Photon-Counting CT Enhances Diagnostic Accuracy in Stable Coronary Artery Disease: A Comparative Study with Conventional CT
Abstract
1. Introduction
2. Materials and Methods
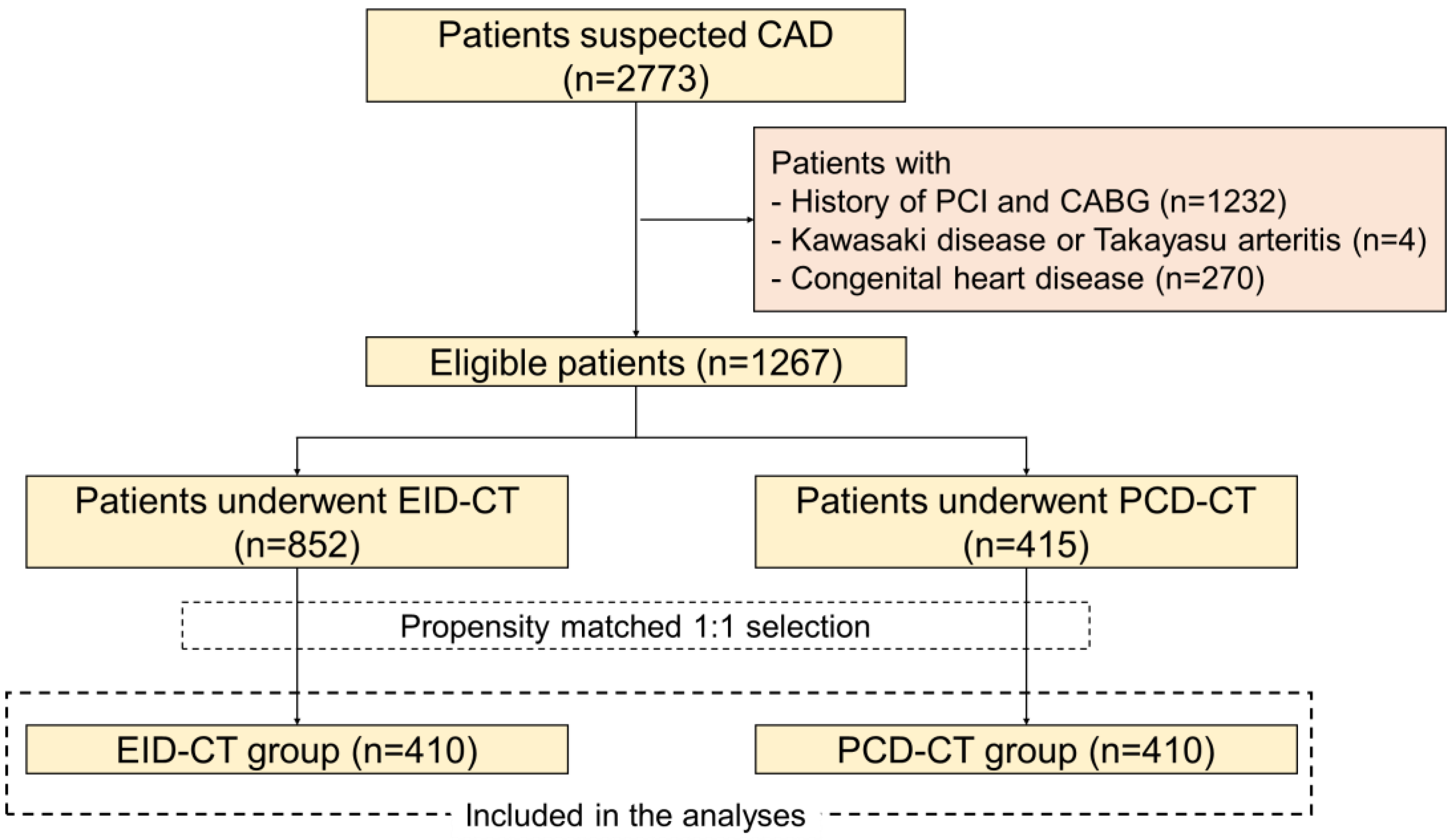
2.1. Study Design and Population
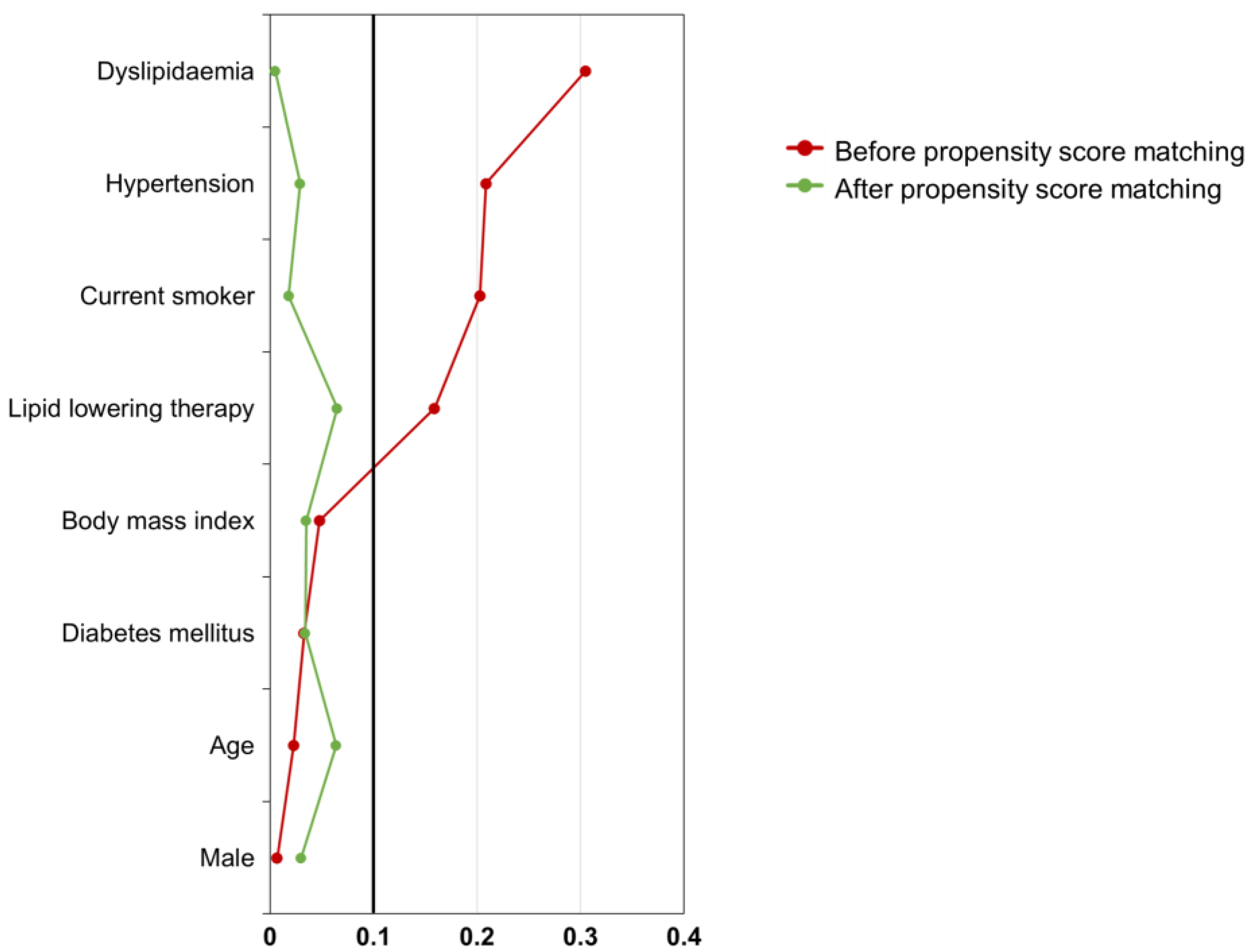
2.2. Patient Selection and Matching
2.3. CT Angiography Acquisition and Analysis
2.4. Clinical Endpoints
2.5. Statistical Analysis
3. Results
3.1. Patient Population
3.2. Coronary CTA Findings
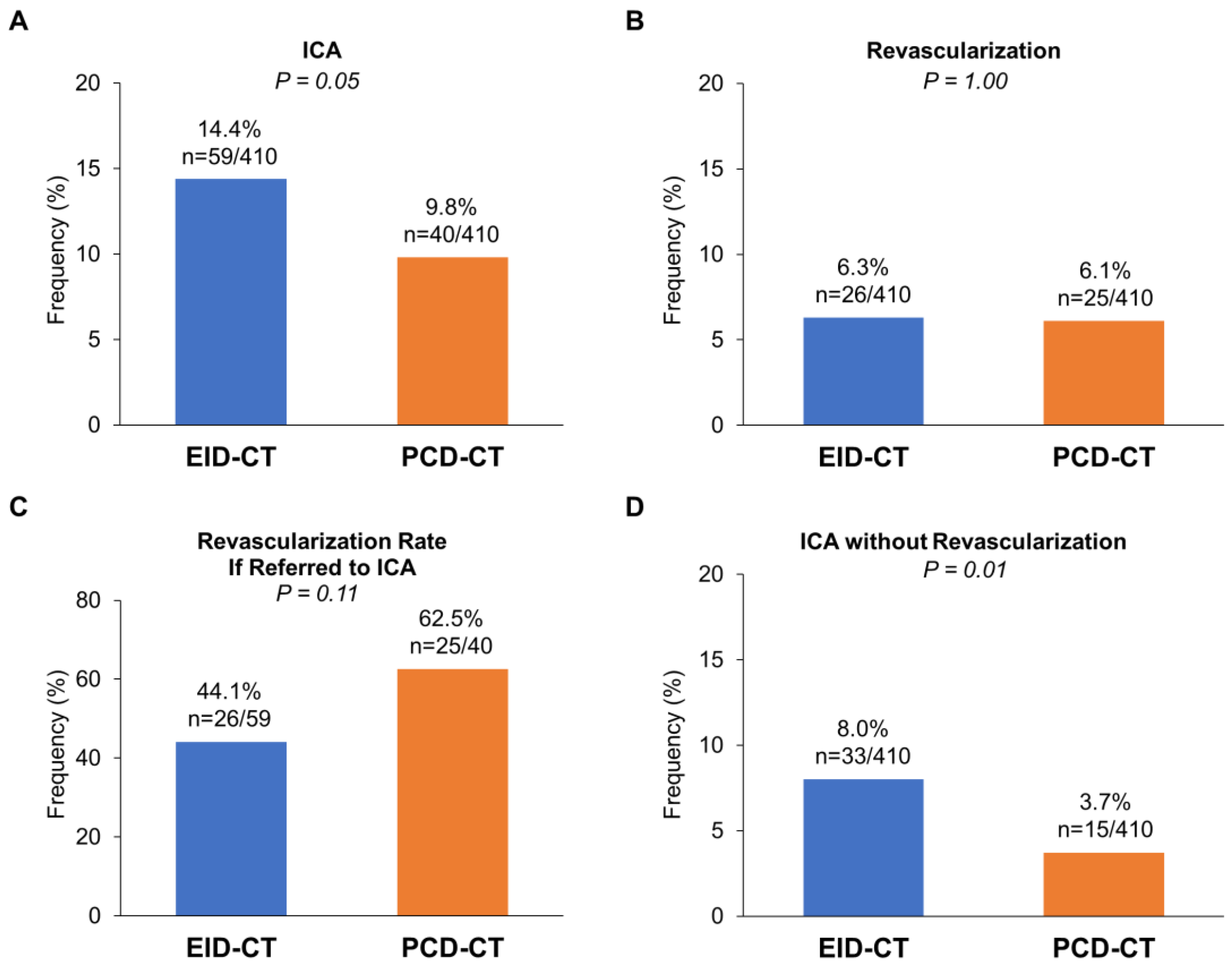
3.3. Downstream ICA and Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Prescott, E.; Storey, R.F.; Deaton, C.; Cuisset, T.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 2020, 41, 407–477. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.M.; Rochitte, C.E.; Dewey, M.; Arbab-Zadeh, A.; Niinuma, H.; Gottlieb, I.; Paul, N.; Clouse, M.E.; Shapiro, E.P.; Hoe, J.; et al. Diagnostic performance of coronary angiography by 64-row CT. N. Engl. J. Med. 2008, 359, 2324–2336. [Google Scholar] [CrossRef] [PubMed]
- Meijboom, W.B.; Meijs, M.F.; Schuijf, J.D.; Cramer, M.J.; Mollet, N.R.; van Mieghem, C.A.; Nieman, K.; van Werkhoven, J.M.; Pundziute, G.; Weustink, A.C.; et al. Diagnostic accuracy of 64-slice computed tomography coronary angiography: A prospective, multicenter, multivendor study. J. Am. Coll. Cardiol. 2008, 52, 2135–2144. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.J.; Blankstein, R.; Bax, J.J.; Ferencik, M.; Bittencourt, M.S.; Min, J.K.; Berman, D.S.; Leipsic, J.; Villines, T.C.; Dey, D.; et al. Society of Cardiovascular Computed Tomography/North American Society of Cardiovascular Imaging—Expert Consensus Document on Coronary CT Imaging of Atherosclerotic Plaque. J. Cardiovasc. Comput. Tomogr. 2021, 15, 93–109. [Google Scholar] [CrossRef] [PubMed]
- Willemink, M.J.; Persson, M.; Pourmorteza, A.; Pelc, N.J.; Fleischmann, D. Photon-counting CT: Technical Principles and Clinical Prospects. Radiology 2018, 289, 293–312. [Google Scholar] [CrossRef] [PubMed]
- Hagar, M.T.; Soschynski, M.; Saffar, R.; Rau, A.; Taron, J.; Weiss, J.; Stein, T.; Faby, S.; von Zur Muehlen, C.; Ruile, P.; et al. Accuracy of Ultrahigh-Resolution Photon-counting CT for Detecting Coronary Artery Disease in a High-Risk Population. Radiology 2023, 307, e223305. [Google Scholar] [CrossRef] [PubMed]
- McCollough, C.H.; Rajiah, P.; Bois, J.P.; Winfree, T.N.; Carter, R.E.; Rajendran, K.; Williamson, E.E.; Thorne, J.E.; Leng, S. Comparison of Photon-counting Detector and Energy-integrating Detector CT for Visual Estimation of Coronary Percent Luminal Stenosis. Radiology 2023, 309, e230853. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Shin, D.; Singh, M.; Malik, S.; Dakroub, A.; Sami, Z.; Weber, J.; Cao, J.J.; Parikh, R.; Chen, L.; et al. Diagnostic Performance and Clinical Impact of Photon-Counting Detector Computed Tomography in Coronary Artery Disease. J. Am. Coll. Cardiol. 2025, 85, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.; Hrenko, A.; Kerkovits, N.M.; Nagy, K.; Vertes, M.; Balogh, H.; Nagy, N.; Munkacsi, T.; Emrich, T.; Varga-Szemes, A.; et al. Photon-counting detector CT reduces the rate of referrals to invasive coronary angiography as compared to CT with whole heart coverage energy-integrating detector. J. Cardiovasc. Comput. Tomogr. 2024, 18, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Vecsey-Nagy, M.; Tremamunno, G.; Schoepf, U.J.; Gnasso, C.; Zsarnoczay, E.; Fink, N.; Kravchenko, D.; Halfmann, M.C.; Laux, G.S.; O’Doherty, J.; et al. Intraindividual Comparison of Ultrahigh-Spatial-Resolution Photon-Counting Detector CT and Energy-Integrating Detector CT for Coronary Stenosis Measurement. Circ. Cardiovasc. Imaging 2024, 17, e017112. [Google Scholar] [CrossRef] [PubMed]
- Nishihara, T.; Miyoshi, T.; Nakashima, M.; Akagi, N.; Morimitsu, Y.; Inoue, T.; Miki, T.; Yoshida, M.; Toda, H.; Nakamura, K.; et al. Diagnostic improvements of calcium-removal image reconstruction algorithm using photon-counting detector CT for calcified coronary lesions. Eur. J. Radiol. 2024, 172, 111354. [Google Scholar] [CrossRef] [PubMed]
- Koons, E.K.; Rajiah, P.S.; Thorne, J.E.; Weber, N.M.; Kasten, H.J.; Shanblatt, E.R.; McCollough, C.H.; Leng, S. Coronary artery stenosis quantification in patients with dense calcifications using ultra-high-resolution photon-counting-detector computed tomography. J. Cardiovasc. Comput. Tomogr. 2024, 18, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.C. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat. Med. 2009, 28, 3083–3107. [Google Scholar] [CrossRef] [PubMed]
- Agatston, A.S.; Janowitz, W.R.; Hildner, F.J.; Zusmer, N.R.; Viamonte, M., Jr.; Detrano, R. Quantification of coronary artery calcium using ultrafast computed tomography. J. Am. Coll. Cardiol. 1990, 15, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Cury, R.C.; Leipsic, J.; Abbara, S.; Achenbach, S.; Berman, D.; Bittencourt, M.; Budoff, M.; Chinnaiyan, K.; Choi, A.D.; Ghoshhajra, B.; et al. CAD-RADS™ 2.0—2022 Coronary Artery Disease-Reporting and Data System: An Expert Consensus Document of the Society of Cardiovascular Computed Tomography (SCCT), the American College of Cardiology (ACC), the American College of Radiology (ACR), and the North America Society of Cardiovascular Imaging (NASCI). J. Cardiovasc. Comput. Tomogr. 2022, 16, 536–557. [Google Scholar] [CrossRef] [PubMed]
- Clark, D.P.; Holbrook, M.; Lee, C.L.; Badea, C.T. Photon-counting cine-cardiac CT in the mouse. PLoS ONE 2019, 14, e0218417. [Google Scholar] [CrossRef] [PubMed]
- Vecsey-Nagy, M.; Tremamunno, G.; Schoepf, U.J.; Gnasso, C.; Zsarnoczay, E.; Fink, N.; Kravchenko, D.; Halfmann, M.C.; O’Doherty, J.; Szilveszter, B.; et al. Coronary Plaque Quantification with Ultrahigh-Spatial-Resolution Photon-counting Detector CT: Intraindividual Comparison with Energy-integrating Detector CT. Radiology 2025, 314, e241479. [Google Scholar] [CrossRef]
- McCollough, C.H.; Rajendran, K.; Baffour, F.I.; Diehn, F.E.; Ferrero, A.; Glazebrook, K.N.; Horst, K.K.; Johnson, T.F.; Leng, S.; Mileto, A.; et al. Clinical applications of photon counting detector CT. Eur. Radiol. 2023, 33, 5309–5320. [Google Scholar] [CrossRef]
- McCollough, C.H.; Rajendran, K.; Leng, S.; Yu, L.; Fletcher, J.G.; Stierstorfer, K.; Flohr, T.G. The technical development of photon-counting detector CT. Eur. Radiol. 2023, 33, 5321–5330. [Google Scholar] [CrossRef] [PubMed]
- Motoyama, S.; Kondo, T.; Sarai, M.; Sugiura, A.; Harigaya, H.; Sato, T.; Inoue, K.; Okumura, M.; Ishii, J.; Anno, H.; et al. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J. Am. Coll. Cardiol. 2007, 50, 319–326. [Google Scholar] [CrossRef] [PubMed]


| Before Propensity Score Matching | After Propensity Score Matching | |||||
|---|---|---|---|---|---|---|
| Variables | EID-CT | PCD-CT | SMD | EID-CT | PCD-CT | SMD |
| N | 852 | 415 | 410 | 410 | ||
| Age, years | 66.9 ± 12.9 | 67.2 ± 13.4 | 0.02 | 68.0 ± 12.5 | 67.1 ± 13.3 | 0.06 |
| Male | 531 (62.3) | 260 (62.7) | <0.01 | 263 (64.1) | 257 (62.7) | 0.03 |
| Body mass index, kg/m2 | 24.2 ± 4.5 | 24.5 ± 6.9 | 0.05 | 24.3 ± 4.5 | 24.2 ± 4.4 | 0.04 |
| Hypertension | 535 (62.8) | 218 (52.5) | 0.21 | 224 (54.6) | 218 (53.2) | 0.03 |
| Diabetes mellitus | 234 (27.5) | 108 (26.0) | 0.03 | 99 (24.1) | 105 (25.6) | 0.03 |
| Dyslipidaemia | 394 (46.2) | 131 (31.6) | 0.31 | 132 (32.2) | 131 (32.0) | <0.01 |
| Current smoker | 127 (14.9) | 35 (8.4) | 0.20 | 33 (8.0) | 35 (8.5) | 0.02 |
| Lipid-lowering therapy | 316 (37.1) | 123 (29.6) | 0.16 | 109 (26.6) | 121 (29.5) | 0.07 |
| c | EID-CT (N = 410) | PCD-CT (N = 410) | p Value |
|---|---|---|---|
| Highest stenosis severity | 0.033 | ||
| <25% (CAD-RAD 0–1) | 222 (54.1) | 244 (59.5) | |
| 25–49% (CAD-RAD 2) | 63 (15.4) | 66 (16.1) | |
| 50–69% (CAD-RAD 3) | 53 (12.9) | 38 (9.3) | |
| 70–99% (CAD-RAD 4) | 64 (15.6) | 53 (12.9) | |
| 100% (CAD-RAD 5) | 8 (2.0) | 9 (2.2) | |
| High-risk plaque | 125 (30.5) | 93 (22.7) | 0.014 |
| CACS | 55 (0, 334) | 65 (0, 435) | 0.60 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakashima, M.; Miyoshi, T.; Hara, S.; Miyagi, R.; Nishihara, T.; Miki, T.; Osawa, K.; Yuasa, S. Photon-Counting CT Enhances Diagnostic Accuracy in Stable Coronary Artery Disease: A Comparative Study with Conventional CT. J. Clin. Med. 2025, 14, 6049. https://doi.org/10.3390/jcm14176049
Nakashima M, Miyoshi T, Hara S, Miyagi R, Nishihara T, Miki T, Osawa K, Yuasa S. Photon-Counting CT Enhances Diagnostic Accuracy in Stable Coronary Artery Disease: A Comparative Study with Conventional CT. Journal of Clinical Medicine. 2025; 14(17):6049. https://doi.org/10.3390/jcm14176049
Chicago/Turabian StyleNakashima, Mitsutaka, Toru Miyoshi, Shohei Hara, Ryosuke Miyagi, Takahiro Nishihara, Takashi Miki, Kazuhiro Osawa, and Shinsuke Yuasa. 2025. "Photon-Counting CT Enhances Diagnostic Accuracy in Stable Coronary Artery Disease: A Comparative Study with Conventional CT" Journal of Clinical Medicine 14, no. 17: 6049. https://doi.org/10.3390/jcm14176049
APA StyleNakashima, M., Miyoshi, T., Hara, S., Miyagi, R., Nishihara, T., Miki, T., Osawa, K., & Yuasa, S. (2025). Photon-Counting CT Enhances Diagnostic Accuracy in Stable Coronary Artery Disease: A Comparative Study with Conventional CT. Journal of Clinical Medicine, 14(17), 6049. https://doi.org/10.3390/jcm14176049






