The Role of Surgery for Stage 0 Adenocarcinoma In Situ of the Lung: A National Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Data Source
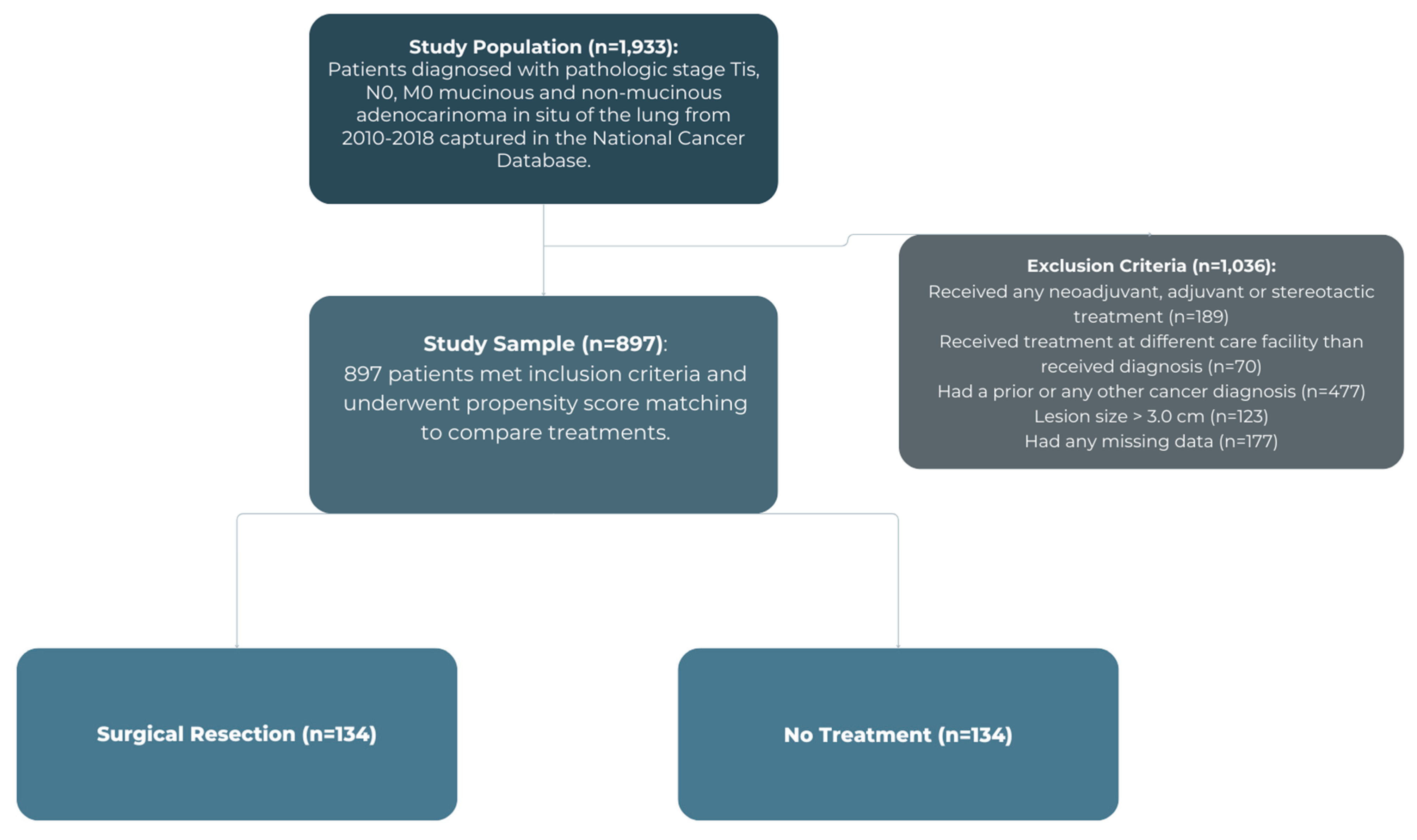
2.3. Study Design
2.4. Statistical Analysis
3. Results
3.1. Predictors of Surgical Intervention and Overall Survival
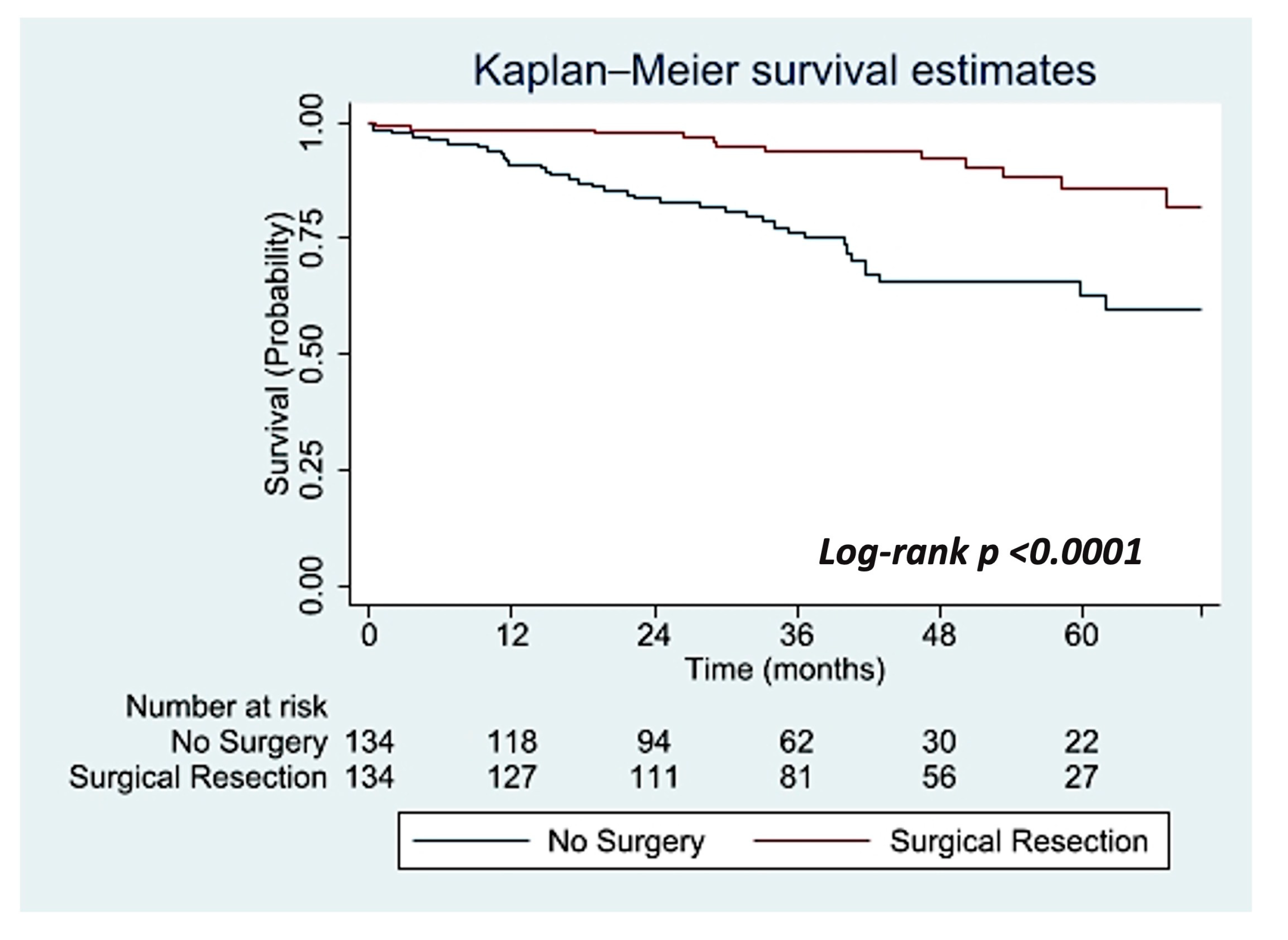
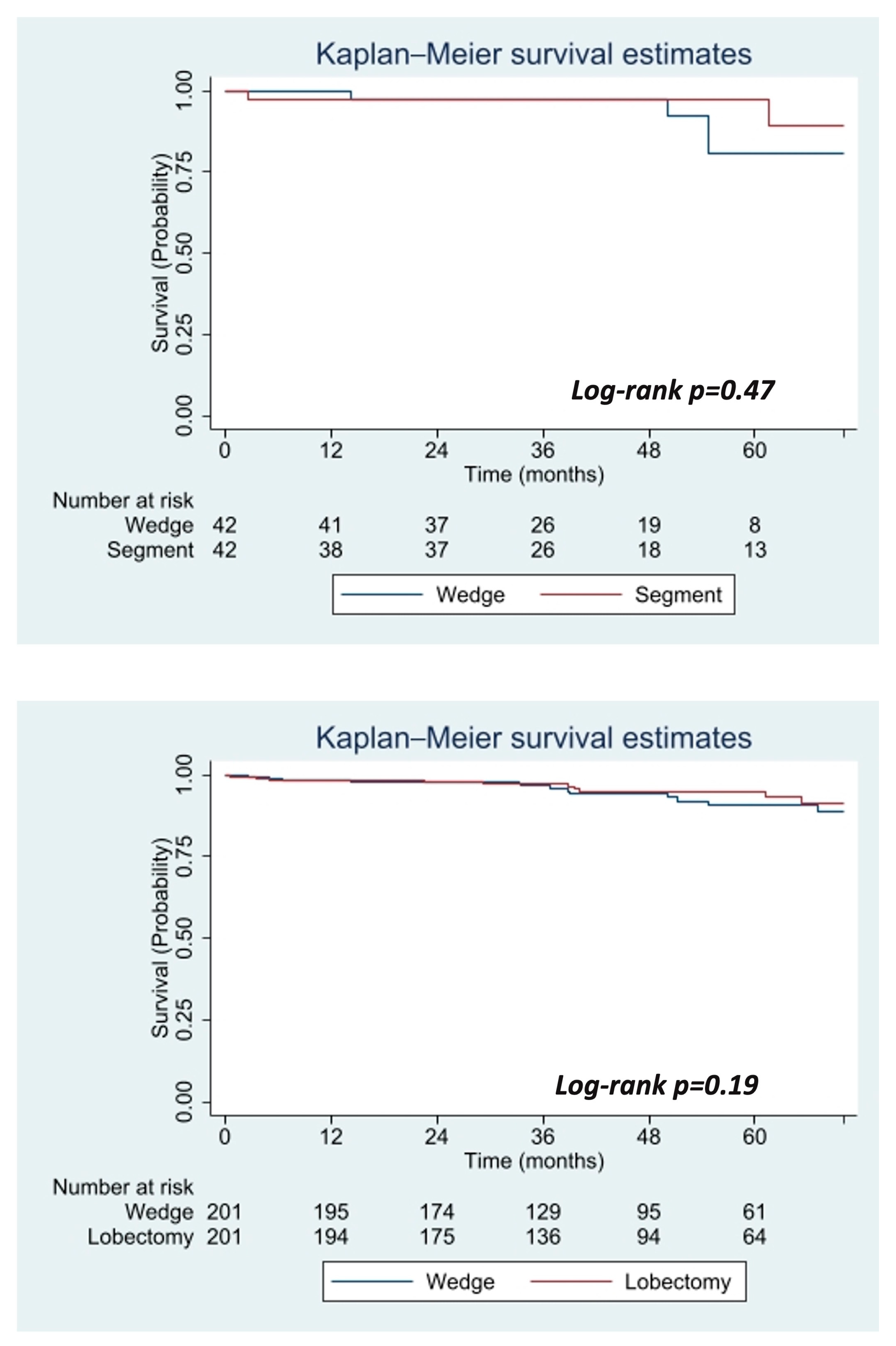
3.2. Propensity Score-Matched Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACS | American College of Surgeons |
| AIS | Adenocarcinoma in Situ |
| CDCC | Charlson–Deyo Comorbidity |
| CoC | Commission on Cancer |
| CT | Computed Tomography |
| GGN | Ground-Glass Nodule |
| ICD-O-3 | International Classification of Diseases for Oncology, 3rd edition |
| NCDB | National Cancer Database |
| NSCLC | Non-Small Cell Lung Cancer |
| OS | Overall Survival |
References
- Travis, W.D.; Brambilla, E.; Noguchi, M.; Nicholson, A.G.; Geisinger, K.R.; Yatabe, Y.; Beer, D.G.; Powell, C.A.; Riely, G.J.; Van Schil, P.E.; et al. International association for the study of lung cancer/American thoracic society/European respiratory society international multidisciplinary classification of lung adenocarcinoma. J. Thorac. Oncol. 2011, 6, 244–285. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-J.; Lee, C.H.; Jeong, Y.J.; Chung, D.H.; Goo, J.M.; Park, C.M.; Austin, J.H.M. IASLC/ATS/ERS International Multi-disciplinary Classification of Lung Adenocarcinoma: Novel Concepts and Radiologic Implications. J. Thorac. Imaging 2012, 27, 340–353. [Google Scholar] [CrossRef]
- MacMahon, H.; Naidich, D.P.; Goo, J.M.; Lee, K.S.; Leung, A.N.C.; Mayo, J.R.; Mehta, A.C.; Ohno, Y.; Powell, C.A.; Prokop, M.; et al. Guidelines for Management of Incidental Pulmonary Nodules Detected on CT Images: From the Fleischner Society 2017. Radiology 2017, 284, 228–243. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wang, R.; Zhang, Y.; Li, Y.; Cheng, C.; Pan, Y.; Xiang, J.; Zhang, Y.; Chen, H.; Sun, Y. Precise Diagnosis of Intraoperative Frozen Section Is an Effective Method to Guide Resection Strategy for Peripheral Small-Sized Lung Adenocarcinoma. J. Clin. Oncol. 2016, 34, 307–313. [Google Scholar] [CrossRef]
- Ito, M.; Miyata, Y.; Kushitani, K.; Yoshiya, T.; Mimae, T.; Ibuki, Y.; Misumi, K.; Takeshima, Y.; Okada, M. Prediction for prognosis of resected pT1a-1bN0M0 adenocarcinoma based on tumor size and histological status: Relationship of TNM and IASLC/ATS/ERS classifications. Lung Cancer 2014, 85, 270–275. [Google Scholar] [CrossRef]
- Ishida, H.; Shimizu, Y.; Sakaguchi, H.; Nitanda, H.; Kaneko, K.; Yamazaki, N.; Yanagihara, A.; Taguchi, R.; Sakai, F.; Yasuda, M.; et al. Distinctive clinicopathological features of adenocarcinoma in situ and minimally invasive adenocarcinoma of the lung: A retrospective study. Lung Cancer 2019, 129, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Behera, M.; Owonikoko, T.K.; Gal, A.A.; Steuer, C.E.; Kim, S.; Pillai, R.N.; Khuri, F.R.; Ramalingam, S.S.; Sica, G.L. Lung Adenocarcinoma Staging Using the 2011 IASLC/ATS/ERS Classification: A Pooled Analysis of Adenocarcinoma in Situ and Minimally Invasive Adenocarcinoma. Clin. Lung Cancer 2016, 17, e57–e64. [Google Scholar] [CrossRef]
- Zhang, Y.; Ma, X.; Shen, X.; Wang, S.; Li, Y.; Hu, H.; Chen, H. Surgery for pre- and minimally invasive lung adenocarcinoma. J. Thorac. Cardiovasc. Surg. 2020, 163, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Li, W.-J.; Weng, J.-J.; Chen, Z.-J.; Wen, Y.-Y.; Deng, T.; Le, H.-B.; Zhang, Y.-K.; Zhang, B.-J. Cancer-associated fibroblasts, matrix metalloproteinase-9 and lymphatic vessel density are associated with progression from adenocarcinoma in situ to invasive adenocarcinoma of the lung. Oncol. Lett. 2020, 20, 130. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, T.; Marushima, H.; Saji, H.; Kojima, K.; Hoshikawa, M.; Takagi, M.; Nakamura, H. PD-L1 Expression in Non-Small-Cell Lung Cancer Including Various Adenocarcinoma Subtypes. Ann Thorac. Cardiovasc. Surg. 2019, 25, 1–9. [Google Scholar] [CrossRef]
- Boland, J.M.; Froemming, A.T.; Wampfler, J.A.; Maldonado, F.; Peikert, T.; Hyland, C.; de Andrade, M.; Aubry, M.C.; Yang, P.; Yi, E.S. Adenocarcinoma in situ, minimally invasive adenocarcinoma, and invasive pulmonary adenocarcinoma—Analysis of interobserver agreement, survival, radiographic characteristics, and gross pathology in 296 nodules. Hum. Pathol. 2016, 51, 41–50. [Google Scholar] [CrossRef]
- Tsubokawa, N.; Mimae, T.; Miyata, Y.; Sasada, S.; Yoshiya, T.; Kushitani, K.; Takeshima, Y.; Murakami, S.; Yokose, T.; Ito, H.; et al. Prognostic significance of vascular invasion in intermediate-grade subtype of lung adenocarcinoma. Jpn. J. Clin. Oncol. 2016, 46, 1015–1021. [Google Scholar] [CrossRef] [PubMed]
- Carretta, A.; Bandiera, A.; Muriana, P.; Viscardi, S.; Ciriaco, P.; Gajate, A.M.S.; Arrigoni, G.; Lazzari, C.; Gregorc, V.; Negri, G. Prognostic role of positron emission tomography and computed tomography parameters in stage I lung adenocarcinoma. Radiol. Oncol. 2020, 54, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Aokage, K.; Miyoshi, T.; Ishii, G.; Kusumoto, M.; Nomura, S.; Katsumata, S.; Sekihara, K.; Tane, K.; Tsuboi, M. Influence of Ground Glass Opacity and the Corresponding Pathological Findings on Survival in Patients with Clinical Stage I Non-Small Cell Lung Cancer. J. Thorac. Oncol. 2018, 13, 533–542. [Google Scholar] [CrossRef]
- Russell, P.A.; Wainer, Z.; Wright, G.M.; Daniels, M.; Conron, M.; Williams, R.A. Does lung adenocarcinoma subtype predict patient survival? A clinicopathologic study based on the new International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary lung adenocarcinoma classification. J. Thorac. Oncol. 2011, 6, 1496–1504. [Google Scholar]
- Bilimoria, K.Y.; Stewart, A.K.; Winchester, D.P.; Ko, C.Y. The National Cancer Data Base: A powerful initiative to improve cancer care in the United States. Ann. Surg. Oncol. 2008, 15, 683–690. [Google Scholar] [CrossRef]
| Predictors | Non-Treatment Group | Surgical Group | p-Value | |
|---|---|---|---|---|
| 181 | 716 | |||
| Sex | Male | 73 (40.3%) | 180 (25.1%) | <0.001 |
| Female | 108 (59.7%) | 536 (74.9%) | ||
| Age, mean years (SD) | 72.8 (9.7) | 66.3 (9.7) | <0.001 | |
| Race, n (%) | White | 155 (85.6%) | 597 (83.4%) | 0.86 |
| Black | 13 (7.2%) | 62 (8.7%) | ||
| Asian | 10 (5.5%) | 46 (6.4%) | ||
| Other | 3 (1.7%) | 9 (1.3%) | ||
| Insurance Status n (%) | Uninsured | 2 (1.1%) | 10 (1.4%) | 0.002 |
| Private | 33 (18.2%) | 235 (32.8%) | ||
| Medicaid | 7 (3.9%) | 30 (4.2%) | ||
| Medicare | 132 (72.9%) | 431 (60.2%) | ||
| Other | 7 (3.9%) | 10 (1.4%) | ||
| Annual Income n (%) | <USD 40,227 | 22 (13.9%) | 80 (12.6%) | 0.55 |
| USD 40,227–50,353 | 33 (20.9%) | 116 (18.3%) | ||
| USD 50,354–63,332 | 39 (24.7%) | 140 (22.1%) | ||
| >USD 63,332 | 64 (40.5%) | 297 (46.9%) | ||
| Education Percentage Without High School Diploma, n (%) | >17.6% | 26 (16.3%) | 105 (16.6%) | 0.15 |
| 10.9–17.5% | 43 (26.9%) | 143 (22.6%) | ||
| 6.3–10.8% | 52 (32.5%) | 175 (27.6%) | ||
| <6.3% | 39 (24.4%) | 211 (33.3%) | ||
| Facility Type, n (%) | Community Cancer Facility | 7 (3.9%) | 24 (3.4%) | 0.024 |
| Comprehensive Cancer Facility | 84 (46.7%) | 247 (34.8%) | ||
| Academic/Research Facility | 57 (31.7%) | 291 (41.0%) | ||
| Integrated Network Facility | 32 (17.8%) | 148 (20.8%) | ||
| Distance from Facility, mean miles (SD) | 30.1 (107.9) | 52.6 (237.6) | 0.24 | |
| Year of Diagnosis, n (%) | 2010 | 1 (0.6%) | 4 (0.6%) | 0.064 |
| 2011 | 2 (1.1%) | 7 (1.0%) | ||
| 2012 | 7 (3.9%) | 39 (5.4%) | ||
| 2013 | 12 (6.6%) | 53 (7.4%) | ||
| 2014 | 12 (6.6%) | 97 (13.5%) | ||
| 2015 | 21 (11.6%) | 120 (16.8%) | ||
| 2016 | 31 (17.1%) | 105 (14.7%) | ||
| 2017 | 48 (26.5%) | 158 (22.1%) | ||
| 2018 | 47 (26.0%) | 133 (18.6%) | ||
| CDCC Score, n (%) | 0 | 93 (51.4%) | 419 (58.5%) | 0.16 |
| 1 | 54 (29.8%) | 205 (28.6%) | ||
| 2 | 25 (13.8%) | 64 (8.9%) | ||
| 3 | 9 (5.0%) | 28 (3.9%) | ||
| Tumor Size, mean size in mm (SD) | 16.7 (6.8) | 13.1 (6.2) | <0.001 |
| Predictors | Odds Ratio (95% CI, p-Value) | Hazards Ratio (95% CI, p-Value) | |
|---|---|---|---|
| Surgery (reference no treatment) | NA | 0.25 (0.16–0.40, <0.001) | |
| Sex (male vs. female) | 1.86 (1.23–2.80, 0.003) | 0.55 (0.36–0.84, 0.005) | |
| Age (per year) | 0.92 (0.90–0.95, <0.001) | 1.05 (1.02–1.08, 0.001) | |
| Race (reference white) | Black | 1.52 (0.68–3.38, 0.31) | 2.08 (1.08–3.98, 0.03) |
| Asian | 1.10 (0.50–2.46, 0.80) | 0.52 (0.15–1.78, 0.30) | |
| Other | 0.74 (0.16–3.55, 0.71) | 2.89 (0.16–52.45, 0.47) | |
| Insurance status (reference uninsured) | Private | 2.38 (0.41–13.73, 0.33) | 0.99 (0.12–8.08, 0.99) |
| Medicaid | 1.33 (0.19–9.36, 0.78) | 1.31 (0.13–12.78, 0.82) | |
| Medicare | 3.3 (0.57–19.44, 0.18) | 1.07 (0.13–8.78, 0.06) | |
| Other | 1.75 (0.19–16.44, 0.63) | 2.01 (0.17–24.02, 0.58) | |
| Annual income (reference < 40,227) | USD 40,227–50,353 | 1.38 (0.68–2.81, 0.38) | 0.55 (0.27–1.14, 0.11) |
| USD 50,354–63,332 | 1.64 (0.79–3.42, 0.19) | 0.68 (0.34–1.38, 0.29) | |
| >USD 63,332 | 1.68 (0.78–3.60, 0.18) | 0.73 (0.35–1.50, 0.39) | |
| Education percentage without high school diploma (reference < 17.6%) | 10.9–17.5% | 0.56 (0.30–1.07, 0.08) | 0.91 (0.46–1.79, 0.79) |
| 6.3–10.8% | 0.67 (0.34–1.31, 0.24) | 1.03 (0.51–2.08, 0.93) | |
| <6.3% | 0.92 (0.43–1.98, 0.83) | 1.04 (0.47–2.32, 0.91) | |
| Facility type (reference community cancer facility) | Comprehensive Cancer Facility | 0.56 (0.20–1.56, 0.27) | 0.75 (0.26–2.18, 0.60) |
| Academic/Research Facility | 0.71 (0.25–1.99, 0.51) | 0.62 (0.21–1.84, 0.39) | |
| Integrated Network Facility | 0.76 (0.25–2.28, 0.63) | 0.97 (0.32–2.96, 0.96) | |
| Distance from facility (per mile) | 1.00 (0.99–1.01, 0.52) | 1.01 (0.99–1.01, 0.55) | |
| Year of diagnosis (per year) | 0.84 (0.75–0.94, 0.003) | 1.05 (0.93–1.20, 0.42) | |
| CDCC score (reference 0) | 1 | 0.90 (0.57–1.42, 0.65) | 1.35 (0.83–2.19, 0.22) |
| 2 | 0.69 (0.38–1.27, 0.24) | 2.18 (0.25–3.81, 0.06) | |
| 3 | 0.90 (0.38–2.21, 0.84) | 1.21 (0.48–3.08, 0.69) | |
| Tumor size (per mm) | 0.94 (0.91–0.97, <0.001) | 1.00 (0.97–1.03, 0.95) |
| Predictors | Non-Treatment Group | Surgical Group | Absolute Standardized Difference (%) | |
|---|---|---|---|---|
| 134 | 134 | |||
| Female Sex, n (%) | 85 (63.4%) | 82 (61.2%) | 4.8 | |
| Age, mean years (SD) | 71.8 (9.5) | 72.2 (7.8) | 3.9 | |
| Race, n (%) | White | 112 (83.6%) | 107 (79.9%) | 6.1 |
| Black | 10 (7.5%) | 13 (9.7%) | 8.3 | |
| Asian | 10 (7.5%) | 11 (8.2%) | 3.1 | |
| Other | 2 (1.5%) | 3 (2.2%) | 6.2 | |
| Insurance Status, n (%) | Uninsured | 1 (0.7%) | 4 (3.0%) | 2.6 |
| Private | 29 (21.6%) | 26 (19.4%) | 5.2 | |
| Medicaid | 4 (3.0%) | 4 (3.0%) | <0.1 | |
| Medicare | 96 (71.6%) | 97 (72.4%) | 1.6 | |
| Other | 4 (3.0%) | 3 (2.2%) | <0.1 | |
| Annual Income, n (%) | <USD 40,227 | 18 (13.4%) | 20 (14.9%) | 4.6 |
| USD 40,227–50,353 | 27 (20.1%) | 30 (22.4%) | 5.6 | |
| USD 50,354–63,332 | 33 (24.6%) | 34 (25.4%) | 1.8 | |
| >USD 63,332 | 56 (41.8%) | 50 (37.3%) | 9.1 | |
| Education Percentage Without High School Diploma, n (%) | >17.6% | 24 (17.9%) | 26 (19.4%) | 4.2 |
| 10.9–17.5% | 32 (23.9%) | 30 (22.4%) | 3.5 | |
| 6.3–10.8% | 42 (31.3%) | 50 (37.3%) | 11 | |
| <6.3% | 36 (26.9%) | 28 (20.9%) | 10.2 | |
| Facility Type, n (%) | Community Cancer Facility | 5 (3.7%) | 3 (2.2%) | 8.4 |
| Comprehensive Cancer Facility | 62 (46.3%) | 59 (44.0%) | 4.6 | |
| Academic/Research Facility | 46 (34.3%) | 49 (36.6%) | 4.7 | |
| Integrated Network Facility | 21 (15.7%) | 23 (17.2%) | 3.8 | |
| Distance from Facility, mean miles (SD) | 31.1 (116.7) | 36.4 (201.4) | 2.8 | |
| CDCC Score, n (%) | 0 | 69 (51.5%) | 69 (51.5%) | <0.1 |
| 1 | 38 (28.4%) | 37 (27.6%) | 1.6 | |
| 2 | 18 (13.4%) | 19 (14.2%) | 2.3 | |
| 3 | 9 (6.7%) | 9 (6.7%) | <0.1 | |
| Year of Diagnosis, (IQR) | 2014 (2011, 2017) | 2014 (2011, 2017) | 9.4 | |
| Tumor Size, mm (SD) | 16.1 (6.8) | 16.6 (7.2) | 6.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Copeland, J.; Neal, E.; Farooq, T.; Orav, E. The Role of Surgery for Stage 0 Adenocarcinoma In Situ of the Lung: A National Analysis. J. Clin. Med. 2025, 14, 6130. https://doi.org/10.3390/jcm14176130
Copeland J, Neal E, Farooq T, Orav E. The Role of Surgery for Stage 0 Adenocarcinoma In Situ of the Lung: A National Analysis. Journal of Clinical Medicine. 2025; 14(17):6130. https://doi.org/10.3390/jcm14176130
Chicago/Turabian StyleCopeland, Jessica, Eliza Neal, Tayyiaba Farooq, and Endel Orav. 2025. "The Role of Surgery for Stage 0 Adenocarcinoma In Situ of the Lung: A National Analysis" Journal of Clinical Medicine 14, no. 17: 6130. https://doi.org/10.3390/jcm14176130
APA StyleCopeland, J., Neal, E., Farooq, T., & Orav, E. (2025). The Role of Surgery for Stage 0 Adenocarcinoma In Situ of the Lung: A National Analysis. Journal of Clinical Medicine, 14(17), 6130. https://doi.org/10.3390/jcm14176130







