Effect of Prophylactic Phenylephrine Infusion Versus Interventional Ephedrine Boluses on Umbilical Blood pH in Cesarean Deliveries Under Spinal Anesthesia: A Retrospective Case-Control Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Spinal Anesthesia Technique and Management
2.3. Hypotension Management
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SBP | Systolic Blood Pressure |
| MAP | Mean Arterial Pressure |
| CI | Confidence Interval |
| HR | Heart Rate |
| PIH | Pregnancy-Induced Hypertension |
References
- Loubert, C. Fluid and vasopressor management for cesarean delivery under spinal anesthesia: Continuing professional development. Can. J. Anaesth. 2012, 59, 604–619. [Google Scholar] [CrossRef] [PubMed]
- Greiss, F.C.; van Wilkes, D. Effects of sympathomimetic drugs and angiotensin on the uterine vascular bed. Obstet. Gynecol. 1964, 23, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Ngan Kee, W.D.; Lee, A.; Khaw, K.S.; Ng, F.F.; Karmakar, M.K.; Gin, T. A randomized double-blinded comparison of phenylephrine and ephedrine infusion combinations to maintain blood pressure during spinal anesthesia for cesarean delivery: The effects on fetal acid-base status and hemodynamic control. Anesth. Analg. 2008, 107, 1295–1302. [Google Scholar] [CrossRef]
- Thomas, D.G.; Robson, S.C.; Redfern, N.; Hughes, D.; Boys, R.J. Randomized trial of bolus phenylephrine or ephedrine for maintenance of arterial pressure during spinal anaesthesia for Caesarean section. Br. J. Anaesth. 1996, 76, 61–65. [Google Scholar] [CrossRef]
- Cooper, D.W.; Carpenter, M.; Mowbray, P.; Desira, W.R.; Ryall, D.M.; Kokri, M.S. Fetal and maternal effects of phenylephrine and ephedrine during spinal anesthesia for cesarean delivery. Anesthesiology 2002, 97, 1582–1590. [Google Scholar] [CrossRef]
- Kinsella, S.M.; Carvalho, B.; Dyer, R.A.; Fernando, R.; McDonnell, N.; Mercier, F.J.; Palanisamy, A.; Sia, A.T.H.; Van de Velde, M.; Vercueil, A.; et al. International consensus statement on the management of hypotension with vasopressors during caesarean section under spinal anaesthesia. Anaesthesia 2018, 73, 71–92. [Google Scholar] [CrossRef]
- Xu, C.; Liu, S.; Huang, Y.; Guo, X.; Xiao, H.; Qi, D. Phenylephrine vs ephedrine in cesarean delivery under spinal anesthesia: A systematic literature review and meta-analysis. Int. J. Surg. 2018, 60, 48–59. [Google Scholar] [CrossRef]
- Cooper, D.W.; Gowni, R.R. Impact of changing from ephedrine to phenylephrine as the first-line vasopressor during urgent caesarean section. Int. J. Obstet. Anesth. 2006, 15, 339–340. [Google Scholar] [CrossRef]
- Prakash, S.; Pramanik, V.; Chellani, H.; Salhan, S.; Gogia, A.R. Maternal and neonatal effects of bolus administration of ephedrine and phenylephrine during spinal anaesthesia for caesarean delivery: A randomised study. Int. J. Obstet. Anesth. 2010, 19, 24–30. [Google Scholar] [CrossRef]
- Higgins, N.; Fitzgerald, P.C.; van Dyk, D.; Dyer, R.A.; Rodriguez, N.; McCarthy, R.J.; Wong, C.A. The effect of prophylactic phenylephrine and ephedrine infusions on umbilical artery blood pH in women with preeclampsia undergoing cesarean delivery with spinal anesthesia: A randomized, double-blind trial. Anesth. Analg. 2018, 126, 1999–2006. [Google Scholar] [CrossRef]
- Chen, Y.; Guo, L.; Shi, Y.; Ma, G.; Xue, W.; He, L.; Ma, S.; Ni, X. Norepinephrine prophylaxis for postspinal anesthesia hypotension in parturients undergoing cesarean section: A randomized, controlled trial. Arch. Gynecol. Obstet. 2020, 302, 829–836. [Google Scholar] [CrossRef]
- Ngan Kee, W.D.; Lee, A.; Khaw, K.S.; Ng, F.F.; Karmakar, M.K.; Gin, T. Prophylactic phenylephrine infusion for preventing hypotension during spinal anaesthesia for cesarean delivery. Anesth. Analg. 2004, 98, 815–821. [Google Scholar] [CrossRef]
- das Neves, J.F.; Monteiro, G.A.; de Almeida, J.R.; Sant’Anna, R.S.; Bonin, H.B.; Macedo, C.F. Phenylephrine for blood pressure control in elective cesarean section: Therapeutic versus prophylactic doses. Rev. Bras. Anestesiol. 2010, 60, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Ngan Kee, W.D.; Gin, T. A quantitative, systematic review of randomized controlled trials of ephedrine versus phenylephrine for the management of hypotension during spinal anesthesia for cesarean delivery. Anesth. Analg. 2002, 94, 920–926. [Google Scholar] [CrossRef]
- Thorp, J.A.; Dildy, G.A.; Yeomans, E.R.; Meyer, B.A.; Parisi, V.M. Umbilical cord blood gas analysis at delivery. Am. J. Obstet. Gynecol. 1996, 175, 517–522. [Google Scholar] [CrossRef] [PubMed]
- Mon, W.; Stewart, A.; Fernando, R.; Ashpole, K.; El-Wahab, N.; MacDonald, S.; Tamilselvan, P.; Columb, M.; Liu, Y.M. Cardiac output changes with phenylephrine and ephedrine infusions during spinal anesthesia for cesarean section: A randomized, double-blind trial. J. Clin. Anesth. 2017, 37, 43–48. [Google Scholar] [CrossRef]
- Yadav, U.; Bharat, K. A clinical comparative study of prophylactic infusions of phenylephrine and ephedrine on maternal hemodynamics and fetal acidosis in elective caesarean section. Int. J. Pharm. Sci. Res. 2012, 3, 5056–5061. [Google Scholar]
- Soxhuku-Isufi, A.; Shpata, V.; Sula, H. Maternal and Neonatal Effects of Vasopressors Used for Treating Hypotension after Spinal Anesthesia for Caesarean Section: A Randomized Controlled Study. Open Access Maced. J. Med. Sci. 2016, 4, 54–58. [Google Scholar] [CrossRef]
- Nazir, I.; Bhat, M.A.; Qazi, S.; Buchh, V.N.; Gurcoo, S.A. Comparison between phenylephrine and ephedrine in preventing hypotension during spinal anesthesia for cesarean section. J. Obstet. Anaesth. Crit. Care 2012, 2, 92–97. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Jain, K.; Arora, S.; Bharti, N. A comparison of three vasopressors for tight control of maternal blood pressure during cesarean section under spinal anesthesia: Effect on maternal and fetal outcome. J. Anaesthesiol. Clin. Pharmacol. 2013, 29, 26–31. [Google Scholar] [CrossRef]
- Cooper, D.W.; Sharma, S.; Orakkan, P.; Gurung, S. Retrospective study of association between choice of vasopressor given during spinal anaesthesia for high-risk caesarean delivery and fetal pH. Int. J. Obstet. Anesth. 2010, 19, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Mohta, M.; Aggarwal, M.; Sethi, A.K.; Harisinghani, P.; Guleria, K. Randomized double-blind comparison of ephedrine and phenylephrine for management of post-spinal hypotension in potential fetal compromise. Int. J. Obstet. Anesth. 2016, 27, 32–40. [Google Scholar] [CrossRef]
- Dyer, R.A.; Emmanuel, A.; Adams, S.C.; Lombard, C.J.; Arcache, M.J.; Vorster, A.; Wong, C.A.; Higgins, N.; Reed, A.R.; James, M.F.; et al. A randomised comparison of bolus phenylephrine and ephedrine for the management of spinal hypotension in patients with severe preeclampsia and fetal compromise. Int. J. Obstet. Anesth. 2018, 33, 23–31. [Google Scholar] [CrossRef]
- Habib, A.S. A review of the impact of phenylephrine administration on maternal hemodynamics and maternal and neonatal outcomes in women undergoing cesarean delivery under spinal anesthesia. Anesth. Analg. 2012, 114, 377–390. [Google Scholar] [CrossRef]
- Cooper, D.W.; Gibb, S.C.; Meek, T.; Owen, S.; Kokri, M.S.; Malik, A.T.; Koneti, K.K. Effect of intravenous vasopressor on spread of spinal anaesthesia and fetal acid-base equilibrium. Br. J. Anaesth. 2007, 98, 649–656. [Google Scholar] [CrossRef]
- Ngan Kee, W.D.; Khaw, K.S.; Tan, P.E.; Ng, F.F.; Karmakar, M.K. Placental transfer and fetal metabolic effects of phenylephrine and ephedrine during spinal anesthesia for cesarean delivery. Anesthesiology 2009, 111, 506–512. [Google Scholar] [CrossRef]
| Ephedrine | Phenylephrine | Standard Difference (95%CI) | p | |
|---|---|---|---|---|
| N | 47 | 47 | ||
| Age, years | 33.80 ± 4.72 | 33.06 ± 4.74 | 0.74 (−1.19; 2.68) | 0.447 |
| Weight, kg | 81.04 ± 11.75 | 78.74 ± 11.39 | 2.3 (−2.44; 7.03) | 0.338 |
| Height, cm | 167.38 ± 6.32 | 165.80 ± 6.29 | 1.58 (−1.01; 4.16) | 0.229 |
| Weight gain, kg | 13.92 ± 4.65 | 12.98 ± 4.69 | 0.93 (−0.97; 2.85) | 0.334 |
| Hemoglobin, g/dL | 12.31 ± 1.10 | 12.27 ± 1.01 | 0.04 (−0.39; 0.47) | 0.846 |
| Gestational age (wk) | 38.57 ± 0.82 | 38.42 ± 1.01 | 0.15 (−0.23; 0.52) | 0.438 |
| Twin pregnancies | 1 | 2 | ||
| Gravidity | 3.00 (4.00; 4.00) | 2.00 (1.00; 3.00) | (54.61; 40.38) * | 0.009 |
| Parity | 2.00 (1.00; 3.00) | 2.00 (1.00; 2.00) | (52.81; 42.18) * | 0.045 |
| Baseline SBP (mmHg) | 130.02 ± 13.83 | 136.76 ± 12.85 | −6.74 (−12.21; −1.27) | 0.016 |
| Baseline MAP (mmHg) | 99.91 ± 12.10 | 101.12 ± 11.39 | −1.21 (−6.05; 3.62) | 0.619 |
| Bupivacaine dose (mg) | 11.97 ± 0.91 | 11.14 ± 1.09 | 0.83 (0.42; 1.24) | <0.001 |
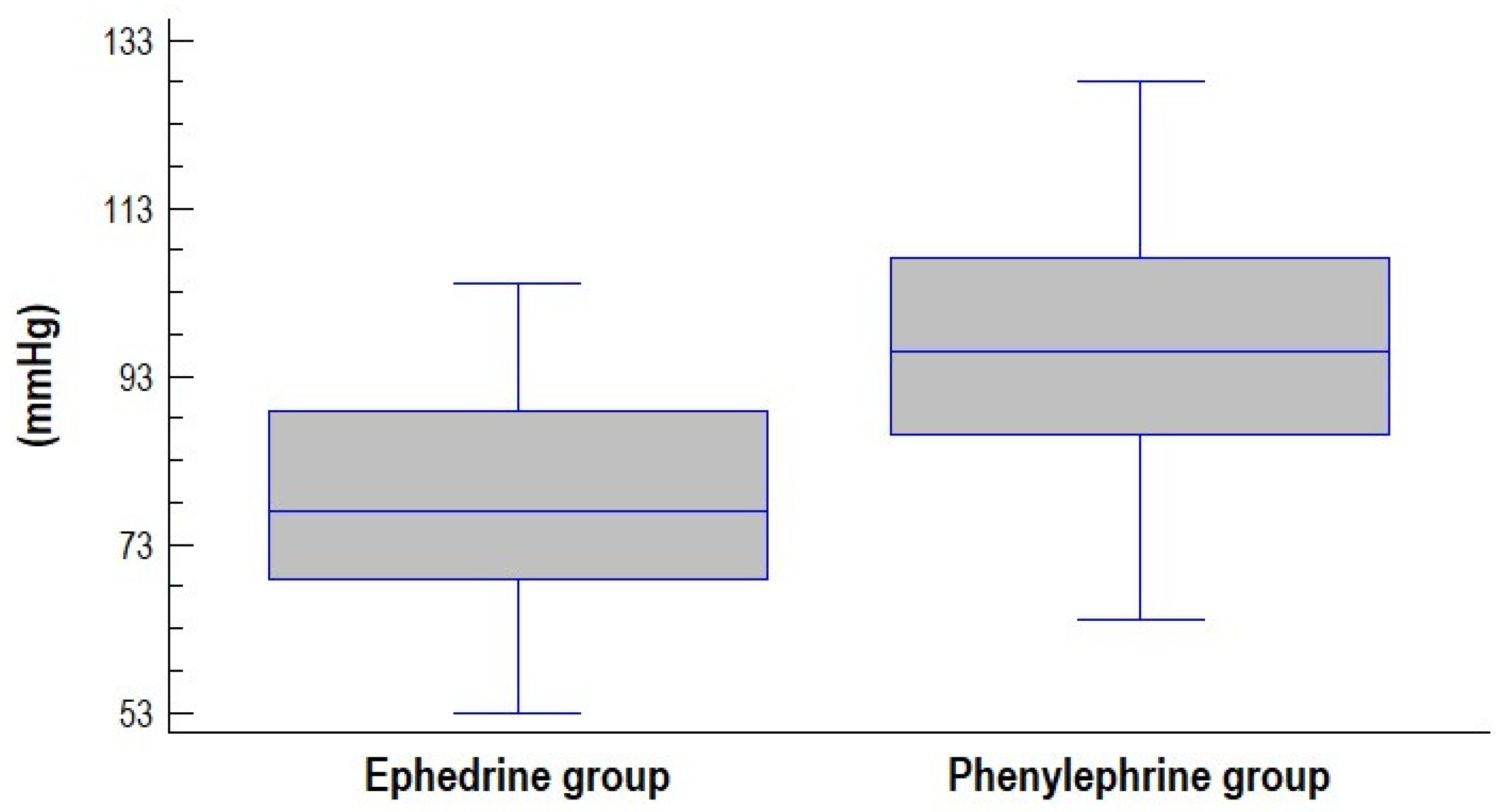
| Lowest SBP | 77.87 ± 12.36 | 96.76 ± 15.16 | −18.89 (−24.56; −13.22) | <0.001 |
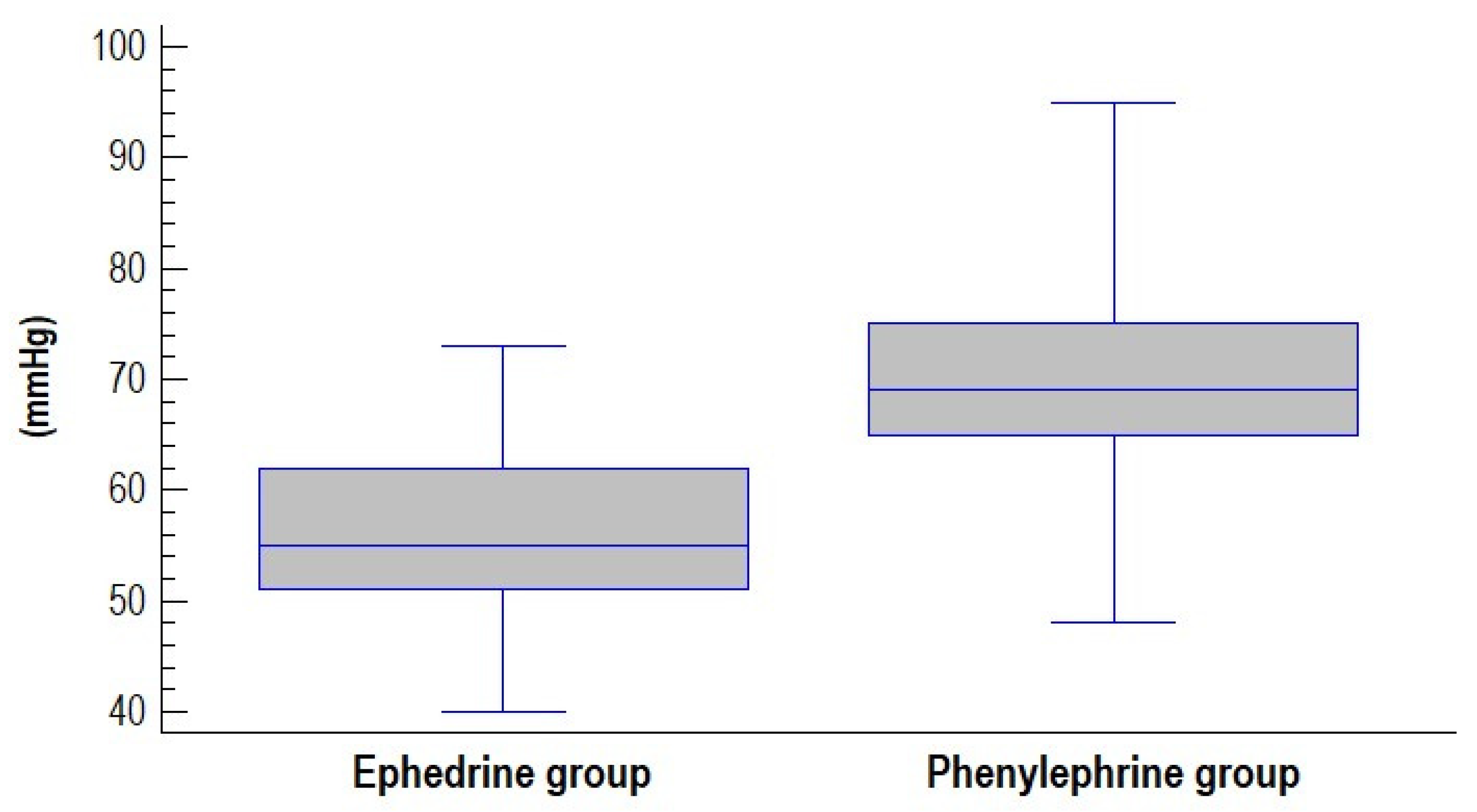
| Lowest MAP | 56.47 ± 8.25 | 69.97 ± 10.77 | −13.50 (−17.46; −9.53) | <0.001 |
| Total vasopressor dose (mg) | 25 (20; 35) | 0.72 (0.428; 1) | ||
| Maximal upper sensory block level | T3 (T4; T2) | T2 (T1; T3) | −1 (−1; 1) | 0.06 |
| Crystalloids volume (mL) | 1397.87 ± 311.02 | 1417.02 ± 312.3 | −19.15 (−146.84; 108.54) | 0.77 |
| Newborn weight (g) | 3540.52 ± 449.95 | 3331.53 ± 484.34 | 208.99 (20.45; 397.52) | 0.03 |
| Bradycardia | 2 (4.2) | 5 (10.6) | −6.4 (−16.9; 4.17) | 0.435 |
| Nausea | 20 (42.5) | 5 (10.6) | 31.9 (15.3; 48.6) | <0.001 |
| Vomiting | 1 (2.1) | 0 (0) | 2.1 (−2.0; 6.3) | 1.0 |
| Ephedrine | Phenylephrine | Standard Difference (95%CI) | p | |
|---|---|---|---|---|
| N | 48 | 49 | ||
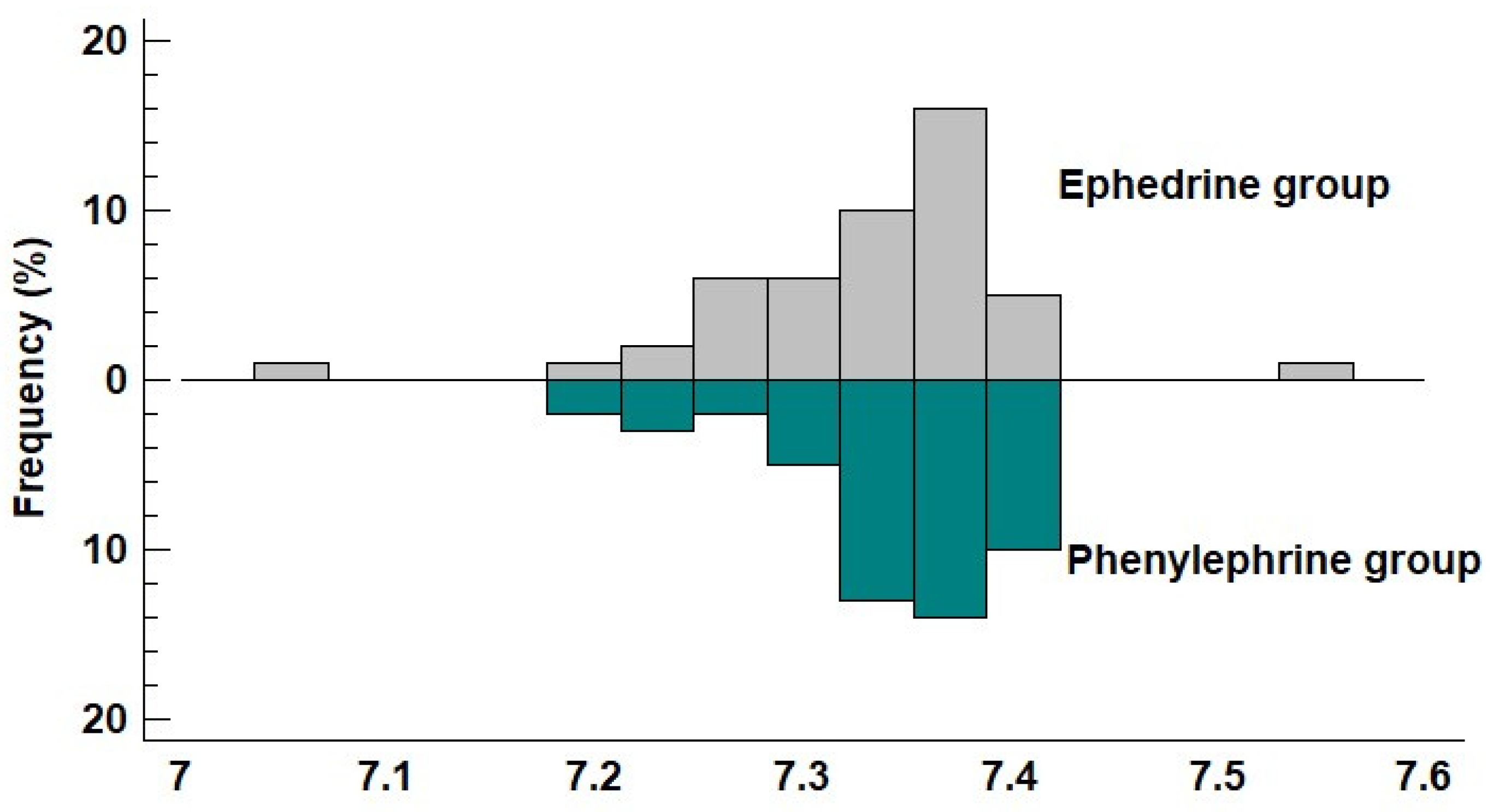
| pH | 7.34 (7.29; 7.37) | 7.35 (7.31; 7.38) | −0.008 (−0.03; 0.01) | 0.396 |
| PaCO2 (mmHg) | 45.3 (41.75; 52.17) | 46.5 (42.05; 52) | −0.54 (−4.12; 3.02) | 0.899 |
| PaO2 (mmHg) | 24.75 (18.22; 30.3) | 21.7 (17.8; 27.75) | 1.90 (−2.35; 6.16) | 0.310 |
| Base excess (mEq/L) | −1.8 (−3.32; −0.6) | −1.4 (−2.5; 0.1) | −0.85 (−1.66; −0.05) | 0.062 |
| Umbilical artery pH < 7.2 | 2 (4.2) | 0 (0) | 4.2 (−1.47; 9.81) | 0.24 |
| Apgar score at 5 min | 10 (10; 10) | 10 (10; 10) | 0 (0; 0) | 0.94 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Horosz, B.; Białowolska-Horosz, K.; Malec-Milewska, M. Effect of Prophylactic Phenylephrine Infusion Versus Interventional Ephedrine Boluses on Umbilical Blood pH in Cesarean Deliveries Under Spinal Anesthesia: A Retrospective Case-Control Study. J. Clin. Med. 2025, 14, 6016. https://doi.org/10.3390/jcm14176016
Horosz B, Białowolska-Horosz K, Malec-Milewska M. Effect of Prophylactic Phenylephrine Infusion Versus Interventional Ephedrine Boluses on Umbilical Blood pH in Cesarean Deliveries Under Spinal Anesthesia: A Retrospective Case-Control Study. Journal of Clinical Medicine. 2025; 14(17):6016. https://doi.org/10.3390/jcm14176016
Chicago/Turabian StyleHorosz, Bartosz, Katarzyna Białowolska-Horosz, and Małgorzata Malec-Milewska. 2025. "Effect of Prophylactic Phenylephrine Infusion Versus Interventional Ephedrine Boluses on Umbilical Blood pH in Cesarean Deliveries Under Spinal Anesthesia: A Retrospective Case-Control Study" Journal of Clinical Medicine 14, no. 17: 6016. https://doi.org/10.3390/jcm14176016
APA StyleHorosz, B., Białowolska-Horosz, K., & Malec-Milewska, M. (2025). Effect of Prophylactic Phenylephrine Infusion Versus Interventional Ephedrine Boluses on Umbilical Blood pH in Cesarean Deliveries Under Spinal Anesthesia: A Retrospective Case-Control Study. Journal of Clinical Medicine, 14(17), 6016. https://doi.org/10.3390/jcm14176016






