Pulmonary and Immune Dysfunction in Pediatric Long COVID: A Case Study Evaluating the Utility of ChatGPT-4 for Analyzing Scientific Articles
Abstract
1. Introduction
2. Methods
Rubric for Evaluating ChatGPT-4-Provided Articles
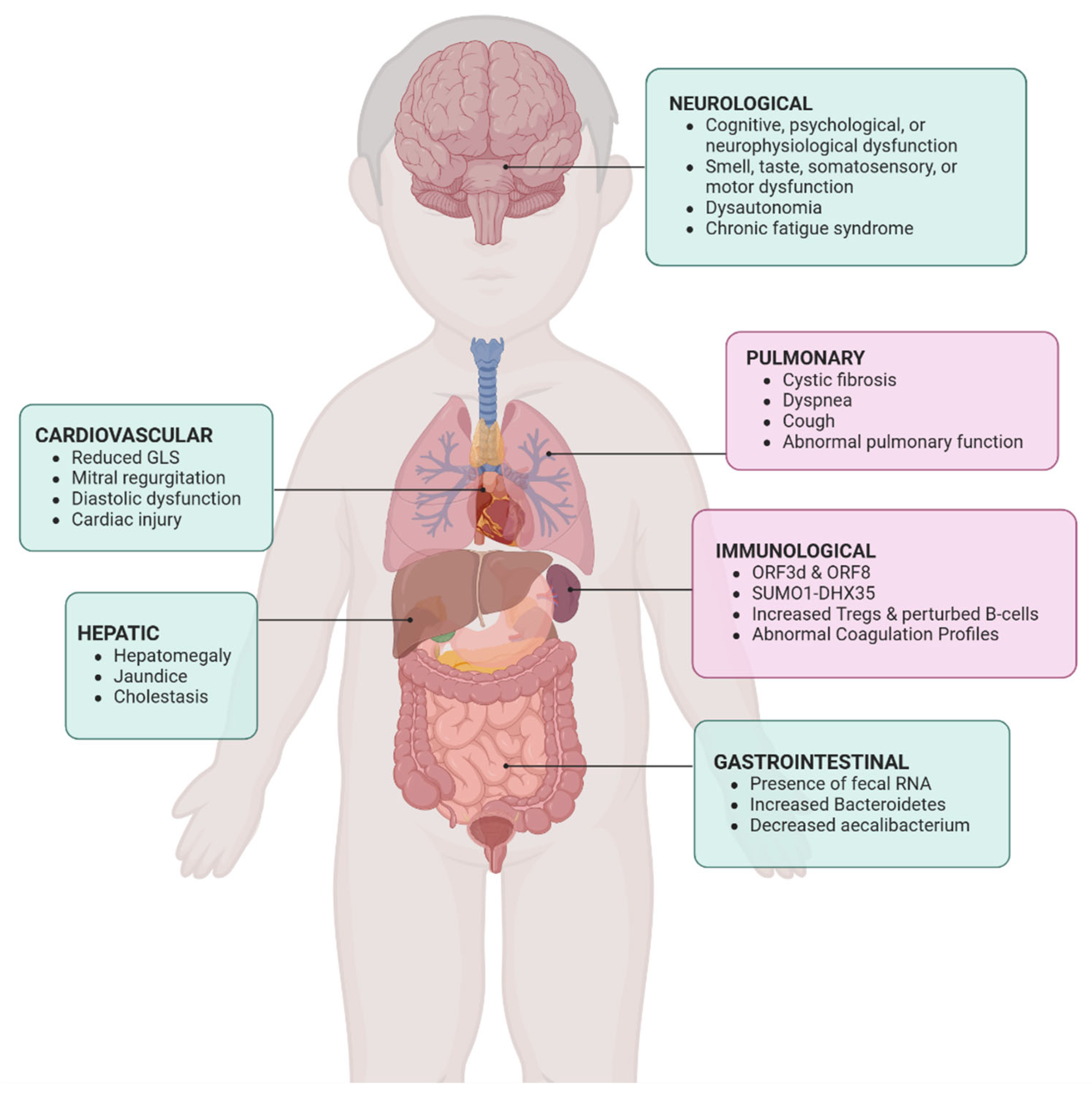
3. Pulmonary Complications
3.1. Persistent Pulmonary Symptoms
3.2. Pulmonary Comorbidities and Risk Factors
3.3. Long-Term Pulmonary Sequelae
3.4. Summary of Pulmonary Consequences
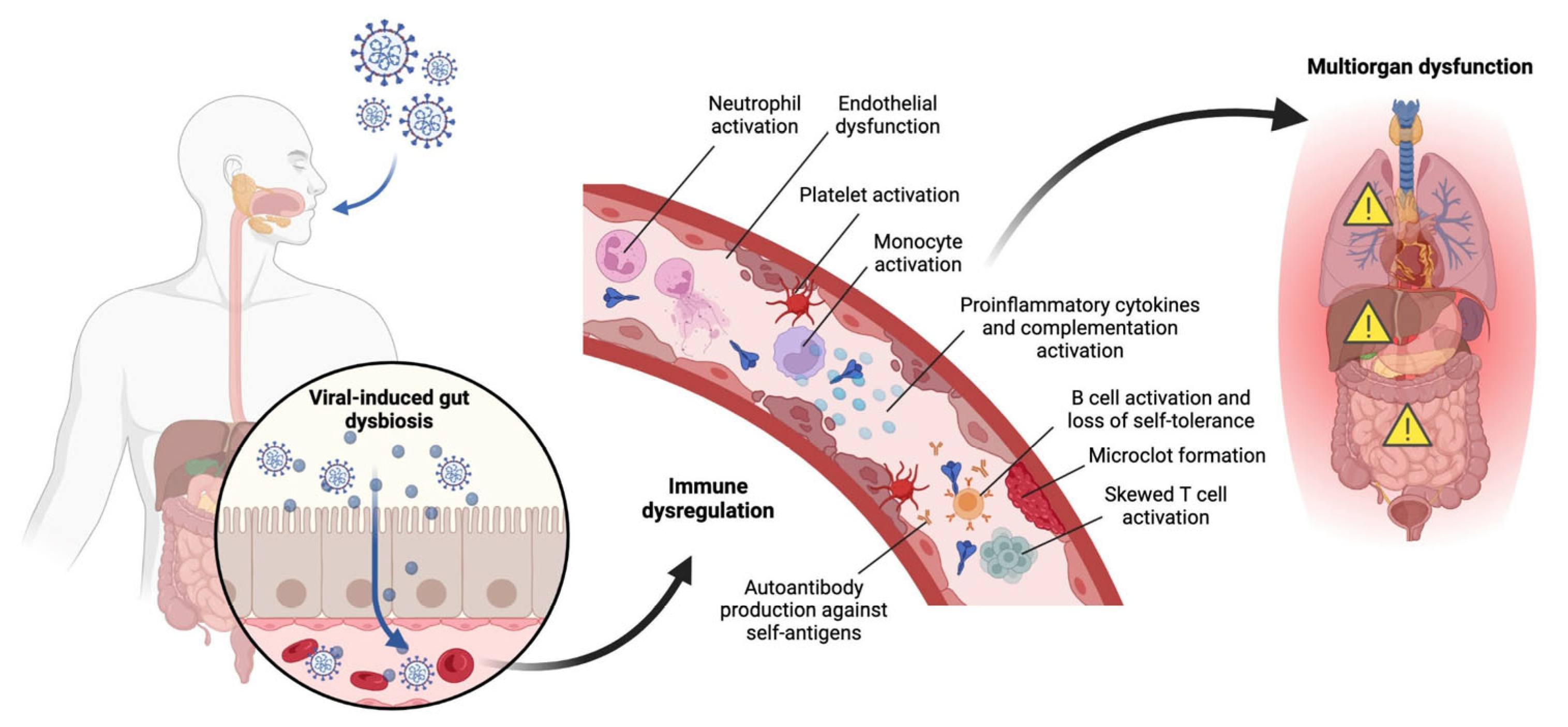
4. Immunology of Pediatric COVID-19
4.1. Immunological Mechanisms
4.2. Summary of Immunological Complications
5. ChatGPT-4 Implementation and Guidelines for Generating Scientific Reviews
Future Direction and Ensuring Research Integrity
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- ONS. Coronavirus (COVID-19) Latest Insights: Infections. Available online: https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/articles/coronaviruscovid19latestinsights/infections (accessed on 21 February 2023).
- Davis, H.E.; McCorkell, L.; Vogel, J.M.; Topol, E.J. Author Correction: Long COVID: Major findings, mechanisms and recommendations. Nat. Rev. Microbiol. 2023, 21, 408. [Google Scholar] [CrossRef]
- Benhood, S.; Newlands, F.; O’Mahoney, L.; Haghighat Ghahfarokhi, M.; Muhid, M.Z.; Dudley, J.; Stephenson, T.; Ladhani, S.N.; Bennett, S.; Viner, R.M.; et al. Persistent symptoms are associated with long term effects of COVID-19 among children and young people: Results from a systematic review and meta-analysis of controlled studies. PLoS ONE 2023, 18, e0293600. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.O.; Gonçalves, L.C.; Silva, P.A.N.; Moreira, A.L.E.; Ito, C.R.M.; Peixoto, F.A.O.; Wastowski, I.J.; Carneiro, L.C.; Avelino, M.A.G. Multisystem inflammatory syndrome (MIS-C): A systematic review and meta-analysis of clinical characteristics, treatment, and outcomes. J. Pediatr. 2022, 98, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, T.; Sivan, M.; Perlowski, A.; Nikolich, J.Z. Long COVID: A clinical update. Lancet 2024, 404, 707–724. [Google Scholar] [CrossRef]
- O’Mahoney, L.L.; Routen, A.; Gillies, C.; Ekezie, W.; Welford, A.; Zhang, A.; Karamchandani, U.; Simms-Williams, N.; Cassambai, S.; Ardavani, A.; et al. The prevalence and long-term health effects of Long COVID among hospitalised and non-hospitalised populations: A systematic review and meta-analysis. EClinicalMedicine 2023, 55, 101762. [Google Scholar] [CrossRef]
- Thompson, E.J.; Williams, D.M.; Walker, A.J.; Mitchell, R.E.; Niedzwiedz, C.L.; Yang, T.C.; Huggins, C.F.; Kwong, A.S.F.; Silverwood, R.J.; Di Gessa, G.; et al. Long COVID burden and risk factors in 10 UK longitudinal studies and electronic health records. Nat. Commun. 2022, 13, 3528. [Google Scholar] [CrossRef] [PubMed]
- Vahratian, A.; Saydah, S.; Bertolli, J.; Unger, E.R.; Gregory, C.O. Prevalence of Post-COVID-19 Condition and Activity-Limiting Post-COVID-19 Condition Among Adults. JAMA Netw. Open 2024, 7, e2451151. [Google Scholar] [CrossRef]
- Peter, R.S.; Nieters, A.; Gopel, S.; Merle, U.; Steinacker, J.M.; Deibert, P.; Friedmann-Bette, B.; Niess, A.; Muller, B.; Schilling, C.; et al. Persistent symptoms and clinical findings in adults with post-acute sequelae of COVID-19/post-COVID-19 syndrome in the second year after acute infection: A population-based, nested case-control study. PLoS Med. 2025, 22, e1004511. [Google Scholar] [CrossRef]
- Buonsenso, D.; Martino, L.; Morello, R.; Mariani, F.; Fearnley, K.; Valentini, P. Viral persistence in children infected with SARS-CoV-2: Current evidence and future research strategies. Lancet Microbe 2023, 4, e745–e756. [Google Scholar] [CrossRef]
- Ha, E.K.; Kim, J.H.; Han, M.Y. Long COVID in children and adolescents: Prevalence, clinical manifestations, and management strategies. Clin. Exp. Pediatr. 2023, 66, 465–474. [Google Scholar] [CrossRef]
- Stephenson, T.; Pinto Pereira, S.M.; Nugawela, M.D.; McOwat, K.; Simmons, R.; Chalder, T.; Ford, T.; Heyman, I.; Swann, O.V.; Fox-Smith, L.; et al. Long COVID-six months of prospective follow-up of changes in symptom profiles of non-hospitalised children and young people after SARS-CoV-2 testing: A national matched cohort study (The CLoCk) study. PLoS ONE 2023, 18, e0277704. [Google Scholar] [CrossRef]
- Naeimi, R.; Sepidarkish, M.; Mollalo, A.; Parsa, H.; Mahjour, S.; Safarpour, F.; Almukhtar, M.; Mechaal, A.; Chemaitelly, H.; Sartip, B.; et al. SARS-CoV-2 seroprevalence in children worldwide: A systematic review and meta-analysis. EClinicalMedicine 2023, 56, 101786. [Google Scholar] [CrossRef]
- Kane, A.S.; Godfrey, M.; Noval Rivas, M.; Arditi, M.; Fasano, A.; Yonker, L.M. The Spectrum of Postacute Sequelae of COVID-19 in Children: From MIS-C to Long COVID. Annu. Rev. Virol. 2024, 11, 327–341. [Google Scholar] [CrossRef] [PubMed]
- La Torre, F.; Elicio, M.P.; Monno, V.A.; Chironna, M.; Moramarco, F.; Campanozzi, A.; Civino, A.; Cecinati, V.; Vairo, U.; Giordano, M.; et al. Incidence and Prevalence of Multisystem Inflammatory Syndrome in Children (MIS-C) in Southern Italy. Children 2023, 10, 766. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Gross, R.S.; Mohandas, S.; Stein, C.R.; Case, A.; Dreyer, B.; Pajor, N.M.; Bunnell, H.T.; Warburton, D.; Berg, E.; et al. Postacute Sequelae of SARS-CoV-2 in Children. Pediatrics 2024, 153, e2023062570. [Google Scholar] [CrossRef]
- Su, S.; Zhao, Y.; Zeng, N.; Liu, X.; Zheng, Y.; Sun, J.; Zhong, Y.; Wu, S.; Ni, S.; Gong, Y.; et al. Epidemiology, clinical presentation, pathophysiology, and management of long COVID: An update. Mol. Psychiatry 2023, 28, 4056–4069. [Google Scholar] [CrossRef]
- Yang, W.H.; Yang, Y.J.; Chen, T.J. ChatGPT’s innovative application in blood morphology recognition. J. Chin. Med. Assoc. 2024, 87, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Hanada, S.; Hayashi, Y.; Subramani, S.; Thenuwara, K. Pioneering the Integration of Artificial Intelligence in Medical Oral Board Examinations. Cureus 2024, 16, e52318. [Google Scholar] [CrossRef]
- Kassab, J.; Hadi El Hajjar, A.; Wardrop, R.M., 3rd; Brateanu, A. Accuracy of Online Artificial Intelligence Models in Primary Care Settings. Am. J. Prev. Med. 2024, 66, 1054–1059. [Google Scholar] [CrossRef]
- Azam, M.; Chen, Y.; Arowolo, M.O.; Liu, H.; Popescu, M.; Xu, D. A Comprehensive Evaluation of Large Language Models in Mining Gene Interactions and Pathway Knowledge. bioRxiv 2024. [Google Scholar] [CrossRef]
- Sheth, S.; Baker, H.P.; Prescher, H.; Strelzow, J.A. Ethical Considerations of Artificial Intelligence in Health Care: Examining the Role of Generative Pretrained Transformer-4. J. Am. Acad. Orthop. Surg. 2024, 32, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Azamfirei, R.; Kudchadkar, S.R.; Fackler, J. Large language models and the perils of their hallucinations. Crit. Care 2023, 27, 120. [Google Scholar] [CrossRef]
- Merma Molina, G.; Peña Alfaro, H.; Peña Alfaro González, S.R. Design and Validation of a Rubric to Assess the Use of American Psychological Association Style in scientific articles. J. New Approaches Educ. Res. 2017, 6, 78–86. [Google Scholar] [CrossRef]
- Ipek, S.; Gungor, S.; Gullu, U.U.; Kizildag, B.; Ozkars, M.Y.; Yurttutan, S.; Kutukcu, M.K.; Demiray, S. Evaluation of Pulmonary Functions After Discharge in Pediatric Patients with COVID-19: A Prospective Study. Sisli Etfal Hastan. Tip. Bul. 2022, 56, 318–322. [Google Scholar] [CrossRef]
- Öztürk, G.K.; Beken, B.; Doğan, S.; Akar, H.H. Pulmonary function tests in the follow-up of children with COVID-19. Eur. J. Pediatr. 2022, 181, 2839–2847. [Google Scholar] [CrossRef]
- Al-Shamrani, A.; Al-Shamrani, K.; Al-Otaibi, M.; Alenazi, A.; Aldosaimani, H.; Aldhalaan, Z.; Alalkami, H.; Yousef, A.A.; Kobeisy, S.; Alharbi, S. Residual Cough and Asthma-like Symptoms Post-COVID-19 in Children. Children 2023, 10, 1031. [Google Scholar] [CrossRef]
- Bode, S.F.N.; Haendly, M.; Fabricius, D.; Mayer, B.; Zernickel, M.; Haddad, A.D.M.; Frieh, P.; Elling, R.; Renk, H.; Stich, M.; et al. Pulmonary Function and Persistent Clinical Symptoms in Children and Their Parents 12 Months After Mild SARS-CoV-2 Infection. Front. Pediatr. 2022, 10, 894331. [Google Scholar] [CrossRef]
- Gross, R.S.; Thaweethai, T.; Kleinman, L.C.; Snowden, J.N.; Rosenzweig, E.B.; Milner, J.D.; Tantisira, K.G.; Rhee, K.E.; Jernigan, T.L.; Kinser, P.A.; et al. Characterizing Long COVID in Children and Adolescents. JAMA 2024, 332, 1174–1188. [Google Scholar] [CrossRef]
- Hazan, G.; Shah, M.Z.; Brennan, S. Markers for severe disease and long-term sequelae in pediatric patients with severe acute respiratory syndrome coronavirus 2 infection. Pediatr. Int. 2022, 64, e15177. [Google Scholar] [CrossRef] [PubMed]
- Borczuk, A.C. Pathogenesis of Pulmonary Long COVID-19. Mod. Pathol. 2024, 37, 100378. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Zhang, B.; Zhang, D.; Wu, Q.; Chen, J.; Li, L.; Lu, Y.; Becich, M.J.; Blecker, S.; Chilukuri, N.; et al. Body Mass Index and Postacute Sequelae of SARS-CoV-2 Infection in Children and Young Adults. JAMA Netw. Open 2024, 7, e2441970. [Google Scholar] [CrossRef]
- Garcia-Garcia, A.; Perez de Diego, R.; Flores, C.; Rinchai, D.; Sole-Violan, J.; Deya-Martinez, A.; Garcia-Solis, B.; Lorenzo-Salazar, J.M.; Hernandez-Brito, E.; Lanz, A.L.; et al. Humans with inherited MyD88 and IRAK-4 deficiencies are predisposed to hypoxemic COVID-19 pneumonia. J. Exp. Med. 2023, 220, e20220170. [Google Scholar] [CrossRef]
- Bogusławski, S.; Strzelak, A.; Gajko, K.; Peradzyńska, J.; Popielska, J.; Marczyńska, M.; Kulus, M.; Krenke, K. The outcomes of COVID-19 pneumonia in children-clinical, radiographic, and pulmonary function assessment. Pediatr. Pulmonol. 2023, 58, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Adrover, J.M.; Carrau, L.; Dassler-Plenker, J.; Bram, Y.; Chandar, V.; Houghton, S.; Redmond, D.; Merrill, J.R.; Shevik, M.; tenOever, B.R.; et al. Disulfiram inhibits neutrophil extracellular trap formation and protects rodents from acute lung injury and SARS-CoV-2 infection. JCI Insight 2022, 7, e157342. [Google Scholar] [CrossRef] [PubMed]
- Palacios, S.; Krivchenia, K.; Eisner, M.; Young, B.; Ramilo, O.; Mejias, A.; Lee, S.; Kopp, B.T. Long-term pulmonary sequelae in adolescents post-SARS-CoV-2 infection. Pediatr. Pulmonol. 2022, 57, 2455–2463. [Google Scholar] [CrossRef]
- Campos, C.; Prokopich, S.; Loewen, H.; Sanchez-Ramirez, D.C. Long-Term Effect of COVID-19 on Lung Imaging and Function, Cardiorespiratory Symptoms, Fatigue, Exercise Capacity, and Functional Capacity in Children and Adolescents: A Systematic Review and Meta-Analysis. Healthcare 2022, 10, 2492. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, L.; Liu, Q.; Zheng, Y.; Tan, X.; Huang, Z.; Guo, M.; Wang, X.; Chen, X.; Liang, S.; et al. The lethal K18-hACE2 knock-in mouse model mimicking the severe pneumonia of COVID-19 is practicable for antiviral development. Emerg. Microbes Infect. 2024, 13, 2353302. [Google Scholar] [CrossRef]
- Bridges, J.P.; Vladar, E.K.; Huang, H.; Mason, R.J. Respiratory epithelial cell responses to SARS-CoV-2 in COVID-19. Thorax 2022, 77, 203–209. [Google Scholar] [CrossRef]
- Ravindra, N.G.; Alfajaro, M.M.; Gasque, V.; Huston, N.C.; Wan, H.; Szigeti-Buck, K.; Yasumoto, Y.; Greaney, A.M.; Habet, V.; Chow, R.D.; et al. Single-cell longitudinal analysis of SARS-CoV-2 infection in human airway epithelium identifies target cells, alterations in gene expression, and cell state changes. PLoS Biol. 2021, 19, e3001143. [Google Scholar] [CrossRef]
- Xiang, M.; Jing, H.; Wang, C.; Novakovic, V.A.; Shi, J. Persistent Lung Injury and Prothrombotic State in Long COVID. Front. Immunol. 2022, 13, 862522. [Google Scholar] [CrossRef] [PubMed]
- Sinha, S.; Castillo, V.; Espinoza, C.R.; Tindle, C.; Fonseca, A.G.; Dan, J.M.; Katkar, G.D.; Das, S.; Sahoo, D.; Ghosh, P. COVID-19 lung disease shares driver AT2 cytopathic features with Idiopathic pulmonary fibrosis. EBioMedicine 2022, 82, 104185. [Google Scholar] [CrossRef]
- Yu, D.; Yin, G.; Lei, J.; Gong, Y.; Zheng, L.; He, D.; Lei, L.; Sun, L. The correlation between serum levels of laminin, type IV collagen, type III procollagen N-terminal peptide and hyaluronic acid with the progression of post-COVID-19 pulmonary fibrosis. Front. Cell Dev. Biol. 2024, 12, 1382244. [Google Scholar] [CrossRef] [PubMed]
- Ambardar, S.R.; Hightower, S.L.; Huprikar, N.A.; Chung, K.K.; Singhal, A.; Collen, J.F. Post-COVID-19 Pulmonary Fibrosis: Novel Sequelae of the Current Pandemic. J. Clin. Med. 2021, 10, 2452. [Google Scholar] [CrossRef]
- Mohammadi, A.; Balan, I.; Yadav, S.; Matos, W.F.; Kharawala, A.; Gaddam, M.; Sarabia, N.; Koneru, S.C.; Suddapalli, S.K.; Marzban, S. Post-COVID-19 Pulmonary Fibrosis. Cureus 2022, 14, e22770. [Google Scholar] [CrossRef] [PubMed]
- Hama Amin, B.J.; Kakamad, F.H.; Ahmed, G.S.; Ahmed, S.F.; Abdulla, B.A.; Mohammed, S.H.; Mikael, T.M.; Salih, R.Q.; Ali, R.K.; Salh, A.M.; et al. Post COVID-19 pulmonary fibrosis; a meta-analysis study. Ann. Med. Surg. 2022, 77, 103590. [Google Scholar] [CrossRef] [PubMed]
- Kramer, D.; Hilton, R.; Roman, J. Pulmonary fibrosis and COVID-19. Am. J. Med. Sci. 2023, 366, 245–253. [Google Scholar] [CrossRef]
- Sanhueza, S.; Vidal, M.A.; Hernandez, M.A.; Henriquez-Beltran, M.E.; Cabrera, C.; Quiroga, R.; Antilef, B.E.; Aguilar, K.P.; Castillo, D.A.; Llerena, F.J.; et al. Clinical and pulmonary function analysis in long-COVID revealed that long-term pulmonary dysfunction is associated with vascular inflammation pathways and metabolic syndrome. Front. Med. 2023, 10, 1271863. [Google Scholar] [CrossRef]
- Riou, M.; Coste, F.; Meyer, A.; Enache, I.; Talha, S.; Charloux, A.; Reboul, C.; Geny, B. Mechanisms of Pulmonary Vasculopathy in Acute and Long-Term COVID-19: A Review. Int. J. Mol. Sci. 2024, 25, 4941. [Google Scholar] [CrossRef]
- Narasimhan, H.; Cheon, I.S.; Qian, W.; Hu, S.S.; Parimon, T.; Li, C.; Goplen, N.; Wu, Y.; Wei, X.; Son, Y.M.; et al. An aberrant immune-epithelial progenitor niche drives viral lung sequelae. Nature 2024, 634, 961–969. [Google Scholar] [CrossRef]
- Kosyreva, A.; Dzhalilova, D.; Lokhonina, A.; Vishnyakova, P.; Fatkhudinov, T. The Role of Macrophages in the Pathogenesis of SARS-CoV-2-Associated Acute Respiratory Distress Syndrome. Front. Immunol. 2021, 12, 682871. [Google Scholar] [CrossRef]
- Sefik, E.; Israelow, B.; Mirza, H.; Zhao, J.; Qu, R.; Kaffe, E.; Song, E.; Halene, S.; Meffre, E.; Kluger, Y.; et al. A humanized mouse model of chronic COVID-19. Nat. Biotechnol. 2022, 40, 906–920. [Google Scholar] [CrossRef]
- Yang, C.; Tan, Y.; Li, Z.; Hu, L.; Chen, Y.; Zhu, S.; Hu, J.; Huai, T.; Li, M.; Zhang, G.; et al. Pulmonary redox imbalance drives early fibroproliferative response in moderate/severe coronavirus disease-19 acute respiratory distress syndrome and impacts long-term lung abnormalities. Ann. Intensive Care 2024, 14, 72. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Yang, G.; Wang, N.; Zeng, M.; Jiang, H.; Yuan, S.; Wu, J.; Zhang, J.; Cui, J.; Zhou, G.; et al. Exploring the prevalence and chest CT predictors of Long COVID in children: A comprehensive study from Shanghai and Linyi. Front. Pediatr. 2024, 12, 1420196. [Google Scholar] [CrossRef] [PubMed]
- Bain, C.C.; Lucas, C.D.; Rossi, A.G. Pulmonary macrophages and SARS-CoV2 infection. Int. Rev. Cell Mol. Biol. 2022, 367, 1–28. [Google Scholar]
- Patterson, B.K.; Francisco, E.B.; Yogendra, R.; Long, E.; Pise, A.; Rodrigues, H.; Hall, E.; Herrera, M.; Parikh, P.; Guevara-Coto, J.; et al. Persistence of SARS CoV-2 S1 Protein in CD16+ Monocytes in Post-Acute Sequelae of COVID-19 (PASC) up to 15 Months Post-Infection. Front. Immunol. 2021, 12, 746021. [Google Scholar] [CrossRef] [PubMed]
- Santana-de Anda, K.; Torres-Ruiz, J.; Mejia-Dominguez, N.R.; Alcala-Carmona, B.; Maravillas-Montero, J.L.; Paez-Franco, J.C.; Vargas-Castro, A.S.; Lira-Luna, J.; Camacho-Moran, E.A.; Juarez-Vega, G.; et al. Novel Clinical, Immunological, and Metabolic Features Associated with Persistent Post-Acute COVID-19 Syndrome. Int. J. Mol. Sci. 2024, 25, 9661. [Google Scholar] [CrossRef]
- Buonsenso, D.; Piazza, M.; Boner, A.L.; Bellanti, J.A. Long COVID: A proposed hypothesis-driven model of viral persistence for the pathophysiology of the syndrome. Allergy Asthma Proc. 2022, 43, 187–193. [Google Scholar] [CrossRef]
- Ehrenfeld, M.; Tincani, A.; Andreoli, L.; Cattalini, M.; Greenbaum, A.; Kanduc, D.; Alijotas-Reig, J.; Zinserling, V.; Semenova, N.; Amital, H.; et al. COVID-19 and autoimmunity. Autoimmun. Rev. 2020, 19, 102597. [Google Scholar] [CrossRef]
- Montes-Cobos, E.; Bastos, V.C.; Monteiro, C.; de Freitas, J.C.R.; Fernandes, H.D.P.; Constancio, C.S.; Rodrigues, D.A.S.; Gama, A.; Vidal, V.M.; Alves, L.S.; et al. Oligosymptomatic long-term carriers of SARS-CoV-2 display impaired innate resistance but increased high-affinity anti-spike antibodies. iScience 2023, 26, 107219. [Google Scholar] [CrossRef]
- Richter, A.G.; Shields, A.M.; Karim, A.; Birch, D.; Faustini, S.E.; Steadman, L.; Ward, K.; Plant, T.; Reynolds, G.; Veenith, T.; et al. Establishing the prevalence of common tissue-specific autoantibodies following severe acute respiratory syndrome coronavirus 2 infection. Clin. Exp. Immunol. 2021, 205, 99–105. [Google Scholar] [CrossRef]
- Wallukat, G.; Hohberger, B.; Wenzel, K.; Furst, J.; Schulze-Rothe, S.; Wallukat, A.; Honicke, A.S.; Muller, J. Functional autoantibodies against G-protein coupled receptors in patients with persistent Long-COVID-19 symptoms. J. Transl. Autoimmun. 2021, 4, 100100. [Google Scholar] [CrossRef]
- Crook, H.; Raza, S.; Nowell, J.; Young, M.; Edison, P. Long Covid-mechanisms, risk factors, and management. BMJ 2021, 374, n1648. [Google Scholar] [CrossRef]
- Sudre, C.H.; Murray, B.; Varsavsky, T.; Graham, M.S.; Penfold, R.S.; Bowyer, R.C.; Pujol, J.C.; Klaser, K.; Antonelli, M.; Canas, L.S.; et al. Attributes and predictors of long COVID. Nat. Med. 2021, 27, 626–631. [Google Scholar] [CrossRef]
- Hachim, A.; Gu, H.; Kavian, O.; Mori, M.; Kwan, M.Y.W.; Chan, W.H.; Yau, Y.S.; Chiu, S.S.; Tsang, O.T.Y.; Hui, D.S.C.; et al. SARS-CoV-2 accessory proteins reveal distinct serological signatures in children. Nat. Commun. 2022, 13, 2951. [Google Scholar] [CrossRef]
- Joshi, D.; Nyhoff, L.E.; Zarnitsyna, V.I.; Moreno, A.; Manning, K.; Linderman, S.; Burrell, A.R.; Stephens, K.; Norwood, C.; Mantus, G.; et al. Infants and young children generate more durable antibody responses to SARS-CoV-2 infection than adults. iScience 2023, 26, 107967. [Google Scholar] [CrossRef]
- Lima, K.; Fontoura, J.C.; de Souza, P.O.; Fazolo, T.; Hilario, G.; Zorzetto, R.; Rodrigues Junior, L.C.; Coimbra, L.D.; Borin, A.; Bispo-Dos-Santos, K.; et al. SARS-CoV-2 infected children form early immune memory responses dominated by nucleocapsid-specific CD8+ T cells and antibodies. Front. Immunol. 2022, 13, 1033364. [Google Scholar] [CrossRef] [PubMed]
- Sieber, J.; Mayer, M.; Schmidthaler, K.; Kopanja, S.; Camp, J.V.; Popovitsch, A.; Dwivedi, V.; Hoz, J.; Schoof, A.; Weseslindtner, L.; et al. Long-Lived Immunity in SARS-CoV-2-Recovered Children and Its Neutralizing Capacity Against Omicron. Front. Immunol. 2022, 13, 882456. [Google Scholar] [CrossRef]
- Ertesvåg, N.U.; Iversen, A.; Blomberg, B.; Özgümüş, T.; Rijal, P.; Fjelltveit, E.B.; Cox, R.J.; Langeland, N.; Bergen COVID-19 research group. Post COVID-19 condition after delta infection and omicron reinfection in children and adolescents. EBioMedicine 2023, 92, 104599. [Google Scholar] [CrossRef] [PubMed]
- Buonsenso, D.; Valentini, P.; De Rose, C.; Tredicine, M.; Pereyra Boza, M.D.C.; Camponeschi, C.; Morello, R.; Zampino, G.; Brooks, A.E.S.; Rende, M.; et al. Recovering or Persisting: The Immunopathological Features of SARS-CoV-2 Infection in Children. J. Clin. Med. 2022, 11, 4363. [Google Scholar] [CrossRef] [PubMed]
- Cheong, J.G.; Ravishankar, A.; Sharma, S.; Parkhurst, C.N.; Grassmann, S.A.; Wingert, C.K.; Laurent, P.; Ma, S.; Paddock, L.; Miranda, I.C.; et al. Epigenetic memory of coronavirus infection in innate immune cells and their progenitors. Cell 2023, 186, 3882–3902.e24. [Google Scholar] [CrossRef]
- Cheong, J.-G.; Ravishankar, A.; Sharma, S.; Parkhurst, C.; Nehar-Belaid, D.; Ma, S.; Paddock, L.; Farou, B.; Karakaslar, O.; Thibodeau, A.; et al. Epigenetic Memory of COVID-19 in Innate Immune Cells and Their Progenitors. Cell 2022, 186, 3882–3902.e24. [Google Scholar] [CrossRef]
- Fedorchenko, Y.; Zimba, O. Long COVID in autoimmune rheumatic diseases. Rheumatol. Int. 2023, 43, 1197–1207. [Google Scholar] [CrossRef]
- Ramasamy, A.; Wang, C.; Brode, W.M.; Verduzco-Gutierrez, M.; Melamed, E. Immunologic and Autoimmune-Related Sequelae of Severe Acute Respiratory Syndrome Coronavirus 2 Infection: Clinical Symptoms and Mechanisms of Disease. Phys. Med. Rehabil. Clin. N. Am. 2023, 34, 623–642. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Julg, B.; Mohandas, S.; Bradfute, S.B.; Force, R.M.P.T. Viral persistence, reactivation, and mechanisms of long COVID. Elife 2023, 12, e86015. [Google Scholar] [CrossRef] [PubMed]
- L’Huillier, A.G.; Pagano, S.; Baggio, S.; Meyer, B.; Andrey, D.O.; Nehme, M.; Guessous, I.; Eberhardt, C.S.; Huttner, A.; Posfay-Barbe, K.M.; et al. Autoantibodies against apolipoprotein A-1 after COVID-19 predict symptoms persistence. Eur. J. Clin. Investig. 2022, 52, e13818. [Google Scholar] [CrossRef] [PubMed]
- Thurner, L.; Fadle, N.; Regitz, E.; Preuss, K.D.; Neumann, F.; Cetin, O.; Schormann, C.; Hoffmann, M.C.; Herr, C.; Kheiroddin, P.; et al. Autoantibodies against SUMO1-DHX35 in long-COVID. J. Transl. Autoimmun. 2022, 5, 100171. [Google Scholar] [CrossRef]
- Sandoni, A.; Schaffrath Rosario, A.; Michel, J.; Kuttig, T.; Wurm, J.; Damerow, S.; Iwanowski, H.; Finkel, B.; Schrick, L.; Buchholz, U.; et al. SARS-CoV-2 viral clearance and viral load kinetics in young children (1–6 years) compared to adults: Results of a longitudinal study in Germany. Front. Pediatr. 2022, 10, 989456. [Google Scholar] [CrossRef]
- Tandel, D.; Sah, V.; Singh, N.K.; Potharaju, P.S.; Gupta, D.; Shrivastava, S.; Sowpati, D.T.; Harshan, K.H. SARS-CoV-2 Variant Delta Potently Suppresses Innate Immune Response and Evades Interferon-Activated Antiviral Responses in Human Colon Epithelial Cells. Microbiol. Spectr. 2022, 10, e0160422. [Google Scholar] [CrossRef]
- Yonker, L.M.; Gilboa, T.; Ogata, A.F.; Senussi, Y.; Lazarovits, R.; Boribong, B.P.; Bartsch, Y.C.; Loiselle, M.; Rivas, M.N.; Porritt, R.A.; et al. Multisystem inflammatory syndrome in children is driven by zonulin-dependent loss of gut mucosal barrier. J. Clin. Investig. 2021, 131, e149633. [Google Scholar] [CrossRef]
- Natarajan, A.; Zlitni, S.; Brooks, E.F.; Vance, S.E.; Dahlen, A.; Hedlin, H.; Park, R.M.; Han, A.; Schmidtke, D.T.; Verma, R.; et al. Gastrointestinal symptoms and fecal shedding of SARS-CoV-2 RNA suggest prolonged gastrointestinal infection. Med 2022, 3, 371–387.e9. [Google Scholar] [CrossRef]
- Cui, L.; Fang, Z.; De Souza, C.M.; Lerbs, T.; Guan, Y.; Li, I.; Charu, V.; Chen, S.Y.; Weissman, I.; Wernig, G. Innate immune cell activation causes lung fibrosis in a humanized model of long COVID. Proc. Natl. Acad. Sci. USA 2023, 120, e2217199120. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Lee, J.; Kim, S.; Lee, Y.W.; Kim, G.C.; Hong, S.M.; An, S.H.; Noh, H.; Kim, K.E.; On, D.; et al. A longitudinal molecular and cellular lung atlas of lethal SARS-CoV-2 infection in K18-hACE2 transgenic mice. EBioMedicine 2024, 99, 104932. [Google Scholar] [CrossRef]
- Dinnon, K.H., 3rd; Leist, S.R.; Okuda, K.; Dang, H.; Fritch, E.J.; Gully, K.L.; De la Cruz, G.; Evangelista, M.D.; Asakura, T.; Gilmore, R.C.; et al. SARS-CoV-2 infection produces chronic pulmonary epithelial and immune cell dysfunction with fibrosis in mice. Sci. Transl. Med. 2022, 14, eabo5070. [Google Scholar] [CrossRef]
- Garcia, G.; Labrouche-Colomer, S.; Duvignaud, A.; Clequin, E.; Dussiau, C.; Tregouet, D.A.; Malvy, D.; Prevel, R.; Zouine, A.; Pellegrin, I.; et al. Impaired balance between neutrophil extracellular trap formation and degradation by DNases in COVID-19 disease. J. Transl. Med. 2024, 22, 246. [Google Scholar] [CrossRef]
- Dean, L.S.; Devendra, G.; Jiyarom, B.; Subia, N.; Tallquist, M.D.; Nerurkar, V.R.; Chang, S.P.; Chow, D.C.; Shikuma, C.M.; Park, J. Phenotypic alteration of low-density granulocytes in people with pulmonary post-acute sequalae of SARS-CoV-2 infection. Front. Immunol. 2022, 13, 1076724. [Google Scholar] [CrossRef]
- Kulczynska-Przybik, A.; Czupryna, P.; Adamczuk, J.; Kruszewska, E.; Mroczko, B.; Moniuszko-Malinowska, A. Clinical usefulness of the serum levels of neuroinflammatory and lung fibrosis biomarkers in the assessment of cognitive dysfunction in post-COVID19 patients. Sci. Rep. 2024, 14, 25798. [Google Scholar] [CrossRef]
- Shekhar Patil, M.; Richter, E.; Fanning, L.; Hendrix, J.; Wyns, A.; Barrero Santiago, L.; Nijs, J.; Godderis, L.; Polli, A. Epigenetic changes in patients with post-acute COVID-19 symptoms (PACS) and long-COVID: A systematic review. Expert Rev. Mol. Med. 2024, 26, e29. [Google Scholar] [CrossRef]
- Salehi, S.; Reddy, S.; Gholamrezanezhad, A. Long-term Pulmonary Consequences of Coronavirus Disease 2019 (COVID-19): What We Know and What to Expect. J. Thorac. Imaging 2020, 35, W87–W89. [Google Scholar] [CrossRef]
- Roig-Marti, C.; Perez-Catalan, I.; Varea-Villanueva, M.; Folgado-Escudero, S.; Navarro-Ballester, A.; Fernandez-Garcia, M.P.; Segura-Fabrega, A.; Herrero-Rodriguez, G.; Dominguez-Bajo, E.; Fabra-Juana, S.; et al. Predictors of the presence of radiological abnormalities 6 months after severe COVID-19 pneumonia. BMC Infect. Dis. 2024, 24, 883. [Google Scholar] [CrossRef] [PubMed]
- Wendisch, D.; Dietrich, O.; Mari, T.; von Stillfried, S.; Ibarra, I.L.; Mittermaier, M.; Mache, C.; Chua, R.L.; Knoll, R.; Timm, S.; et al. SARS-CoV-2 infection triggers profibrotic macrophage responses and lung fibrosis. Cell 2021, 184, 6243–6261.e27. [Google Scholar] [CrossRef] [PubMed]
- Kevian, D.; Syed, U.; Guo, X.; Havens, A.; Dullerud, G.; Seiler, P.; Qin, L.; Hu, B. Capabilities of Large Language Models in Control Engineering: A Benchmark Study on GPT-4, Claude 3 Opus, and Gemini 1.0 Ultra. arXiv 2024, arXiv:2404.03647. [Google Scholar] [CrossRef]

| Criterion | 0—Not at All | 1—Very Poor | 2—Poor | 3—Fair | 4—Good | 5—Excellent |
|---|---|---|---|---|---|---|
| Summaries of articles provided by ChatGPT-4 accurately reflect the source material. | Summary contains no accurate elements from the source | Summary is highly inaccurate, misrepresents key findings | Summary has major inaccuracies and omits key points | Summary partially accurate but missing several important details | Summary mostly accurate with only minor omissions or errors | Summary fully accurate, clearly represents source material with no errors |
| Articles directly address the specific research topic. | Article is unrelated to the research topic | Article is minimally related, highly off-topic | Article is tangentially related with little relevance | Article moderately related but lacks direct focus on topic | Article is relevant and addresses most aspects of topic | Article is fully relevant and directly addresses all aspects of the topic |
| Articles are listed in PubMed. | Not listed in any database | Not listed in PubMed | Incorrect listing in PubMed | Listed but with incorrect citation or incomplete metadata | Correctly listed in PubMed but with minor citation inconsistencies | Fully and correctly listed in PubMed with accurate citation |
| Articles are found on publisher’s website. | Not found on any website | Not found on publisher’s website | Incorrect listing on publisher’s website | Found but behind paywall without citation details | Found with correct citation but missing some metadata | Fully available on publisher’s site with complete citation and metadata |
| Criterion | Mean ± SD | Median (IQR) | % Scoring ≥ 4 | % Scoring 5 |
|---|---|---|---|---|
| Accuracy of summaries | 4.2 ± 0.9 | 4 (1–5) | 82% | 51% |
| Relevance to topic | 4.6 ± 0.7 | 5 (2–5) | 89% | 77% |
| PubMed listing | 4.9 ± 0.8 | 5 (0–5) | 96% | 96% |
| Publisher website availability | 4.9 ± 0.4 | 5 (2–5) | 98% | 98% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Var, S.R.; Maeser, N.; Blake, J.; Zahs, E.; Deep, N.; Vasilakos, Z.; McKay, J.; Johnson, S.; Strell, P.; Chang, A.; et al. Pulmonary and Immune Dysfunction in Pediatric Long COVID: A Case Study Evaluating the Utility of ChatGPT-4 for Analyzing Scientific Articles. J. Clin. Med. 2025, 14, 6011. https://doi.org/10.3390/jcm14176011
Var SR, Maeser N, Blake J, Zahs E, Deep N, Vasilakos Z, McKay J, Johnson S, Strell P, Chang A, et al. Pulmonary and Immune Dysfunction in Pediatric Long COVID: A Case Study Evaluating the Utility of ChatGPT-4 for Analyzing Scientific Articles. Journal of Clinical Medicine. 2025; 14(17):6011. https://doi.org/10.3390/jcm14176011
Chicago/Turabian StyleVar, Susanna R., Nicole Maeser, Jeffrey Blake, Elise Zahs, Nathan Deep, Zoey Vasilakos, Jennifer McKay, Sether Johnson, Phoebe Strell, Allison Chang, and et al. 2025. "Pulmonary and Immune Dysfunction in Pediatric Long COVID: A Case Study Evaluating the Utility of ChatGPT-4 for Analyzing Scientific Articles" Journal of Clinical Medicine 14, no. 17: 6011. https://doi.org/10.3390/jcm14176011
APA StyleVar, S. R., Maeser, N., Blake, J., Zahs, E., Deep, N., Vasilakos, Z., McKay, J., Johnson, S., Strell, P., Chang, A., Korthas, H., Krishna, V., Narayanan, M., Arju, T., Natera-Rodriguez, D. E., Roman, A., Schulz, S. J., Shetty, A., Vernekar, M., ... Low, W. C. (2025). Pulmonary and Immune Dysfunction in Pediatric Long COVID: A Case Study Evaluating the Utility of ChatGPT-4 for Analyzing Scientific Articles. Journal of Clinical Medicine, 14(17), 6011. https://doi.org/10.3390/jcm14176011







