1. Introduction
Auto-inflammatory diseases (AID) constitute a group of rare, hereditary disorders characterized by dysregulation of the innate immune system, leading to chronic systemic inflammation [
1]. Pro-inflammatory cytokines, particularly interleukin-1 (IL-1), play a pivotal role in driving inflammatory cascades that result in recurrent clinical manifestations such as fever, rash, arthralgia, and, notably, fatigue [
1,
2,
3]. The advent of IL-1-targeted therapies have revolutionized disease management, effectively controlled systemic inflammation, and ameliorated many clinical features [
2,
4,
5].
Fatigue, however, has emerged as one of the most pervasive and disabling symptoms in AID. Unlike ordinary tiredness, it is severe, persistent, and disproportionate to exertion or rest, substantially impairing quality of life and daily functioning [
6]. Importantly, fatigue often persists even when systemic inflammation is adequately controlled by IL-1 blockade, suggesting that its etiology is complex and extends beyond direct inflammatory activity [
7,
8]. This residual fatigue contributes to long-term disability and diminished productivity. It results in a considerable societal burden [
8].
In parallel, depressive symptoms have increasingly been recognized in patients with AID; they often remain underdiagnosed and undertreated. Chronic inflammation has been implicated in the pathogenesis of depression, with cytokines such as IL-1, IL-6, and TNF-α linked to altered mood regulation, neuroplasticity, and hypothalamic–pituitary–adrenal axis function [
9]. Moreover, the psychosocial consequences of living with a chronic, unpredictable illness further heighten vulnerability to depression and other mental health disorders [
10]. Depression in this context not only worsens overall disease burden but also interferes with adherence, functional capacity, and quality of life [
7,
10,
11].
Despite the high prevalence of fatigue and depression in AID, their interrelationship has not been systematically examined to date. This knowledge gap hinders comprehensive patient management. It underscores the need for studies that characterize both symptoms, explore their correlation, and identify disease- and patient-related factors that contribute to their persistence.
Therefore, the aims of this study were as follows: (1) to characterize fatigue and depression in a longitudinal cohort of pediatric and adult patients with AIDs on effective therapy including global measures of fatigue severity, (2) to compare Overall Fatigue and its domains and depression across diseases, and (3) to identify modifiable risk factors associated with debilitating fatigue in patients with treated auto-inflammatory diseases.
2. Materials and Methods
A single-center cohort study of consecutive pediatric and adult patients with a confirmed diagnosis of an auto-inflammatory disease, including CAPS and Familial Mediterranean Fever (FMF), between January 2007 and June 2024 was performed. The inclusion criteria were as follows: (1) an established diagnosis of CAPS or FMF according to published criteria [
1,
4,
12]; (2) age ≥ 8 years at the last visit [
13], and (3) evidence of inactive disease either on or off treatment at the last follow-up visit [
14]. Patients with comorbidities known to be independently associated with fatigue, including diagnosed malignancies and mental health disorders, were excluded. All clinical and laboratory data were prospectively recorded in the institutional electronic registry (Arthritis and Rheumatism Database and Information System). Ethical approval for the study protocol was obtained from the Ethics Committee of the Medical Faculty, Eberhard Karls University of Tuebingen, and the University Hospital Tuebingen (Project Number: 070/2024BO2). All participants (or their legal guardians) provided written informed consent prior to inclusion in the study.
2.1. Data and Assessments
Patient- and disease-related variables included biological sex, ethnicity, age at symptom onset, at diagnosis, and at treatment start. Follow-up intervals were captured. Disease-associated clinical manifestations and phenotypes were recorded. Laboratory parameters comprised inflammatory markers, including serum amyloid A (SAA) and C-reactive protein (CRP), as well as identified gene variants and their classification according to the American College of Medical Genetics and Genomics (ACMG) guidelines [
15]. Therapy data were documented, including medication type, dosage, and administration frequency.
The baseline visit was defined as the initial patient assessment conducted prior to or at initiation of therapy; the last follow-up visit was defined as the final patient assessment during the study period. Disease duration was defined as the interval from symptom onset to the last clinical visit, and treatment delay as the interval from symptom onset to initiation of effective anti-inflammatory therapy. For the analysis, both timeframes were dichotomized into ≤10 years and >10 years.
2.2. Concepts, Definitions, and Instruments
(1)
Disease activity was assessed using validated instruments, including the Physician Global Assessment (PGA) and the Patient/Parent Global Assessment (PPGA), with both quantifying disease activity along a 10 cm visual analog scale (VAS), where 0 indicated no disease activity and 10 represented maximal disease activity. Categories of clinical disease activity were determined by PGA on a 10 cm VAS and categorized as mild (<2), moderate (2–4), and high (>4), as described previously (REF). Complete remission was defined as PGA ≤ 2, with CRP ≤ 0.5 mg/dl and/or SAA ≤ 10 mg/L. Partial remission was PGA > 2 and ≤5, with CRP > 0.5 mg/dl but ≤3 mg/dl and/or SAA > 10 mg/L but ≤30 mg/L. Non-remission was defined as PGA > 5, with CRP > 3 mg/dl and/or SAA > 30 mg/L [
14].
(2)
Fatigue was evaluated and quantified using two approaches: (a) A 10 cm VAS embedded within the PedsQL Present Functioning Visual Analogue Scales questionnaire [
16], which comprises six parameters, including fatigue, and is routinely completed by patients prior to their outpatient visit. A score of 0 indicated no fatigue, whereas a score of 10 indicated maximal fatigue. Fatigue was considered present if the VAS score was ≥1. (b) The PedsQL Multidimensional Fatigue Scale (MFS) [
17], an 18-item questionnaire comprising three subscales—General Fatigue, Sleep/Rest Fatigue, and Cognitive Fatigue—scored on a 0–100 scale, with higher scores indicating lower fatigue and better functioning. For the statistical analyses, scores were reverse-transformed (100—original score) so that higher values consistently reflected greater fatigue burden. The instrument has been validated for both child self-report (ages 5–18 years) and parent proxy-report (ages 2–18 years) [
17].
(3)
Depressive symptoms were assessed using the Center for Epidemiologic Studies Depression Scale (CES-D) [
11] and its revised version, the CESD-R [
18]. Both are 20-item self-report questionnaires measuring depressive symptomatology in the general population. While the original CES-D employs a 0–3 scoring system, the CESD-R uses a 0–4 scale. In accordance with previously described and widely applied practice, responses scored as 4 on the CESD-R were recoded to 3 to facilitate comparability and simplify the assessment of clinical depression risk [
18]. A cut-off score of ≥16 was applied to define clinically relevant depressive symptoms [
19].
Disease activity (PGA/PPGA) and fatigue VAS from the PedsQL Present Functioning Scales were recorded at every visit. PedsQL-MFS and CES-D/CESD-R were obtained only at the last follow-up. Each patient, therefore, contributed multiple measurements for disease activity and fatigue VAS, and one measurement for PedsQL-MFS and depression.
2.3. Statistical Analysis
Continuous variables were presented as median (IQR) and categorical variables as counts and percentages. Group comparisons used the Mann–Whitney U test for continuous data and Chi-square or Fisher’s exact tests for categorical data. Paired baseline–last visit comparisons employed the Wilcoxon signed-rank test. Correlations were assessed using Spearman’s ρ for non-parametric and Pearson’s r for parametric data (|ρ|/|r| < 0.3 = weak, 0.3–0.5 = moderate, and >0.5 = strong). To identify independent determinants of fatigue, relevant variables including age group, pathogenicity of variants, disease duration, treatment delay, and depression risk were first tested in univariable analyses. Those showing significant associations were subsequently included in multivariable linear regression models. Analyses were conducted in IBM SPSS Statistics v28.0.1.1, with p < 0.05 considered statistically significant.
3. Study Endpoints
The primary endpoint was Overall Fatigue measured by the PedsQL-MFS at the last follow-up visit. Secondary endpoints, all assessed at the last follow-up visit, included the following: (1) PedsQL-MFS subdomain scores (General, Sleep/Rest, and Cognitive Fatigue), (2) fatigue assessed by VAS, and (3) depression scores measured by CES-D/CESD-R.
4. Results
4.1. Patient Characteristics
The cohort consisted of 66 individuals affected by auto-inflammatory diseases, with a balanced distribution regarding biological sex and age. CAPS and FMF were represented in similar proportions. Genetic testing revealed a heterogeneous spectrum of variants, including pathogenic mutations as well as variants of uncertain significance, while more than one-third of patients showed no identifiable variant. Most patients had a German background, with a smaller proportion of Turkish origin. An overview of the demographic and genetic characteristics is provided in
Table 1.
4.2. Clinical Presentation
Symptoms generally began in early childhood, and treatment was initiated across a wide age range. During long-term follow-up, all patients achieved complete remission at the last visit. Most remained in remission while receiving anti-inflammatory therapy, most commonly colchicine or IL-1 inhibitors. Treatment and follow-up characteristics are presented in
Table 1.
In FMF patients (n = 31), the most common findings were abdominal pain (97%) and febrile attacks (94%), followed by arthritis/arthralgia (48%), stress- or infection-triggered attacks (45%), myalgia (42%), headache (39%), irritability (36%), and lymphadenopathy (23%). All CAPS patients (n = 35) had flares triggered by stress, cold, or infection. Urticarial rash (89%), arthralgia (74%), headache and conjunctivitis (71%, respectively), and myalgia and irritability (66%, respectively) were frequently seen. Complications included hearing loss (40%), tinnitus (26%), amyloidosis and skeletal abnormalities (14%, respectively), and aseptic meningitis (6%).
4.3. Disease Activity
Baseline PGA and PPGA values were consistent with moderate disease activity and declined to zero at the final assessment, indicating complete disease inactivity. A statistically significant disparity in fatigue severity between CAPS and FMF was evident, as presented in
Table 2. Adults showed a trend toward higher baseline PGA scores compared with children (median PGA 5 (range 3–9) vs. 3 (range 2–9),
p = 0.08).
4.3.1. Fatigue
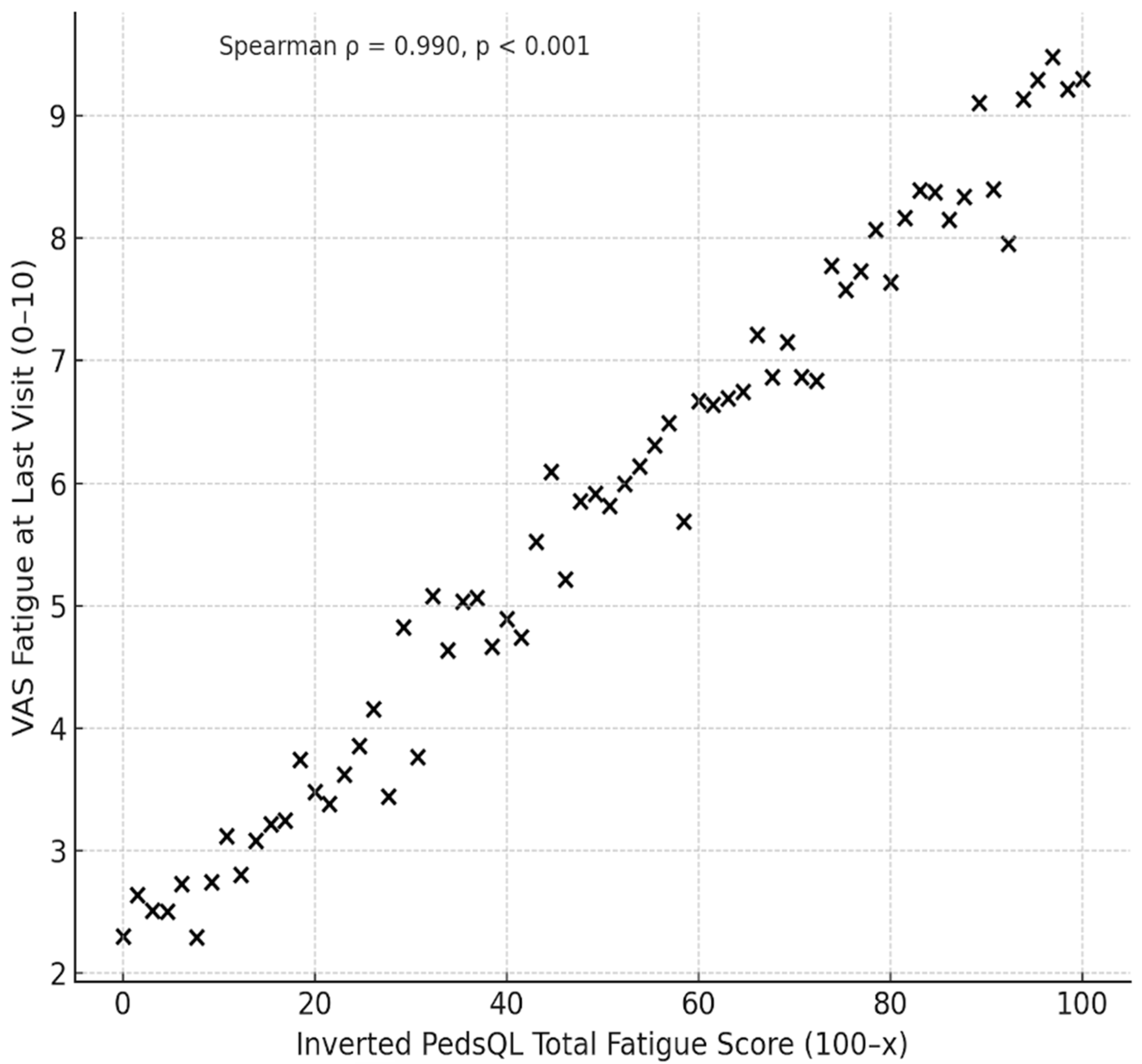
At baseline, Overall Fatigue levels were high and remained within the moderate-to-high range at the last visit despite well-controlled disease activity. Inverted PedsQL-MFS scores indicated substantial impairment across all fatigue domains, with values markedly higher than those of the general population (
Table 2). A significant correlation between the inverted PedsQL Overall Fatigue Score (100–x) and VAS Fatigue at the last visit was observed (Pearson’s r = 0.918,
p < 0.001; Spearman’s ρ = 0.942,
p < 0.001) (
Figure 1).
In the comparison of CAPS and FMF patients, significant differences were confined to fatigue-related measures. CAPS patients demonstrated greater Cognitive Fatigue and substantially higher baseline VAS Fatigue scores. No additional between-group differences reached statistical significance, as detailed in
Table 2.
4.3.2. Depression
Depressive symptoms, as measured by CES-D/CESD-R, were generally elevated across the cohort, indicating a notable psychological burden in patients with inactive auto-inflammatory disease. Although CAPS patients tended to report slightly higher levels of depressive symptoms compared with FMF patients, this difference did not reach statistical significance (
Table 2). Adults had slightly higher scores than children, with median values of 15 (0–42) versus 13 (0–45). This difference did not reach statistical significance (ρ = 0.244).
4.3.3. Risk Factors Associated with Increased Fatigue
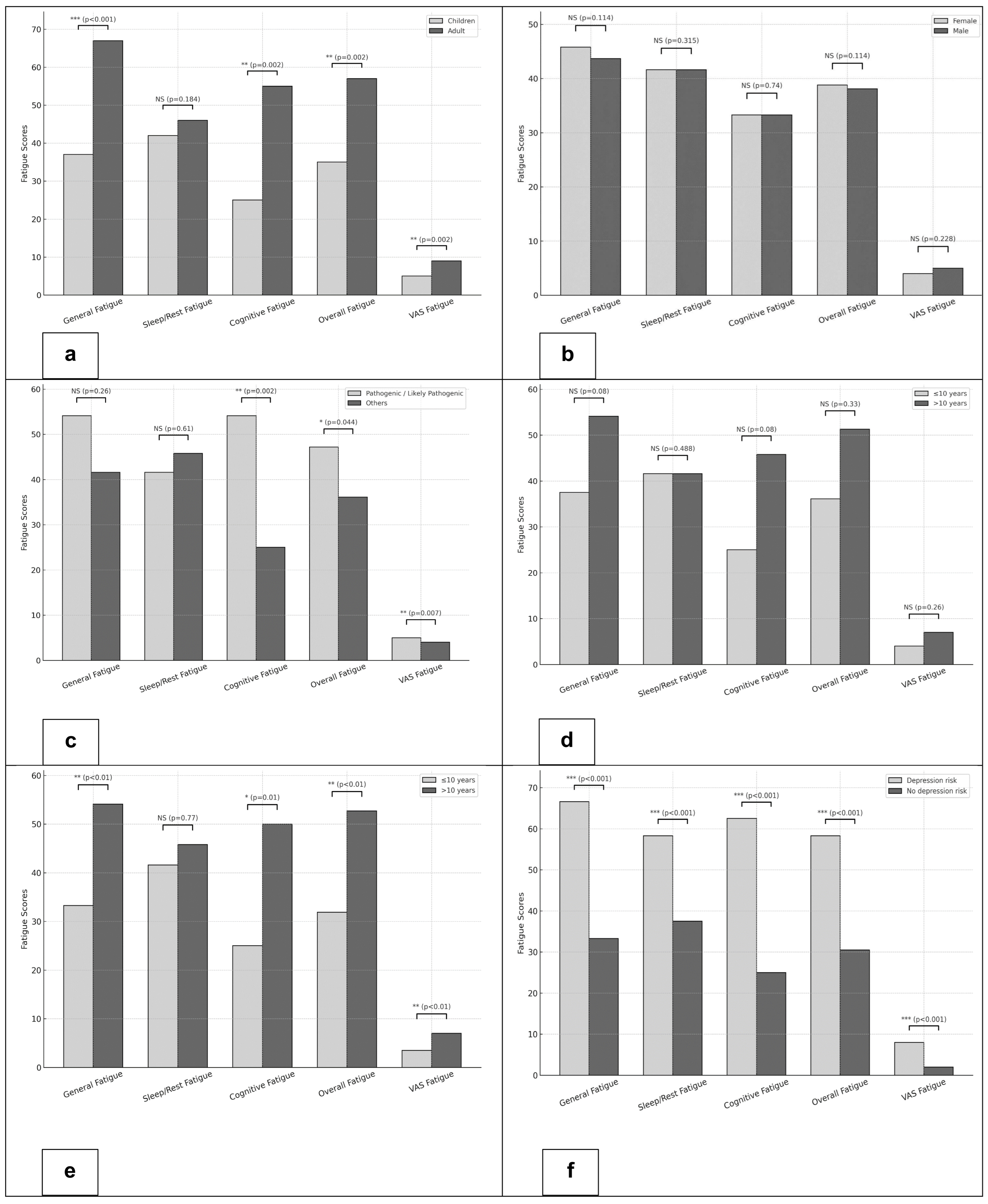
(1) Age: Adults reported significantly higher fatigue scores across multiple domains compared to children. Median inverted PedsQL-MFS scores were 66.6 vs. 37.5 for General Fatigue (
p < 0.001), 54.1 vs. 25.0 for Cognitive Fatigue (
p = 0.002), and 56.9 vs. 34.7 for Overall Fatigue (
p = 0.002). Similarly, the VAS Fatigue was significantly higher among adults (median 7 vs. 4;
p = 0.002) (
Figure 2a).
(2) Biological sex: No significant differences in fatigue were observed. Median inverted PedsQL-MFS scores for General, Sleep/Rest, Cognitive, and Overall Fatigue as well as fatigue VAS scores were comparable between females and males (
Figure 2b).
(3) Pathogenicity of the gene variant: Patients with pathogenic/likely pathogenic variants exhibited significantly higher levels of fatigue compared to those with other or no variants. Median inverted PedsQL-MFS scores were 54.1 vs. 25.0 for Cognitive Fatigue (
p = 0.002) and 47.2 vs. 36.1 for Overall Fatigue (
p = 0.044). VAS Fatigue scores were also higher in the pathogenic/likely pathogenic group (median 5 vs. 4;
p = 0.007) (
Figure 2c).
(4) Delay to effective treatment: Patients with a treatment delay longer than 10 years tended to report higher fatigue levels. This trend was observed across PedsQL-MFS domains, particularly General and Cognitive Fatigue, as well as in VAS Fatigue (
Figure 2d)
(5) Disease duration: Patients with a disease duration of more than 10 years reported significantly higher fatigue scores. Median inverted PedsQL-MFS scores were 54.1 vs. 33.3 for General Fatigue (
p < 0.01), 50.0 vs. 25.0 for Cognitive Fatigue (
p = 0.01), and 52.7 vs. 31.9 for Overall Fatigue (
p < 0.01). Correspondingly, the fatigue VAS scores were higher in the group with longer disease duration (median 7 vs. 3.5;
p < 0.01) (
Figure 2e).
(6) Depression: Overall, 27 of 66 patients (41%) met the threshold for high depression risk, defined as a CES-D/CESD-R score ≥ 16. Patients with a high depression risk had markedly higher fatigue scores across all domains of the PedsQL-MFS compared with those without an elevated depression risk. Median scores were 66.6 vs. 33.3 for General Fatigue, 58.3 vs. 37.5 for Sleep/Rest Fatigue, 62.5 vs. 25.0 for Cognitive Fatigue, and 58.3 vs. 30.5 for Overall Fatigue (all
p < 0.001). Consistently, VAS Fatigue scores were significantly higher in the high depression risk group (median 8 vs. 2;
p < 0.001) (
Figure 2f).
4.3.4. Risk Model for Fatigue in Inactive AID
Depression consistently emerged as the strongest independent predictor of fatigue in multivariable regression analyses accounting for age group (adult vs. child), pathogenicity of the gene variant, delay to effective treatment, and disease duration. Depression risk was significantly associated with higher fatigue burden across all PedsQL domains and VAS Fatigue (all
p < 0.001). Adult age was independently associated with greater Cognitive Fatigue (B = 15.7,
p = 0.039), and disease duration showed a trend towards higher Sleep/Rest and General Fatigue (
Table 3). The overall model explained 52% of the variance in fatigue (R
2 = 0.52).
5. Discussion
This study provided a systematic evaluation of fatigue and depression in patients with inactive auto-inflammatory diseases. Despite controlled disease activity, fatigue scores were significantly elevated, reflecting the ongoing burden. Importantly, disease-specific impacts on the distinct fatigue domains such as Cognitive Fatigue in CAPS patients were documented. The demonstrated strong correlation between VAS Fatigue and PedsQL-MFS highlights the clinical utility of both measures in capturing highly relevant patient-reported outcomes. Depression emerged as the strongest independent determinant of fatigue. Other putative risk factors such as age, disease duration, and pathogenic variants were found to be less relevant in well-controlled auto-inflammatory disease. These findings underscore the need for a precision health approach that addresses fatigue as a distinct and clinically relevant burden in auto-inflammatory diseases, beyond the control of inflammatory disease activity, to prevent overtreatment and ensure optimal patient care.
Fatigue persisted in auto-inflammatory diseases despite adequate disease control. Our findings demonstrate consistently higher levels of fatigue across all domains in inactive disease compared with the general population [
20]. Distinct disease-related fatigue patterns were identified. CAPS patients reported significantly higher Cognitive Fatigue than those with FMF, supporting the critical biological role of the central nervous system as a disease target in CAPS—including aseptic meningitis, brain atrophy, and sensorineural hearing loss [
2]—and contributing to attentional and memory-related impairment. Fatigue needs to be explored and addressed at all stages of disease including its distinct domains to enable targeted strategies through precision health approaches. Taken together, these findings highlight the importance of comprehensive diagnostic and therapeutic strategies to adequately address fatigue in auto-inflammatory diseases.
Depression emerged as the strongest independent determinant of fatigue in patients with inactive AID. Factors such as adult age, longer disease duration and pathogenic variants were also found to be associated with higher fatigue in univariable analyses; however, their explanatory power diminished in multivariable models. Meta-analyses in rheumatoid arthritis and systemic lupus erythematosus have demonstrated a high prevalence of depression and strong associations with greater fatigue, impaired quality of life, and reduced adherence to therapy [
7,
21,
22]. While an inflammatory etiology of depression has been proposed [
9], depression in inactive inflammation remains poorly understood. In pediatric cohorts, depression and fatigue often remain underdiagnosed; nevertheless, their presence strongly predicted impaired school attendance, reduced social participation, and adverse developmental outcomes [
8]. Our findings indicate that depressive symptoms constitute a major determinant of fatigue in auto-inflammatory diseases, independent of underlying inflammatory activity. This highlights the importance of integrating the assessment and management of modifiable psychological factors into routine clinical care. Multivariable modeling further suggests that, beyond depression, additional factors such as psychosocial stressors or sleep-related disturbances may also contribute to the Overall Fatigue burden.
There were several limitations to this study. The small sample size from a single center limits its generalizability. Socioeconomic factors and life circumstances, as well as their impact, were not fully captured; however, the data reflect a real-life cohort within a generally well-supported health and social care system in Germany. In addition, a contemporaneous healthy control group was lacking; however, large normative population data for the PedsQL-MFS [
20] provided validated comparators.
Given the substantial burden of fatigue and its close link to depressive symptoms, targeted and multidimensional management approaches are warranted. Early psychological assessment, evidence-based psychotherapeutic interventions, optimization of anti-inflammatory treatment, structured physical activity, and coordinated care between rheumatology and mental health services may help address both fatigue and depressive comorbidities. Routine screening and stepped-care strategies could further facilitate timely identification and individualized management in this population.
6. Conclusions
Fatigue is a key symptom of active and inactive auto-inflammatory disease, significantly contributing to its individual and societal burden. Depression was found to be the strongest risk factor for debilitating fatigue in patients with inactive disease. There is an urgent need for diagnostic and therapeutic precision health approaches for depression and fatigue when caring for children and adults living with auto-inflammatory diseases. These data emphasize the urgent need for systematic screening and integrated approaches targeting both inflammatory and psychological domains of disease.
Author Contributions
The authors contributed to the work as follows: Conceptualization: Y.S., Ö.S., S.M.B. and J.B.K.-D.; Methodology: Y.S., Ö.S., S.M.B. and J.B.K.-D.; Formal analysis: Y.S. and J.B.K.-D.; Writing—original draft preparation: Y.S.; Review and Editing: Ö.S., S.M.B. and J.B.K.-D. Each author made substantial contributions to the manuscript and approved the submitted version. All authors have read and agreed to the published version of the manuscript.
Funding
Infrastructural funding was provided by the University Hospital Tuebingen. No other funding was received for this study.
Institutional Review Board Statement
Ethics approval was obtained from the ethics committee of Karl Eberhard University Tuebingen (Project Number: 070/2024BO2; Approval Date: 26 June 2024).
Informed Consent Statement
All participants (or their legal guardians) provided written informed consent prior to inclusion in the study.
Data Availability Statement
Upon reasonable request and with obtained ethical approval, the dataset can be made available by the corresponding author.
Acknowledgments
We acknowledge support from the Open Access Publication Fund of the University of Tübingen.
Conflicts of Interest
J.B.K.-D. has received research grants and speaker’s fees from Novartis and SOBI. The other authors declare no competing interests.
References
- Gattorno, M.; Hofer, M.; Federici, S.; Vanoni, F.; Bovis, F.; Aksentijevich, I.; Anton, J.; Arostegui, J.I.; Barron, K.; Ben-Cherit, E.; et al. Classification criteria for autoinflammatory recurrent fevers. Ann. Rheum. Dis. 2019, 78, 1025–1032. [Google Scholar] [CrossRef]
- Welzel, T.; Kuemmerle-Deschner, J.B. Diagnosis and Management of the Cryopyrin-Associated Periodic Syndromes (CAPS): What Do We Know Today? J. Clin. Med. 2021, 10, 128. [Google Scholar] [CrossRef] [PubMed]
- Cardona Gloria, Y.; Weber, A.N.R. Inflammasome Activation in Human Macrophages: IL-1beta Cleavage Detection by Fully Automated Capillary-Based Immunoassay. Methods Mol. Biol. 2023, 2696, 239–256. [Google Scholar] [CrossRef]
- Ozen, S.; Demirkaya, E.; Erer, B.; Livneh, A.; Ben-Chetrit, E.; Giancane, G.; Ozdogan, H.; Abu, I.; Gattorno, M.; Hawkins, P.N.; et al. EULAR recommendations for the management of familial Mediterranean fever. Ann. Rheum. Dis. 2016, 75, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Kone-Paut, I.; Piram, M.; Benseler, S.; Kuemmerle-Deschner, J.B.; Jansson, A.; Rosner, I.; Tommasini, A.; Murias, S.; Karadag, O.; Levy, J.; et al. Use of the Auto-inflammatory Disease Activity Index to monitor disease activity in patients with colchicine-resistant Familial Mediterranean Fever, Mevalonate Kinase Deficiency, and TRAPS treated with canakinumab. Jt. Bone Spine 2022, 89, 105448. [Google Scholar] [CrossRef] [PubMed]
- Duruoz, M.T.; Unal, C.; Bingul, D.K.; Ulutatar, F. Fatigue in familial Mediterranean fever and its relations with other clinical parameters. Rheumatol. Int. 2018, 38, 75–81. [Google Scholar] [CrossRef]
- Gouda, W.; Mokhtar, M.; Elazab, S.A.; Alreefi, R.; Alomar, T.; Kushk, F.; Alahmadi, R.; Khalil, M.; Kamal, M. Sleep disorders in patients with rheumatoid arthritis: Association with quality of life, fatigue, depression levels, functional disability, disease duration, and activity: A multicentre cross-sectional study. J. Int. Med. Res. 2023, 51, 3000605231204477. [Google Scholar] [CrossRef]
- Satirer, Ö.; Satirer, Y.; Gellner, A.-K.; Benseler, S.M.; Kuemmerle-Deschner, J.B. Burden of fatigue in cryopyrin-associated periodic syndromes. EULAR Rheumatol. Open 2025, in press. [Google Scholar] [CrossRef]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef]
- Tsuboi, H.; Takakura, Y.; Tsujiguchi, H.; Miyagi, S.; Suzuki, K.; Nguyen, T.T.T.; Pham, K.O.; Shimizu, Y.; Kambayashi, Y.; Yoshida, N.; et al. Validation of the Japanese Version of the Center for Epidemiologic Studies Depression Scale-Revised: A Preliminary Analysis. Behav. Sci. 2021, 11, 107. [Google Scholar] [CrossRef]
- Siddaway, A.P.; Wood, A.M.; Taylor, P.J. The Center for Epidemiologic Studies-Depression (CES-D) scale measures a continuum from well-being to depression: Testing two key predictions of positive clinical psychology. J. Affect. Disord. 2017, 213, 180–186. [Google Scholar] [CrossRef]
- Kuemmerle-Deschner, J.B.; Ozen, S.; Tyrrell, P.N.; Kone-Paut, I.; Goldbach-Mansky, R.; Lachmann, H.; Blank, N.; Hoffman, H.M.; Weissbarth-Riedel, E.; Hugle, B.; et al. Diagnostic criteria for cryopyrin-associated periodic syndrome (CAPS). Ann. Rheum. Dis. 2017, 76, 942–947. [Google Scholar] [CrossRef] [PubMed]
- Faulstich, M.E.; Carey, M.P.; Ruggiero, L.; Enyart, P.; Gresham, F. Assessment of depression in childhood and adolescence: An evaluation of the Center for Epidemiological Studies Depression Scale for Children (CES-DC). Am. J. Psychiatry 1986, 143, 1024–1027. [Google Scholar] [CrossRef]
- Lachmann, H.J.; Kone-Paut, I.; Kuemmerle-Deschner, J.B.; Leslie, K.S.; Hachulla, E.; Quartier, P.; Gitton, X.; Widmer, A.; Patel, N.; Hawkins, P.N.; et al. Use of canakinumab in the cryopyrin-associated periodic syndrome. N. Engl. J. Med. 2009, 360, 2416–2425. [Google Scholar] [CrossRef]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424. [Google Scholar] [CrossRef]
- Sherman, S.A.; Eisen, S.; Burwinkle, T.M.; Varni, J.W. The PedsQL Present Functioning Visual Analogue Scales: Preliminary reliability and validity. Health Qual. Life Outcomes 2006, 4, 75. [Google Scholar] [CrossRef] [PubMed]
- Panepinto, J.A.; Torres, S.; Bendo, C.B.; McCavit, T.L.; Dinu, B.; Sherman-Bien, S.; Bemrich-Stolz, C.; Varni, J.W. PedsQL Multidimensional Fatigue Scale in sickle cell disease: Feasibility, reliability, and validity. Pediatr. Blood Cancer 2014, 61, 171–177. [Google Scholar] [CrossRef]
- Eaton, W.W.; Smith, C.; Ybarra, M.; Muntaner, C.; Tien, A. Center for Epidemiologic Studies Depression Scale: Review and Revision (CESD and CESD-R). In The Use of Psychological Testing for Treatment Planning and Outcomes Assessment: Instruments for Adults; Maruish, M.E., Ed.; Routledge: New York, NY, USA, 2004; Volume 3. [Google Scholar]
- Lewinsohn, P.M.; Seeley, J.R.; Roberts, R.E.; Allen, N.B. Center for Epidemiologic Studies Depression Scale (CES-D) as a screening instrument for depression among community-residing older adults. Psychol. Aging 1997, 12, 277–287. [Google Scholar] [CrossRef]
- Haverman, L.; Limperg, P.F.; van Oers, H.A.; van Rossum, M.A.; Maurice-Stam, H.; Grootenhuis, M.A. Psychometric properties and Dutch norm data of the PedsQL Multidimensional Fatigue Scale for Young Adults. Qual. Life Res. 2014, 23, 2841–2847. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, R.; Bernardes, M.; Terroso, G.; de Sousa, M.; Figueiredo-Braga, M. Silent Burdens in Disease: Fatigue and Depression in SLE. Autoimmune Dis. 2014, 2014, 790724. [Google Scholar] [CrossRef]
- Kawada, T. The prevalence of depression in rheumatoid arthritis: A systematic review and meta-analysis. Rheumatology 2014, 53, 578. [Google Scholar] [CrossRef] [PubMed]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).