The Long-Term Efficacy of Cephalosporin in Elderly Hip Fracture Patients: A Comprehensive Analysis
Abstract
1. Introduction
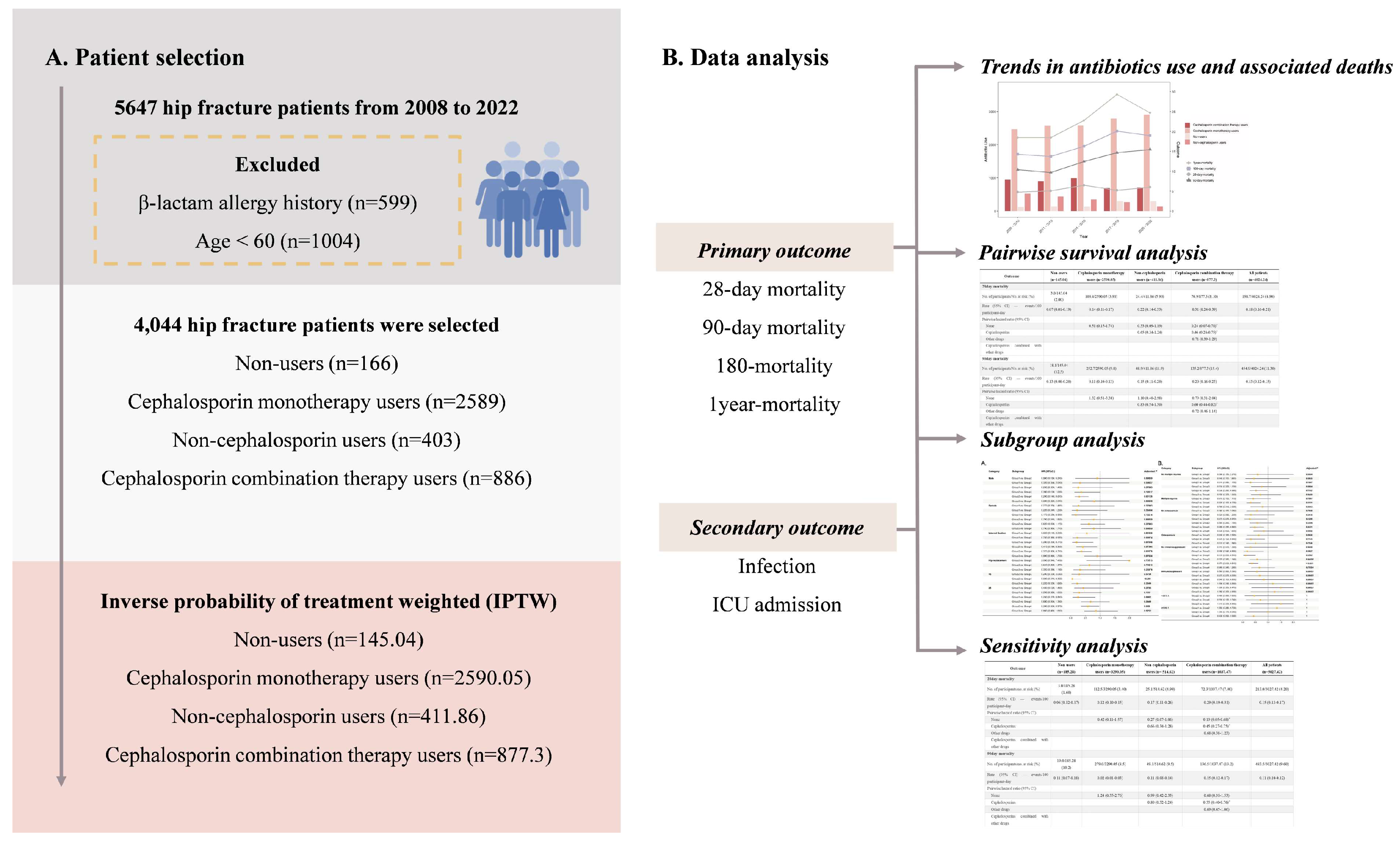
2. Materials and Methods
2.1. Study Population
2.2. Data Collection
2.3. Outcome Definition
2.4. Statistical Analysis
3. Results
3.1. Population Characteristics
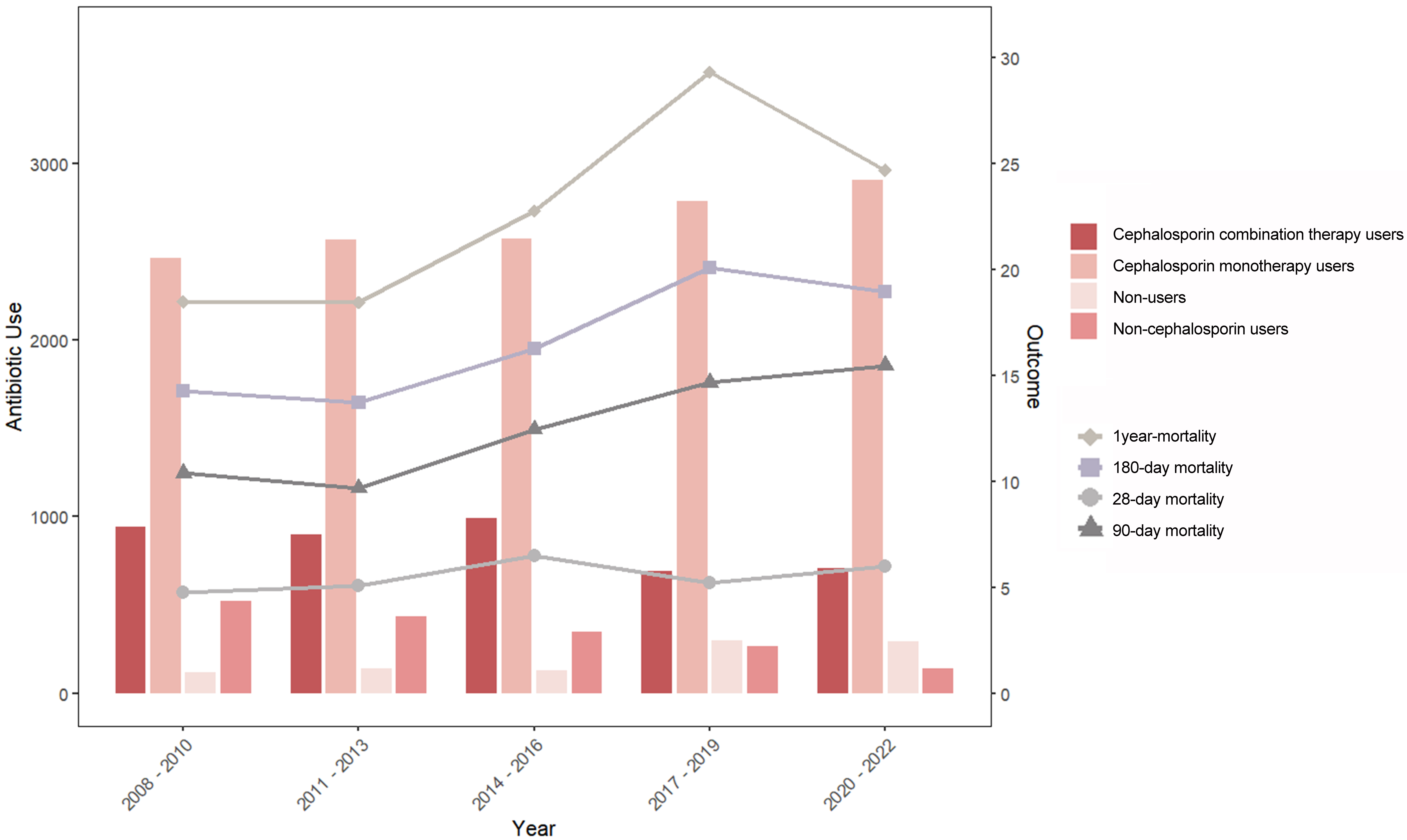
3.2. Antibiotic Use and Mortality Trends from 2008 to 2022
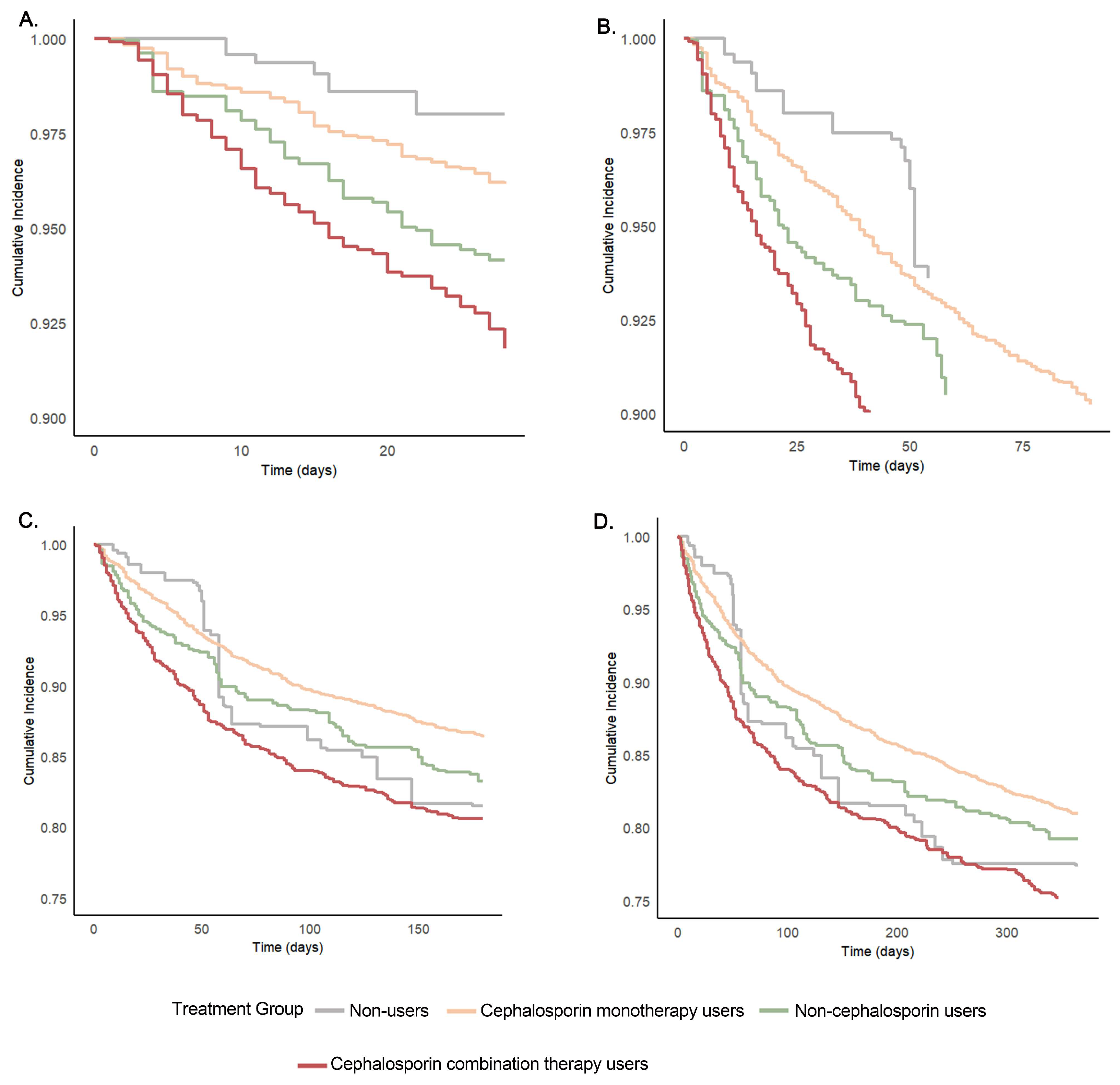
3.3. Association of Cephalosporins with Primary Outcomes
3.4. Association of Cephalosporins with Secondary Outcomes
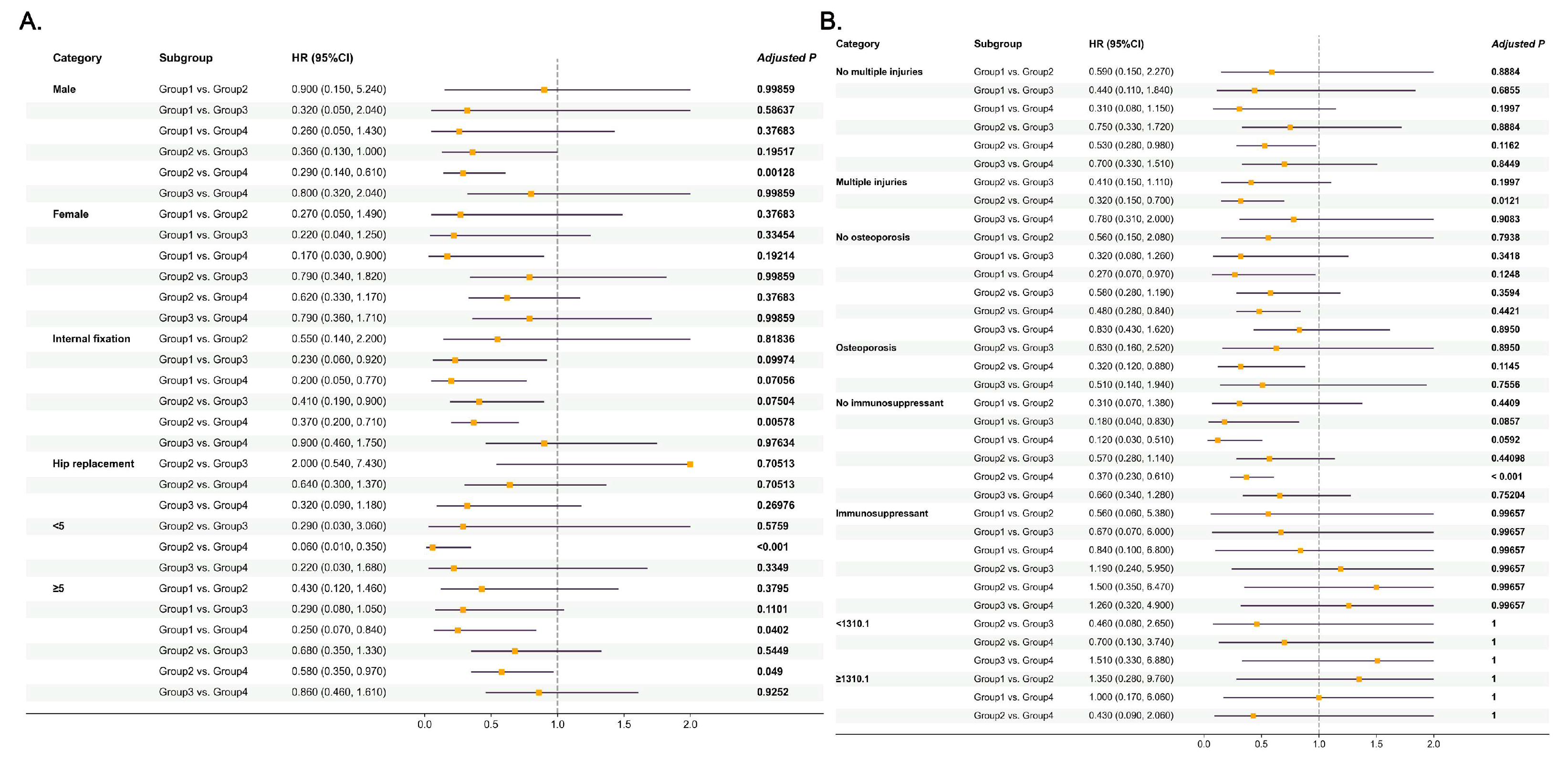
3.5. Subgroup Analysis
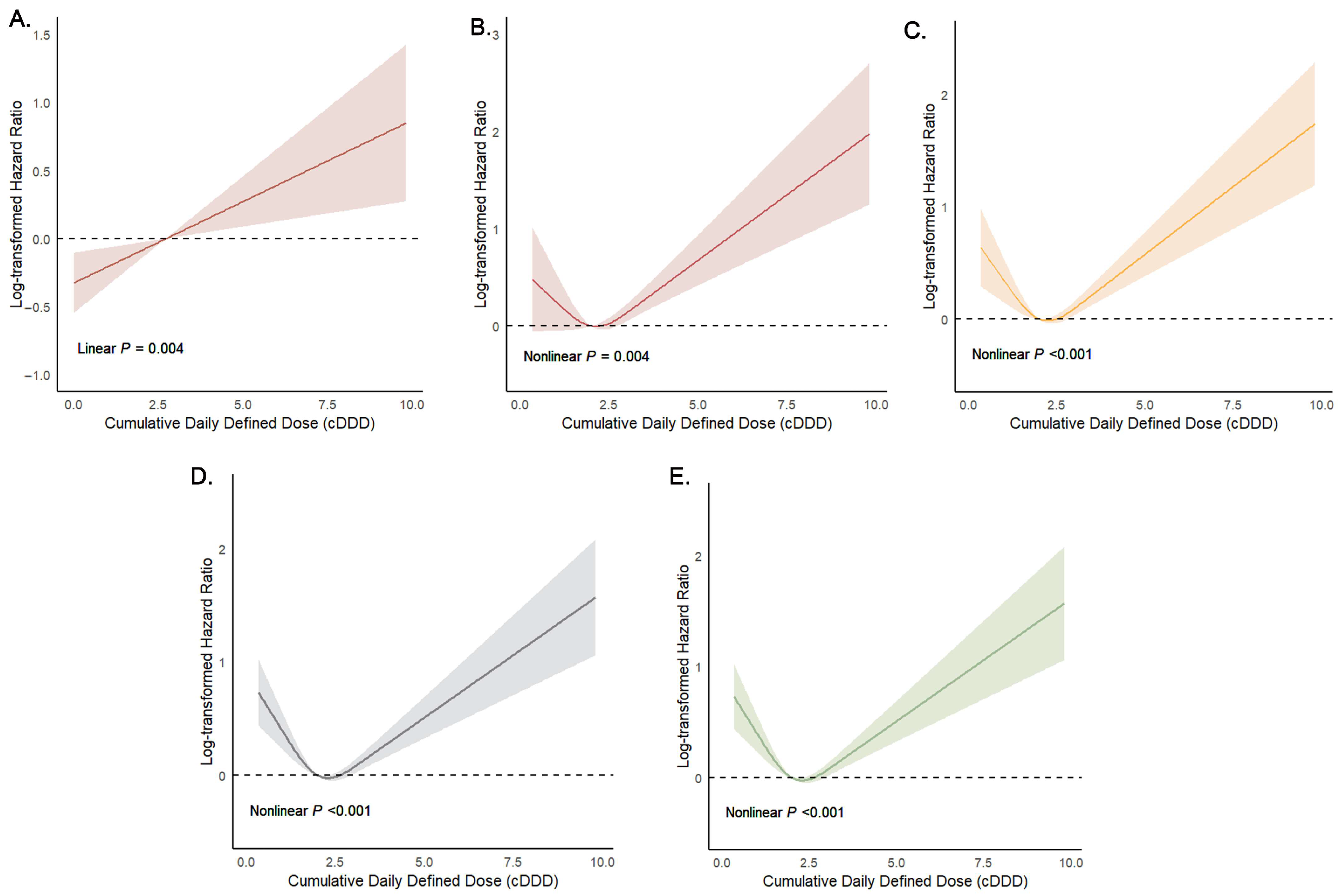
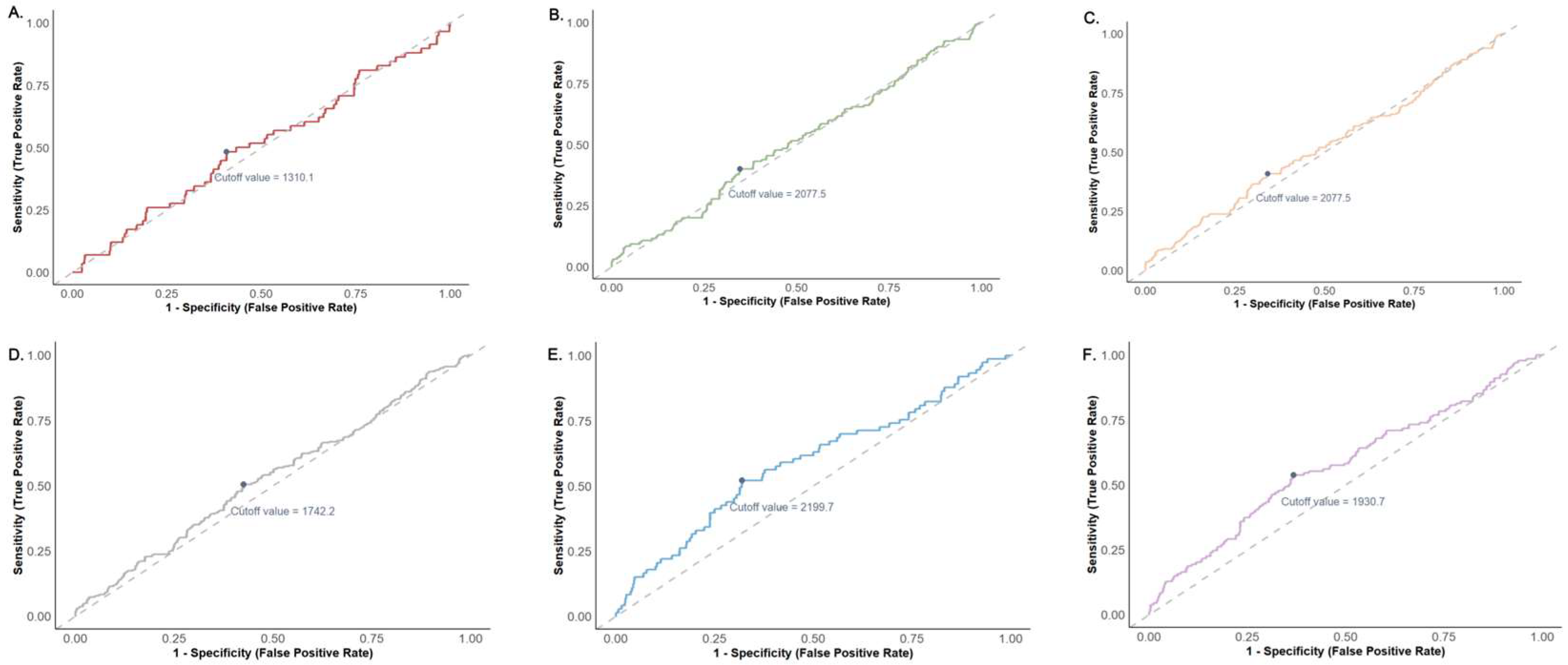
3.6. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brauer, C.A.; Coca-Perraillon, M.; Cutler, D.M.; Rosen, A.B. Incidence and mortality of hip fractures in the United States. Jama 2009, 302, 1573–1579. [Google Scholar] [CrossRef]
- Lewiecki, E.M.; Wright, N.C.; Curtis, J.R.; Siris, E.; Gagel, R.F.; Saag, K.G.; Singer, A.J.; Steven, P.M.; Adler, R.A. Hip fracture trends in the United States, 2002 to 2015. Osteoporos. Int. 2018, 29, 717–722. [Google Scholar] [CrossRef]
- Onizuka, N.; Quatman, C. Global barriers to hip-fracture care. Lancet Healthy Longev. 2024, 5, e510–e511. [Google Scholar] [CrossRef]
- Boxma, H.; Broekhuizen, T.; Patka, P.; Oosting, H. Randomised controlled trial of single-dose antibiotic prophylaxis in surgical treatment of closed fractures: The Dutch Trauma Trial. Lancet 1996, 347, 1133–1137. [Google Scholar] [CrossRef]
- Southwell-Keely, J.P.; Russo, R.R.; March, L.; Cumming, R.; Cameron, I.; Brnabic, A.J. Antibiotic prophylaxis in hip fracture surgery: A metaanalysis. Clin. Orthop. Relat. Res. 2004, 419, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Bratzler, D.W.; Dellinger, E.P.; Olsen, K.M.; Perl, T.M.; Auwaerter, P.G.; Bolon, M.K.; Fish, D.N.; Napolitano, L.M.; Sawyer, R.G.; Slain, D.; et al. Clinical Practice Guidelines for Antimicrobial Prophylaxis in Surgery. Surg. Infect. 2013, 14, 73–156. [Google Scholar] [CrossRef]
- Lian, T.; Dybvik, E.; Gjertsen, J.E.; Dale, H.; Westberg, M.; Nordsletten, L.; Figved, W. Compliance with national guidelines for antibiotic prophylaxis in hip fracture patients: A quality assessment study of 13 329 patients in the Norwegian Hip Fracture Register. BMJ Open 2020, 10, e035598. [Google Scholar] [CrossRef]
- Bush, K.; Bradford, P.A. β-Lactams and β-Lactamase Inhibitors: An Overview. Cold Spring Harb. Perspect. Med. 2016, 6, a025247. [Google Scholar] [CrossRef]
- Yahav, D.; Giske, C.G.; Grāmatniece, A.; Abodakpi, H.; Tam, V.H.; Leibovici, L. New β-Lactam-β-Lactamase Inhibitor Combinations. Clin. Microbiol. Rev. 2020, 34, e00115-20. [Google Scholar] [CrossRef] [PubMed]
- Baggs, J.; Fridkin, S.K.; Pollack, L.A.; Srinivasan, A.; Jernigan, J.A. Estimating National Trends in Inpatient Antibiotic Use Among US Hospitals from 2006 to 2012. JAMA Intern. Med. 2016, 176, 1639–1648. [Google Scholar] [CrossRef] [PubMed]
- Iskandar, K.; Hanna, P.A.; Salameh, P.; Raad, E.B. Antibiotic consumption in non-teaching Lebanese hospitals: A cross-sectional study. J. Infect. Public Health 2016, 9, 618–625. [Google Scholar] [CrossRef][Green Version]
- Global burden of bacterial antimicrobial resistance 1990-2021: A systematic analysis with forecasts to 2050. Lancet 2024, 404, 1199–1226. [CrossRef]
- Hu, Y.; Li, L.; Li, W.; Xu, H.; He, P.; Yan, X.; Dai, H. Combination antibiotic therapy versus monotherapy for Pseudomonas aeruginosa bacteraemia: A meta-analysis of retrospective and prospective studies. Int. J. Antimicrob. Agents 2013, 42, 492–496. [Google Scholar] [CrossRef]
- Starks, I.; Ayub, G.; Walley, G.; Orendi, J.; Roberts, P.; Maffulli, N. Single-dose cefuroxime with gentamicin reduces Clostridium difficile-associated disease in hip-fracture patients. J. Hosp. Infect. 2008, 70, 21–26. [Google Scholar] [CrossRef]
- AlBuhairan, B.; Hind, D.; Hutchinson, A. Antibiotic prophylaxis for wound infections in total joint arthroplasty: A systematic review. J. Bone Joint Surg. Br. 2008, 90, 915–919. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, W.J.; Walenkamp, G.H. Antibiotic prophylaxis for surgery for proximal femoral and other closed long bone fractures. Cochrane Database Syst. Rev. 2010, 2010, Cd000244. [Google Scholar] [CrossRef]
- Johnson, A.E.W.; Bulgarelli, L.; Shen, L.; Gayles, A.; Shammout, A.; Horng, S.; Pollard, T.J.; Hao, S.; Moody, B.; Gow, B.; et al. MIMIC-IV, a freely accessible electronic health record dataset. Sci. Data 2023, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Lancet 2007, 370, 1453–1457. [Google Scholar] [CrossRef]
- World Health Organization. International Classification of Diseases Eleventh Revision (ICD-11); World Health Organization: Geneva, Switzerland, 2022. [Google Scholar]
- Qin, Z.; Li, H.; Wang, L.; Geng, J.; Yang, Q.; Su, B.; Liao, R. Systemic Immune-Inflammation Index Is Associated with Increased Urinary Albumin Excretion: A Population-Based Study. Front. Immunol. 2022, 13, 863640. [Google Scholar] [CrossRef]
- World Health Organization. WHO Collaborating Centre for Drug Statistics Methodology. Available online: https://www.whocc.no/ (accessed on 10 June 2025).
- Zhu, X.; Xue, J.; Liu, Z.; Dai, W.; Xiang, J.; Xu, H.; Zhou, Q.; Zhou, Q.; Wei, X.; Chen, W. The effects of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers in critically ill patients with acute kidney injury: An observational study using the MIMIC database. Front. Pharmacol. 2022, 13, 918385. [Google Scholar] [CrossRef] [PubMed]
- Bettega, F.; Mendelson, M.; Leyrat, C.; Bailly, S. Use and reporting of inverse-probability-of-treatment weighting for multicategory treatments in medical research: A systematic review. J. Clin. Epidemiol. 2024, 170, 111338. [Google Scholar] [CrossRef] [PubMed]
- Harrell, F.E., Jr.; Lee, K.L.; Pollock, B.G. Regression models in clinical studies: Determining relationships between predictors and response. J. Natl. Cancer Inst. 1988, 80, 1198–1202. [Google Scholar] [CrossRef]
- Muhoro, A.M.; Ochomo, E.O.; Kinyua, I.N.; Kosgei, J.J.; Rasaki, L.A.; Farkas, E. A study on the effectiveness of (+)-usnic acid as oral toxic sugar bait against adult male and female Anopheles gambiae. Malar. J. 2024, 23, 311. [Google Scholar] [CrossRef]
- Lachin, J.M.; Bebu, I. Closed testing of each group versus the others combined in a multiple group analysis. Clin. Trials 2020, 17, 77–86. [Google Scholar] [CrossRef]
- Çelen, Z.E. Predictive value of the systemic immune-inflammation index on one-year mortality in geriatric hip fractures. BMC Geriatr. 2024, 24, 340. [Google Scholar] [CrossRef]
- Charlson, M.E.; Carrozzino, D.; Guidi, J.; Patierno, C. Charlson Comorbidity Index: A Critical Review of Clinimetric Properties. Psychother. Psychosom. 2022, 91, 8–35. [Google Scholar] [CrossRef]
- Nelson, C.P.; Goel, A.; Butterworth, A.S.; Kanoni, S.; Webb, T.R.; Marouli, E.; Zeng, L.; Ntalla, I.; Lai, F.Y.; Hopewell, J.C.; et al. Association analyses based on false discovery rate implicate new loci for coronary artery disease. Nat. Genet. 2017, 49, 1385–1391. [Google Scholar] [CrossRef] [PubMed]
- Savitz, M.H.; Malis, L.I.; Savitz, S.I. Efficacy of prophylactic antibiotic therapy in spinal surgery: A meta-analysis. Neurosurgery 2003, 53, 243–244; author reply 244–245. [Google Scholar] [CrossRef]
- Wu, C.Y.; Tsai, C.F.; Hsu, Y.H.; Yang, H.Y. Exploring mortality risk factors and specific causes of death within 30 days after hip fracture hospitalization. Sci. Rep. 2024, 14, 27544. [Google Scholar] [CrossRef] [PubMed]
- Kjørholt, K.E.; Kristensen, N.R.; Prieto-Alhambra, D.; Johnsen, S.P.; Pedersen, A.B. Increased risk of mortality after postoperative infection in hip fracture patients. Bone 2019, 127, 563–570. [Google Scholar] [CrossRef]
- Guastaldi, R.B.; Reis, A.M.; Figueras, A.; Secoli, S.R. Prevalence of potential drug-drug interactions in bone marrow transplant patients. Int. J. Clin. Pharm. 2011, 33, 1002–1009. [Google Scholar] [CrossRef] [PubMed]
- Jongmans, C.; Muller, A.E.; Van Den Broek, P.; Cruz De Almeida, B.M.; Van Den Berg, C.; Van Oldenrijk, J.; Bos, P.K.; Koch, B.C.P. An Overview of the Protein Binding of Cephalosporins in Human Body Fluids: A Systematic Review. Front. Pharmacol. 2022, 13, 900551. [Google Scholar] [CrossRef]
- Vella-Brincat, J.W.; Begg, E.J.; Kirkpatrick, C.M.; Zhang, M.; Chambers, S.T.; Gallagher, K. Protein binding of cefazolin is saturable in vivo both between and within patients. Br. J. Clin. Pharmacol. 2007, 63, 753–757. [Google Scholar] [CrossRef]
- Celestin, M.N.; Musteata, F.M. Impact of Changes in Free Concentrations and Drug-Protein Binding on Drug Dosing Regimens in Special Populations and Disease States. J. Pharm. Sci. 2021, 110, 3331–3344. [Google Scholar] [CrossRef]
- Pan, J.; Ye, Z.; Cai, X.; Wang, L.; Cao, Z. Biophysical study on the interaction of ceftriaxone sodium with bovine serum albumin using spectroscopic methods. J. Biochem. Mol. Toxicol. 2012, 26, 487–492. [Google Scholar] [CrossRef]
- Shi, C.; Xiao, Y.; Zhang, Q.; Li, Q.; Wang, F.; Wu, J.; Lin, N. Efficacy and safety of cefazolin versus antistaphylococcal penicillins for the treatment of methicillin-susceptible Staphylococcus aureus bacteremia: A systematic review and meta-analysis. BMC Infect. Dis. 2018, 18, 508. [Google Scholar] [CrossRef]
- Mondorf, A.W. Investigations on the potential nephrotoxicity of cefazedone and gentamicin and of their combination, in comparison with the combination of cefazolin and cephalothin with gentamicin. Arzneimittelforschung 1979, 29, 449–452. [Google Scholar]
- Miller, M.A.; Fish, D.N.; Barber, G.R.; Barron, M.A.; Goolsby, T.A.; Moine, P.; Mueller, S.W. A comparison of safety and outcomes with cefazolin versus nafcillin for methicillin-susceptible Staphylococcus aureus bloodstream infections. J. Microbiol. Immunol. Infect. 2020, 53, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Peel, T.N.; Astbury, S.; Cheng, A.C.; Paterson, D.L.; Buising, K.L.; Spelman, T.; Tran-Duy, A.; Adie, S.; Boyce, G.; McDougall, C.; et al. Trial of Vancomycin and Cefazolin as Surgical Prophylaxis in Arthroplasty. N. Engl. J. Med. 2023, 389, 1488–1498. [Google Scholar] [CrossRef]
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Bouman, A.; Heineman, M.J.; Faas, M.M. Sex hormones and the immune response in humans. Hum. Reprod. Update 2005, 11, 411–423. [Google Scholar] [CrossRef]
- Trigunaite, A.; Dimo, J.; Jørgensen, T.N. Suppressive effects of androgens on the immune system. Cell Immunol. 2015, 294, 87–94. [Google Scholar] [CrossRef]
- Farkouh, A.; Riedl, T.; Gottardi, R.; Czejka, M.; Kautzky-Willer, A. Sex-Related Differences in Pharmacokinetics and Pharmacodynamics of Frequently Prescribed Drugs: A Review of the Literature. Adv. Ther. 2020, 37, 644–655. [Google Scholar] [CrossRef]
- Borse, H.R. Antimicrobial Utilization Patterns and Postoperative Outcomes in Otorhinolaryngology: A Prospective Observational Study. Res. J. Med. Sci. 2024, 18, 22–27. [Google Scholar] [CrossRef]
- Schröder, W.; Sommer, H.; Gladstone, B.P.; Foschi, F.; Hellman, J.; Evengard, B.; Tacconelli, E. Gender differences in antibiotic prescribing in the community: A systematic review and meta-analysis. J. Antimicrob. Chemother. 2016, 71, 1800–1806. [Google Scholar] [CrossRef]
- Gu, J.; Chen, X.; Yang, Z.; Bai, Y.; Zhang, X. Gender differences in the microbial spectrum and antibiotic sensitivity of uropathogens isolated from patients with urinary stones. J. Clin. Lab. Anal. 2022, 36, e24155. [Google Scholar] [CrossRef] [PubMed]
- Rivera, A.; Sánchez, A.; Luque, S.; Mur, I.; Puig, L.; Crusi, X.; González, J.C.; Sorlí, L.; González, A.; Horcajada, J.P.; et al. Intraoperative Bacterial Contamination and Activity of Different Antimicrobial Prophylaxis Regimens in Primary Knee and Hip Replacement. Antibiotics 2020, 10, 18. [Google Scholar] [CrossRef] [PubMed]
- Swoboda, S.M.; Merz, C.; Kostuik, J.; Trentler, B.; Lipsett, P.A. Does intraoperative blood loss affect antibiotic serum and tissue concentrations? Arch. Surg. 1996, 131, 1165–1171; discussion 1162–1171. [Google Scholar] [CrossRef] [PubMed]
- Si, K.; Zhang, Y.; Wang, G. A short-term efficacy comparison between the FNS and THA as interventions for unstable femoral neck fracture. Front. Surg. 2025, 12, 1537335. [Google Scholar] [CrossRef]
- Liu, H.; Xing, F.; Jiang, J.; Chen, Z.; Xiang, Z.; Duan, X. Random forest predictive modeling of prolonged hospital length of stay in elderly hip fracture patients. Front. Med. 2024, 11, 1362153. [Google Scholar] [CrossRef]
- Wyles, C.C.; Hevesi, M.; Osmon, D.R.; Park, M.A.; Habermann, E.B.; Lewallen, D.G.; Berry, D.J.; Sierra, R.J. 2019 John Charnley Award: Increased risk of prosthetic joint infection following primary total knee and hip arthroplasty with the use of alternative antibiotics to cefazolin: The value of allergy testing for antibiotic prophylaxis. Bone Joint J. 2019, 101-B (Suppl. S6B), 9–15. [Google Scholar] [CrossRef]
- Siverino, C.; Metsemakers, W.J.; Sutter, R.; Della Bella, E.; Morgenstern, M.; Barcik, J.; Ernst, M.; D’Este, M.; Joeris, A.; Chittò, M.; et al. Clinical management and innovation in fracture non-union. Expert. Opin. Biol. Ther. 2024, 24, 973–991. [Google Scholar] [CrossRef]
- Joosten, S.C.M.; Wiersinga, W.J.; Poll, T.V. Dysregulation of Host-Pathogen Interactions in Sepsis: Host-Related Factors. Semin. Respir. Crit. Care Med. 2024, 45, 469–478. [Google Scholar] [CrossRef]
- Domnich, A.; Manini, I.; Calabrò, G.E.; Waure, C.; Montomoli, E. Mapping Host-Related Correlates of Influenza Vaccine-Induced Immune Response: An Umbrella Review of the Available Systematic Reviews and Meta-Analyses. Vaccines 2019, 7, 215. [Google Scholar] [CrossRef]
- Gadgaard, N.R.; Varnum, C.; Nelissen, R.; Vandenbroucke-Grauls, C.; Sørensen, H.T.; Pedersen, A.B. Comorbidity and risk of infection among patients with hip fracture: A Danish population-based cohort study. Osteoporos. Int. 2023, 34, 1739–1749. [Google Scholar] [CrossRef] [PubMed]
- Peel, T.N.; Buising, K.L.; Choong, P.F. Diagnosis and management of prosthetic joint infection. Curr. Opin. Infect. Dis. 2012, 25, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Tomelleri, A.; Dejaco, C.; Bond, M. To prophylax or not to prophylax? The role of trimethoprim/sulfamethoxazole as a prophylactic agent in systemic vasculitis: The case of antineutrophil cytoplasmic antibody-associated vasculitis and giant cell arteritis. Reumatismo 2025, 77, 1791. [Google Scholar] [CrossRef]
- Amzallag, N.; Ashkenazi, I.; Factor, S.; Abadi, M.; Morgan, S.; Graif, N.; Snir, N.; Gold, A.; Warschawski, Y. Addition of gentamicin for antibiotic prophylaxis in hip hemiarthroplasty does not decrease the rate of surgical site infection. Eur. J. Trauma. Emerg. Surg. 2024, 50, 867–873. [Google Scholar] [CrossRef]
- Verrest, L.; Wilthagen, E.A.; Beijnen, J.H.; Huitema, A.D.R.; Dorlo, T.P.C. Influence of Malnutrition on the Pharmacokinetics of Drugs Used in the Treatment of Poverty-Related Diseases: A Systematic Review. Clin. Pharmacokinet. 2021, 60, 1149–1169. [Google Scholar] [CrossRef]
- de Jonge, S.W.; Boldingh, Q.J.J.; Solomkin, J.S.; Dellinger, E.P.; Egger, M.; Salanti, G.; Allegranzi, B.; Boermeester, M.A. Effect of postoperative continuation of antibiotic prophylaxis on the incidence of surgical site infection: A systematic review and meta-analysis. Lancet Infect. Dis. 2020, 20, 1182–1192. [Google Scholar] [CrossRef] [PubMed]
- Angus, D.C.; van der Poll, T. Severe sepsis and septic shock. N. Engl. J. Med. 2013, 369, 840–851. [Google Scholar] [CrossRef]
- Rhinehart, E.; Smith, N.E.; Wennersten, C.; Gorss, E.; Freeman, J.; Eliopoulos, G.M.; Moellering, R.C., Jr.; Goldmann, D.A. Rapid dissemination of beta-lactamase-producing, aminoglycoside-resistant Enterococcus faecalis among patients and staff on an infant-toddler surgical ward. N. Engl. J. Med. 1990, 323, 1814–1818. [Google Scholar] [CrossRef]
- Cohen, S.H.; Gerding, D.N.; Johnson, S.; Kelly, C.P.; Loo, V.G.; McDonald, L.C.; Pepin, J.; Wilcox, M.H. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the society for healthcare epidemiology of America (SHEA) and the infectious diseases society of America (IDSA). Infect. Control Hosp. Epidemiol. 2010, 31, 431–455. [Google Scholar] [CrossRef] [PubMed]
- Becattini, S.; Taur, Y.; Pamer, E.G. Antibiotic-Induced Changes in the Intestinal Microbiota and Disease. Trends Mol. Med. 2016, 22, 458–478. [Google Scholar] [CrossRef] [PubMed]
- Idasiak-Piechocka, I.; Lewandowski, D.; Świgut, W.; Kalinowski, J.; Mikosza, K.; Suchowiejski, P.; Szałek, E.; Karbownik, A.; Miedziaszczyk, M. Effect of hypoalbuminemia on drug pharmacokinetics. Front. Pharmacol. 2025, 16, 1546465. [Google Scholar] [CrossRef] [PubMed]
- Richardson, D.B.; Costello, S.; Kaufman, J.S.; Kelly-Reif, K.; Lewis, S.; Steenland, K.; Tchetgen, E.J.T. Confounding: A Routine Concern in the Interpretation of Epidemiological Studies. In Statistical Methods in Cancer Research Volume V: Bias Assessment in Case–Control and Cohort Studies for Hazard Identification; International Agency for Research on Cancer: Lyon, France, 2024. [Google Scholar]
- Basile, G.; Gallina, M.; Passeri, A.; Gaudio, R.M.; Castelnuovo, N.; Ferrante, P.; Calori, G.M. Prosthetic joint infections and legal disputes: A threat to the future of prosthetic orthopedics. J. Orthop. Traumatol. 2021, 22, 44. [Google Scholar] [CrossRef]
| All Patients (n = 4024.24) | Non-Users (n = 145.04) | Cephalosporin Monotherapy Users (n = 2590.05) | Non-Cephalosporin Users (n = 411.86) | Cephalosporin Combination Therapy Users (n = 877.30) | p-Value | |
|---|---|---|---|---|---|---|
| Age (years) | 80.00 ± 9.86 | 81.45 ± 9.85 | 79.92 ± 9.84 | 80.16 ± 9.50 | 79.92 ± 10.06 | 0.509 |
| Gender, n (%) | 0.916 | |||||
| Female | 2771.4 (68.9) | 102.9 (71.0) | 1782.1 (68.8) | 288.2 (70.0) | 598.1 (68.2) | |
| Male | 1252.8 (31.1) | 42.1 (29.0) | 807.9 (31.2) | 123.6 (30.0) | 279.2 (31.8) | |
| Anchor year, n (%) | 0.999 | |||||
| 2008–2010 | 1548.2 (38.5) | 55.3 (38.1) | 996.0 (38.5) | 158.1 (38.4) | 338.8 (38.6) | |
| 2011–2013 | 877.3 (21.8) | 28.7 (19.8) | 565.6 (21.8) | 90.0 (21.8) | 193.2 (22.0) | |
| 2014–2016 | 716.4 (17.8) | 22.2 (15.3) | 463.9 (17.9) | 74.2 (18.0) | 156.1 (17.8) | |
| 2017–2019 | 495.7 (12.3) | 22.7 (15.7) | 316.2 (12.2) | 51.1 (12.4) | 105.7 (12.1) | |
| 2020–2022 | 386.7 (9.6) | 16.2 (11.2) | 248.4 (9.6) | 38.6 (9.4) | 83.5 (9.5) | |
| Race, n (%) | 0.999 | |||||
| White | 3414.2 (84.8) | 125.6 (86.6) | 2190.7 (84.6) | 355.3 (86.3) | 742.6 (84.6) | |
| Asian | 66.4 (1.7) | 2.5 (1.7) | 43.0 (1.7) | 5.9 (1.4) | 14.9 (1.7) | |
| Black | 221.9 (5.5) | 9.1 (6.3) | 143.3 (5.5) | 20.1 (4.9) | 49.4 (5.6) | |
| Hispanic | 67.0 (1.7) | 1.3 (0.9) | 45.6 (1.8) | 6.4 (1.6) | 13.7 (1.6) | |
| Other | 254.6 (6.3) | 6.4 (4.4) | 167.4 (6.5) | 24.1 (5.9) | 56.7 (6.5) | |
| Admission type, n (%) | 0.960 | |||||
| Emergence | 2188.7 (54.4) | 81.9 (56.5) | 1412.3 (54.5) | 223.6 (54.3) | 470.9 (53.7) | |
| Elective | 79.7 (2.0) | 0.0 (0.0) | 53.7 (2.1) | 8.2 (2.0) | 17.8 (2.0) | |
| Observation | 880.0 (21.9) | 35.0 (24.2) | 567.7 (21.9) | 90.0 (21.9) | 187.2 (21.3) | |
| Same-day surgery | 551.7 (13.7) | 14.2 (9.8) | 350.3 (13.5) | 58.8 (14.3) | 128.4 (14.6) | |
| Urgent | 324.2 (8.1) | 13.9 (9.6) | 206.1 (8.0) | 31.2 (7.6) | 73.0 (8.3) | |
| Total mortality, n (%) | 1422.7 (35.4) | 53.4 (36.8) | 875.2 (33.8) | 142.3 (34.6) | 351.7 (40.1) | 0.039 |
| 28-day mortality, n (%) | 198.7 (4.9) | 3.0 (2.0) | 100.4 (3.9) | 24.4 (5.9) | 70.9 (8.1) | <0.001 |
| 90-day mortality, n (%) | 454.9 (11.3) | 18.1 (12.5) | 252.7 (9.8) | 48.9 (11.9) | 135.2 (15.4) | 0.005 |
| 180-day mortality, n (%) | 618.9 (15.4) | 26.3 (18.2) | 353.3 (13.6) | 70.2 (17.0) | 169.0 (19.3) | 0.009 |
| 1-year mortality, n (%) | 834.7 (20.7) | 32.2 (22.2) | 494.0 (19.1) | 87.4 (21.2) | 221.1 (25.2) | 0.016 |
| Infection, n (%) | 226.3 (5.6) | 0.8 (0.5) | 149.9 (5.8) | 22.7 (5.5) | 52.9 (6.0) | 0.088 |
| ICU admission, n (%) | 490.3 (12.2) | 9.4 (6.5) | 316.4 (12.2) | 52.8 (12.8) | 111.7 (12.7) | 0.220 |
| Length of hospital stay (days) | 6.05 ± 4.32 | 4.78 ± 2.12 | 5.44 ± 3.36 | 6.53 ± 5.40 | 6.84 ± 5.74 | 0.369 |
| Charlson comorbidity index | 5.77 ± 2.24 | 5.85 ± 2.13 | 5.76 ± 2.29 | 5.77 ± 2.17 | 5.81 ± 2.15 | 0.937 |
| Osteoporosis, n (%) | 1200.2 (23.9) | 54.6 (29.5) | 763.7 (23.2) | 143.1 (27.8) | 238.7 (23.0) | 0.096 |
| Multiple injuries, n (%) | 2254.6 (44.9) | 79.0 (42.7) | 1461.5 (44.5) | 262.9 (51.1) | 451.2 (43.5) | 0.780 |
| Vital signs at presentation | ||||||
| BMI (kg/m2) | 27.60 ± 15.70 | 26.26 ± 5.01 | 27.51 ± 17.90 | 27.02 ± 5.38 | 28.35 ± 12.93 | 0.054 |
| Systolic blood pressure (mmHg) | 131.91 ± 14.42 | 133.15 ± 13.39 | 131.95 ± 14.48 | 131.21 ± 14.97 | 131.92 ± 14.14 | 0.671 |
| Diastolic blood pressure (mmHg) | 73.03 ± 8.78 | 72.58 ± 8.11 | 73.02 ± 8.79 | 73.07 ± 9.10 | 73.08 ± 8.69 | 0.959 |
| Laboratory-based data | ||||||
| Red blood cell (109/L) | 3.49 ± 0.63 | 3.49 ± 0.69 | 3.49 ± 0.62 | 3.47 ± 0.64 | 3.49 ± 0.61 | 0.971 |
| White blood cell (109/L) | 10.63 ± 7.05 | 10.52 ± 5.01 | 10.44 ± 4.23 | 12.14 ± 18.01 | 10.47 ± 4.24 | 0.844 |
| Platelet (109/L) | 212.04 ± 84.29 | 204.21 ± 73.59 | 212.09 ± 84.67 | 214.48 ± 87.36 | 212.06 ± 83.37 | 0.796 |
| Hemoglobin (g/dL) | 10.54 ± 1.79 | 10.49 ± 1.95 | 10.55 ± 1.80 | 10.50 ± 1.73 | 10.54 ± 1.76 | 0.963 |
| Creatinine (mg/dL) | 1.12 ± 0.93 | 1.14 ± 0.87 | 1.12 ± 0.92 | 1.14 ± 1.02 | 1.13 ± 0.92 | 0.983 |
| BUN (mg/dL) | 22.50 ± 12.88 | 23.20 ± 11.98 | 22.42 ± 12.88 | 22.45 ± 13.02 | 22.65 ± 12.96 | 0.928 |
| Chloride (mmol/L) | 102.56 ± 4.20 | 102.86 ± 3.93 | 102.54 ± 4.12 | 102.62 ± 4.62 | 102.57 ± 4.30 | 0.887 |
| Bicarbonate (mmol/L) | 25.11 ± 3.44 | 24.93 ± 3.71 | 25.14 ± 3.45 | 24.93 ± 3.43 | 25.12 ± 3.38 | 0.791 |
| Potassium (mmol/L) | 4.26 ±0.59 | 4.29 ± 0.55 | 4.26 ± 0.58 | 4.25 ± 0.62 | 4.25 ± 0.59 | 0.944 |
| Sodium (mmol/L) | 138.14 ± 3.77 | 138.67 ± 3.36 | 138.12 ± 3.68 | 138.05 ± 4.24 | 138.17 ± 3.85 | 0.428 |
| Anion gap (mmol/L) | 13.42 ± 3.14 | 13.46 ± 3.01 | 13.41 ± 3.22 | 13.36 ± 3.03 | 13.46 ± 2.97 | 0.962 |
| Glucose (mg/dl) | 135.60 ± 47.45 | 137.02 ± 49.18 | 135.41 ± 45.12 | 136.05 ± 53.09 | 135.73 ± 51.02 | 0.986 |
| Lymphocyte count (109/L) | 3.58 ± 5.23 | 1.06 ± 5.53 | 4.70 ± 3.84 | 4.03 ± 4.24 | 1.29 ± 1.62 | 0.249 |
| Neutrophil count (109/L) | 8.87 ± 4.62 | 10.14 ± 8.55 | 8.84 ± 4.03 | 7.97 ± 4.89 | 8.87 ± 4.96 | 0.571 |
| Treatment information, n (%) | ||||||
| Mechanical ventilation | 434.2 (10.8) | 7.8 (5.4) | 281.6 (10.9) | 47.7 (11.6) | 97.1 (11.1) | 0.226 |
| Renal replacement therapy | 1.5 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 1.5 (0.2) | 0.167 |
| Surgery | 0.441 | |||||
| Internal fixation | 2456.2 (61.0) | 99.7 (68.7) | 1579.9 (61.0) | 246.0 (59.7) | 530.6 (60.5) | |
| Hip replacement | 1568.1 (39.0) | 45.4 (31.3) | 1010.1 (39.0) | 165.9 (40.3) | 346.7 (39.5) | |
| Drug use, n (%) | ||||||
| Dopamine | 15.4 (0.4) | 0.0 (0.0) | 11.8 (0.5) | 0.9 (0.2) | 2.8 (0.3) | 0.725 |
| Epinephrine | 5.6 (0.1) | 0.0 (0.0) | 3.6 (0.1) | 0.5 (0.1) | 1.5 (0.2) | 0.891 |
| Furosemide | 130.3 (3.2) | 1.1 (0.8) | 83.3 (3.2) | 14.7 (3.6) | 31.2 (3.6) | 0.434 |
| Norepinephrine | 68.5 (1.7) | 0.0 (0.0) | 45.8 (1.8) | 6.3 (1.5) | 16.3 (1.9) | 0.588 |
| Phenylephrine | 132.9 (3.3) | 2.1 (1.5) | 87.0 (3.4) | 12.8 (3.1) | 31.0 (3.5) | 0.591 |
| Immunosuppressant | 452 (9.0) | 20 (9.7) | 204 (6.2) | 67 (13.3) | 161 (15.3) | 0.049 |
| Outcome | Non-Users (n = 145.04) | Cephalosporin Monotherapy Users (n = 2590.05) | Non-Cephalosporin Users (n = 411.86) | Cephalosporin Combination Therapy Users (n = 877.3) | All Patients (n = 4024.24) |
|---|---|---|---|---|---|
| 28-day mortality | |||||
| No. of participants/No. at risk (%) | 3.0/145.04 (2.00) | 100.4/2590.05 (3.90) | 24.4/411.86 (5.90) | 70.9/877.3 (8.10) | 198.7/4024.24 (4.90) |
| Rate (95% CI)―events/100 participant-day | 0.07 (0.01–0.19) | 0.14 (0.11–0.17) | 0.22 (0.14–0.33) | 0.31 (0.24–0.39) | 0.18 (0.16–0.21) |
| Pairwise hazard ratio (95% CI) | |||||
| None | 0.51 (0.15–1.74) | 0.33 (0.09–1.19) | 0.24 (0.07–0.78) * | ||
| Cephalosporins | 0.65 (0.34–1.24) | 0.46 (0.28–0.75) * | |||
| Other drugs | 0.71 (0.39–1.29) | ||||
| Cephalosporins combined with other drugs | |||||
| 90-day mortality | |||||
| No. of participants/No. at risk (%) | 18.1/145.04 (12.5) | 252.7/2590.05 (9.8) | 48.9/411.86 (11.9) | 135.2/877.3 (15.4) | 454.9/4024.24 (11.30) |
| Rate (95% CI)―events/100 participant-day | 0.13 (0.08–0.20) | 0.11 (0.10–0.13) | 0.15 (0.11–0.20) | 0.20 (0.16–0.23) | 0.13 (0.12–0.15) |
| Pairwise hazard ratio (95% CI) | |||||
| None | 1.32 (0.51–3.38) | 1.10 (0.40–2.98) | 0.79 (0.31–2.04) | ||
| Cephalosporins | 0.83 (0.54–1.30) | 0.60 (0.44–0.82) * | |||
| Other drugs | 0.72 (0.46–1.14) | ||||
| Cephalosporins combined with other drugs | |||||
| 180-day mortality | |||||
| No. of participants/No. at risk (%) | 26.3/145.04 (18.2) | 353.3/2590.05 (13.6) | 70.2/411.86 (17.0) | 169.0/877.3 (19.3) | 618.9/4024.24 (15.4) |
| Rate (95% CI)―events/100 participant-day | 0.10 (0.06–0.14) | 0.08 (0.07–0.09) | 0.11 (0.09–0.14) | 0.13 (0.11–0.15) | 0.09 (0.08–0.10) |
| Pairwise hazard ratio (95% CI) | |||||
| None | 1.37 (0.65–2.88) | 1.09 (0.49–2.43) | 0.92 (0.43–1.95) | ||
| Cephalosporins | 0.80 (0.55–1.16) | 0.67 (0.51–0.87) * | |||
| Other drugs | 0.84 (0.57–1.24) | ||||
| Cephalosporins combined with other drugs | |||||
| 1-year mortality | |||||
| No. of participants/No. at risk (%) | 32.2/145.04 (22.2) | 494.0/2590.05 (19.1) | 87.4/411.86 (21.2) | 221.1/877.3 (25.2) | 834.7/4024.24 (20.7) |
| Rate (95% CI)―events/100 participant-day | 0.06 (0.04–0.09) | 0.06 (0.05–0.07) | 0.07 (0.06–0.09) | 0.06 (0.05–0.07) | 0.09 (0.78–0.10) |
| Pairwise hazard ratio (95% CI) | |||||
| None | 1.22 (0.63–2.33) | 1.09 (0.54–2.21) | 0.86 (0.44–1.67) | ||
| Cephalosporins | 0.90 (0.64–1.26) | 0.71 (0.57–0.89) * | |||
| Other drugs | 0.79 (0.55–1.13) | ||||
| Cephalosporins combined with other drugs |
| Outcome | Non-Users (45.04) | Cephalosporin Monotherapy Users (n = 2590.05) | Non-Cephalosporin Users (n = 411.86) | Cephalosporin Combination Therapy Users (n = 877.3) | All Patients (n = 4024.24) |
|---|---|---|---|---|---|
| Infection | |||||
| No. of participants/No. at risk (%) | 0.8/145.04 (0.5) | 149.9/2590.05 (5.8) | 22.7/411.86 (5.5) | 52.9/877.3 (6.0) | 226.3/4024.24 (5.6) |
| Rate (95% CI)―events/100 participant-day | 0.11 (0.003–0.64) | 1.13 (0.96–1.33) | 0.81 (0.53–1.26) | 0.66 (0.50–0.87) | 0.91 (0.80–1.04) |
| Odds ratio (95% CI) | |||||
| None | 0.10 (0.01–1.38) | 0.10 (0.01–1.36) | 0.10 (0.01–1.33) | ||
| Cephalosporins | 0.94 (0.45–1.98) | 0.98 (0.61–1.55) | |||
| Other drugs | 1.04 (0.53–2.02) | ||||
| Cephalosporins combined with other drugs | |||||
| ICU admission | |||||
| No. of participants/No. at risk (%) | 9.4/145.04 (6.5) | 316.4/2590.05 (12.2) | 52.8/411.86 (12.8) | 111.7/877.3 (12.7) | 490.3/4024.24 (12.2) |
| Rate (95% CI)―events/100 participant-day | 1.34 (0.61–2.52) | 2.60 (2.33–2.90) | 2.36 (1.77–3.08) | 2.07 (1.71–2.49) | 2.40 (2.19–2.61) |
| Odds ratio (95% CI) | |||||
| None | 0.50 (0.26–0.96) | 0.44 (0.21–0.96) | 0.48 (0.25–0.91) | ||
| Cephalosporins | 0.89 (0.52–1.51) | 0.96 (0.70–1.32) | |||
| Other drugs | 1.08 (0.64–1.81) | ||||
| Cephalosporins combined with other drugs |
| Outcome | Non-Users (n = 185.28) | Cephalosporin Monotherapy Users (n = 3290.05) | Non-Cephalosporin Users (n = 514.62) | Cephalosporin Combination Therapy Users (n = 1037.47) | All Patients (n = 5027.42) |
|---|---|---|---|---|---|
| 28-day mortality | |||||
| No. of participants/no. at risk (%) | 3.0/185.28 (1.60) | 112.5/3290.05 (3.40) | 25.1/514.62 (4.90) | 72.3/1037.47 (7.00) | 213.0/5027.42 (4.20) |
| Rate (95% CI)―events/100 participant-day | 0.06 (0.12–0.17) | 0.12 (0.10–0.15) | 0.17 (0.11–0.26) | 0.29 (0.19–0.31) | 0.15 (0.13–0.17) |
| Pairwise hazard ratio (95% CI) | |||||
| None | 0.42 (0.11–1.57) | 0.27 (0.07–1.06) | 0.19 (0.05–0.68) * | ||
| Cephalosporins | 0.66 (0.34–1.28) | 0.45 (0.27–0.75) * | |||
| Other drugs | 0.68 (0.38–1.23) | ||||
| Cephalosporins combined with other drugs | |||||
| 90-day mortality | |||||
| No. of participants/no. at risk (%) | 19.0/185.28 (10.2) | 279.0/3290.05 (8.5) | 49.1/514.62 (9.5) | 136.5/1037.47 (13.2) | 483.5/5027.42 (9.60) |
| Rate (95% CI)―events/100 participant-day | 0.11 (0.07–0.18) | 0.02 (0.01–0.03) | 0.11 (0.08–0.14) | 0.15 (0.12–0.17) | 0.11 (0.10–0.12) |
| Pairwise hazard ratio (95% CI) | |||||
| None | 1.24 (0.55–2.76) | 0.99 (0.42–2.33) | 0.68 (0.31–1.53) | ||
| Cephalosporins | 0.80 (0.52–1.24) | 0.55 (0.40–0.76) * | |||
| Other drugs | 0.69 (0.45–1.06) | ||||
| Cephalosporins combined with other drugs | |||||
| 180-day mortality | |||||
| No. of participants/no. at risk (%) | 29.0/185.28 (15.7) | 392.0/3290.05 (11.9) | 71.9/514.62 (14.0) | 171.7/1037.47 (16.6) | 664.6/5027.42 (13.2) |
| Rate (95% CI)―events/100 participant-day | 0.09 (0.06–0.12) | 0.07 (0.06–0.08) | 0.08 (0.06–0.10) | 0.09 (0.08–0.11) | 0.07 (0.06–0.08) |
| Pairwise hazard ratio (95% CI) | |||||
| None | 1.33 (0.71–2.50) | 1.03 (0.52–2.03) | 0.80 (0.42–1.50) | ||
| Cephalosporins | 0.77 (0.53–1.11) | 0.60 (0.46–0.78) * | |||
| Other drugs | 1.13 (0.85–1.50) | ||||
| Cephalosporins combined with other drugs | |||||
| 1-year mortality | |||||
| No. of participants/no. at risk (%) | 41.8/185.28 (22.6) | 548.8/3290.05 (16.7) | 91.0/514.62 (17.7) | 226.8/1037.47 (21.9) | 908.4/5027.42 (18.1) |
| Rate (95% CI)―events/100 participant-day | 0.06 (0.04–0.08) | 0.05 (0.04–0.06) | 0.05 (0.04–0.06) | 0.06 (0.05–0.07) | 0.05 (0.04–0.053) |
| Pairwise hazard ratio (95% CI) | |||||
| None | 1.44 (0.83–2.52) | 1.18 (0.64–2.18) | 0.90 (0.51–1.59) | ||
| Cephalosporins | 0.82 (0.60–1.13) | 0.63 (0.50–0.78) * | |||
| Other drugs | 0.76 (0.54–1.07) | ||||
| Cephalosporins combined with other drugs |
| Outcome | Non-Users (n = 185.28) | Cephalosporin Monotherapy Users (n = 3290.05) | Non-Cephalosporin Users (n = 514.62) | Cephalosporin Combination Therapy Users (n = 1037.47) | All Patients (n = 5027.42) |
|---|---|---|---|---|---|
| Infection | |||||
| No. of participants/No. at risk (%) | 1.9/185.28 (1.0) | 209.5/3290.05 (6.4) | 36.8/514.62 (7.1) | 68.4/1037.47 (6.6) | 316.5/5027.42 (6.3) |
| Rate (95% CI)―events/100 participant-day | 0.17 (0.02–0.64) | 1.31 (1.15–1.51) | 1.31 (0.87–1.69) | 0.90 (0.69–1.13) | 1.14 (1.02–1.27) |
| Odds ratio (95% CI) | |||||
| None | 2.94 (0.41–21.38) | 8.97 (1.21–66.42) | 13.72 (1.90–99.31) | ||
| Cephalosporins | 1.14 (0.66–1.99) | 1.01 (0.74–1.38) | |||
| Other drugs | 0.87 (0.55–1.38) | ||||
| Cephalosporins combined with other drugs | |||||
| ICU admission | |||||
| No. of participants/No. at risk (%) | 17.4/185.28 (9.4) | 433.0/3290.05 (13.2) | 72.2 /514.62 (14.0) | 146.9/1037.47 (14.2) | 669.5/5027.42 (13.3) |
| Rate (95% CI)―events/100 participant-day | 2.01 (1.15–3.13) | 2.90 (2.64–3.19) | 2.65 (2.07–3.31) | 2.32 (1.96–2.72) | 2.69 (2.62–3.03) |
| Odds ratio (95% CI) | |||||
| None | 0.90 (0.50–1.64) | 1.38 (0.82–2.34) | 2.48 (1.56–3.96) | ||
| Cephalosporins | 1.15 (0.80–1.66) | 1.08 (0.86–1.35) | |||
| Other drugs | 0.95 (0.66–1.35) | ||||
| Cephalosporins combined with other drugs |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, H.; Wang, X.; Ou, Q.; Wang, J.; Ai, Z. The Long-Term Efficacy of Cephalosporin in Elderly Hip Fracture Patients: A Comprehensive Analysis. J. Clin. Med. 2025, 14, 6086. https://doi.org/10.3390/jcm14176086
Pan H, Wang X, Ou Q, Wang J, Ai Z. The Long-Term Efficacy of Cephalosporin in Elderly Hip Fracture Patients: A Comprehensive Analysis. Journal of Clinical Medicine. 2025; 14(17):6086. https://doi.org/10.3390/jcm14176086
Chicago/Turabian StylePan, Huiqing, Xiao Wang, Qingjian Ou, Juan Wang, and Zisheng Ai. 2025. "The Long-Term Efficacy of Cephalosporin in Elderly Hip Fracture Patients: A Comprehensive Analysis" Journal of Clinical Medicine 14, no. 17: 6086. https://doi.org/10.3390/jcm14176086
APA StylePan, H., Wang, X., Ou, Q., Wang, J., & Ai, Z. (2025). The Long-Term Efficacy of Cephalosporin in Elderly Hip Fracture Patients: A Comprehensive Analysis. Journal of Clinical Medicine, 14(17), 6086. https://doi.org/10.3390/jcm14176086






