Prevention and Treatment of Bone Metastases in Breast Cancer
Abstract
:1. Introduction
| Radiotherapy | The management of painful metastatic bone disease requires the use of multidisciplinary therapies such as hormone therapy or chemotherapy, external radiotherapy to the painful area or at time of risk of fracture or spinal cord compression, orthopaedic surgery, bisphosphonates, radionuclides and radiofrequency. Analgesics should be prescribed at any time. |
| Hormone therapy | |
| Chemotherapy | |
| Orthopedic surgery | |
| Analgesics | |
| Bisphosphonates | |
| Denosumab | |
| Interventional radiology (cementoplasty) | |
| Radionuclides | |
| Radiofrequency |
2. Clinical Presentation and Diagnostic Workup
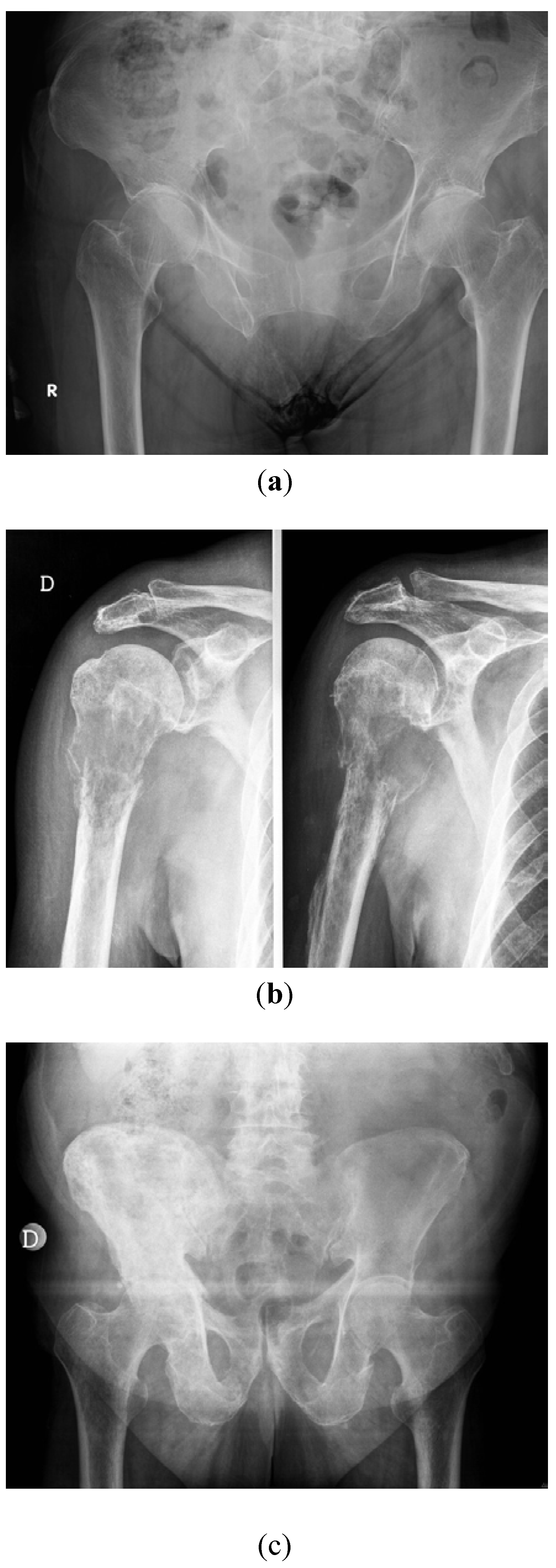
3. Medical Strategies for Bone Metastases
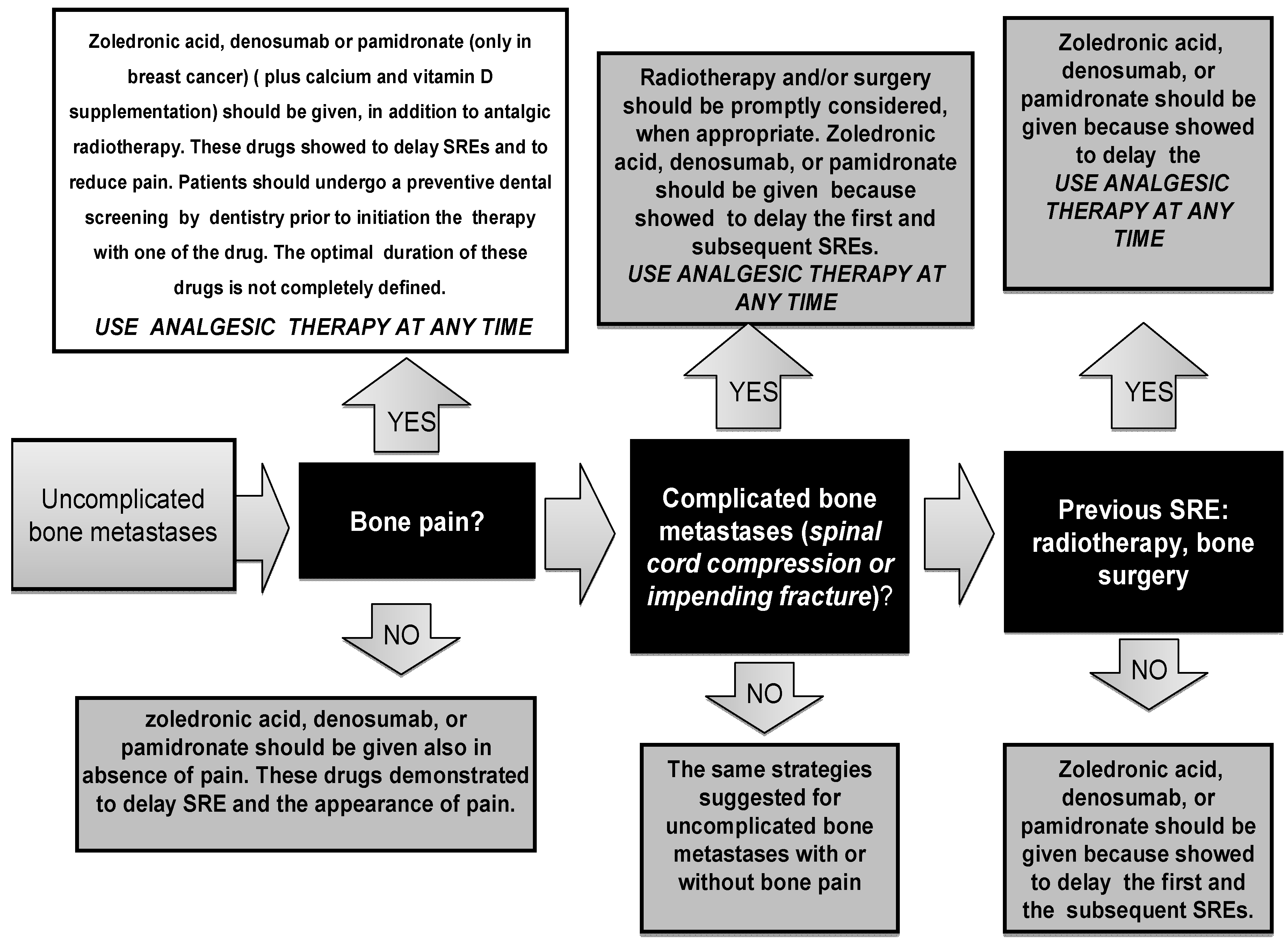
3.1. Bisphosphonates: Prevention of Skeletal Related Events, Bone Loss and Metastasis
3.2. Bone Pain: Assessment and Treatment during All the Illness Trajectories
| 1. Inform the patients about pain and pain management and encourage them to take an active role in their pain management. |
| 2. Prevent the onset of pain by means of the “by the clock” administration, taking into account the half-life, bioavailability and duration of action of the different drugs; thus analgesics for chronic pain should be prescribed on a regular basis and not on “as required” schedule. |
| 3. Prescribe a therapy which is simple to be administered and easy to be managed by the patient himself and his family, especially when the patient is cared for at home. The oral route appears to be the most suitable to meet this requirement, and, if well tolerated, it should be advocated as the 1st choice. |
| 4. Prescribe rescue dose of medications (as required) other than the regular basal therapy episodic or breakthrough pain episodes. |
| 5. Tailor the dosage, the type and the route of drugs administered according to each patient’s needs. |
3.3. Pain at Rest and Pain on Movement
4. Radiotherapy
4.1. Radiotherapy for Bone Pain Relief
4.2. Radiotherapy for Bone Recalcification
4.3. Radiotherapy for Metastatic Spinal Cord Compression
Steroids for Metastatic Spinal Cord Compression
4.4. Radioisotope for Bone Metastases
4.5. New Radiotherapy Technologies for Bone Metastases
5. Surgical Approaches for Bone Metastases and Metastatic Spinal Cord Compression
6. Conclusions
Conflicts of Interest
References
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Thun, M.J. Cancer statistics, 2009. CA Cancer J. Clin. 2009, 59, 225–249. [Google Scholar] [CrossRef]
- Coleman, R.E.; Rubens, R.D. The clinical course of bone metastases from breast cancer. Br. J. Cancer 1987, 55, 61–66. [Google Scholar] [CrossRef]
- Coleman, R.E. Skeletal complications of malignancy. Cancer 1997, 80, 1588–1594. [Google Scholar] [CrossRef]
- Hoskin, P.J. Bisphosphonates and radiation therapy for palliation of metastastic bone disease. Cancer Treat. Rev. 2003, 29, 321–327. [Google Scholar] [CrossRef]
- Early Breast Cancer Trialists’ Collaborative Group. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: An overview of the randomised trials. Lancet 2005, 365, 1687–1717. [CrossRef]
- Romond, E.; Perez, E.A.; Bryant, J.; Suman, V.J.; Geyer, C.E., Jr.; Davidson, N.E.; Tan-Chiu, E.; Martino, S.; Paik, S.; Kaufman, P.A. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N. Engl. J. Med. 2005, 353, 1673–1684. [Google Scholar] [CrossRef]
- Perez, E.A.; Romond, E.H.; Suman, V.J.; Jeong, J.H.; Davidson, N.E.; Geyer, C.E., Jr.; Martino, S.; Mamounas, E.P.; Kaufman, P.A.; Wolmark, N. Four-year follow-up of trastuzumab plus adjuvant chemotherapy for operable human epidermal growth factor receptor 2-positive breast cancer: Joint analysis of data from NCCTG N9831 and NSABP B-31. J. Clin. Oncol. 2011, 29, 3366–3373. [Google Scholar] [CrossRef]
- Perez, E.A.; Suman, V.J.; Davidson, N.E.; Gralow, J.R.; Kaufman, P.A.; Visscher, D.W.; Chen, B.; Ingle, J.N.; Dakhil, S.R.; Zujewski, J.; et al. Sequential versus concurrent trastuzumab in adjuvant chemotherapy for breast cancer. J. Clin. Oncol. 2011, 29, 4491–4497. [Google Scholar] [CrossRef]
- Piccart, M.; Procter, M.; Leyland-Jones, B.; Goldhirsch, A.; Untch, M.; Smith, I.; Gianni, L.; Baselga, J.; Bell, R.; Jackisch, C. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. First result of HERA trial. N. Engl. J. Med. 2005, 353, 1659–1672. [Google Scholar] [CrossRef]
- Gianni, L.; Dafni, U.; Gelber, R.D.; Azambuja, E.; Muehlbauer, S.; Goldhirsch, A.; Untch, M.; Smith, I.; Baselga, J.; Jackisch, C.; et al. Treatment with trastuzumab for 1 year after adjuvant chemotherapy in patients with HER2-positive early breast cancer: A 4-year follow-up of a randomised controlled trial. Lancet Oncol. 2011, 12, 236–244. [Google Scholar] [CrossRef]
- Slamon, D.; Eiermann, W.; Robert, N.; Pienkowski, T.; Martin, M.; Press, M.; Mackey, J.; Glaspy, J.; Chan, A.; Pawlicki, M.; et al. Adjuvant trastuzumab in HER2-positive breast cancer. N. Engl. J. Med. 2011, 365, 1273–1283. [Google Scholar] [CrossRef]
- Joensuu, H.; Kellokumpu-Lehtinen, P.L.; Bono, P.; Alanko, T.; Kataja, V.; Asola, R.; Utriainen, T.; Kokko, R.; Hemminki, A.; Tarkkanen, M.; et al. Adjuvant docetaxel or vinorelbine with or without trastuzumab for breast cancer. N. Engl. J. Med. 2006, 354, 809–820. [Google Scholar] [CrossRef]
- Joensuu, H.; Bono, P.; Kataja, V.; Alanko, T.; Kokko, R.; Asola, R.; Utriainen, T.; Turpeenniemi-Hujanen, T.; Jyrkkiö, S.; Möykkynen, K.; et al. Fluorouracil, epirubicina, and cyclophosphamide with either docetaxel or vinorelbine, with and without trastuzumab, as adjuvant treatments of breast cancer: Final results of the FinHer trial. J. Clin. Oncol. 2009, 27, 5685–5692. [Google Scholar] [CrossRef]
- Spielmann, M.; Rochè, H.; Delozier, T.; Canon, J.L.; Romieu, G.; Bourgeois, H.; Extra, J.M.; Serin, D.; Kerbrat, P.; Machiels, J.P. Trastuzumab for patients with axillary-node-positive breast cancer: Results of the FNCLCC-PACS 04 trial. J. Clin. Oncol. 2009, 27, 6129–6134. [Google Scholar] [CrossRef]
- The Arimidex, Tamoxifen, Alone or in Combination (ATAC) Trialists’ Group; Forbes, J.F.; Cuzick, J.; Buzdar, A.; Howell, A.; Tobias, J.S.; Baum, M. Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 100-Month analysis of the ATAC trial. Lancet Oncol. 2008, 9, 45–53. [Google Scholar] [CrossRef]
- BIG 1-98 Collaborative Group; Mouridsen, H.; Giobbie-Hurder, A.; Goldhirsch, A.; Thürlimann, B.; Paridaens, R.; Smith, I.; Mauriac, L.; Forbes, J.; Price, K.N. Letrozole therapy alone or in sequence with tamoxifene in women with breast cancer. N. Engl. J. Med. 2009, 361, 766–776. [Google Scholar] [CrossRef]
- Dowsett, M.; Cuzick, J.; Ingle, J.; Coates, A.; Forbes, J.; Bliss, J.; Buyse, M.; Baum, M.; Buzdar, A.; Colleoni, M. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J. Clin. Oncol. 2010, 28, 509–518. [Google Scholar] [CrossRef]
- LHRH-agonists in Early Breast Cancer Overview group; Cuzick, J.; Ambroisine, L.; Davidson, N.; Jakesz, R.; Kaufmann, M.; Regan, M.; Sainsbury, R. Use of luteinising-hormone-releasing hormone agonists as adjuvant treatment in premenopausal patients with hormonereceptor-positive breast cancer: A meta-analysis of individual patient data from randomised adjuvant trials. Lancet 2007, 369, 1711–1723. [Google Scholar] [CrossRef]
- Klijn, J.G.; Beex, L.V.; Mauriac, L.; van Zijl, J.A.; Veyret, C.; Wildiers, J.; Jassem, J.; Piccart, M.; Burghouts, J.; Becquart, D. Combined treatment with buserelin and tamoxifen in premenopausal metastatic breast cancer: A randomized study. J. Natl. Cancer Inst. 2000, 92, 903–911. [Google Scholar] [CrossRef]
- Peto, R.; Davies, C.; Godwin, J.; Gray, R.; Pan, H.C.; Clarke, M.; Cutter, D.; Darby, S.; McGale, P.; Taylor, C. Comparisons between different polychemotherapy regimens for early breast cancer: Meta-analyses of long-term outcome among 100,000 women in 123 randomised trials. Lancet 2012, 379, 432–444. [Google Scholar] [CrossRef]
- De Laurentiis, M.; Cancello, G.; D’Agostino, D.; Giuliano, M.; Giordano, A.; Montagna, E.; Lauria, R.; Forestieri, V.; Esposito, A.; Silvestro, L. Taxane-based combinations as adjuvant chemotherapy of early breast cancer: A meta-analysis of randomized trials. J. Clin. Oncol. 2008, 26, 44–53. [Google Scholar] [CrossRef]
- Jones, S.E.; Savin, M.A.; Holmes, F.A.; O’Shaughnessy, J.A.; Blum, J.L.; Vukelja, S.; McIntyre, K.J.; Pippen, J.E.; Bordelon, J.H.; Kirby, R. Phase III trial comparing doxorubicin plus cyclophosphamide with docetaxel plus cyclophosphamide as adjuvant therapy for operable breast cancer. J. Clin. Oncol. 2006, 24, 5381–5387. [Google Scholar] [CrossRef]
- Jones, S.; Holmes, F.; O’Shaughnessy, J.; Blum, J.L.; Vukelja, S.J.; McIntyre, K.J.; Pippen, J.E.; Bordelon, J.H.; Kirby, R.L.; Sandbach, J. Docetaxel with cyclophosphamide is associated with an overall survival benefit compared with doxorubicin and cyclophosphamide: 7-Year follow-up of US Oncology Research trial 9735. J. Clin. Oncol. 2009, 27, 1177–1183. [Google Scholar] [CrossRef]
- AIOM Guidelines on Breast Cancer 2012. Available online: http://www.aiom.com (accessed on 24 July 2013).
- Body, J.J.; Diel, I.J.; Lichinitser, M.R.; Kreuser, E.D.; Dornoff, W.; Gorbunova, V.A.; Budde, M.; Bergström, B.; MF 4265 Study Group. Intravenous ibandronate reduces the incidence of skeletal complications in patients with breast cancer and bone metastases. Ann. Oncol. 2003, 14, 1399–1405. [Google Scholar] [CrossRef]
- Kohno, N.; Aogi, K.; Minami, H.; Nakamura, S.; Asaga, T.; Iino, Y.; Watanabe, T.; Goessl, C.; Ohashi, Y.; Takashima, S. Zoledronic acid significantly reduces skeletal complications compared with placebo in Japanese women with bone metastases from breast cancer: A randomized, placebo-controlled trial. J. Clin. Oncol. 2005, 23, 3314–3321. [Google Scholar] [CrossRef]
- Lipton, A.; Theriault, R.L.; Hortobagyi, G.N.; Simeone, J.; Knight, R.D.; Mellars, K.; Reitsma, D.J.; Heffernan, M.; Seaman, J.J. Pamidronate prevents skeletal complications and is effective palliative treatment in women with breast carcinoma and osteolytic bone metastases: Long term follow-up of two randomized, placebo-controlled trials. Cancer 2000, 88, 1082–1090. [Google Scholar] [CrossRef]
- Paterson, A.H.; Powles, T.J.; Kanis, J.A.; McCloskey, E.; Hanson, J.; Ashley, S. Double-blind controlled trial of oral clodronate in patients with bone metastases from breast cancer. J. Clin. Oncol. 1993, 11, 59–65. [Google Scholar] [PubMed]
- Rosen, L.S.; Gordon, D.; Kaminski, M.; Howell, A.; Belch, A.; Mackey, J.; Apffelstaedt, J.; Hussein, M.A.; Coleman, R.E.; Reitsma, D.J.; et al. Long-term efficacy and safety of zoledronic acid compared with pamidronate disodium in the treatment of skeletal complications in patients with advanced multiple myeloma or breast carcinoma: A randomized, double-blind, multicenter, comparative trial. Cancer 2003, 98, 1735–1744. [Google Scholar] [CrossRef]
- Saarto, T.; Blomqvist, C.; Valimaki, M.; Makela, P.; Sarna, S.; Elomaa, I. Clodronate improves bone mineral density in post-menopausal breast cancer patients treated with adjuvant antioestrogens. Br. J. Cancer 1997, 75, 602–605. [Google Scholar] [CrossRef]
- Van Poznak, C.H.; Temin, S.; Yee, G.C.; Janjan, N.A.; Barlow, W.E.; Biermann, J.S.; Bosserman, L.D.; Geoghegan, C.; Hillner, B.E.; Theriault, R.L.; et al. American Society of Clinical Oncology executive summary of the clinical practice guideline update on the role of bone-modifying agents in metastatic breast cancer. J. Clin. Oncol. 2011, 29, 1221–1227. [Google Scholar] [CrossRef]
- Fizazi, K.; Carducci, M.; Smith, M.; Damião, R.; Brown, J.; Karsh, L.; Milecki, P.; Shore, N.; Rader, M.; Wang, H.; et al. Denosumab versus zoledronic acid for treatment of bone metastases in men with castration-resistent prostate cancer: A randomized, double-blind study. Lancet 2011, 377, 813–822. [Google Scholar] [CrossRef]
- Stopeck, A.T.; Lipton, A.; Body, J.J.; Steger, G.G.; Tonkin, K.; de Boer, R.H.; Lichinitser, M.; Fujiwara, Y.; Yardley, D.A.; Viniegra, M.; et al. Denosumab compared with zoledronic acid for the treatment of bone metastases in patients with advanced breast cancer: A randomized, double-blind study. J. Clin. Oncol. 2010, 28, 5132–5139. [Google Scholar] [CrossRef]
- Amadori, D.; Aglietta, M.; Alessi, B.; Gianni, L.; Ibrahim, T.; Farina, G.; Gaion, F.; Bertoldo, F.; Santini, D.; Rondena, R.; et al. Efficacy and safety of 12-wwkly versus 4-weekly zoledronic acid for prolonged treatment of patients with bone metastases from breast cancer (ZOOM): A phase 3, open-label, randomised, non-inferiority trial. Lancet Oncol. 2013, 14, 663–670. [Google Scholar] [CrossRef]
- Coleman, R.E.; Major, P.; Lipton, A.; Brown, J.E.; Lee, K.A.; Smith, M.; Saad, F.; Zheng, M.; Hei, Y.J.; Seaman, J.; et al. Predictive value of bone resorption and formation markers in cancer patients with bone metastases receiving the bisphosphonate zoledronicacid. J. Clin. Oncol. 2005, 23, 4925–4935. [Google Scholar] [CrossRef]
- American Association of Oral and Maxillofacial Surgeons. Position paper on bisphosphonate-related osteonecrosis of the jaws. J. Oral Maxillofac. Surg. 2007, 65, 369–376. [CrossRef]
- Ripamonti, C.I.; Maniezzo, M.; Campa, T.; Fagnoni, E.; Brunelli, C.; Saibene, G.; Bareggi, C.; Ascani, L.; Cislaghi, E. Decreased occurrence of osteonecrosis of the jaw after implementation of dental preventive measures in solid tumour patients with bone metastases treated with bisphosphonates. The experience of the National Cancer Institute of Milan. Ann. Oncol. 2009, 20, 137–145. [Google Scholar] [PubMed]
- American Association of Oral and Maxillofacial Surgeons. Position paper on bisphosphonate-related osteonecrosis of the jaw—2009 Update. Available online: http://www.aaoms.org/docs/position_papers/bronj_update.pdf (accessed on 14 June 2010).
- Ripamonti, C.I.; Cislaghi, E.; Mariani, L.; Maniezzo, M. Efficay and safety of medical ozone (O3) delivered in oil suspension applications for the treatment of osteonecrosis of the jaw in patients with bone metastases treated with bisphosphonates. Preliminary results of a phase I–II study. Oral Oncol. 2011, 47, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Ripamonti, C.; Maniezzo, M.; Pessi, M.A.; Cislaghi, E.; Mariani, L.; Boldini, S. Efficacy and tolerability of medical ozone (O3) gas insufflations in patients with osteonecrosis of the jaw treated with bisphosphonates—Preliminary data: Medical ozone gas insufflation in treating ONJ lesions. J. Bone Oncol. 2012, 1, 81–87. [Google Scholar] [CrossRef]
- Gnant, M.; Dubsky, P.; Hadji, P. Bisphosphonates: Prevention of bone metastases in breast cancer. Recent Results Cancer Res. 2012, 192, 65–91. [Google Scholar] [PubMed]
- Lustberg, M.B.; Reinbolt, R.E.; Shapiro, C.L. Bone health in adult cancer survivorship. J. Clin. Oncol. 2012, 30, 3665–3674. [Google Scholar] [CrossRef] [PubMed]
- Winter, M.C.; Coleman, R.E. Bisphosphonates in the adjuvant treatment of breast cancer. Clin. Oncol. 2013, 25, 135–145. [Google Scholar] [CrossRef]
- Coleman, R.E. Adjuvant bone-targeted therapy to prevent metastasis: Lessons from the AZURE study. Curr. Opin. Support. Palliat. Care 2012, 6, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Young, R.J.; Coleman, R.E. Zoledronic acid to prevent and treat cancer metastasis: New prospects for an old drug. Future Oncol. 2013, 9, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Powles, T.; Paterson, S.; Kanis, J.A.; McCloskey, E.; Ashley, S.; Tidy, A.; Rosenqvist, K.; Smith, I.; Ottestad, L.; Legault, S.; et al. Randomized placebo-controlled trial of clodronate in patients with primary operable breast cancer. J. Clin. Oncol. 2002, 20, 3219–3224. [Google Scholar] [CrossRef] [PubMed]
- Saarto, T.; Blomqvist, C.; Virkkunen, P.; Elomaa, I. Adjuvant clodronate treatment does not reduce the frequency of skeletal metastases in node positive breast cancer patients: 5-Year results of a randomized controlled trial. J. Clin. Oncol. 2001, 19, 10–17. [Google Scholar] [PubMed]
- Diel, I.J.; Jaschke, A.; Solomayer, E.F.; Gollan, C.; Bastert, G.; Sohn, C.; Schuetz, F. Adjuvant oral clodronate improves the overall survival of primary breast cancer patients with micrometastases to the bone marrow—A long term follow-up. Ann. Oncol. 2008, 19, 2007–2011. [Google Scholar] [CrossRef] [PubMed]
- Gnant, M.; Mlineritsch, B.; Schipping, W.; Luschin-Ebengreuth, G.; Pöstlberger, S.; Menzel, C.; Jakesz, R.; Seifert, M.; Hubalek, M.; Bjelic-Radisic, V.; et al. Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N. Engl. J. Med. 2009, 360, 679–691. [Google Scholar] [CrossRef]
- Gnant, M.; Mlineritsch, B.; Schippinger, W.; Luschin-Ebengreuth, G.; Pöstlberger, S.; Menzel, C.; Jakesz, R.; Seifert, M.; Hubalek, M.; Bjelic-Radisic, V.; et al. Adjuvant endocrine therapy plus zoledronic acid in premenopausal women with early-stage breast cancer: 62-Month follow-up from the ABCSG-12 randomized trial. Lancet Oncol. 2011, 12, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Gnant, M.; Mlineritsch, B.; Stoeger, H.; Luschin-Ebengreuth, G.; Heck, D.; Menzel, C.; Jakesz, R.; Seifert, M.; Hubalek, M.; Pristauz, G.; et al. Long-term follow-up in ABCSG-12: Significant improved overall survival with adjuvant zoledronic acid in pre-menopausal patients with endocrine-receptor-positive early breast cancer. Cancer Res. 2011, 71, S1–S2. [Google Scholar]
- Coleman, R.E.; Marshall, H.; Cameron, D.; Dodwell, D.; Burkinshaw, R.; Keane, M.; Gil, M.; Houston, S.J.; Grieve, R.J.; Barrett-Lee, P.J.; et al. Breast cancer adjuvant therapy with zoledronic acid. N. Engl. J. Med. 2011, 365, 1396–1405. [Google Scholar] [CrossRef]
- Eidtmann, H.; de Boer, R.; Bundred, N.; Llombart-Cussac, A.; Davidson, N.; Neven, P.; von Minckwitz, G.; Miller, J.; Schenk, N.; Coleman, R. Efficacy of zoledronic acid in postmenopausal women with early breast cancer adjuvant letrozole: 36-Month results of the ZO-FAST study. Ann. Oncol. 2010, 21, 2188–2194. [Google Scholar] [CrossRef]
- Ripamonti, C.; Santini, D.; Maranzano, E.; Berti, M.; Roila, F.; ESMO Guidelines Working Group. Management of cancer pain: ESMO clinical practice guidelines. Ann. Oncol. 2012, 23, 139–154. [Google Scholar] [CrossRef]
- Word Health Organization, World Health Organization Cancer Pain Relief Geneva; Word Health Organization: Geneve, Switzerland, 1986.
- Word Health Organization, World Health Organization Cancer Pain Relief, 2nd Geneva ed; Word Health Organization: Geneve, Switzerland, 1996.
- Mercadante, S.; Radbruch, L.; Caraceni, A.; Cherny, N.; Kaasa, S.; Nauck, F.; Ripamonti, C.; de Conno, F. Episodic breakthrough pain: Consensus conference of an export working group of the European Association for Palliative Care. Cancer 2002, 94, 832–839. [Google Scholar] [CrossRef]
- Wong, R.K.S.; Wiffen, P.J. Bisphosphonates for the relief of pain secondary to bone metastases. Cochrane Database Syst. Rev. 2002, 2. [Google Scholar] [CrossRef]
- Cleeland, C.S.; Body, J.J.; Stopeck, A.; von Moos, R.; Fallowfield, L.; Mathias, S.D.; Patrick, D.L.; Clemons, M.; Tonkin, K.; Masuda, N.; et al. Pain outcomes in patients with advanced breast cancer and bone metastases: Results from a randomized, double-blind study of denosumab and zoledronic acid. Cancer 2013, 119, 832–838. [Google Scholar] [CrossRef]
- Wu, J.S.; Wong, R.K.; Lloyd, N.S.; Johnston, M.; Bezjak, A.; Whelan, T.; Supportive Care Guidelines Group of Cancer Care Ontario. Radiotherapy fractionation for the palliation of uncomplicated painful bone metastases—An evidence-based practice guideline. BMC Cancer 2004, 4. [Google Scholar] [CrossRef]
- Koswig, S.; Budach, V. Remineralization and pain relief in bone metastases after different radiotherapy fractions (10 times 3 Gy vs. 1 time 8 Gy). A prospective study. Strahlenther. Onkol. 1999, 175, 500–508. [Google Scholar] [CrossRef]
- Chow, E.; Harris, K.; Fan, G.; Tsao, M.; Sze, W.M. Palliative radiotherapy trials for bone metastases: A systematic review. J. Clin. Oncol. 2007, 10, 1423–1436. [Google Scholar]
- Fairchild, A.; Barnes, E.; Ghosh, S.; Ben-Josef, E.; Roos, D.; Hartsell, W.; Holt, T.; Wu, J.; Janjan, N.; Chow, E. International pattern of practice in palliative radiotherapy for painful bone metastases: Evidence-based practice? Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 1501–1510. [Google Scholar] [CrossRef]
- Lutz, S.; Berk, L.; Chang, E.; Lutz, S.; Berk, L.; Chang, E.; Chow, E.; Hahn, C.; Hoskin, P.; Howell, D.; et al. Palliative radiotherapy for bone metastases: An ASTRO evidence-based guideline. Int. J. Radiat. Oncol. Biol. Phys. 2011, 79, 965–976. [Google Scholar] [CrossRef]
- Howell, D.D.; James, J.L.; Hartsell, W.F.; Suntharalingam, M.; Machtay, M.; Suh, J.H.; Demas, W.F.; Sandler, H.M.; Kachnic, L.A.; Berk, L.B. Single-fraction radiotherapy versus multifraction radiotherapy for palliation of painful vertebral bone metastases-equivalent efficacy, less toxicity, more convenient. Cancer 2013, 119, 888–896. [Google Scholar] [CrossRef]
- Van der Linden, Y.M.; Lok, J.J.; Steenland, E.; Martijn, H.; van Houwelingen, H.; Marijnen, C.A.; Leer, J.W. Single fraction radiotherapy is efficacious: A further analysis of the Dutch Bone Metastasis Study controlling for the influence of retreatment. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, 528–537. [Google Scholar] [CrossRef]
- Chow, E.; Hoskin, P.J.; Wu, J.; Roos, D.; van der Linden, Y.; Hartsell, W.; Vieth, R.; Wilson, C.; Pater, J. A phase III international randomised trial comparing single with multiple fractions for re-irradiation of painful bone metastases: National Cancer Institute of Canada Clinical Trials Group (NCICCTG) SC20. Clin.Oncol. 2006, 18, 125–128. [Google Scholar] [CrossRef]
- Huisman, M.; van den Bosch, M.A.; Wijlemans, J.W.; van Vulpen, M.; van der Linden, Y.M.; Verkooijen, H.M. Effectiveness of reirradiation for painful bonemetastases: A systematic review and meta-analysis. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, 8–14. [Google Scholar] [CrossRef]
- Cai, B.; Nickman, N.A.; Gaffney, D.K. The role of palliative external beam radiation therapy in boney metastases pain management. J. Pain Palliat. Care Pharmacother. 2013, 27, 28–34. [Google Scholar] [CrossRef]
- Dennis, K.; Makhani, L.; Zeng, L.; Lam, H.; Chow, E. Single fraction conventional external beam radiation therapy for bone metastases: A systematic review of randomised controlled trials. Radiother.Oncol. 2013, 106, 5–14. [Google Scholar] [CrossRef]
- Souchon, R.; Wenz, F.; Sedlmayer, F.; Budach, W.; Dunst, J.; Feyer, P.; Haase, W.; Harms, W.; Sautter-Bihl, M.L.; Sauer, R. DEGRO practice guidelines for palliative radiotherapy of metastatic breast cancer: Bone metastases and metastatic spinal cord compression (MSCC). Strahlenther. Onkol. 2009, 185, 417–424. [Google Scholar] [CrossRef]
- Falkmer, U.; Järhult, J.; Wersäll, P.; Cavallin-Ståhl, E. A systematic overview of radiation therapy effects in skeletal metastases. Acta Oncol. 2003, 42, 620–633. [Google Scholar] [CrossRef]
- Kouloulias, E.V.; Kouvaris, R.J.; Antypas, C.; Mystakidou, K.; Matsopoulos, G.; Uzunoglu, C.N.; Moulopoulos, A.; Vlahos, J.L. An intra-patient dose escalation study of disodium pamidronate plus radiotherapy versus radiotherapy alone for the treatment of osteolytic metastasis. Monitoring of recalcification using image-processing. Strahlenther. Onkol. 2003, 179, 471–479. [Google Scholar] [PubMed]
- Maranzano, E.; Trippa, F.; Chirico, L.; Basagni, M.L.; Rossi, R. Management of metastatic spinal cord compression. Tumori 2003, 89, 469–475. [Google Scholar] [PubMed]
- Loblaw, D.A.; Mitera, G.; Ford, M.; Laperriere, N.J. A 2011 updated systematic review and clinical practice guideline for the management of malignant extradural spinal cord compression. Int.J.Radiat.Oncol.Biol.Phys. 2012, 84, 312–317. [Google Scholar] [CrossRef]
- Byrne, T.N. Spinal cord compression from epidural metastases. N. Engl. J. Med. 1992, 327, 614–619. [Google Scholar] [CrossRef]
- Rades, D.; Heidenreich, F.; Karstens, J.H. Final results of a prospective study of the prognostic value of the time to develop motor deficits before irradiation in metastatic spinal cord compression. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 975–979. [Google Scholar] [CrossRef]
- Patchell, R.A.; Tibbs, P.A.; Regine, W.F.; Payne, R.; Saris, S.; Kryscio, R.J.; Mohiuddin, M.; Young, B. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer: A randomised trial. Lancet 2005, 366, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Maranzano, E.; Bellavita, R.; Rossi, R.; de Angelis, V.; Frattegiani, A.; Bagnoli, R.; Mignogna, M.; Beneventi, S.; Lupattelli, M.; Ponticelli, P.; et al. Short-course versus split-course radiotherapy in metastatic spinal cord compression: Results of a phase III, randomized, multicenter trial. J. Clin. Oncol. 2005, 23, 3358–3365. [Google Scholar] [CrossRef]
- Maranzano, E.; Trippa, F.; Casale, M.; Costantini, S.; Lupattelli, M.; Bellavita, R.; Marafioti, L.; Pergolizzi, S.; Santacaterina, A.; Mignogna, M.; et al. 8 Gy single-dose radiotherapy is effective in metastatic spinal cord compression: Results of a phase III randomized multicentre Italian trial. Radiother. Oncol. 2009, 93, 174–179. [Google Scholar] [CrossRef]
- Prewett, S.; Venkitaraman, R. Metastatic spinal cord compression: Review of the evidence for a radiotherapy dose fractionation schedule. Clin. Oncol. 2010, 22, 222–230. [Google Scholar] [CrossRef]
- Holt, T.; Hoskin, P.; Maranzano, E.; Sahgal, A.; Schild, S.; Ryu, S.; Loblaw, A. Malignant epidural spinal cord compression: The role of external beam radiotherapy. Curr. Opin. Support. Palliat. Care 2012, 6, 103–108. [Google Scholar] [CrossRef]
- Rades, D.; Lange, M.; Veninga, T.; Stalpers, L.J.; Bajrovic, A.; Adamietz, I.A.; Rudat, V.; Schild, S.E. Final results of a prospective study comparing the local control of short-course and long-course radiotherapy for metastatic spinal cord compression. Int. J. Radiat. Oncol. Biol.Phys. 2011, 79, 524–530. [Google Scholar] [CrossRef]
- Sorensen, S.; Helweg-Larsen, S.; Mouridsen, H.; Hansen, H.H. Effect of high-dose dexamethasone in carcinomatous metastatic spinal cord compression treated with radiotherapy: A randomized trial. Eur. J. Cancer 1994, 1, 22–27. [Google Scholar]
- Maranzano, E.; Latini, P.; Beneventi, S.; Perrucci, E.; Panizza, B.M.; Aristei, C.; Lupattelli, M.; Tonato, M. Radiotherapy without steroids in selected metastatic spinal cord compression patients. A phase II trial. Am. J. Clin. Oncol. 1996, 19, 179–184. [Google Scholar] [CrossRef]
- Sciuto, R.; Festa, A.; Pasqualoni, R.; Semprebene, A.; Rea, S.; Bergomi, S.; Maini, C.L. Metastatic bone pain palliation with 89-Sr and 186-Re-HeDP in breast cancer patients. Breast Cancer Res. Treat. 2001, 66, 101–109. [Google Scholar] [CrossRef]
- Leondi, A.H.; Souvatzoglou, M.A.; Rapti, A.S.; Leontopoulou, S.A.; Papadaki, E.K.; Datseris, E.I.; Anagnostopoulou, U.S.; Zerva, C.J. Palliative treatment of painful disseminated bone metastases with 186Rhenium-HeDP in patients with lung cancer. J. Nucl. Med. 2004, 48, 211–219. [Google Scholar]
- Han, S.H.; de Klerk, J.M.; Tan, S.; van Het Schip, A.D.; Derksen, B.H.; van Dijk, A.; Kruitwagen, C.L.; Blijham, G.H.; van Rijk, P.P.; Zonnenberg, B.A. The PLACORHeN study: A double-blind, placebo-controlled, randomized radionuclide study with (186)Re-etidronate in hormone-resistant prostate cancer patients with painful bone metastases. J. Nucl. Med. 2002, 43, 1150–1156. [Google Scholar] [PubMed]
- Roqué, I.; Figuls, M.; Martinez-Zapata, M.J.; Scott-Brown, M.; Alonso-Coello, P. Radioisotopes for metastatic bone pain. Cochrane Database Syst. Rev. 2011, 7. [Google Scholar] [CrossRef]
- Saghal, A.; Larson, D.A.; Chang, E.L. Stereotactic body radiosurgery for spinal metastases: A critical review. Int. J. Radiat. Oncol. Biol. Phys. 2008, 71, 652–665. [Google Scholar] [CrossRef]
- Regine, W.; Ryu, S.; Chang, E.L. Spine radiosurgery for spinal cord compression: The radiation oncologist’s perspective. J. Radiosurg. SBRT 2011, 1, 55–61. [Google Scholar]
- Chang, E.L.; Shiu, A.S.; Mendel, E.; Mathews, L.A.; Mahajan, A.; Allen, P.K.; Weinberg, J.S.; Brown, B.W.; Wang, X.S.; Woo, S.Y.; et al. Phase I/II study of stereotactic body radiotherapy for spinal metastasis and its pattern of failure. J. Neurosurg. Spine 2007, 7, 151–160. [Google Scholar] [CrossRef]
- Attar, S.; Steffner, R.J.; Avedian, R.; Hussain, W.M. Surgicalintervention of non-vertebral osseousmetastasis. Cancer Control 2012, 19, 113–121. [Google Scholar] [PubMed]
- Townsend, P.W.; Rosenthal, H.G.; Smalley, S.R.; Cozad, S.C.; Hassanein, R.E. Impact of postoperative radiation therapy and other perioperative factors on outcome after orthopedic stabilization of impending or pathologic fractures due to metastatic disease. J. Clin. Oncol. 1994, 12, 2345–2350. [Google Scholar] [PubMed]
- Fourney, D.R.; Schomer, D.F.; Nader, R.; Chlan-Fourney, J.; Suki, D.; Ahrar, K.; Rhines, L.D.; Gokaslan, Z.L. Percutaneous vertebroplasty and kyphoplasty for painful vertebral body fractures in cancer patients. J. Neurosurg. 2003, 98, 21–30. [Google Scholar] [CrossRef]
- Pflugmacher, R.; Taylor, R.; Agarwal, A.; Melcher, I.; Disch, A.; Haas, N.P.; Klostermann, C. Balloon kyphoplasty in the treatment of metastatic disease of the spine: A 2-year prospective evaluation. Eur. Spine J. 2008, 17, 1042–1048. [Google Scholar] [CrossRef]
- Gerszten, P.C.; Germanwala, A.; Burton, S.; Welch, W.C.; Ozhasoglu, C.; Vogel, W. Combination kyphoplasty and spinal radiosurgery: A new treatment paradigm for pathological fractures. J. Neurosurg. Spine 2005, 3, 296–301. [Google Scholar] [CrossRef]
- Ashamalla, H.; Cardoso, E.; Macedon, M.; Guirguis, A.; Weng, L.; Ali, S.; Mokhtar, B.; Ashamalla, M.; Panigrahi, N. Phase I trial of Vertebral Intracavitary Cement and Samarium (VICS): Novel technique for treatment of painful vertebral metastasis. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 836–842. [Google Scholar] [CrossRef]
- Fourney, D.R.; Abi-Said, D.; Lang, F.F.; McCutcheon, I.E.; Gokaslan, Z.L. Use of pedicle screw fixation in the management of malignant spinal disease: Experience in 100 consecutive procedures. J. Neurosurg. 2001, 94, 25–37. [Google Scholar] [PubMed]
- Gokaslan, Z.L.; York, J.E.; Walsh, G.L.; McCutcheon, I.E.; Lang, F.F.; Putnam, J.B., Jr.; Wildrick, D.M.; Swisher, S.G.; Abi-Said, D.; Sawaya, R. Transthoracic vertebrectomy for metastatic spinal tumors. J. Neurosurg. 1998, 89, 599–609. [Google Scholar] [CrossRef]
- Klimo, P., Jr.; Thompson, C.J.; Kestle, J.R.; Schmidt, M.H. A meta-analysis of surgery versus conventional radiotherapy for the treatment of metastatic spinal epidural disease. Neuro Oncol. 2005, 7, 64–76. [Google Scholar] [CrossRef]
- Young, R.F.; Post, E.M.; King, G.A. Treatment of spinal epidural metastases: Randomized prospective comparison of laminectomy and radiotherapy. J. Neurosurg. 1980, 53, 741–748. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ripamonti, C.; Trippa, F.; Barone, G.; Maranzano, E. Prevention and Treatment of Bone Metastases in Breast Cancer. J. Clin. Med. 2013, 2, 151-175. https://doi.org/10.3390/jcm2030151
Ripamonti C, Trippa F, Barone G, Maranzano E. Prevention and Treatment of Bone Metastases in Breast Cancer. Journal of Clinical Medicine. 2013; 2(3):151-175. https://doi.org/10.3390/jcm2030151
Chicago/Turabian StyleRipamonti, Carla, Fabio Trippa, Gloria Barone, and Ernesto Maranzano. 2013. "Prevention and Treatment of Bone Metastases in Breast Cancer" Journal of Clinical Medicine 2, no. 3: 151-175. https://doi.org/10.3390/jcm2030151
APA StyleRipamonti, C., Trippa, F., Barone, G., & Maranzano, E. (2013). Prevention and Treatment of Bone Metastases in Breast Cancer. Journal of Clinical Medicine, 2(3), 151-175. https://doi.org/10.3390/jcm2030151





