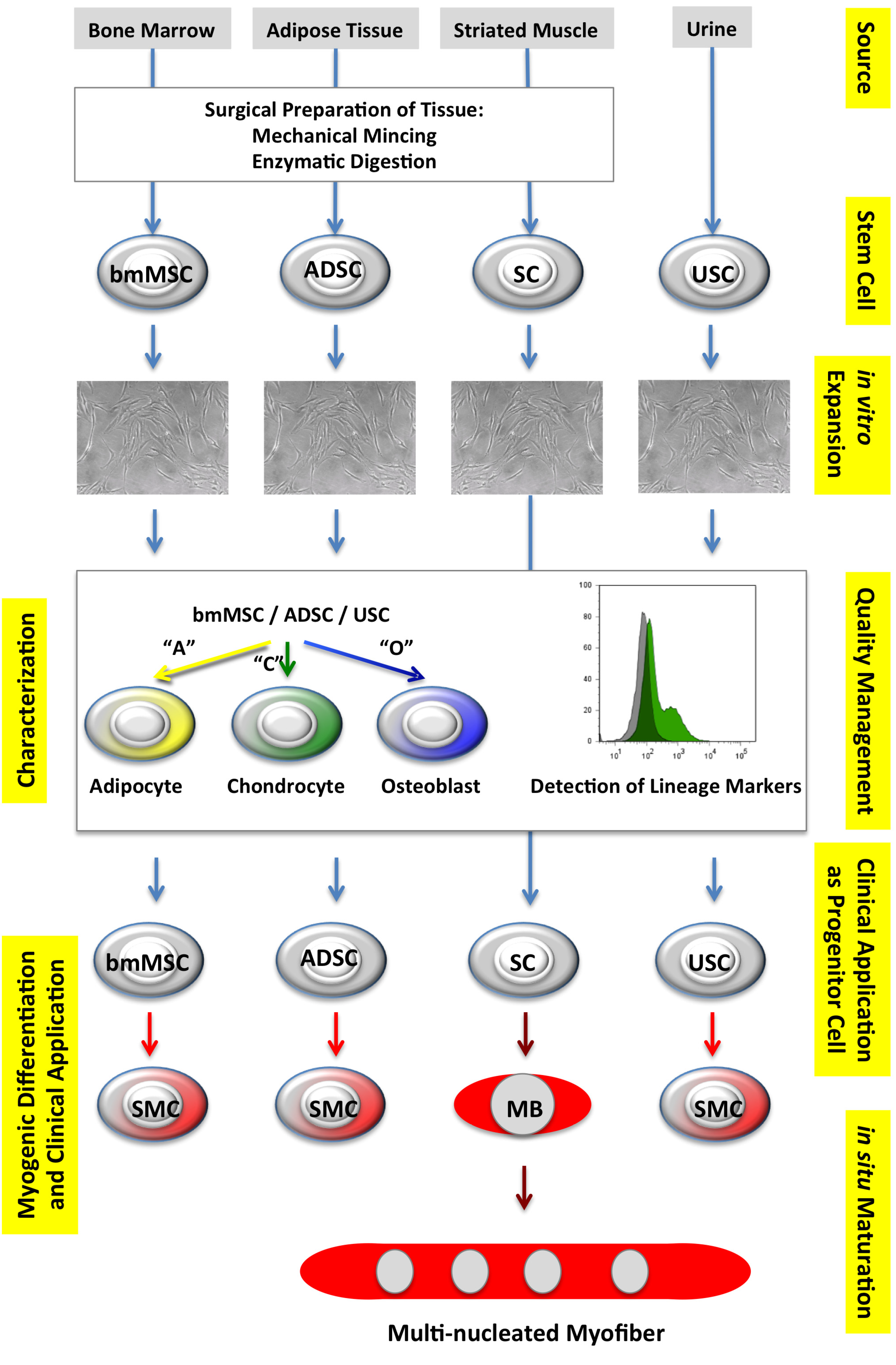
2.1.2. Urine-Derived Stem Cells
2.1.3. Muscle-Derived Satellite Cells
2.2. Navigation and Improved Cell Application Techniques
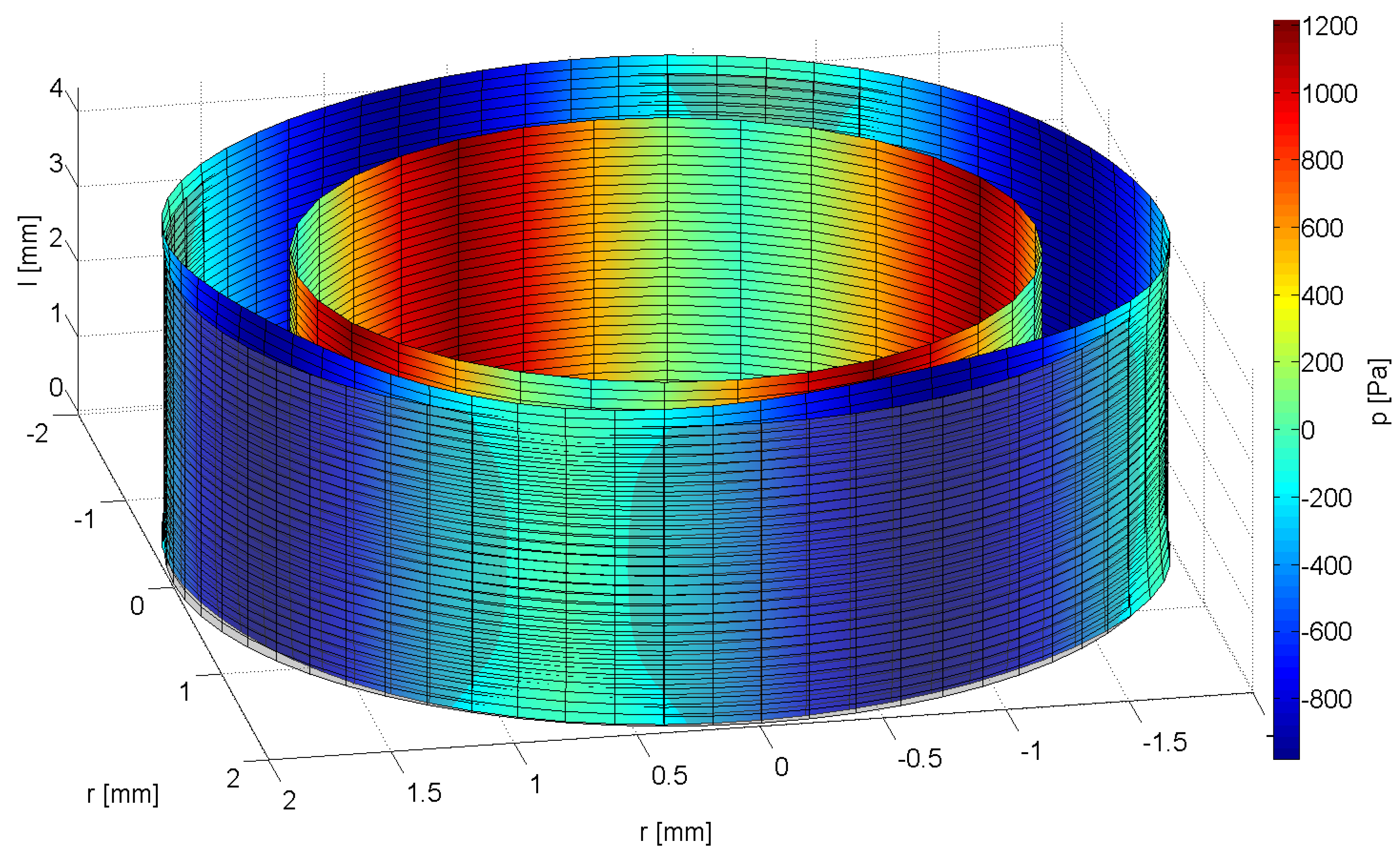
2.3. Signal Processing to Evaluate the Regeneration of the Urinary Sphincter Muscle in Situ

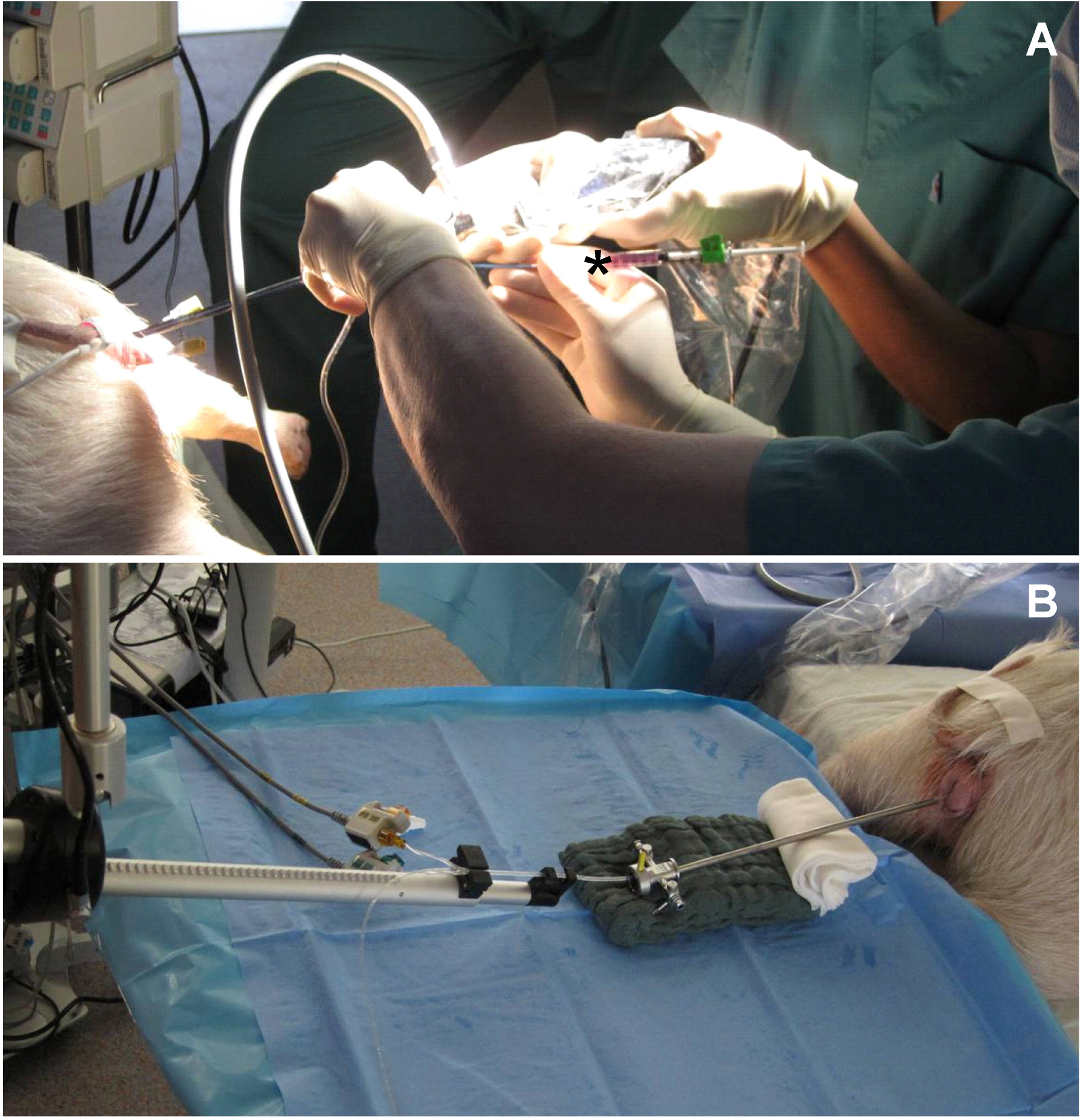
2.4. Pre-Clinical Model
3. Conclusions
Acknowledgments
Conflicts of Interest
References
- Subak, L.L.; Brubaker, L.; Chai, T.C.; Creasman, J.M.; Diokno, A.C.; Goode, P.S.; Kraus, S.R.; Kusek, J.W.; Leng, W.W.; Lukacz, E.S.; et al. High costs of urinary incontinence among women electing surgery to treat stress incontinence. Obstet. Gynecol. 2008, 111, 899–907. [Google Scholar] [CrossRef]
- Markland, A.D.; Goode, P.S.; Redden, D.T.; Borrud, L.G.; Burgio, K.L. Prevalence of urinary incontinence in men: Results from the national health and nutrition examination survey. J. Urol. 2010, 184, 1022–1027. [Google Scholar] [CrossRef]
- Delancey, J.O. Why do women have stress urinary incontinence? Neurourol. Urodyn. 2010, 29, 13–17. [Google Scholar] [CrossRef]
- Chermansky, C.J.; Winters, J.C. Complications of vaginal mesh surgery. Curr. Opin. Urol. 2012, 22, 287–291. [Google Scholar] [CrossRef]
- Kerr, L.A. Bulking agents in the treatment of stress urinary incontinence: History, outcomes, patient populations, and reimbursement profile. Rev. Urol. 2005, 7, 3–11. [Google Scholar]
- Scott, F.B.; Bradley, W.E.; Timm, G.W. Treatment of urinary incontinence by implantable prosthetic sphincter. Urology 1973, 1, 252–259. [Google Scholar] [CrossRef]
- Vakalopoulos, I.; Kampantais, S.; Laskaridis, L.; Chachopoulos, V.; Koptsis, M.; Toutziaris, C. New artificial urinary sphincter devices in the treatment of male iatrogenic incontinence. Adv. Urol. 2012, 2012. [Google Scholar] [CrossRef]
- Dissaranan, C.; Cruz, M.A.; Couri, B.M.; Goldman, H.B.; Damaser, M.S. Stem cell therapy for incontinence: Where are we now? What is the realistic potential? Curr. Urol. Rep. 2011, 12, 336–344. [Google Scholar] [CrossRef]
- Staack, A.; Rodriguez, L.V. Stem cells for the treatment of urinary incontinence. Curr. Urol. Rep. 2011, 12, 41–46. [Google Scholar] [CrossRef]
- Wang, H.J.; Chuang, Y.C.; Chancellor, M.B. Development of cellular therapy for the treatment of stress urinary incontinence. Int. Urogynecol. J. 2011, 22, 1075–1083. [Google Scholar] [CrossRef]
- Cannon, T.W.; Lee, J.Y.; Somogyi, G.; Pruchnic, R.; Smith, C.P.; Huard, J.; Chancellor, M.B. Improved sphincter contractility after allogenic muscle-derived progenitor cell injection into the denervated rat urethra. Urology 2003, 62, 958–963. [Google Scholar]
- Kwon, D.; Kim, Y.; Pruchnic, R.; Jankowski, R.; Usiene, I.; de Miguel, F.; Huard, J.; Chancellor, M.B. Periurethral cellular injection: Comparison of muscle-derived progenitor cells and fibroblasts with regard to efficacy and tissue contractility in an animal model of stress urinary incontinence. Urology 2006, 68, 449–454. [Google Scholar]
- Praud, C.; Sebe, P.; Biérinx, A.S.; Sebille, A. Improvement of urethral sphincter deficiency in female rats following autologous skeletal muscle myoblasts grafting. Cell Transplant. 2007, 16, 741–749. [Google Scholar]
- Fu, Q.; Song, X.F.; Liao, G.L.; Deng, C.L.; Cui, L. Myoblasts differentiated from adipose-derived stem cells to treat stress urinary incontinence. Urology 2010, 75, 718–723. [Google Scholar] [CrossRef]
- Lin, G.; Wang, G.; Banie, L.; Ning, H.; Shindel, A.W.; Fandel, T.M.; Lue, T.F.; Lin, C.S. Treatment of stress urinary incontinence with adipose tissue-derived stem cells. Cytotherapy 2010, 12, 88–95. [Google Scholar] [CrossRef]
- Nitta, M.; Tamaki, T.; Tono, K.; Okada, Y.; Masuda, M.; Akatsuka, A.; Hoshi, A.; Usui, Y.; Terachi, T. Reconstitution of experimental neurogenic bladder dysfunction using skeletal muscle-derived multipotent stem cells. Transplantation 2010, 89, 1043–1049. [Google Scholar] [CrossRef]
- Goldman, H.B.; Sievert, K.D.; Damaser, M.S. Will we ever use stem cells for the treatment of SUI? ICI-RS 2011. Neurourol. Urodyn. 2012, 31, 386–389. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, S.-R.; Song, Y.S.; Lee, H.J. Stem cell therapy in bladder dysfunction: Where are we? And where do we have to go? Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Strasser, H.; Marksteiner, R.; Margreiter, E.; Pinggera, G.M.; Mitterberger, M.; Frauscher, F.; Ulmer, H.; Fussenegger, M.; Kofler, K.; Bartsch, G. Autologous myoblasts and fibroblasts versus collagen for treatment of stress urinary incontinence in women: A randomised controlled trial. Lancet 2007, 369, 2179–2186. [Google Scholar] [CrossRef]
- Strasser, H.; Tiefenthaler, M.; Steinlechner, M.; Bartsch, G.; Konwalinka, G. Urinary incontinence in the elderly and age-dependent apoptosis of rhabdosphincter cells. Lancet 1999, 354, 918–919. [Google Scholar] [CrossRef]
- Carr, L.K.; Steele, D.; Steele, S.; Wagner, D.; Pruchnic, R.; Jankowski, R.; Erickson, J.; Huard, J.; Chancellor, M.B. 1-Year follow-up of autologous muscle-derived stem cell injection pilot study to treat stress urinary incontinence. Int. Urogynecol. J. Pelvic Floor Dysfunct. 2008, 19, 881–883. [Google Scholar] [CrossRef]
- Kajbafzadeh, A.M.; Elmi, A.; Payabvash, S.; Salmasi, A.H.; Saeedi, P.; Mohamadkhani, A.; Sadeghi, Z.; Nikfarjam, L. Transurethral autologous myoblast injection for treatment of urinary incontinence in children with classic bladder exstrophy. J. Urol. 2008, 180, 1098–1105. [Google Scholar] [CrossRef]
- Mitterberger, M.; Marksteiner, R.; Margreiter, E.; Pinggera, G.M.; Frauscher, F.; Ulmer, H.; Fussenegger, M.; Bartsch, G.; Strasser, H. Myoblast and fibroblast therapy for post-prostatectomy urinary incontinence: 1-Year followup of 63 patients. J. Urol. 2008, 179, 226–231. [Google Scholar]
- Yamamoto, T.; Gotoh, M.; Hattori, R.; Toriyama, K.; Kamei, Y.; Iwaguro, H.; Matsukawa, Y.; Funahashi, Y. Periurethral injection of autologous adipose-derived stem cells for the treatment of stress urinary incontinence in patients undergoing radical prostatectomy: Report of two initial cases. Int. J. Urol. 2010, 17, 75–82. [Google Scholar] [CrossRef]
- Gerullis, H.; Eimer, C.; Georgas, E.; Homburger, M.; El-Baz, A.G.; Wishahi, M.; Borós, M.; Ecke, T.H.; Otto, T. Muscle-derived cells for treatment of iatrogenic sphincter damage and urinary incontinence in men. Sci. World J. 2012, 2012. [Google Scholar] [CrossRef]
- Garcia-Arranz, M.; Gregorio, S.A. Phase II Clinical Trial to Study Feasibility And Safety of the Expanded Autologous MSC Derived from Adipose Tissue for the Local Feminine Stress Urinary Incontinence. 2012. Available online: http://clinicaltrials.gov/ (accessed on 1 October 2014).
- Kleinert, S.; Horton, R. Retraction—Autologous myoblasts and fibroblasts versus collagen [corrected] for treatment of stress urinary incontinence in women: A [corrected] randomised controlled trial. Lancet 2008, 372, 789–790. [Google Scholar] [CrossRef]
- Friedenstein, A.J.; Gorskaja, J.F.; Kulagina, N.N. Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp. Hematol. 1976, 4, 267–274. [Google Scholar]
- Ashton, B.A.; Allen, T.D.; Howlett, C.R.; Eaglesom, C.C.; Hattori, A.; Owen, M. Formation of bone and cartilage by marrow stromal cells in diffusion chambers in vivo. Clin. Orthop. Relat. Res. 1980, 151, 294–307. [Google Scholar]
- Bab, I.; Ashton, B.A.; Gazit, D.; Marx, G.; Williamson, M.C.; Owen, M.E. Kinetics and differentiation of marrow stromal cells in diffusion chambers in vivo. J. Cell Sci. 1986, 84, 139–151. [Google Scholar]
- Friedenstein, A.J.; Chailakhyan, C.A.; Gerasimov, U.V. Bone marrow osteogenic stem cells: In vitro cultivation and transplantation in diffusion chambers. Cell Tissue Kinet. 1987, 20, 263–272. [Google Scholar]
- Caplan, A.I. Mesenchymal stem cells. J. Orthop. Res. 1991, 9, 641–650. [Google Scholar] [CrossRef]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef]
- Zannettino, A.C.W.; Paton, S.; Arthur, A.; Khor, F.; Itescu, S.; Gimble, J.M.; Gronthos, S. Multipotential human adipose-derived stromal stem cells exhibit a perivascular phenotype in vitro and in vivo. J. Cell Physiol. 2008, 214, 413–421. [Google Scholar] [CrossRef]
- Parolini, O.; Alviano, F.; Bergwerf, I.; Boraschi, D.; de Bari, C.; de Waele, P.; Dominici, M.; Evangelista, M.; Falk, W.; Hennerbichler, S.; Hess, D.C.; et al. Toward cell therapy using placenta-derived cells: Disease mechanisms, cell biology, preclinical studies, and regulatory aspects at the round table. Stem Cells Dev. 2010, 19, 143–154. [Google Scholar] [CrossRef]
- Covas, D.T.; Panepucci, R.A.; Fontes, A.M.; Silva, W.A., Jr.; Orellana, M.D.; Freitas, M.C.; Neder, L.; Santos, A.R.; Peres, L.C.; Jamur, M.C.; Zago, M.A. Multipotent mesenchymal stromal cells obtained from diverse human tissues share functional properties and gene-expression profile with CD146+ perivascular cells and fibroblasts. Exp. Hematol. 2008, 36, 642–654. [Google Scholar] [CrossRef]
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 2008, 3, 301–313. [Google Scholar] [CrossRef]
- Aggarwal, S.; Pittenger, M.F. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005, 105, 1815–1822. [Google Scholar] [CrossRef]
- Puissant, B.; Barreau, C.; Bourin, P.; Clavel, C.; Corre, J.; Bousquet, C.; Taureau, C.; Cousin, B.; Abbal, M.; Laharrague, P.; et al. Immunomodulatory effect of human adipose tissue-derived adult stem cells: Comparison with bone marrow mesenchymal stem cells. Br. J. Haematol. 2005, 129, 118–129. [Google Scholar]
- Sudres, M.; Norol, F.; Trenado, A.; Grégoire, S.; Charlotte, F.; Levacher, B.; Lataillade, J.J.; Bourin, P.; Holy, X.; Vernant, J.P.; et al. Bone marrow mesenchymal stem cells suppress lymphocyte proliferation in vitro but fail to prevent graft-versus-host disease in mice. J. Immunol. 2006, 176, 7761–7767. [Google Scholar]
- Jang, M.J.; Kim, H.S.; Lee, H.G.; Kim, G.J.; Jeon, H.G.; Shin, H.S.; Chang, S.K.; Hur, G.H.; Chong, S.Y.; Oh, D.; Chung, H.M. Placenta-derived mesenchymal stem cells have an immunomodulatory effect that can control acute graft-versus-host disease in mice. Acta Haematol. 2013, 129, 197–206. [Google Scholar] [CrossRef]
- Chen, L.; Tredget, E.E.; Wu, P.Y.G.; Wu, Y. Paracrine factors of mesenchymal stem cells recruit macrophages and endothelial lineage cells and enhance wound healing. PLoS One 2008, 3, e1886. [Google Scholar] [CrossRef]
- Salgado, A.J.; Reis, R.L.; Sousa, N.J.; Gimble, J.M. Adipose tissue derived stem cells secretome: Soluble factors and their roles in regenerative medicine. Curr. Stem Cell Res. Ther. 2010, 5, 103–110. [Google Scholar] [CrossRef]
- Minguell, J.J.; Erices, A. Mesenchymal stem cells and the treatment of cardiac disease. Exp. Biol. Med. 2006, 231, 39–49. [Google Scholar]
- Rackwitz, L.; Eden, L.; Reppenhagen, S.; Reichert, J.C.; Jakob, F.; Walles, H.; Pullig, O.; Tuan, R.S.; Rudert, M.; Nöth, U. Stem cell- and growth factor-based regenerative therapies for avascular necrosis of the femoral head. Stem Cell Res. Ther. 2012, 3. [Google Scholar] [CrossRef]
- Johnson, K.; Zhu, S.; Tremblay, M.S.; Payette, J.N.; Wang, J.; Bouchez, C.; Meeusen, S.; Althage, A.; Cho, C.Y.; Wu, X.; Schultz, P.J. A stem cell-based approach to cartilage repair. Science 2012, 336, 717–721. [Google Scholar] [CrossRef]
- Hughey, C.C.; Johnsen, V.L.; Ma, L.; James, F.D.; Young, P.P.; Wasserman, D.H.; Rottman, J.N.; Hittel, D.S.; Shearer, J. Mesenchymal stem cell transplantation for the infarcted heart: A role in minimizing abnormalities in cardiac-specific energy metabolism. Am. J. Physiol. Endocrinol. Metab. 2012, 302, 163–172. [Google Scholar] [CrossRef]
- Quevedo, H.C.; Hatzistergos, K.E.; Oskouei, B.N.; Feigenbaum, G.S.; Rodriguez, J.E.; Valdes, D.; Pattany, P.M.; Zambrano, J.P.; Hu, Q.; McNiece, I.; et al. Allogeneic mesenchymal stem cells restore cardiac function in chronic ischemic cardiomyopathy via trilineage differentiating capacity. Proc. Natl. Acad. Sci. USA 2009, 106, 14022–14027. [Google Scholar] [CrossRef]
- Gu, W.; Zhang, F.; Xue, Q.; Ma, Z.; Lu, P.; Yu, B. Transplantation of bone marrow mesenchymal stem cells reduces lesion volume and induces axonal regrowth of injured spinal cord. Neuropathology 2010, 30, 205–217. [Google Scholar]
- Fukuda, K. Use of adult marrow mesenchymal stem cells for regeneration of cardiomyocytes. Bone Marrow Transplant. 2003, 32, 25–27. [Google Scholar] [CrossRef]
- Kanematsu, A.; Yamamoto, S.; Iwai-Kanai, E.; Kanatani, I.; Imamura, M.; Adam, R.M.; Tabata, Y.; Ogawa, O. Induction of smooth muscle cell-like phenotype in marrow-derived cells among regenerating urinary bladder smooth muscle cells. Am. J. Pathol. 2005, 166, 565–573. [Google Scholar] [CrossRef]
- Drost, A.C.; Weng, S.; Feil, G.; Schäfer, J.; Baumann, S.; Kanz, L.; Sievert, K.D.; Stenzl, A.; Möhle, R. In vitro myogenic differentiation of human bone marrow-derived mesenchymal stem cells as a potential treatment for urethral sphincter muscle repair. Ann. N. Y. Acad. Sci. 2009, 1176, 135–143. [Google Scholar]
- Kinebuchi, Y.; Aizawa, N.; Imamura, T.; Ishizuka, O.; Igawa, Y.; Nishizawa, O. Autologous bone-marrow-derived mesenchymal stem cell transplantation into injured rat urethral sphincter. Int. J. Urol. 2010, 17, 359–368. [Google Scholar] [CrossRef]
- Gunetti, M.; Tomasi, S.; Giammò, A.; Boido, M.; Rustichelli, D.; Mareschi, K.; Errichiello, E.; Parola, M.; Ferrero, I.; Fagioli, F.; Vercelli, A.; et al. Myogenic potential of whole bone marrow mesenchymal stem cells in vitro and in vivo for usage in urinary incontinence. PLoS One 2012, 7, e45538. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.J.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef]
- Lang, R.; Liu, G.; Shi, Y.; Bharadwaj, S.; Leng, X.; Zhou, X.; Liu, H.; Atala, A.; Zhang, Y. Self-renewal and differentiation capacity of urine-derived stem cells after urine preservation for 24 hours. PLoS One 2013, 8, e53980. [Google Scholar]
- Liu, G.; Pareta, R.A.; Wu, R.; Shi, Y.; Zhou, X.; Liu, H.; Deng, C.; Sun, X.; Atala, A.; Opara, E.C.; Zhang, Y. Skeletal myogenic differentiation of urine-derived stem cells and angiogenesis using microbeads loaded with growth factors. Biomaterials 2013, 34, 1311–1326. [Google Scholar] [CrossRef]
- Montarras, D.; Morgan, J.; Collins, C.; Relaix, F.; Zaffran, S.; Cumano, A.; Partridge, T.; Buckingham, M. Direct isolation of satellite cells for skeletal muscle regeneration. Science 2005, 309, 2064–2067. [Google Scholar] [CrossRef]
- Chapman, M.R.; Balakrishnan, K.R.; Li, J.; Conboy, M.J.; Huang, H.; Mohanty, S.K.; Jabart, E.; Hack, J.; Conboy, I.M.; Sohn, L.L. Sorting single satellite cells from individual myofibers reveals heterogeneity in cell-surface markers and myogenic capacity. Integr. Biol. (Camb.) 2013, 5, 692–702. [Google Scholar] [CrossRef]
- Muller, I.; Vaegler, M.; Holzwarth, C.; Tzaribatchev, N.; Pfister, S.M.; Schütt, B.; Reize, P.; Greil, J.; Handgretinger, R.; Rudert, M. Secretion of angiogenic proteins by human multipotent mesenchymal stromal cells and their clinical potential in the treatment of avascular osteonecrosis. Leukemia 2008, 22, 2054–2061. [Google Scholar] [CrossRef]
- Felka, T.; Schäfer, R.; de Zwart, P.; Aicher, W.K. Animal serum-free expansion and differentiation of human mesenchymal stromal cells. Cytotherapy 2010, 12, 143–153. [Google Scholar] [CrossRef]
- Persson, J.; Wolner-Hanssen, P.; Rydhstroem, H. Obstetric risk factors for stress urinary incontinence: A population-based study. Obstet. Gynecol. 2000, 96, 440–445. [Google Scholar] [CrossRef]
- Pilz, G.A.; Ulrich, C.; Ruh, M.; Abele, H.; Schäfer, R.; Kluba, T.; Bühring, H.J.; Rolauffs, B.; Aicher, W.K. Human term placenta-derived mesenchymal stromal cells are less prone to osteogenic differentiation than bone marrow-derived mesenchymal stromal cells. Stem Cells Dev. 2011, 20, 635–646. [Google Scholar] [CrossRef]
- Ulrich, C.; Rolauffs, B.; Abele, H.; Bonin, M.; Nieselt, K.; Hart, M.L.; Aicher, W.K. Low osteogenic differentiation potential of placenta-derived mesenchymal stromal cells correlates with low expression of the transcription factors Runx2 and Twist2. Stem Cells Dev. 2013, 22, 2859–2872. [Google Scholar] [CrossRef]
- Zhang, Y.; McNeill, E.; Tian, H.; Soker, S.; Andersson, K.E.; Yoo, J.J.; Atala, A. Urine derived cells are a potential source for urological tissue reconstruction. J. Urol. 2008, 180, 2226–2233. [Google Scholar] [CrossRef]
- Bodin, A.; Bharadwaj, S.; Wu, S.; Gatenholm, P.; Atala, A.; Zhang, Y. Tissue-engineered conduit using urine-derived stem cells seeded bacterial cellulose polymer in urinary reconstruction and diversion. Biomaterials 2010, 31, 8889–8901. [Google Scholar] [CrossRef]
- Wu, S.; Wang, Z.; Bharadwaj, S.; Hodges, S.J.; Atala, A.; Zhang, Y. Implantation of autologous urine derived stem cells expressing vascular endothelial growth factor for potential use in genitourinary reconstruction. J. Urol. 2011, 186, 640–647. [Google Scholar] [CrossRef]
- Wu, S.; Liu, Y.; Bharadwaj, S.; Atala, A.; Zhang, Y. Human urine-derived stem cells seeded in a modified 3D porous small intestinal submucosa scaffold for urethral tissue engineering. Biomaterials 2011, 32, 1317–1326. [Google Scholar] [CrossRef]
- Phinney, D.G.; Prockop, D.J. Concise review: Mesenchymal stem/multipotent stromal cells: The state of transdifferentiation and modes of tissue repair—Current views. Stem Cells 2007, 25, 2896–2902. [Google Scholar] [CrossRef]
- Wallner, C.; Dabhoiwala, N.F.; DeRuiter, M.C.; Lamers, W.H. The anatomical components of urinary continence. Eur. Urol. 2009, 55, 932–943. [Google Scholar] [CrossRef]
- Gang, E.J.; Jeong, J.A.; Hong, S.H.; Hwang, S.H.; Kim, S.W.; Yang, I.H.; Ahn, C.; Han, H.; Kim, H. Skeletal myogenic differentiation of mesenchymal stem cells isolated from human umbilical cord blood. Stem Cells 2004, 22, 617–624. [Google Scholar] [CrossRef]
- Dezawa, M.; Ishikawa, H.; Itokazu, Y.; Yoshihara, T.; Hoshino, M.; Takeda, S.; Ide, C.; Nabeshima, Y. Bone marrow stromal cells generate muscle cells and repair muscle degeneration. Science 2005, 309, 314–317. [Google Scholar] [CrossRef]
- Warren, L.; Manos, P.D.; Ahfeldt, T.; Loh, Y.H.; Li, H.; Lau, F.; Ebina, W.; Mandal, P.K.; Smith, Z.D.; Meissner, A. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 2010, 7, 618–630. [Google Scholar] [CrossRef]
- Mauro, A. Satellite cells of skeletal muscle fibers. J. Biophys. Biochem. Cytol. 1961, 9, 493–495. [Google Scholar] [CrossRef]
- Wagers, A.J.; Conboy, I.M. Cellular and molecular signatures of muscle regeneration: Current concepts and controversies in adult myogenesis. Cell 2005, 122, 659–667. [Google Scholar] [CrossRef]
- Chancellor, M.B.; Yokoyama, T.; Tirney, S.; Mattes, C.E.; Ozawa, H.; Yoshimura, N.; de Groat, W.C.; Huard, J. Preliminary results of myoblast injection into the urethra and bladder wall: A possible method for the treatment of stress urinary incontinence and impaired detrusor contractility. Neurourol. Urodyn. 2000, 19, 279–287. [Google Scholar] [CrossRef]
- Yiou, R.; Lefaucheur, J.P.; Atala, A. The regeneration process of the striated urethral sphincter involves activation of intrinsic satellite cells. Anat. Embryol. (Berl.) 2003, 206, 429–435. [Google Scholar]
- Kwon, D.; Minnery, B.; Kim, Y.; Kim, J.H.; de Miguel, F.; Yoshimura, N.; Chancellor, M.B. Neurologic recovery and improved detrusor contractility using muscle-derived cells in rat model of unilateral pelvic nerve transection. Urology 2005, 65, 1249–1253. [Google Scholar] [CrossRef]
- Elmi, A.; Kajbafzadeh, A.M.; Tourchi, A.; Talab, S.S.; Esfahani, S.A. Safety, efficacy and health related quality of life of autologous myoblast transplantation for treatment of urinary incontinence in children with bladder exstrophy-epispadias complex. J. Urol. 2011, 186, 2021–2026. [Google Scholar] [CrossRef]
- Winkler, T.; von Roth, P.; Matziolis, G.; Mehta, M.; Perka, C.; Duda, G.N. Dose-response relationship of mesenchymal stem cell transplantation and functional regeneration after severe skeletal muscle injury in rats. Tissue Eng. A 2009, 15, 487–492. [Google Scholar] [CrossRef]
- Lee, J.Y.; Cannon, T.W.; Pruchnic, R.; Fraser, M.O.; Huard, J.; Chancellor, M.B. The effects of periurethral muscle-derived stem cell injection on leak point pressure in a rat model of stress urinary incontinence. Int. Urogynecol. J. 2003, 14, 31–37. [Google Scholar] [CrossRef]
- Eberli, D.; Aboushwareb, T.; Soker, S.; Yoo, J.J.; Atala, A. Muscle precursor cells for the restoration of irreversibly damaged sphincter function. Cell Transplant. 2012, 21, 2089–2098. [Google Scholar] [CrossRef]
- Jiang, H.H.; Damaser, M.S. Animal models of stress urinary incontinence. Handb. Exp. Pharmacol. 2011, 202, 45–67. [Google Scholar] [CrossRef]
- Conway, D.A.; Kamo, I.; Yoshimura, N.; Chancellor, M.B.; Cannon, T.W. Comparison of leak point pressure methods in an animal model of stress urinary incontinence. Int. Urogynecol. J. Pelvic Floor Dysfunct. 2005, 16, 359–363. [Google Scholar] [CrossRef]
- Schultz-Lampel, D.; Goepel, M.; Haferkamp, A. Urodynamik; Springer: Berlin, Germany, 2012. [Google Scholar]
- Rossier, A.B.; Fam, B.A. 5-Microtransducer catheter in evaluation of neurogenic bladder function. Urology 1986, 27, 371–378. [Google Scholar] [CrossRef]
- Griffiths, D. The pressure within a collapsed tube, with special reference to urethral pressure. Med. Biol. 1985, 30, 951–963. [Google Scholar]
- Hadamard, J. Lectures on Cauchy’s Problem: In Linear Partial Differential Equations; Courier Dover Publications: New York, NY, USA, 2003. [Google Scholar]
- Bonnet, M.; Constantinescu, A. Inverse problems in elasticity. Inverse Probl. 2005, 21. [Google Scholar] [CrossRef]
- Balocco, S.; Basset, O.; Courbebaisse, G.; Boni, E.; Frangi, A.F.; Tortoli, P.; Cachard, C. Estimation of the viscoelastic properties of vessel walls using a computational model and doppler ultrasound. Phys. Med. Biol. 2010, 55, 3557–3575. [Google Scholar] [CrossRef]
- Perego, M.; Veneziani, A.; Vergara, C. A variational approach for estimating the compliance of the cardiovascular tissue: An inverse fluidstructure interaction problem. SIAM J. Sci. Comput. 2011, 33, 1181–1211. [Google Scholar] [CrossRef]
- Bertoglio, C.; Moireau, P.; Gerbeau, J. Sequential parameter estimation for fluid—Structure problems: Application to hemodynamics. Int. J. Numerical. Meth. Biomed. Eng. 2012, 28, 434–455. [Google Scholar] [CrossRef]
- Norton, P.; Brubaker, L. Urinary incontinence in women. Lancet 2006, 367, 57–67. [Google Scholar] [CrossRef]
- Schafer, R.; Bantleon, R.; Kehlbach, R.; Siegel, G.; Wiskirchen, J.; Wolburg, H.; Kluba, T.; Eibofner, F.; Northoff, H.; Claussen, C.D.; Schlemmer, H.P. Functional investigations on human mesenchymal stem cells exposed to magnetic fields and labeled with clinically approved iron nanoparticles. BMC Cell Biol. 2010, 11. [Google Scholar] [CrossRef]
- Guan, X.; Delo, D.M.; Atala, A.; Soker, S. In vitro cardiomyogenic potential of human amniotic fluid stem cells. J. Tissue Eng. Regen. Med. 2011, 5, 220–228. [Google Scholar] [CrossRef]
- Karnoub, A.E.; Dash, A.B.; Vo, A.P.; Sullivan, A.; Brooks, M.W.; Bell, G.W.; Richardson, A.L.; Polyak, K.; Tubo, R.; Weinberg, R.A. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 2007, 449, 557–563. [Google Scholar] [CrossRef]
- Pearson, T.; Greiner, D.L.; Shultz, L.D. Humanized SCID mouse models for biomedical research. Curr. Top. Microbiol. Immunol. 2008, 324, 25–51. [Google Scholar]
- Caplan, A.I.; Correa, D. The MSC: An injury drugstore. Cell Stem Cell 2011, 9, 11–15. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Aicher, W.K.; Hart, M.L.; Stallkamp, J.; Klünder, M.; Ederer, M.; Sawodny, O.; Vaegler, M.; Amend, B.; Sievert, K.D.; Stenzl, A. Towards a Treatment of Stress Urinary Incontinence: Application of Mesenchymal Stromal Cells for Regeneration of the Sphincter Muscle. J. Clin. Med. 2014, 3, 197-215. https://doi.org/10.3390/jcm3010197
Aicher WK, Hart ML, Stallkamp J, Klünder M, Ederer M, Sawodny O, Vaegler M, Amend B, Sievert KD, Stenzl A. Towards a Treatment of Stress Urinary Incontinence: Application of Mesenchymal Stromal Cells for Regeneration of the Sphincter Muscle. Journal of Clinical Medicine. 2014; 3(1):197-215. https://doi.org/10.3390/jcm3010197
Chicago/Turabian StyleAicher, Wilhelm K., Melanie L. Hart, Jan Stallkamp, Mario Klünder, Michael Ederer, Oliver Sawodny, Martin Vaegler, Bastian Amend, Karl D. Sievert, and Arnulf Stenzl. 2014. "Towards a Treatment of Stress Urinary Incontinence: Application of Mesenchymal Stromal Cells for Regeneration of the Sphincter Muscle" Journal of Clinical Medicine 3, no. 1: 197-215. https://doi.org/10.3390/jcm3010197
APA StyleAicher, W. K., Hart, M. L., Stallkamp, J., Klünder, M., Ederer, M., Sawodny, O., Vaegler, M., Amend, B., Sievert, K. D., & Stenzl, A. (2014). Towards a Treatment of Stress Urinary Incontinence: Application of Mesenchymal Stromal Cells for Regeneration of the Sphincter Muscle. Journal of Clinical Medicine, 3(1), 197-215. https://doi.org/10.3390/jcm3010197