1. Introduction
Inguinal hernia repair is among the most frequently performed surgical procedures worldwide, with millions of cases annually. The widespread adoption of mesh-based techniques has led to a substantial reduction in recurrence rates—estimated at between 50% and 75% [
1]. Consequently, clinical attention has shifted from recurrence prevention to the management of postoperative complications, particularly chronic groin pain.
Several surgical techniques are currently employed for inguinal hernia repair. Open procedures, such as Lichtenstein, Shouldice, and Bassini, remain widely used but carry risks of nerve injury and mesh-related inflammation [
2]. Minimally invasive approaches, including Trans Abdominal Pre Peritoneal (TAPP) and Totally Extra Peritoneal (TEP) laparoscopy, are associated with faster recovery and, in most series, lower rates of chronic pain, although specific complications may occur [
3,
4]. Robotic-assisted repair, a more recent innovation, offers enhanced precision and ergonomics, but current evidence remains limited [
5,
6,
7]. Each technique has its own risk profile, and postoperative pain has emerged as one of the most relevant complications, sometimes persisting beyond the expected recovery period.
While post-herniorrhaphy discomfort typically resolves within two months [
2], a subset of patients continues to experience persistent pain beyond this period. This condition is known as chronic postoperative inguinal pain (CPIP), also referred to in pain literature as Persistent Postsurgical Pain (PPSP) after inguinal hernia repair [
3,
4]. Originally defined by the International Association for the Study of Pain (IASP) in 1986 as pain persisting for more than three months after surgery, CPIP was later refined by the HerniaSurge Group [2018] [
7]. The updated definition includes pain that is moderate to severe, lasts beyond three months, and interferes with daily activities such as movement, sleep, or social interaction [
5,
6,
7]. However, due to ongoing mesh-related inflammation beyond the three-month period, some experts advocate extending the diagnostic threshold to six months [
8].
Recent studies have further expanded the understanding of CPIP, highlighting the importance of individualized surgical strategies [
9]. Huy et al. (2025) demonstrated that tailored interventions, including neurectomy and mesh removal, can significantly improve outcomes in patients with refractory CPIP [
9]. Kwee et al. (2024) emphasized the role of selective and triple neurectomy in neuropathic CPIP, reporting pain relief in over two-thirds of treated patients [
10]. Moreover, Reichert et al. (2025) investigated the inflammatory profiles of different mesh types, suggesting that material selection may influence long-term pain outcomes [
11]. Li et al. (2025) compared robotic and laparoscopic techniques, showing that robotic-assisted repair may reduce reoperation rates and enhance postoperative recovery, although further validation is needed [
12].
Despite consensus definitions, the diagnostic threshold remains debated, particularly in cases of prolonged mesh-related inflammation [
13]. A review of the literature offers a comprehensive overview of CPIP following inguinal hernioplasty, across various studies and geographic regions (
Table 1), with incidence rates ranging from 6% to 64.3% across different countries and surgical approaches [
13,
14,
15,
16,
17,
18,
19,
20,
21,
22,
23,
24,
25,
26,
27,
28,
29,
30,
31,
32]. Importantly, the risk of chronic postoperative inguinal pain (CPIP) is not uniform across all hernia repair techniques. Open mesh-based procedures, such as Lichtenstein or Shouldice, tend to show higher CPIP rates due to increased nerve exposure and mesh fixation [
33]. In contrast, laparoscopic approaches like TEP and TAPP generally yield lower pain incidence, although exceptions exist and technique-specific complications may arise [
34]. Robotic-assisted repairs, while promising, remain under investigated [
35]. Therefore, any analysis of CPIP must be contextualized within the specific surgical technique employed, to avoid misleading associations and ensure accurate interpretation of outcomes [
36].
2. Methodology
This narrative review was conducted through a comprehensive literature search using PubMed, Scopus, and Web of Science databases. Keywords included “chronic postoperative inguinal pain”, “CPIP”, “Persistent Postsurgical Pain”, “hernia repair”, “neuropathic pain”, “nociceptive pain”, and “neurectomy”. Studies published between January 2000 and April 2025 were considered.
Inclusion criteria were the following: sample size ≥100 patients, clear subclassification of pain types, relevance to surgical technique and postoperative pain outcomes, and full-text availability in English. Exclusion criteria included case reports, editorials, and studies lacking pain classification or follow-up data. Reference lists of selected articles were manually screened to identify additional relevant studies.
3. Epidemiological Landscape
The international literature on CPIP reveals a complex and heterogeneous picture. Incidence rates vary widely—from 6% in Forester’s UK-based study (2021) to an exceptional 64.3% in Niebuhr’s German cohort (2018)—highlighting the multifactorial nature of CPIP [
15,
22]. This variability reflects a dynamic interplay of factors, including surgical technique, follow-up duration, patient selection criteria, regional healthcare practices, and methodological consistency.
A clear trend emerges regarding follow-up duration. Studies with shorter postoperative evaluations [3–6 months], such as Lo (2021: 8.7%) and Forester [2021: 6%], tend to report lower CPIP rates [
14,
15]. In contrast, studies with extended follow-up periods [≥12 months], including Lundström (2018: 15.2%) and Jeroukhimov (2014: 32.8%), consistently report higher pain prevalence [
20,
29]. This pattern suggests that early assessments may underestimate the true burden of chronic pain, reinforcing the importance of long-term surveillance in clinical research. Recent evidence supports this view. Gutlic et al. (2024) conducted an 8-year follow-up comparing TEP and Lichtenstein repairs, showing that CPIP rates remain stable over time and may persist well beyond the first postoperative year [
40]. Narita et al. (2024) reported that 88% of patients undergoing surgery for CPIP experienced excellent or good outcomes after a median of 24.6 months, reinforcing the need for extended monitoring and individualized treatment strategies [
41]. Chu et al. (2024) performed a meta-analysis of 29,466 patients and found a pooled CPIP incidence of 17.01%, with higher rates in Europe (18.65%) compared to Asia (14.70%) and North America (6.04%), highlighting geographic variability and the importance of regional surveillance protocols [
42]. These findings underscore the need for standardized long-term follow-up protocols to accurately assess CPIP prevalence and guide postoperative care.
4. Surgical Technique and Geographic Variation
Surgical technique plays a pivotal role in CPIP outcomes. Laparoscopic repairs generally yield lower CPIP rates compared to open approaches. For example, Lo (2021) and Gutlic (2019) report rates below 9% in laparoscopic cohorts, supporting the hypothesis that minimally invasive techniques may reduce nerve trauma and mesh-related inflammation [
14,
27]. However, this advantage is not absolute. Niebuhr’s 2018 study presents a striking anomaly: a CPIP rate of 64.3%, despite exclusive use of laparoscopy, with a notably large sample size of 20,004 patients [
22]. This outlier raises methodological concerns, and suggests the influence of confounding variables such as non-standardized surgical protocols, selection bias, or inconsistencies in pain assessment. It serves as a reminder that no technique is inherently superior without rigorous execution and individualized patient care.
A recent study by Liu et al. (2025) further supports this perspective, reporting significantly higher chronic pain rates in the open repair group compared to the laparoscopic group (4.8% vs. 1.88%,
p < 0.05) [
43]. Similarly, in a prospective cohort by Thulasilingam et al. (2023), patients undergoing laparoscopic TAPP repair experienced significantly lower pain scores and faster return to activity, compared to those receiving open mesh repairs, reinforcing the role of technique in postoperative recovery [
44]. A randomized controlled trial by Ulutaş and Yilmaz (2025) comparing laparoscopic TEP and open Lichtenstein repair in elderly patients found substantially lower chronic pain rates in the laparoscopic group (1.7% vs. 10%,
p = 0.05), along with faster recovery and reduced analgesic use [
45].
The interpretation of CPIP incidence must be contextualized within the specific surgical technique employed. As shown in
Table 1, studies reporting CPIP outcomes vary in their use of open, laparoscopic, or mixed approaches, each of which significantly influences postoperative pain profiles. Open mesh-based repairs, such as the Lichtenstein technique, are generally associated with higher rates of chronic pain, due to increased nerve exposure and fixation methods. In contrast, laparoscopic techniques like TEP and TAPP tend to yield lower CPIP rates, likely due to reduced tissue dissection and minimized nerve handling. Nevertheless, exceptions exist—such as the elevated pain rates observed in certain laparoscopic cohorts—which underscore the importance of surgical precision, adherence to standardized protocols, and individualized patient care. Therefore, any analysis of CPIP must carefully distinguish between surgical modalities, to avoid misleading associations and ensure accurate clinical interpretation.
Geographic variation further complicates the CPIP landscape. European studies often report moderate-to-high CPIP rates—Fränneby (2006, Sweden): 31% [
38], Nienhuijs (2005, Netherlands): 43.3% [
39] suggest chronic postoperative pain may affect up to 30% of patients across various procedures, highlighting the multifactorial nature of pain outcomes [
3]. In contrast, Asian studies such as Lo (2021, Taiwan) [
14] and Min (2020, China: 26.8%) [
16] tend to report lower or intermediate rates, raising questions about regional differences in clinical practice, mesh selection, perioperative care, and even genetic predisposition to chronic pain. A recent meta-analysis by Chu et al. (2024) confirmed this trend, showing CPIP rates of 18.65% in Europe, 14.70% in Asia, and 6.04% in North America across 29,466 patients [
42].
These discrepancies indicate that CPIP is not solely a surgical issue, but also reflects broader systemic and cultural determinants. Interestingly, surgical expertise does not appear to significantly influence CPIP rates. As demonstrated by de la Croix (2025), the incidence was nearly identical between patients operated on by specialist surgeons and those treated by surgical residents (15.4% vs. 15.5%) [
46].
This finding aligns with the results of Lange et al. (2016), who found no statistically significant difference in CPIP rates between expert and non-expert surgeons in Lichtenstein repairs, despite a trend toward better outcomes in high-volume operators [
47].
5. Strengths and Limitations of the Literature
From a critical standpoint, the dataset presents several strengths. It encompasses a wide range of countries, surgical techniques, and follow-up durations, offering a broad overview of CPIP across diverse clinical settings. The inclusion of large patient samples—such as Niebuhr’s 2018 cohort of over 20,000 individuals—enhances the statistical reliability of reported outcomes [
22]. Recent multicenter studies, such as Liu et al. (2025) and Chu et al. (2024), have further expanded the evidence base by incorporating diverse geographic cohorts and standardized pain metrics, improving external validity and comparative potential [
42,
43]. However, limitations are equally evident. The heterogeneity in study designs—particularly regarding follow-up intervals and CPIP definitions—complicates direct comparisons. The presence of “NR” (Not Reported) values in several studies further obscures the timeline of pain evaluation, while outlier data—such as the unexpectedly high CPIP rate in Niebuhr’s laparoscopic series—requires deeper methodological scrutiny to identify potential sources of bias or inconsistency.
Clinically, these findings underscore the urgent need for standardized pain assessment tools to improve data consistency and comparability. Instruments such as the DN4 questionnaire should be routinely employed to distinguish between neuropathic and nociceptive pain, thereby refining diagnostic accuracy. A 2025 systematic review by Madani et al. confirmed the DN4’s strong psychometric properties, with pooled sensitivity of 82% and specificity of 76.1%, supporting its use in chronic pain cohorts, including CPIP [
48]. Hardt et al. (2023) validated the English 7-item DN4 version, demonstrating excellent short- and long-term test–retest reliability (weighted kappa > 0.85), reinforcing its applicability in longitudinal CPIP studies [
49]. Although laparoscopic approaches generally appear to yield better outcomes in terms of CPIP reduction, the presence of elevated pain rates in certain laparoscopic cohorts highlights the need for further investigation into surgical technique, perioperative management, and patient-specific variables. Moreover, regional differences in CPIP prevalence should be explored in greater depth, to develop tailored pain prevention strategies that align with local healthcare infrastructure and demographic profiles.
Chu et al. (2024) highlighted significant geographic variation, with CPIP rates of 18.65% in Europe, 14.70% in Asia, and 6.04% in North America, suggesting that cultural, systemic, and clinical factors play a substantial role in pain outcomes [
42].
Future research should prioritize harmonization of study designs and the adoption of validated pain assessment tools to enable meaningful comparisons and guide evidence-based interventions. Routine use of instruments such as the DN4 questionnaire should become standard practice in both research and clinical settings.
6. Neuropathic vs. Nociceptive CPIP: Diagnostic Challenges and Clinical Implications
Understanding the nature of CPIP is essential for effective treatment. Differentiating between neuropathic and nociceptive pain is not merely academic—it directly influences therapeutic choices. In routine clinical practice, distinguishing between neuropathic and non-neuropathic CPIP remains a significant challenge. Although the underlying mechanisms differ, their clinical manifestations often overlap, making accurate classification difficult [
10,
50]. A re-analysis of selected studies reporting CPIP incidence reveals that only a limited number of authors have attempted to differentiate between neuropathic and nociceptive groin pain—highlighting a significant gap in diagnostic precision (
Table 2).
Recent evidence confirms this diagnostic gap. Kwee et al. (2024) conducted a systematic review of surgical treatments for neuropathic CPIP, and found that only 10 studies explicitly classified pain type prior to intervention, underscoring the lack of standardized diagnostic frameworks [
10]. Hardt et al. (2023) validated the English version of the DN4 questionnaire, showing excellent reliability (weighted kappa > 0.85), and recommended its routine use in CPIP studies to distinguish neuropathic components [
49]. Nijs et al. (2023) emphasized the importance of pain phenotyping in chronic groin pain, proposing a stratified approach based on predominant pain type—nociceptive, neuropathic, or nociplastic—to guide individualized treatment [
51].
Most of these studies focus on open surgical techniques, with only Loos (2007) including laparoscopic procedures [
37]. The inclusion of laparoscopy in this cohort may influence CPIP outcomes, potentially shifting the balance between neuropathic and nociceptive pain.
The absence of consistent pain subclassification across studies limits the development of targeted treatment strategies. A standardized diagnostic framework is urgently needed, to improve patient outcomes.
Van Veenendaal et al. (2023) highlighted the fact that most non-surgical CPIP studies fail to report pain phenotype, making it difficult to assess treatment efficacy and select appropriate interventions [
52]. Emerging approaches such as targeted muscle reinnervation (TMR) show promise in neuropathic CPIP, but require precise preoperative pain classification, to optimize outcomes [
10].
7. Neuropathic and Nociceptive Pain Profiles
Neuropathic CPIP rates vary widely. Ergönenç (2017) reports the highest proportion [73.7%], suggesting a predominant neuropathic component likely associated with limited nerve preservation during open repair [
23]. Bande (2020) and Nienhuijs (2005) report similar rates (38.5% and 40.3%, respectively), possibly reflecting comparable surgical techniques or perioperative protocols [
13,
39]. Loos (2007), with a rate of 46.5%, occupies a mid-range position—potentially influenced by the inclusion of laparoscopic procedures, which may mitigate nerve trauma [
37].
Recent evidence from Kwee et al. (2024) confirms the predominance of neuropathic pain in CPIP, with pooled estimates showing neuropathic features in up to 68% of patients undergoing surgical treatment for refractory groin pain [
10].
Charitakis et al. (2025) reported that triple neurectomy yielded pain improvement in 98.2% of neuropathic CPIP cases, reinforcing the importance of accurate pain classification in surgical decision-making [
53].
Conversely, non-neuropathic CPIP rates show an inverse trend. Ergönenç (2017) reports the lowest rate (26.3%), while Bande (2020) and Nienhuijs (2005) report higher rates (61.5% and 59.7%) [
13,
23,
39]. These discrepancies may be attributed to variations in pain classification criteria, diagnostic methodology, or postoperative nerve handling. Van Veenendaal et al. (2023) emphasized that most non-surgical CPIP studies fail to report pain phenotype, making it difficult to assess treatment efficacy and select appropriate interventions [
52]. The relatively balanced distribution in Loos (2007) further supports the hypothesis that surgical approach—particularly laparoscopy—may influence the type of pain experienced [
37].
These findings are consistent with the broader framework of Persistent Postsurgical Pain (PPSP), which integrates both neuropathic and nociceptive components and is increasingly recognized in the pain literature as a distinct clinical entity.
Fuller et al. (2023) described PPSP as affecting 10–50% of surgical patients, with central sensitization and neuroimmune mechanisms contributing to chronicity, especially in procedures involving nerve-rich regions like the groin [
54]. Moka et al. (2024) highlighted the need for transitional pain services and early phenotyping to prevent PPSP evolution, particularly in high-risk surgeries such as hernia repair [
55].
8. Limitations and Methodological Considerations
Several limitations must be acknowledged. Sample sizes vary significantly—from 61 patients in Ergönenç (2017) to 239 in Bande (2020)—which may affect the statistical reliability and generalizability of findings [
13,
23]. Latenstein et al. (2021) highlighted substantial hospital-level variation in inguinal hernia repair practices, suggesting that surgical outcomes may be influenced by institutional preferences rather than standardized evidence-based protocols [
56].
Additionally, the lack of precise differentiation between open techniques (e.g., Lichtenstein vs. Shouldice), as well as between minimally invasive approaches such as TAPP (Transabdominal Preperitoneal) and TEP (Totally Extraperitoneal), may obscure subtle trends and hinder direct comparisons across surgical modalities [
43].
Henriksen et al. (2024) emphasized the fact that laparoscopic techniques, while technically demanding, are associated with lower surgical-site infection rates and improved postoperative quality of life, though they require greater surgical expertise [
57]. Crepaz et al. (2025), in the ACTIVE study, demonstrated that laparoscopic approaches in emergency settings resulted in fewer complications and shorter hospital stays, compared to open surgery [
58].
Inconsistencies in pain classification—particularly in distinguishing neuropathic from nociceptive pain—further limit cross-study comparability.
Pedersen et al. (2021) proposed a simplified clinical algorithm for diagnosing and surgically treating chronic post-herniorrhaphy pain, incorporating tools such as PainDETECT and Quantitative Sensory Testing (QST) [
59]. Van Veenendaal et al. (2023) noted that most non-surgical CPIP studies fail to report pain phenotype, limiting the ability to assess treatment efficacy and select appropriate interventions [
52].
From a clinical standpoint, the high prevalence of neuropathic pain in Ergönenç (2017) underscores the importance of meticulous nerve identification and preservation during hernia repair [
23]. The potential protective effect of laparoscopy, as suggested by Loos (2007), merits further investigation, to clarify whether minimally invasive techniques reduce the risk of neuropathic complications [
37].
Sun et al. (2025) found that laparoscopic obturator hernia repair significantly reduced systemic inflammation and hospital stay duration compared to open surgery, suggesting a broader benefit in postoperative pain outcomes [
60].
Ultimately, pain management strategies should be tailored to individual patient risk profiles, with careful consideration of surgical technique, intraoperative nerve handling, and structured postoperative monitoring.
Jensen et al. (2025) initiated the STRONG-Hernia trial, evaluating the impact of personalized prehabilitation programs on postoperative pain and complications in patients with modifiable risk factors [
61]. Alzatari et al. (2024) demonstrated that non-opioid analgesic regimens were non-inferior to opioid-based protocols in managing post-herniorrhaphy pain, supporting a safer multimodal approach [
62].
9. Toward a Structured Diagnostic Approach
These findings offer valuable insights into the relative prevalence of neuropathic pain among CPIP cases. Notably, Ergönenç (2017) reported a striking 73.7% incidence of neuropathic CPIP in an open repair cohort, suggesting nerve injury as a predominant mechanism in certain surgical contexts [
23]. Other studies show a more balanced distribution, indicating that both neuropathic and nociceptive pathways contribute meaningfully to postoperative pain.
Recent evidence confirms this dual-pathway model. Charitakis et al. (2025) found that triple neurectomy led to pain improvement in over 98% of patients with neuropathic CPIP, while double neurectomy offered complete relief in 80% of cases, albeit with a higher complication rate [
53]. Kwee et al. (2024) reported that targeted muscle reinnervation yielded the highest success rate among surgical techniques for refractory neuropathic CPIP, with 88% of patients experiencing significant pain relief [
10].
Despite these observations, the limited number of studies performing this subclassification highlights a critical gap in the literature
—Van Veenendaal et al. (2023) emphasized that most non-surgical CPIP studies fail to report pain phenotype, limiting treatment personalization and outcome assessment [
52]. Wang et al. (2023) demonstrated that CXCL13/CXCR5 signaling contributes to mechanical allodynia and neuroinflammation in CPIP models, suggesting a molecular basis for nociceptive pain persistence [
63].
In the absence of consistent use of validated diagnostic tools and standardized definitions, the true burden of neuropathic CPIP remains difficult to quantify. Further research is essential, to elucidate risk factors, refine diagnostic criteria, and optimize surgical techniques, to minimize both forms of chronic pain.
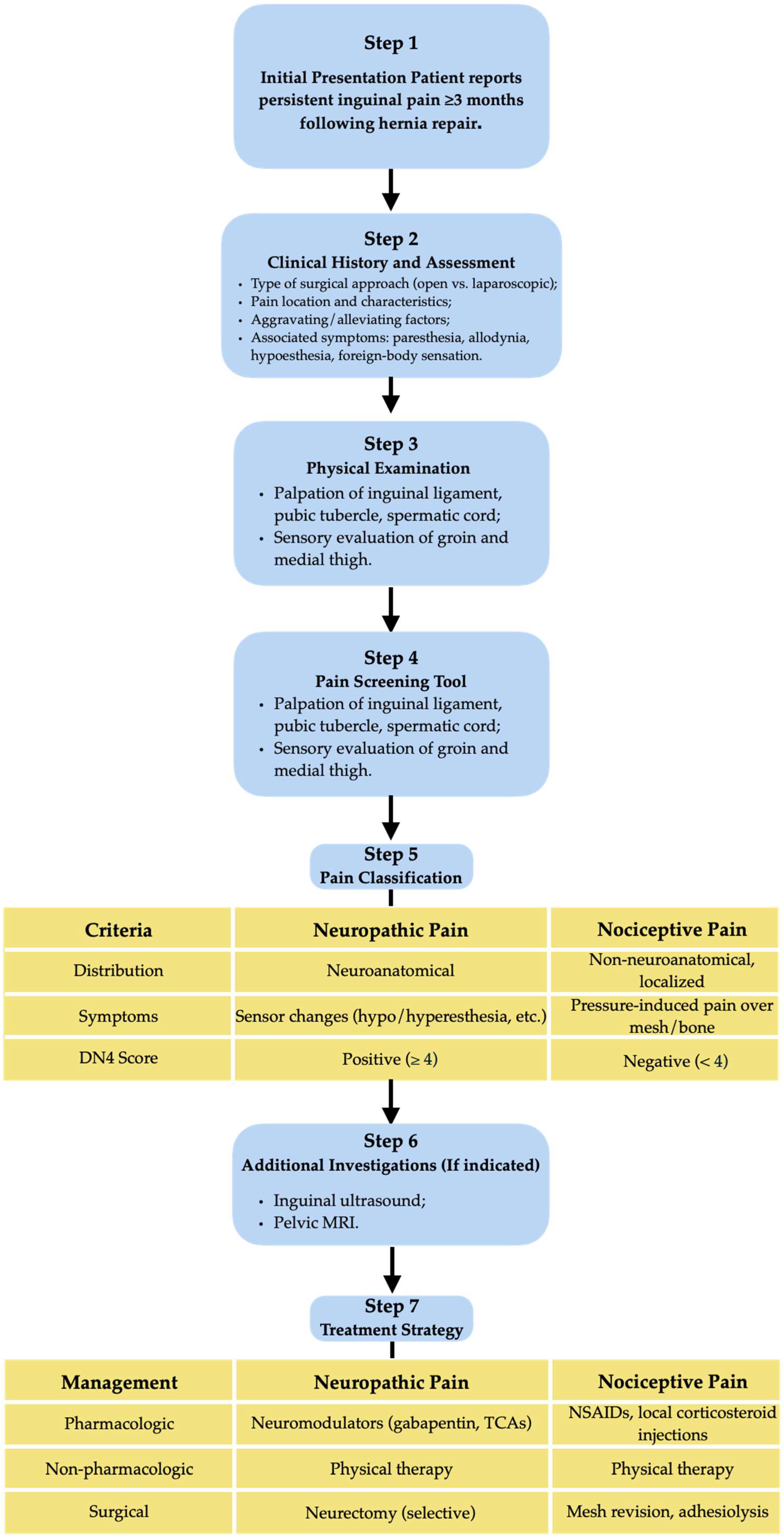
Given the diagnostic complexity and multifactorial nature of CPIP, a structured clinical algorithm may assist surgeons and pain specialists in navigating evaluation and treatment. Pedersen et al. (2021) proposed a simplified algorithm integrating PainDETECT and Quantitative Sensory Testing (QST) to distinguish pain types and guide surgical decision-making [
59]. Pawlak et al. (2022) advocated for multidisciplinary team (MDT) clinics to personalize CPIP management, especially in cases with overlapping pain mechanisms [
64]. Such a pathway should integrate current evidence and clinical reasoning to distinguish between pain types, guide appropriate investigations, and tailor management strategies to individual patient profiles. This approach emphasizes early recognition, standardized assessment tools, and a stepwise therapeutic plan aimed at minimizing long-term morbidity (
Figure 1).
Validation of this algorithm through prospective studies and its incorporation into clinical guidelines could significantly enhance CPIP management and reduce long-term disability.
10. Neurectomy in CPIP Management
Neurectomy—particularly during open inguinal hernia repair—has been proposed as both a preventive and therapeutic strategy for chronic postoperative inguinal pain (CPIP). Selective neurectomy involves intentional transection of one or more inguinal nerves (e.g., ilioinguinal, iliohypogastric, or genitofemoral) to mitigate the risk of postoperative neuropathic pain.
The technique of triple neurectomy, although more aggressive, has shown promising results in specific patient cohorts, with significant reductions in pain scores, and improvements in quality of life. In a multicenter study by Charitakis et al., 98.2% of patients undergoing triple neurectomy reported substantial clinical improvement [
53].
Kwee et al. (2024) confirmed that targeted muscle reinnervation yielded the highest success rate among surgical techniques for refractory neuropathic CPIP, with 88% of patients reporting significant pain relief [
10]. Moore et al. (2025) demonstrated that retroperitoneal laparoscopic triple neurectomy led to durable pain reduction and improved sensory thresholds over a 3-year follow-up, supporting its long-term efficacy in selected patients [
65].
However, the procedure remains controversial. While some studies report favorable outcomes, others highlight potential complications such as sensory loss, neuroma formation, and chronic dysesthesia [
10]. Cirocchi et al. conducted a meta-analysis of 16 RCTs involving 1550 patients, and found a significant reduction in groin pain at 6 months in the neurectomy group, but no clear long-term advantage over nerve preservation [
66]. This aligns with findings from Singh et al. (2024), who also noted short-term benefits but questioned the durability of prophylactic neurectomy outcomes [
67].
The decision to perform neurectomy should be individualized, based on intraoperative findings (e.g., nerve entrapment or inflammation), patient risk factors, and surgeon expertise. Recently, Messias et al. proposed ten recommendations to optimize outcomes in Lichtenstein hernia repair, including pragmatic neurectomy as a strategy to reduce chronic postoperative pain in selected patients [
68].
Recent guidelines suggest that prophylactic neurectomy may be considered in high-risk patients undergoing open repair, particularly when nerve preservation is not technically feasible [
6,
7,
69]. Conversely, therapeutic neurectomy should be reserved for patients with refractory neuropathic CPIP unresponsive to conservative measures [
70].
Minimally invasive approaches such as laparoscopic triple neurectomy are emerging as viable options. A case report by Castillo et al. documented complete pain resolution following laparoscopic triple neurectomy, opening new avenues for CPIP treatment [
71].
Further research is needed to standardize indications, refine surgical techniques, and evaluate long-term outcomes of neurectomy in CPIP management.
11. Conclusions
Chronic postoperative inguinal pain (CPIP) remains a clinically significant and multifactorial complication of hernia surgery. The available evidence highlights substantial heterogeneity in reported incidence, largely attributable to variations in diagnostic criteria, follow-up protocols, and methodological design. Despite these inconsistencies, a consistent finding is that CPIP often encompasses both neuropathic and nociceptive components, underscoring the importance of accurate subclassification in clinical assessment. Minimally invasive approaches are generally associated with lower pain rates, although this advantage is not universal and must be interpreted with caution. What clearly emerges from the literature is the urgent need for standardized diagnostic definitions, validated assessment tools, and long-term prospective studies. These elements are essential to enable reliable comparisons and to support the development of tailored management strategies. Advancing in this direction is crucial to reduce the burden of CPIP and improve long-term outcomes and quality of life for patients undergoing inguinal hernia repair.
Author Contributions
Conceptualization, R.C. and P.B.; methodology, R.C. and B.C.; validation, G.C., F.B. and M.L.; investigation, P.B., P.F. and R.C.; resources, S.L.; data curation, R.C. and M.C.R.; writing—original draft preparation, R.C. and P.B.; writing—review and editing, L.T.; visualization, P.F. and L.T.; supervision, R.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CPIP | Chronic postoperative inguinal pain |
| IASP | International Association for the Study of Pain |
| NR | Not Reported |
| DN4 | Douleur Neuropathique 4 |
| TAPP | Transabdominal Preperitoneal |
| TEP | Totally Extraperitoneal |
| CI | Confidence Interval |
| PMID | PubMed Identifier |
References
- Scott, N.W.; McCormack, K.; Graham, P.; Go, P.M.; Ross, S.J.; Grant, A.M. Open mesh versus non-mesh for repair of femoral and inguinal hernia. Cochrane Database Syst. Rev. 2002, 4, CD002197. [Google Scholar]
- Bay-Nielsen, M.; Perkins, F.M.; Kehlet, H.; Danish Hernia Database. Pain and functional impairment 1 year after inguinal herniorrhaphy: A nationwide questionnaire study. Ann. Surg. 2001, 233, 1–7. [Google Scholar] [CrossRef]
- Poobalan, A.S.; Bruce, J.; Smith, W.C.S.; King, P.M.; Krukowski, Z.H.; Chambers, W.A. A review of chronic pain after inguinal herniorrhaphy. Clin. J. Pain 2003, 19, 48–54. [Google Scholar] [CrossRef]
- Chapman, C.R.; Vierck, C.J. The Transition of Acute Postoperative Pain to Chronic Pain: An Integrative Overview of Research on Mechanisms. J. Pain 2017, 18, 359.e1–359.e38. [Google Scholar] [CrossRef]
- Classification of chronic pain. Descriptions of chronic pain syndromes and definitions of pain terms. Prepared by the International Association for the Study of Pain, Subcommittee on Taxonomy. Pain Suppl. 1986, 3, S1–S226.
- Stabilini, C.; van Veenendaal, N.; Aasvang, E.; Agresta, F.; Aufenacker, T.; Berrevoet, F.; Burgmans, I.; Chen, D.; de Beaux, A.; East, B.; et al. Update of the international HerniaSurge guidelines for groin hernia management. BJS Open 2023, 7, zrad080. [Google Scholar] [CrossRef]
- HerniaSurge Group. International guidelines for groin hernia management. Hernia 2018, 22, 1–165. [Google Scholar] [CrossRef] [PubMed]
- Faessen, J.L.; Stoot, J.H.M.B.; van Vugt, R. Safety and efficacy in inguinal hernia repair: A retrospective study comparing TREPP, TEP and Lichtenstein (SETTLE). Hernia 2021, 25, 1309–1315. [Google Scholar] [CrossRef]
- Huy, T.C.; Lu, Y.; Weitzner, Z.; Lau, S.W.; DeJesus, M.S.; Hu, T.; MacQueen, I.T.; Chen, D.C. Reoperation for Chronic Postoperative Inguinal Pain. JAMA Surg. 2025, e252502. [Google Scholar] [CrossRef] [PubMed]
- Kwee, E.; Langeveld, M.; Duraku, L.S.; Hundepool, C.A.; Zuidam, M. Surgical Treatment of Neuropathic Chronic Postherniorrhaphy Inguinal Pain: A Systematic Review and Meta-Analysis. J. Clin. Med. 2024, 13, 2812. [Google Scholar] [CrossRef]
- Reichert, M.; Massambo, B.; Amati, A.L.; Grau, V.; Richter, K.; Hecker, A. Acute inflammation triggered by two lightweight hernia meshes: A comparative in vitro and retrospective cohort study. Hernia 2025, 29, 205. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, S.; Wei, L.; Li, H. Efficacy and safety of robot-assisted versus endo-laparoscopic ventral hernia repair: A meta-analysis of randomized controlled trials. BMC Surg. 2025, 25, 255. [Google Scholar] [CrossRef] [PubMed]
- Bande, D.; Moltó, L.; Pereira, J.A.; Montes, A. Chronic pain after groin hernia repair: Pain characteristics and impact on quality of life. BMC Surg. 2020, 20, 147. [Google Scholar] [CrossRef]
- Lo, C.-W.; Chen, Y.-T.; Jaw, F.-S.; Yu, C.-C.; Tsai, Y.-C. Predictive factors of post-laparoscopic inguinal hernia acute and chronic pain: Prospective follow-up of 807 patients from a single experienced surgeon. Surg. Endosc. 2021, 35, 148–158. [Google Scholar] [CrossRef]
- Forester, B.; Attaar, M.; Chirayil, S.; Kuchta, K.; Denham, W.; Linn, J.G.; Haggerty, S.P.; Ujiki, M. Predictors of chronic pain after laparoscopic inguinal hernia repair. Surgery 2021, 169, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Min, L.; Yong, P.; Yun, L.; Balde, A.I.; Chang, Z.; Qian, G.; He, L.; Fang, P. Propensity score analysis of outcomes between the transabdominal preperitoneal and open Lichtenstein repair techniques for inguinal hernia repair: A single-center experience. Surg. Endosc. 2020, 34, 5338–5345. [Google Scholar] [CrossRef]
- Köckerling, F.; Lorenz, R.; Hukauf, M.; Grau, H.; Jacob, D.; Fortelny, R.; Koch, A. Influencing Factors on the Outcome in Female Groin Hernia Repair: A Registry-based Multivariable Analysis of 15,601 Patients. Ann. Surg. 2019, 270, 1–9. [Google Scholar] [CrossRef]
- Chinchilla-Hermida, P.A.; Baquero-Zamarra, D.R.; Guerrero-Nope, C.; Bayter-Mendoza, E.F. Incidence of chronic post-surgical pain and its associated factors in patients taken to inguinal hernia repair. Colomb. J. Anesthesiol. 2017, 45, 291–299. [Google Scholar] [CrossRef]
- Andercou, O.; Olteanu, G.; Stancu, B.; Mihaileanu, F.; Chiorescu, S.; Dorin, M. Risk factors for and prevention of chronic pain and sensory disorders following inguinal hernia repair. Ann. Ital. Chir. 2019, 90, 442–446. [Google Scholar]
- Lundström, K.J.; Holmberg, H.; Montgomery, A.; Nordin, P. Patient-reported rates of chronic pain and recurrence after groin hernia repair. Br. J. Surg. 2018, 105, 106–112. [Google Scholar] [CrossRef]
- Matikainen, M.; Aro, E.; Vironen, J.; Kössi, J.; Hulmi, T.; Silvasti, S.; Ilves, I.; Hertsi, M.; Mustonen, K.; Paajanen, H. Factors predicting chronic pain after open inguinal hernia repair: A regression analysis of randomized trial comparing three different meshes with three fixation methods (FinnMesh Study). Hernia 2018, 22, 813–818. [Google Scholar] [CrossRef]
- Niebuhr, H.; Wegner, F.; Hukauf, M.; Lechner, M.; Fortelny, R.; Bittner, R.; Schug-Pass, C.; Köckerling, F. What are the influencing factors for chronic pain following TAPP inguinal hernia repair: An analysis of 20,004 patients from the Herniamed Registry. Surg. Endosc. 2018, 32, 1971–1983. [Google Scholar] [CrossRef] [PubMed]
- Ergönenç, T.; Beyaz, S.G.; Özocak, H.; Palabıyık, O.; Altıntoprak, F. Persistent postherniorrhaphy pain following inguinal hernia repair: A cross-sectional study of prevalence, pain characteristics, and effects on quality of life. Int. J. Surg. 2017, 46, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Olsson, A.; Sandblom, G.; Fränneby, U.; Sondén, A.; Gunnarsson, U.; Dahlstrand, U. Impact of postoperative complications on the risk for chronic groin pain after open inguinal hernia repair. Surgery 2017, 161, 509–516. [Google Scholar] [CrossRef]
- Andresen, K.; Fenger, A.Q.; Burcharth, J.; Pommergaard, H.C.; Rosenberg, J. Mesh fixation methods and chronic pain after transabdominal preperitoneal (TAPP) inguinal hernia surgery: A comparison between fibrin sealant and tacks. Surg. Endosc. 2017, 31, 4077–4084. [Google Scholar] [CrossRef]
- Pierides, G.A.; Paajanen, H.E.; Vironen, J.H. Factors predicting chronic pain after open mesh based inguinal hernia repair: A prospective cohort study. Int. J. Surg. 2016, 29, 165–170. [Google Scholar] [CrossRef]
- Gutlic, N.; Rogmark, P.; Nordin, P.; Petersson, U.; Montgomery, A. Impact of Mesh Fixation on Chronic Pain in Total Extraperitoneal Inguinal Hernia Repair (TEP): A Nationwide Register-based Study. Ann. Surg. 2016, 263, 1199–1206. [Google Scholar] [CrossRef]
- Langeveld, H.R.; Klitsie, P.; Smedinga, H.; Eker, H.; Riet, M.V.; Weidema, W.; Vergouwe, Y.; Bonjer, H.J.; Jeekel, J.; Lange, J.F. Prognostic value of age for chronic postoperative inguinal pain. Hernia 2015, 19, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Jeroukhimov, I.; Wiser, I.; Karasic, E.; Nesterenko, V.; Poluksht, N.; Lavy, R.; Halevy, A. Reduced postoperative chronic pain after tension-free inguinal hernia repair using absorbable sutures: A single-blind randomized clinical trial. J. Am. Coll. Surg. 2014, 218, 102–107. [Google Scholar] [CrossRef]
- Nikkolo, C.; Murruste, M.; Vaasna, T.; Seepter, H.; Tikk, T.; Lepner, U. Three-year results of randomised clinical trial comparing lightweight mesh with heavyweight mesh for inguinal hernioplasty. Hernia 2012, 16, 555–559. [Google Scholar] [CrossRef]
- Reinpold, W.M.J.; Nehls, J.; Eggert, A. Nerve management and chronic pain after open inguinal hernia repair: A prospective two phase study. Ann. Surg. 2011, 254, 163–168. [Google Scholar] [CrossRef]
- Hompes, R.; Vansteenkiste, F.; Pottel, H.; Devriendt, D.; Van Rooy, F. Chronic pain after Kugel inguinal hernia repair. Hernia 2008, 12, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Reinpold, W.; Chen, D. Chronic Postoperative Inguinal Pain (CPIP). In Laparo-Endoscopic Hernia Surgery: Evidence Based Clinical Practice; Bittner, R., Köckerling, F., Fitzgibbons, R.J., Jr., LeBlanc, K.A., Mittal, S.K., Chowbey, P., Eds.; Springer: Berlin/Heidelberg, Germany, 2018; pp. 201–214. [Google Scholar] [CrossRef]
- Hidalgo, N.J.; Guillaumes, S.; Bachero, I.; Butori, E.; Espert, J.J.; Ginestà, C.; Vidal, Ó.; Momblán, D. Bilateral inguinal hernia repair by laparoscopic totally extraperitoneal (TEP) vs. laparoscopic transabdominal preperitoneal (TAPP). BMC Surg. 2023, 23, 270. [Google Scholar] [CrossRef]
- Wes Love, M.; Carbonell, A.M. Robotic Inguinal Hernia Repair: Current Status. Curr. Surg. Rep. 2024, 12, 448–454. [Google Scholar] [CrossRef]
- Santos, I.; Simões, J.F.F.; Dias, C.C.; Alves, M.S.; Azevedo, J.; Cunha, M.; João, A.A.; Nobre, J.G.; Picciochi, M.; Soares, A.S.; et al. Surgical Technique and Chronic Postoperative Inguinal Pain in Patients Undergoing Open Inguinal Hernioplasty in Portugal: A Prospective Multicentric Cohort Study. Acta Med. Port. 2024, 37, 507–517. [Google Scholar] [CrossRef]
- Loos, M.J.A.; Roumen, R.M.H.; Scheltinga, M.R.M. Classifying post-herniorrhaphy pain syndromes following elective inguinal hernia repair. World J. Surg. 2007, 31, 1760–1765. [Google Scholar] [CrossRef]
- Fränneby, U.; Sandblom, G.; Nordin, P.; Nyrén, O.; Gunnarsson, U. Risk factors for long-term pain after hernia surgery. Ann. Surg. 2006, 244, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Nienhuijs, S.W.; Boelens, O.B.A.; Strobbe, L.J.A. Pain after anterior mesh hernia repair. J. Am. Coll. Surg. 2005, 200, 885–889. [Google Scholar] [CrossRef]
- Gutlic, A.; Petersson, U.; Rogmark, P.; Montgomery, A. Long term inguinal pain comparing TEP to Lichtenstein repair: The TEPLICH RCT 8 years follow-up. Hernia 2024, 29, 49. [Google Scholar] [CrossRef] [PubMed]
- Narita, M.; Yamaoka, R.; Hata, H. The long-term outcome of surgical treatment in patients with chronic postoperative inguinal pain. Br. J. Surg. 2024, 111 (Suppl. S5), znae122.400. [Google Scholar] [CrossRef]
- Chu, Z.; Zheng, B.; Yan, L. Incidence and predictors of chronic pain after inguinal hernia surgery: A systematic review and meta-analysis. Hernia 2024, 28, 967–987. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shen, J.; Liu, N.; Liu, Z.; Zhu, X.; Zhong, M.; Tang, R. Comparison of open and laparoscopic preperitoneal tension-free repair of groin hernia: A prospective nonrandomized controlled study. Surg. Endosc. 2025. [Google Scholar] [CrossRef]
- Thulasilingam, K.; Akhter, J. A comparative study on short term surgical outcome between laparoscopic inguinal hernia mesh repair vs. open inguinal hernia mesh repair for primary unilateral inguinal hernias. Int. Surg. J. 2023, 10, 599–605. [Google Scholar] [CrossRef]
- Czarnota, M.; Adamowicz, M.; Górniak, A. Minimally Invasive Surgery in Modern Medicine: A Review of Laparoscopic Techniques, Indications, and Postoperative Outcomes. J. Educ. Health Sport 2025, 83, 61761. [Google Scholar] [CrossRef]
- de la Croix, H.; Montgomery, A.; Holmberg, H.; Melkemichel, M.; Nordin, P. Surgical expertise and risk of long-term complications following groin hernia mesh repair in Sweden: A prospective, patient-reported, nationwide register study. Int. J. Surg. 2025. [Google Scholar] [CrossRef]
- Lange, J.F.M.; Meyer, V.M.; Voropai, D.A.; Keus, E.; Wijsmuller, A.R.; Ploeg, R.J.; Pierie, J.P.E.N. The role of surgical expertise with regard to chronic postoperative inguinal pain (CPIP) after Lichtenstein correction of inguinal hernia: A systematic review. Hernia 2016, 20, 349–356. [Google Scholar] [CrossRef]
- Madani, S.; Bagherzadeh Cham, M.; Sajadi, S.; Mansouri, K.; Samadaeian, N.; Zare, N. Assessing the Validity and Reliability of the DN4 Neuropathic Pain Questionnaire: A Systematic Review. Ann. Mil. Health Sci. Res. 2025, 23, e154462. [Google Scholar] [CrossRef]
- Hardt, S.; Bergau, S.-A.; Jacques, A.; Tampin, B. Short- and long-term test-retest reliability of the English version of the 7-item DN4 questionnaire—A screening tool for neuropathic pain. Scand. J. Pain 2023, 23, 494–500. [Google Scholar] [CrossRef]
- Taha-Mehlitz, S.; Taha, A.; Janzen, A.; Saad, B.; Hendie, D.; Ochs, V.; Krähenbühl, L. Is pain control for chronic neuropathic pain after inguinal hernia repair using endoscopic retroperitoneal neurectomy effective? A meta-analysis of 142 patients from 1995 to 2022. Langenbecks Arch. Surg. 2023, 408, 39. [Google Scholar] [CrossRef] [PubMed]
- Nijs, J.; De Baets, L.; Hodges, P. Phenotyping nociceptive, neuropathic, and nociplastic pain: Who, how, & why? Braz. J. Phys. Ther. 2023, 27, 100537. [Google Scholar] [CrossRef] [PubMed]
- van Veenendaal, N.; Foss, N.B.; Miserez, M.; Pawlak, M.; Zwaans, W.A.R.; Aasvang, E.K. A narrative review on the non-surgical treatment of chronic postoperative inguinal pain: A challenge for both surgeon and anaesthesiologist. Hernia 2023, 27, 5–14. [Google Scholar] [CrossRef]
- Charitakis, E.; Haj-Ali, E.; Al Hasani-Pfister, F.; Saad, B.; Ortlieb, N.; Haberstroh, A.; Ponholzer, F.; Taha-Mehlitz, S.; Schupp, L.-M.; Bauer, R.C.; et al. Impact of different neurectomy techniques on managing chronic pain after inguinal hernia repair: A meta-analysis and systematic review. Hernia 2025, 29, 249. [Google Scholar] [CrossRef]
- Fuller, A.M.; Bharde, S.; Sikandar, S. The mechanisms and management of persistent postsurgical pain. Front. Pain Res. 2023, 4, 1154597. [Google Scholar] [CrossRef]
- Moka, E.; Aguirre, J.A.; Sauter, A.R.; Lavand’homme, P.; European Society of Regional Anaesthesia and Pain Therapy (ESRA). Chronic postsurgical pain and transitional pain services: A narrative review highlighting European perspectives. Reg. Anesth. Pain Med. 2025, 50, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Latenstein, C.S.S.; Thunnissen, F.M.; Harker, M.; Groenewoud, S.; Noordenbos, M.W.; Atsma, F.; de Reuver, P.R. Variation in practice and outcomes after inguinal hernia repair: A nationwide observational study. BMC Surg. 2021, 21, 45. [Google Scholar] [CrossRef]
- Henriksen, N.A.; Bougard, H.; Gonçalves, M.R.; Hope, W.; Khare, R.; Shao, J.; Quiroga-Centeno, A.C.; Deerenberg, E.B. Primary ventral and incisional hernias: Comprehensive review. BJS Open 2024, 9, zrae145. [Google Scholar] [CrossRef] [PubMed]
- Crepaz, L.; Sartori, A.; Olmi, S.; Podda, M.; Di Leo, A.; Stabilini, C.; Carlucci, M.; Ortenzi, M. The ACTIVE study: Surgical outcomes for minimally invasive and open approach to incisional ventral hernias in a non-elective setting. Updates Surg. 2025. [Google Scholar] [CrossRef]
- Pedersen, K.F.; Chen, D.C.; Kehlet, H.; Stadeager, M.W.; Bisgaard, T. A Simplified clinical algorithm for standardized surgical treatment of chronic pain after inguinal hernia repair: A quality assessment study. Scand. J. Surg. 2021, 110, 359–367. [Google Scholar] [CrossRef]
- Sun, Z.; Tang, L.; Wu, Z.; Hu, G.; Xu, M. Retrospective comparison of laparoscopic and open surgery for obturator hernia: A single-center experience. Eur. J. Med. Res. 2025, 30, 396. [Google Scholar] [CrossRef] [PubMed]
- Jensen, S.A.M.S.; Lauridsen, S.V.; Fonnes, S.; Rosenberg, J.; Tønnesen, H. Effect of tailored, intensive prehabilitation for risky lifestyles before ventral hernia repair on postoperative outcomes, health, and costs—Study protocol for a randomised controlled trial (STRONG-Hernia). PLoS ONE 2025, 20, e0324002. [Google Scholar] [CrossRef]
- Alzatari, R.; Huang, L.C.; Poulose, B.K. The impact of opioid versus non-opioid analgesics on postoperative pain level, quality of life, and outcomes in ventral hernia repair. Hernia 2024, 28, 1599–1607. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yin, C.; Pan, Y.; Yang, Y.; Li, W.; Ni, H.; Liu, B.; Nie, H.; Xu, R.; Wei, H.; et al. CXCL13 contributes to chronic pain of a mouse model of CRPS-I via CXCR5-mediated NF-κB activation and pro-inflammatory cytokine production in spinal cord dorsal horn. J. Neuroinflamm. 2023, 20, 109. [Google Scholar] [CrossRef]
- Pawlak, M.; Alabbasi, I.; Cocciolo, F.; Wei, R. P-155 ROLE OF MDT IN CPIP MANAGEMENT: LITERATURE REVIEW. Br. J. Surg. 2022, 109 (Suppl. S7), znac308.252. [Google Scholar] [CrossRef]
- Moore, A.M.; Bjurstrom, M.F.; Hiatt, J.R.; Amid, P.K.; Chen, D.C. Efficacy of retroperitoneal triple neurectomy for refractory neuropathic inguinodynia. Am. J. Surg. 2016, 212, 1126–1132. [Google Scholar] [CrossRef]
- Cirocchi, R.; Sutera, M.; Fedeli, P.; Anania, G.; Covarelli, P.; Suadoni, F.; Boselli, C.; Carlini, L.; Trastulli, S.; D’Andrea, V.; et al. Ilioinguinal Nerve Neurectomy is better than Preservation in Lichtenstein Hernia Repair: A Systematic Literature Review and Meta-analysis. World J. Surg. 2021, 45, 1750–1760. [Google Scholar] [CrossRef]
- Singh, A.; Khan, N.; Krasicka, D.; Gulab, A.; Singh, K.; Sajid, M. WP1.7—A systematic review and a meta-analysis of randomised control trials of prophylactic ilioinguinal neurectomy versus ilioinguinal nerve preservation for open inguinal hernia repair. Br. J. Surg. 2024, 111 (Suppl. S8), znae197.117. [Google Scholar] [CrossRef]
- Messias, B.A.; Nicastro, R.G.; Mocchetti, E.R.; Waisberg, J.; Roll, S.; Junior, M.A.F.R. Lichtenstein technique for inguinal hernia repair: Ten recommendations to optimize surgical outcomes. Hernia 2024, 28, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.P.; Aufenacker, T.; Bay-Nielsen, M.; Bouillot, J.L.; Campanelli, G.; Conze, J.; de Lange, D.; Fortelny, R.; Heikkinen, T.; Kingsnorth, A.; et al. European Hernia Society guidelines on the treatment of inguinal hernia in adult patients. Hernia 2009, 13, 343–403. [Google Scholar] [CrossRef]
- Lange, J.F.M.; Kaufmann, R.; Wijsmuller, A.R.; Pierie, J.P.E.N.; Ploeg, R.J.; Chen, D.C.; Amid, P.K. An international consensus algorithm for management of chronic postoperative inguinal pain. Hernia 2015, 19, 33–43. [Google Scholar] [CrossRef]
- Castillo Ade, J.R.; Muñoz, C.A.P.; Juarez, J.A.I.; Diaz, J.G.V. Laparoscopic triple neurectomy as a treatment for chronic postoperative inguinal pain: A case report. Int. Surg. J. 2025, 12, 1354–1357. [Google Scholar] [CrossRef]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).