Pharmacological Modulation of Pupil Size in Presbyopia: Optical Modeling and Clinical Implications
Abstract
1. Introduction
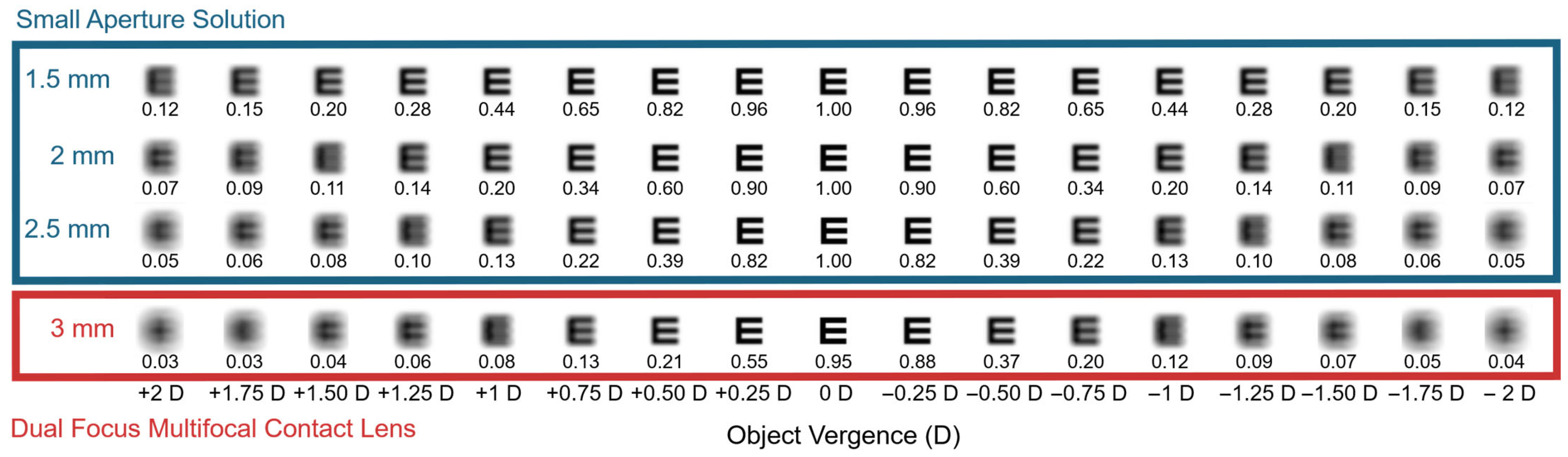
2. Theoretical Pupil Size Targets
3. Pupil Sizes Achieved in Presbyopia Pilocarpine Clinical Studies
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Markoulli, M.; Fricke, T.R.; Arvind, A.; Frick, K.D.; Hart, K.M.; Joshi, M.R.; Kandel, H.; Macedo, A.F.; Makrynioti, D.; Retallic, N.; et al. BCLA CLEAR Presbyopia: Epidemiology and impact. Contact Lens Anterior Eye 2024, 47, 102157. [Google Scholar] [CrossRef]
- Wolffsohn, J.S.; Naroo, S.A.; Bullimore, M.A.; Craig, J.P.; Davies, L.N.; Markoulli, M.; Schnider, C.; Morgan, P.B. BCLA CLEAR Presbyopia: Definitions. Contact Lens Anterior Eye 2024, 47, 102155. [Google Scholar] [CrossRef]
- Davies, L.N.; Biswas, S.; Bullimore, M.; Cruickshank, F.; Estevez, J.J.; Khanal, S.; Kollbaum, P.; Marcotte-Collard, R.; Montani, G.; Plainis, S.; et al. BCLA CLEAR presbyopia: Mechanism and optics. Contact Lens Anterior Eye 2024, 47, 102185. [Google Scholar] [CrossRef] [PubMed]
- Stokes, J.; Shirneshan, E.; Graham, C.A.; Paulich, M.; Johnson, N. Exploring the Experience of Living with and Managing Presbyopia. Optom. Vis. Sci. 2022, 99, 635–644. [Google Scholar] [CrossRef]
- Brujic, M.; Kruger, P.; Todd, J.; Barnes, E.; Wuttke, M.; Perna, F.; Aliò, J. Living with presbyopia: Experiences from a virtual roundtable dialogue among impacted individuals and healthcare professionals. BMC Ophthalmol. 2022, 22, 204. [Google Scholar] [CrossRef]
- Katz, J.A.; Karpecki, P.M.; Dorca, A.; Chiva-Razavi, S.; Floyd, H.; Barnes, E.; Wuttke, M.; Donnenfeld, E. Presbyopia-A Review of Current Treatment Options and Emerging Therapies. Clin. Ophthalmol. 2021, 15, 2167–2178. [Google Scholar] [CrossRef]
- Pepose, J.S.; Hartman, P.J.; DuBiner, H.B.; Abrams, M.A.; Smyth-Medina, R.J.; Moroi, S.E.; Meyer, A.R.; Sooch, M.P.; Jaber, R.M.; Charizanis, K.; et al. Phentolamine mesylate ophthalmic solution provides lasting pupil modulation and improves near visual acuity in presbyopic glaucoma patients in a randomized phase 2b clinical trial. Clin. Ophthalmol. 2021, 15, 79–91. [Google Scholar] [CrossRef]
- Abdelkader, A. Improved Presbyopic Vision With Miotics. Eye Contact Lens 2015, 41, 323–327. [Google Scholar] [CrossRef]
- Abdelkader, A.; Kaufman, H.E. Clinical outcomes of combined versus separate carbachol and brimonidine drops in correcting presbyopia. Eye Vis. 2016, 3, 31. [Google Scholar] [CrossRef]
- Mousavi, S.A.; Rajavi, Z.; Sabbaghi, H.; Abdi, S.; Montazerin, N.; Kheiri, B.; Monfared, A.H.; Sheibani, K.; Jaldian, H.B. Pharmacological Treatment of Presbyopia Using Pilocarpine 1.25% Eye Drops. J. Ophthalmic Vis. Res. 2024, 19, 468–475. [Google Scholar] [CrossRef]
- Ehlers, J.P.; Shah, C.P.; Wills Eye Hospital (Philadelphia Pa.). The Wills Eye Manual: Office and Emergency Room Diagnosis and Treatment of Eye Disease, 5th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2008. [Google Scholar]
- Haghpanah, N.; Alany, R. Pharmacological treatment of presbyopia: A systematic review. Eur. J. Transl. Myol. 2022, 32, 10781. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Sebastian, S.C.; Yadav, H.; Khan, I.J.; Kamath, V. The effect of 2% pilocarpine on depth of field in different time intervals among presbyopic subjects. Indian J. Ophthalmol. 2024, 72, 1795–1797. [Google Scholar] [CrossRef] [PubMed]
- Kannarr, S.; El-Harazi, S.M.; Moshirfar, M.; Lievens, C.; Kim, J.L.; Peace, J.H.; Safyan, E.; Liu, H.; Zheng, S.; Robinson, M.R. Safety and Efficacy of Twice-Daily Pilocarpine HCl in Presbyopia: The Virgo Phase 3, Randomized, Double-Masked, Controlled Study. Am. J. Ophthalmol. 2023, 253, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Farid, M.; Rowen, S.L.; Moshirfar, M.; Cunningham, D.; Gaddie, I.B.; Smits, G.; Ignacio, T.; Gupta, P.K. Combination Low-Dose Pilocarpine/Diclofenac Sodium and Pilocarpine Alone for Presbyopia: Results of a Randomized Phase 2b Clinical Trial. Clin. Ophthalmol. 2024, 18, 3425–3439. [Google Scholar] [CrossRef]
- Holland, E.; Karpecki, P.; Fingeret, M.; Schaeffer, J.; Gupta, P.; Fram, N.; Smits, G.; Ignacio, T.; Lindstrom, R. Efficacy and Safety of CSF-1 (0.4% Pilocarpine Hydrochloride) in Presbyopia: Pooled Results of the NEAR Phase 3 Randomized, Clinical Trials. Clin. Ther. 2024, 46, 104–113. [Google Scholar] [CrossRef]
- Cobb, P.W. The influence of illumination of the eye on visual acuity. J. Physiol. 1911, 29, 76–99. [Google Scholar] [CrossRef]
- Campbell, F.W.; Gregory, A.H. Effect of Size of Pupil on Visual Acuity. Nature 1960, 187, 1121–1123. [Google Scholar] [CrossRef]
- Atchison, D.; Smith, G.; Efron, N. The effect of pupil size on visual acuity in uncorrected and corrected myopia. Am J Optom Physiol Opt. 1979, 56, 315–323. [Google Scholar] [CrossRef]
- Xu, R.; Thibos, L.; Bradley, A. Effect of Target Luminance on Optimum Pupil Diameter for Presbyopic Eyes. Optom. Vis. Sci. 2016, 93, 1409–1419. [Google Scholar] [CrossRef]
- Xu, R.; Wang, H.; Jaskulski, M.; Kollbaum, P.; Bradley, A. Small-pupil versus multifocal strategies for expanding depth of focus of presbyopic eyes. J. Cataract. Refract. Surg. 2019, 45, 647–655. [Google Scholar] [CrossRef]
- De Gracia, P. Fourier tools for the evaluation of refractive multifocal designs. Sci. Rep. 2023, 13, 22585. [Google Scholar] [CrossRef]
- Lazar, R.; Degen, J.; Fiechter, A.-S.; Monticelli, A.; Spitschan, M. Regulation of pupil size in natural vision across the human lifespan. R. Soc. Open Sci. 2024, 11, 191613. [Google Scholar] [CrossRef]
- Benozzi, J.; Benozzi, G.; Orman, B. Presbyopia: A new potential pharmacological treatment. Med. Hypothesis Discov. Innov. Ophthalmol. 2012, 1, 3–5. [Google Scholar] [PubMed]
- Benozzi, G.; Perez, C.; Leiro, J.; Facal, S.; Orman, B. Presbyopia Treatment With Eye Drops: An Eight Year Retrospective Study. Transl. Vis. Sci. Technol. 2020, 9, 25. [Google Scholar] [CrossRef] [PubMed]
- Benozzi, G.; Cortina, M.E.; Gimeno, E.; Vantesone, D.L.; Solas, A.E.; Lorda, G.M.; Facal, S.; Leiro, J.; Orman, B. A multicentric study of pharmacological treatment for presbyopia. Graefe’s Arch. Clin. Exp. Ophthalmol. 2021, 259, 2441–2450. [Google Scholar] [CrossRef] [PubMed]
- Waring, G.O.; Price, F.W.; Wirta, D.; McCabe, C.; Moshirfar, M.; Guo, Q.; Gore, A.; Liu, H.; Safyan, E.; Robinson, M.R. Safety and Efficacy of AGN-190584 in Individuals With Presbyopia: The GEMINI 1 Phase 3 Randomized Clinical Trial. JAMA Ophthalmol. 2022, 140, 363–371. [Google Scholar] [CrossRef]
- Lievens, C.W.; Hom, M.M.O.; McLaurin, E.B.; Yuan, J.; Safyan, E.B.; Liu, H. Pilocarpine HCl 1.25% for treatment of presbyopia after laser vision correction: Pooled analysis of two phase 3 randomized trials (GEMINI 1 and 2). J. Cataract. Refract. Surg. 2024, 50, 57–63. [Google Scholar] [CrossRef]
- Waring, G.O.; Brujic, M.; McGee, S.; Micheletti, J.M.; Zhao, C.; Schachter, S.; Liu, H.; Safyan, E. Impact of presbyopia treatment pilocarpine hydrochloride 1.25% on night-driving performance. Clin. Exp. Optom. 2024, 107, 665–672. [Google Scholar] [CrossRef]
- Vargas, V.; Vejarano, F.; Alio, J.L. Near Vision Improvement with the Use of a New Topical Compound for Presbyopia Correction: A Prospective, Consecutive Interventional Non-Comparative Clinical Study. Ophthalmol. Ther. 2019, 8, 31–39. [Google Scholar] [CrossRef]
- Jones, L.; Downie, L.E.; Korb, D.; Benitez-Del-Castillo, J.M.; Dana, R.; Deng, S.X.; Dong, P.N.; Geerling, G.; Hida, R.Y.; Liu, Y.; et al. TFOS DEWS II Management and Therapy Report. Ocul. Surf. 2017, 15, 575–628. [Google Scholar] [CrossRef]
- Marsack, J.D.; Thibos, L.N.; Applegate, R.A. Metrics of optical quality derived from wave aberrations predict visual performance. J. Vis. 2004, 4, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Bradley, A.; Thibos, L.N. Predicting subjective judgment of best focus with objective image quality metrics. J. Vis. 2004, 4, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Kiel, M.; Grabitz, S.D.; Hopf, S.; Koeck, T.; Wild, P.S.; Schmidtmann, I.; Lackner, K.J.; Münzel, T.; Beutel, M.E.; Pfeiffer, N.; et al. Distribution of Pupil Size and Associated Factors: Results from the Population-Based Gutenberg Health Study. J. Ophthalmol. 2022, 2022, 9520512. [Google Scholar] [CrossRef] [PubMed]

| Study | Inclusion Criteria—Age (Years) | Inclusion Criteria—Refractive Error (D) | Sample Size | Treatment | Pupil Size (Baseline) | Pupil Size (Post-Treatment) | Significant | Post-Drop Application Time (Hours) | Lighting Conditions | Quality |
|---|---|---|---|---|---|---|---|---|---|---|
| Benozzi Method | ||||||||||
| Benozzi et al., 2012 [24] | 45–50 | Emmetropic Eyes | 100 | 1.0% Pilocarpine + 0.1% Diclofenac, (BID) | Not Collected | Not Collected | N/A | Not Collected | N/A | Level 3 |
| Benozzi et al., 2020 [25] | 40–60 | −0.50 D to +1.00 D with ≤1.00 D of cylinder | 910 | 1.0% Pilocarpine + 0.1% Diclofenac, (BID) | Not Collected | Not Collected | N/A | Not Collected | N/A | Level 3 |
| Benozzi et al., 2021 [26] | 40–60 | −0.50 D to +2.00 D with ≤1.50 D of cylinder | 148 | 1.0% Pilocarpine + 0.1% Diclofenac, (BID) | Not Collected | Not Collected | N/A | Not Collected | N/A | Level 3 |
| 1.25% pilocarpine (VUITY®; AbbVie) | ||||||||||
| Waring et al., 2022 [27] | 40–55 | −4.00 D to +1.00 D with ≤2.00 D of cylinder | 163 | 1.25% pilocarpine HCl, (QD) | 3.40 # | 1.86 # | N/T | 1 | Photopic | Level 1 |
| 160 | Vehicle, (QD) | 3.21 # | 3.09 # | N/T | ||||||
| Kannarr et al., 2023 [14] | 40–55 | −4.00 D to +1.00 D with ≤2.00 D of cylinder | 114 | 1.25% pilocarpine HCl, (BID) | 3.21 # | 1.97 # | Yes | 1 | Photopic | Level 1 |
| 116 | Vehicle, (BID) | 3.04 # | 3.03 # | No | ||||||
| Lievens et al., 2024 † [28] | 40–55 | −4.00 D to +1.00 D with ≤2.00 D of cylinder | 39 | 1.25% pilocarpine HCl, (QD) | Not Reported | Not Reported | N/A | Not Reported | N/A | Level 1 |
| 41 | Vehicle, (QD) | Not Reported | Not Reported | N/A | ||||||
| Mousavi et al., 2024 [10] | 40–60 | ≤1.00 D | 45 | 1.25% pilocarpine HCl, (QD) ‡ (Unilateral Treatment) | 2.68 ± 0.81 | 1.23 ± 0.58 | Yes | 1–2 | Mesopic | Level 2 |
| 30 | 1.25% pilocarpine HCl, (QD) (Unilateral Treatment) | 2.78 ± 0.43 | 1.68 ± 0.50 | Yes | ||||||
| Waring et al., 2024 [29] | 40–55 | −4.00 D to +1.00 D with ≤2.00 D of cylinder | 43 | 1.25% pilocarpine HCl, (QD) | Not Collected | Not Collected | N/A | Not Collected | N/A | Level 1 |
| 43 | Placebo | Not Collected | Not Collected | N/A | ||||||
| 0.4% Pilocarpine (Qlosi; Orasis Pharmaceuticals) | ||||||||||
| Farid et al., 2024 [15] | 45–64 | −4.50 D to +2.00 D with <2.00 D of cylinder | 55 | 0.2% pilocarpine HCl (BID), days 8 | 3.17 # | 2.37 # | Yes | 1 | Mesopic | Level 1 |
| 0.4% pilocarpine HCl (CSF-1), (BID), days 15 | 3.02 # | 2.12 # | Yes | |||||||
| 53 | 0.2% pilocarpine HCl/0.006% diclofenac sodium (BID), days 8 | 3.28 # | 2.46 # | Yes | ||||||
| 0.4% pilocarpine HCl/0.006% diclofenac sodium (BID), days 15 | 3.13 # | 2.22 # | Yes | |||||||
| 58 | 0.006% diclofenac sodium (BID), days 8 | 3.46 # | 3.37 # | No | ||||||
| 0.006% diclofenac sodium (BID), days 15 | 3.42 # | 3.30 # | No | |||||||
| Holland et al., 2024 [16] | 45–64 | −4.50 D to +2.00 D with <2.00 D of cylinder | 309 | 0.4% pilocarpine HCl (CSF-1), (BID) | 3.36 | 2.30 ± 0.59 | Yes | 1 | Scotopic | Level 1 |
| 304 | Vehicle, (BID) | 3.51 | 3.43 ± 0.99 | No | ||||||
| Additional Pilocarpine Formulations | ||||||||||
| Vargas et al., 2019 [30] | 41–65 | −0.50 D to +1.50 D with <1.50 D of cylinder | 117 | 0.247% pilocarpine + 0.78% phenylephrine + 0.09% polyethyleneglycol + 0.023% nepafenac + 0.034% pheniramine + 0.003% naphazoline, (QD) | 3.30 | 3.05 | Yes | 2 | Photopic | Level 2 |
| Tripathi et al., 2024 [13] | 40–55 | Not Reported | 120 | 2.0% pilocarpine | 2.34 ± 0.37 | 1.57 ± 0.34 | Yes | 0.75 | Photopic | Level 2 |
| Charbachol Formulations | ||||||||||
| Abdelkader et al., 2015 [8] | 43–56 | ±0.25 D (spherical equivalent) with ≤0.25 D of cylinder | 30 | 2.25% Carbachol + 0.2% Brimonidine, (QD) | ≥50 Years: 4.77 <50 Years: 4.72 | ≥50 Years: 2.50 <50 Years: 2.61 | Yes Yes | 1 | Not Reported | Level 1 |
| 18 | Placebo | ≥50 Years: 4.57 <50 Years: 4.80 | ≥50 Years: 4.48 <50 Years: 4.78 | No No | ||||||
| Abdelkader et al., 2016 [9] | 42–58 | ±0.25 D (spherical equivalent) with ≤0.25 D of cylinder | 10 | 3.0% Carbachol + 0.2% Brimonidine, (QD) | 4.30 ± 0.50 | 1.20 ± 0.30 | Yes | 1 | Not Reported | Level 1 |
| 10 | 3.0% Carbachol, (QD) followed by 0.2% Brimonidine, (QD) | 4.30 ± 0.50 | 1.90 ± 0.30 | Yes | ||||||
| 10 | 3.0% Carbachol, (QD) | 4.30 ± 0.50 | 2.80 ± 0.50 | Yes | ||||||
| 10 | 0.2% Brimonidine, (QD) | 4.30 ± 0.50 | 3.95 ± 0.50 | No | ||||||
| Phentolamine Mesylate Formulations | ||||||||||
| Pepose et al., 2021 [7] | ≥18 Years δ | Not Reported | 19 | 1.0% Phentolamine Mesylate, (QD) | 4.69 ± 0.95 | 3.71 ± 0.81 | Yes | 10–12 | Photopic | Level 1 |
| 20 | Vehicle | Non-Significant Change | N/A | |||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Gracia, P.; Pucker, A.D. Pharmacological Modulation of Pupil Size in Presbyopia: Optical Modeling and Clinical Implications. J. Clin. Med. 2025, 14, 6040. https://doi.org/10.3390/jcm14176040
De Gracia P, Pucker AD. Pharmacological Modulation of Pupil Size in Presbyopia: Optical Modeling and Clinical Implications. Journal of Clinical Medicine. 2025; 14(17):6040. https://doi.org/10.3390/jcm14176040
Chicago/Turabian StyleDe Gracia, Pablo, and Andrew D. Pucker. 2025. "Pharmacological Modulation of Pupil Size in Presbyopia: Optical Modeling and Clinical Implications" Journal of Clinical Medicine 14, no. 17: 6040. https://doi.org/10.3390/jcm14176040
APA StyleDe Gracia, P., & Pucker, A. D. (2025). Pharmacological Modulation of Pupil Size in Presbyopia: Optical Modeling and Clinical Implications. Journal of Clinical Medicine, 14(17), 6040. https://doi.org/10.3390/jcm14176040





