Efficacy of Upadacitinib in Treating Alopecia Areata, Atopic Dermatitis, and Th1 Comorbidities in Pediatric Patients: A Comprehensive Case Series and Literature Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patients
2.2. Therapeutic Regimen
2.3. Data Collection and Outcomes
2.4. Literature Review
3. Results
3.1. Case 1
3.2. Case 2
3.3. Case 3
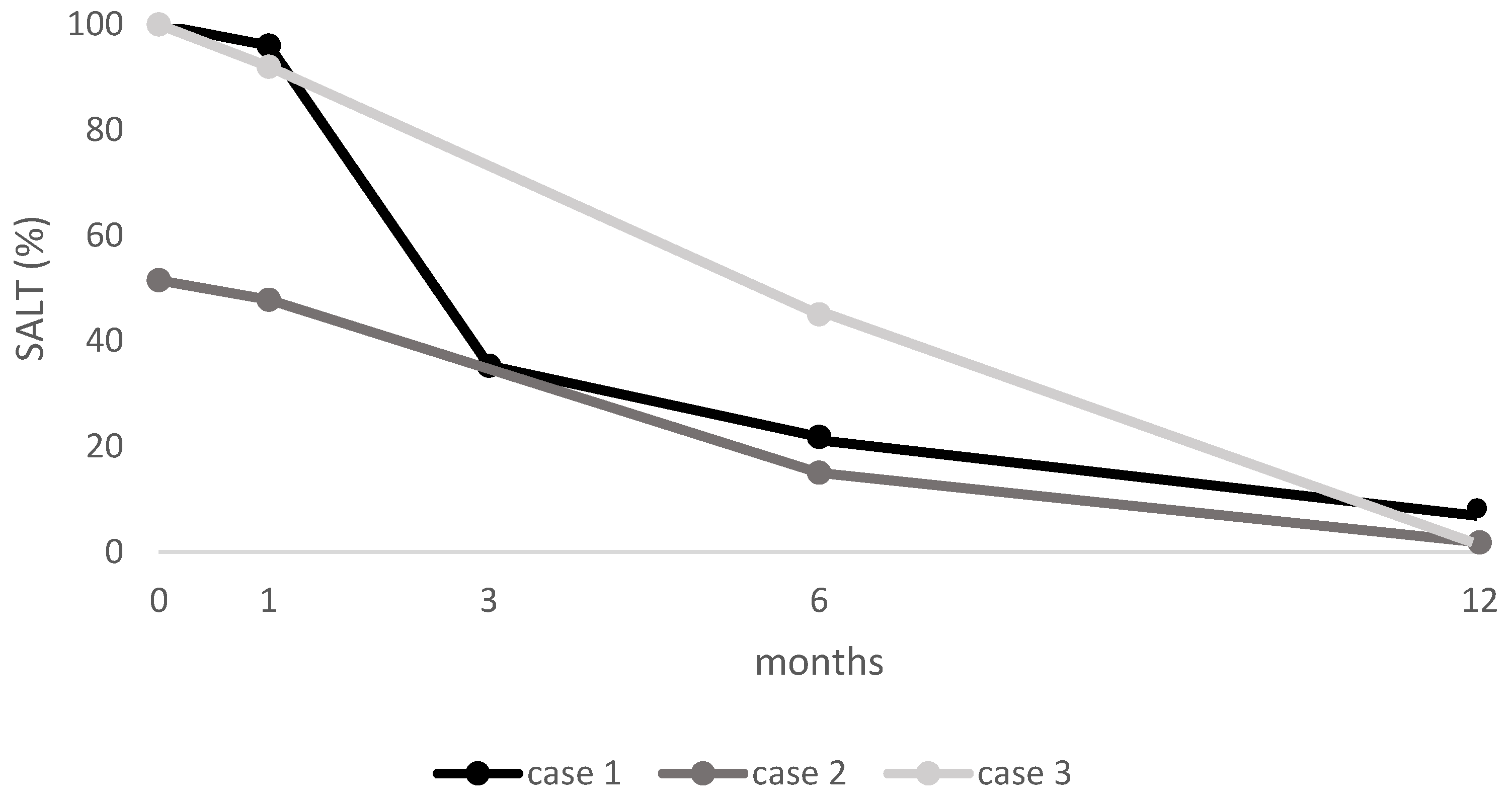
3.4. SALT Score
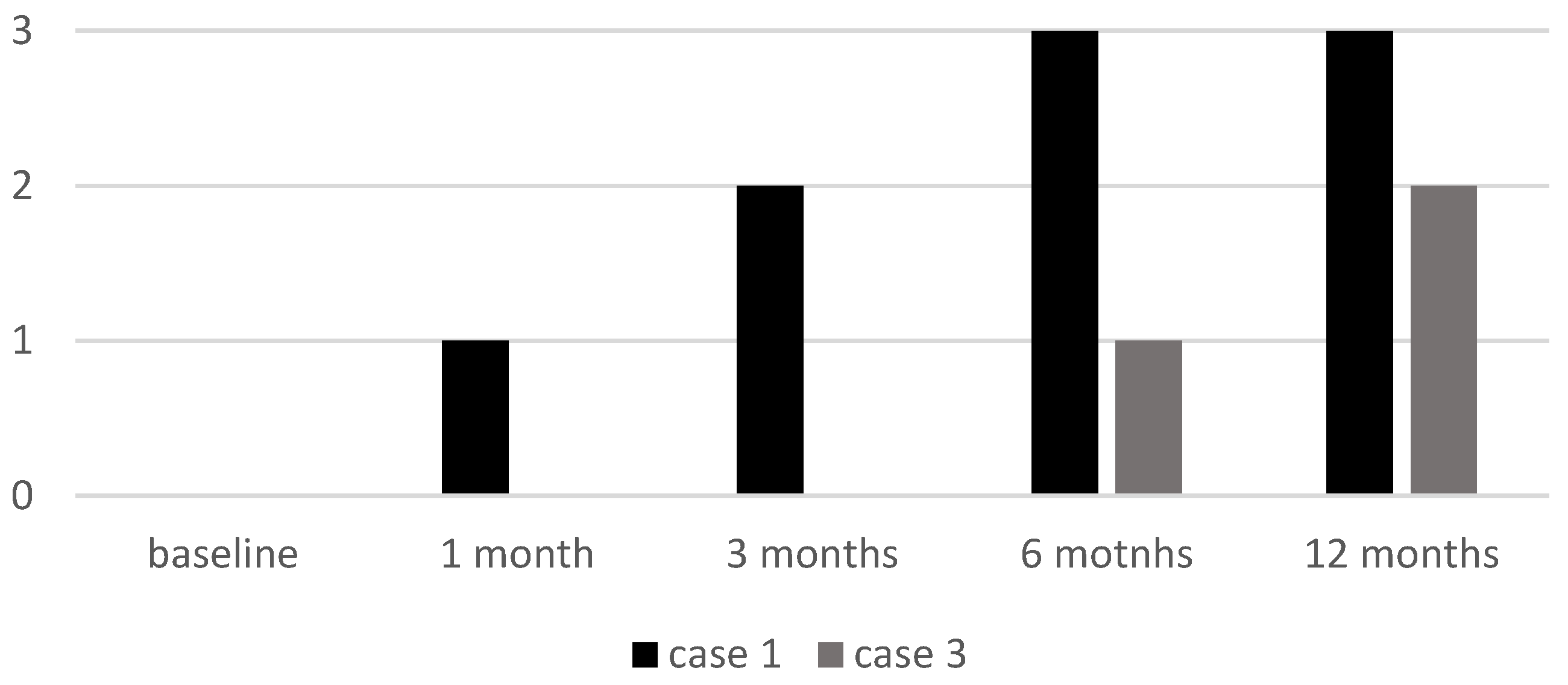
3.5. EBA Score
3.6. ELA Score
3.7. Adverse Events
3.8. Literature Review
4. Discussion
4.1. Pathogenesis and Therapeutic Challenges in Pediatric AA and AD
4.2. The Role of the JAK-STAT Pathway and Rationale for JAK Inhibitors
4.3. Efficacy and Safety of Upadacitinib in Pediatric Population
4.4. Literature Review
4.5. Our Findings
4.6. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Seneschal, J.; Boniface, K.; Jacquemin, C. Alopecia areata: Recent advances and emerging therapies. Ann. Dermatol. Vénéréologie 2022, 149, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Lepe, K.; Syed, H.A.; Zito, P.M. Alopecia Areata. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Dainichi, T.; Iwata, M.; Kaku, Y. Alopecia areata: What’s new in the epidemiology, comorbidities, and pathogenesis? J. Dermatol. Sci. 2023, 112, 120–127. [Google Scholar] [CrossRef]
- Sroka-Tomaszewska, J.; Trzeciak, M. Molecular Mechanisms of Atopic Dermatitis Pathogenesis. Int. J. Mol. Sci. 2021, 22, 4130. [Google Scholar] [CrossRef]
- Schuler, C.F.; Tsoi, L.C.; Billi, A.C.; Harms, P.W.; Weidinger, S.; Gudjonsson, J.E. Genetic and Immunological Pathogenesis of Atopic Dermatitis. J. Investig. Dermatol. 2024, 144, 954–968. [Google Scholar] [CrossRef]
- Brunner, P.M.; Guttman-Yassky, E.; Leung, D.Y.M. The immunology of atopic dermatitis and its reversibility with broad-spectrum and targeted therapies. J. Allergy Clin. Immunol. 2017, 139, S65–S76. [Google Scholar] [CrossRef]
- Criado, P.R.; Miot, H.A.; Bueno-Filho, R.; Ianhez, M.; Criado, R.F.J.; de Castro, C.C.S. Update on the pathogenesis of atopic dermatitis. An. Bras. Dermatol. 2024, 99, 895–915. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Cai, J.; Li, Z.; Lin, Y. Association of atopic dermatitis with autoimmune diseases: A bidirectional and multivariable two-sample mendelian randomization study. Front. Immunol. 2023, 14, 1132719. [Google Scholar] [CrossRef]
- Maliyar, K.; Sibbald, C.; Pope, E.; Sibbald, R.G. Diagnosis and Management of Atopic Dermatitis: A Review. Adv. Ski. Wound Care 2018, 31, 538–550. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Muscianese, M.; Piraccini, B.M.; Starace, M.; Carlesimo, M.; Mandel, V.D.; Alessandrini, A.; Calvieri, S.; Caro, G.; D’Arino, A.; et al. Italian Guidelines in diagnosis and treatment of alopecia areata. Ital. J. Dermatol. Venereol. 2019, 154, 609–623. [Google Scholar] [CrossRef]
- Sanofi. Press Release: FDA Approves Dupixent® (Dupilumab) as First Biologic Medicine for Children Aged 6 Months to 5 Years with Moderate-to-Severe Atopic Dermatitis. 2022. Available online: https://www.sanofi.com/en/media-room/press-releases/2022/2022-06-07-20-45-00-2458243 (accessed on 25 July 2024).
- Stefanis, A.J. Janus Kinase Inhibitors in the Treatment of Alopecia Areata. Prague Med. Rep. 2023, 124, 5–15. [Google Scholar] [CrossRef]
- Magdaleno-Tapial, J.; Valenzuela-Oñate, C.; García-Legaz-Martínez, M.; Martínez-Domenech, Á.; Pérez-Ferriols, A. Improvement of alopecia areata with Dupilumab in a patient with severe atopic dermatitis and review the literature. Australas. J. Dermatol. 2020, 61, e223–e225. [Google Scholar] [CrossRef] [PubMed]
- Haughton, R.D.; Herbert, S.M.; Ji-Xu, A.; Downing, L.; Raychaudhuri, S.P.; Maverakis, E. Janus kinase inhibitors for alopecia areata: A narrative review. Indian J. Dermatol. Venereol. Leprol. 2023, 89, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.-E.F.; Bhatnagar, S.; Parmentier, J.M.; Nakasato, P.; Wung, P. Upadacitinib: Mechanism of action, clinical, and translational science. Clin. Transl. Sci. 2024, 17, e13688. [Google Scholar] [CrossRef] [PubMed]
- Grieco, T.; Caviglia, M.; Cusano, G.; Sernicola, A.; Chello, C.; Del Duca, E.; Cantisani, C.; Taliano, A.; Sini, N.; Ianiro, G.; et al. Atopic Dermatitis and Ulcerative Colitis Successfully Treated with Upadacitinib. Medicina 2023, 59, 542. [Google Scholar] [CrossRef]
- Rinvoq. Summary of Product Characteristics. AbbVie Deutschland GmbH. 2022. Available online: https://www.ema.europa.eu/en/documents/product-information/rinvoq-epar-product-information_en.pdf (accessed on 14 December 2022).
- Bourkas, A.N.; Sibbald, C. Upadacitinib for the treatment of alopecia areata and severe atopic dermatitis in a paediatric patient: A case report. SAGE Open Med. Case Rep. 2022, 10, 2050313X221138452. [Google Scholar] [CrossRef]
- Kołcz, K.; Żychowska, M.; Sawińska, E.; Reich, A. Alopecia Universalis in an Adolescent Successfully Treated with Upadacitinib—A Case Report and Review of the Literature on the Use of JAK Inhibitors in Pediatric Alopecia Areata. Dermatol. Ther. 2023, 13, 843–856. [Google Scholar] [CrossRef]
- Yu, D.; Ren, Y. Upadacitinib for Successful Treatment of Alopecia Universalis in a Child: A Case Report and Literature Review. Acta Derm. Venereol. 2023, 103, adv5578. [Google Scholar] [CrossRef]
- Ha, G.U.; Kim, J.H.; Jang, Y.H. Improvement of severe alopecia areata in an adolescent patient on upadacitinib. Pediatr. Dermatol. 2024, 41, 356–358. [Google Scholar] [CrossRef]
- Zhou, C.; Li, X.; Wang, C.; Zhang, J. Alopecia Areata: An Update on Etiopathogenesis, Diagnosis, and Management. Clin. Rev. Allergy Immunol. 2021, 61, 403–423. [Google Scholar] [CrossRef]
- Adhanom, R.; Ansbro, B.; Castelo-Soccio, L. Epidemiology of Pediatric Alopecia Areata. Pediatr. Dermatol. 2025, 1, 12–23. [Google Scholar] [CrossRef]
- Miot, H.A.; Criado, P.R.; de Castro, C.C.S.; Ianhez, M.; Talhari, C.; Ramos, P.M. JAK-STAT pathway inhibitors in dermatology. An. Bras. Dermatol. 2023, 98, 656–677. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Li, J.; Fu, M.; Zhao, X.; Wang, W. The JAK/STAT signaling pathway: From bench to clinic. Signal Transduct. Target. Ther. 2021, 6, 402. [Google Scholar] [CrossRef] [PubMed]
- Donovan, J. The Evolving Story of JAK Inhibitors for Treating Alopecia Areata: A Review of Current Progress and Future Directions. Ski. Ther. Lett. 2023, 28, 1–7. [Google Scholar]
- Yamaguchi, Y.; Peeva, E.; Del Duca, E.; Facheris, P.; Bar, J.; Shore, R.; Cox, L.A.; Sloan, A.; Thaçi, D.; Ganesan, A.; et al. Ritlecitinib, a JAK3/TEC family kinase inhibitor, stabilizes active lesions and repigments stable lesions in vitiligo. Arch. Dermatol. Res. 2024, 316, 478. [Google Scholar] [CrossRef]
- Padda, I.S.; Bhatt, R.; Patel, P.; Parmar, M. StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar] [PubMed]
- Byikova, A.P.; Kozlova, I.V. Mechanisms of Development and Characteristics the Pathology of the Gastrointestinal Tract in Chronic Pancreatitis. Eksp. Klin. Gastroenterol. 2016, 11, 69–75, (In English, Russian). [Google Scholar] [PubMed]
- Paller, A.S.; Ladizinski, B.; Mendes-Bastos, P.; Siegfried, E.; Soong, W.; Prajapati, V.H.; Lio, P.; Thyssen, J.P.; Simpson, E.L.; Platt, A.M.; et al. Efficacy and Safety of Upadacitinib Treatment in Adolescents With Moderate-to-Severe Atopic Dermatitis: Analysis of the Measure Up 1, Measure Up 2, and AD Up Randomized Clinical Trials. JAMA Dermatol. 2023, 159, 526–535. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, T.-Y.; Zeng, Y.-P. Off-label use of JAK1 inhibitor upadacitinib in dermatology. Arch. Dermatol. Res. 2025, 317, 363. [Google Scholar] [CrossRef]
- Spano, F.; Donovan, J.C. Alopecia areata: Part 1: Pathogenesis, diagnosis, and prognosis. Can. Fam. Physician 2015, 61, 751–755. [Google Scholar]
- Olsen, E.A.; Hordinsky, M.K.; Price, V.H.; Roberts, J.L.; Shapiro, J.; Canfield, D.; Duvic, M.; King, L.E.; McMichael, A.J.; Randall, V.A. Alopecia areata investigational assessment guidelines—Part II. J. Am. Acad. Dermatol. 2004, 51, 440–447. [Google Scholar] [CrossRef]
- King, B.; Ohyama, M.; Kwon, O.; Zlotogorski, A.; Ko, J.; Mesinkovska, N.A.; Hordinsky, M. For the BRAVE-AA Investigators. Two Phase 3 Trials of Baricitinib for Alopecia Areata. N. Engl. J. Med. 2022, 386, 1687–1699. [Google Scholar] [CrossRef]
- Hanifin, J.M.; Thurston, M.; Omoto, M.; Cherill, R.; Tofte, S.J.; Graeber, M. The eczema area and severity index (EASI): Assessment of reliability in atopic dermatitis. EASI Evaluator Group. Exp. Dermatol. 2001, 10, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Yosipovitch, G.; Reaney, M.; Mastey, V.; Eckert, L.; Abbé, A.; Nelson, L.; Clark, M.; Williams, N.; Chen, Z.; Ardeleanu, M.; et al. Peak Pruritus Numerical Rating Scale: Psychometric validation and responder definition for assessing itch in moderate-to-severe atopic dermatitis. Br. J. Dermatol. 2019, 181, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Lewis-Jones, M.S.; Finlay, A.Y. The Children’s Dermatology Life Quality Index (CDLQI): Initial validation and practical use. Br. J. Dermatol. 1995, 132, 942–949. [Google Scholar] [CrossRef] [PubMed]
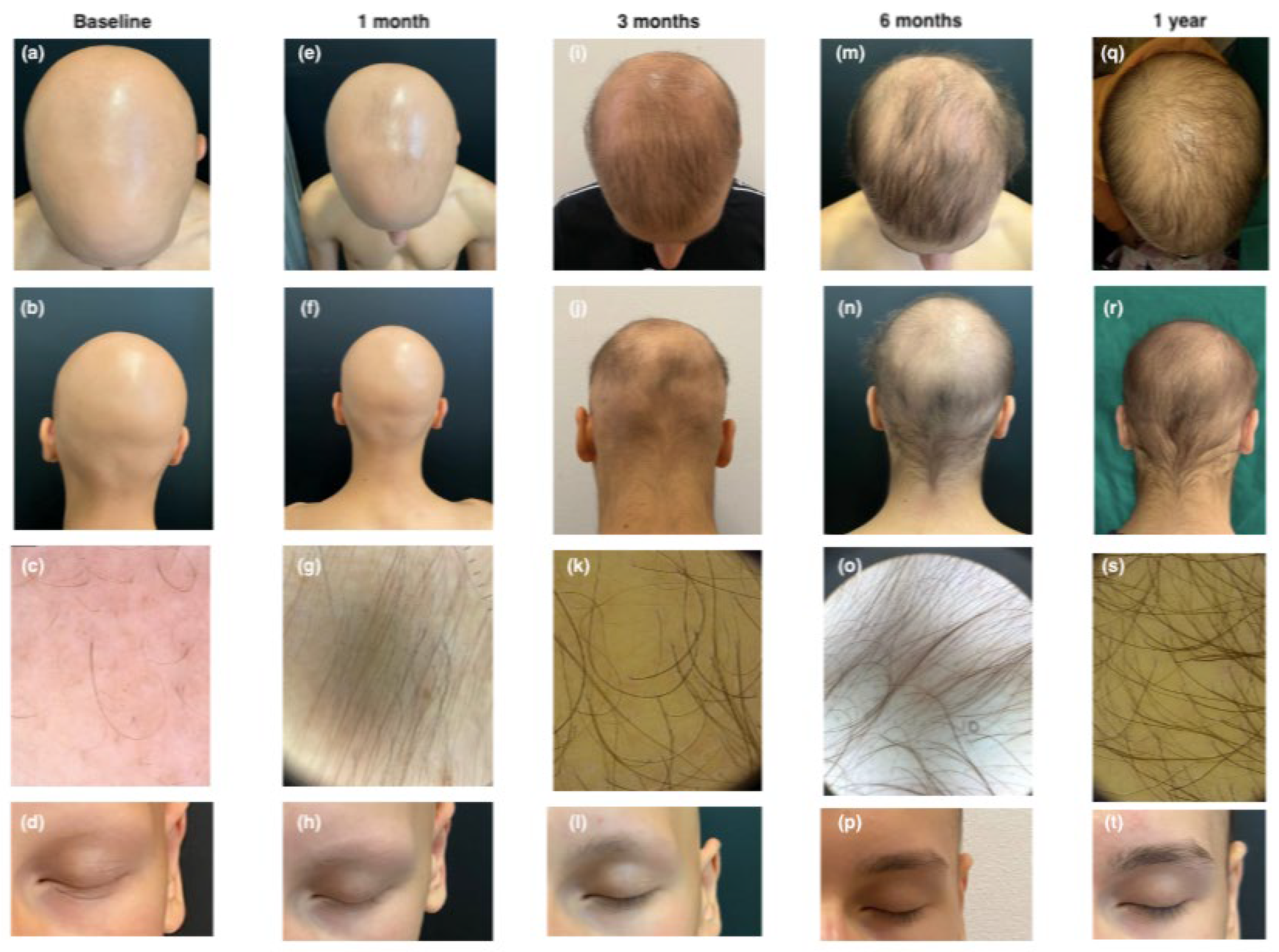

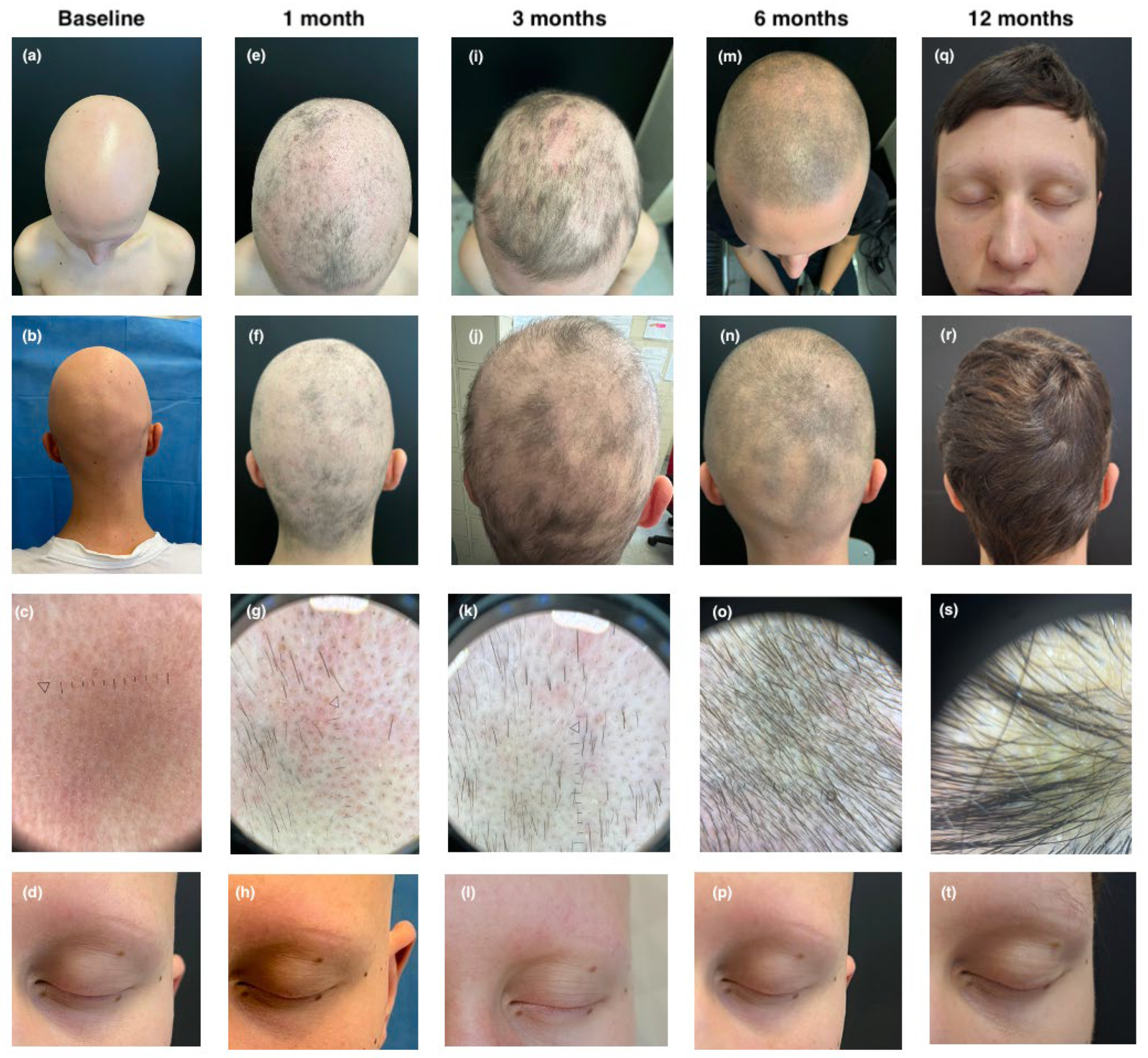
| Case Number | Age (Years)/Sex (M/F) | Atopic Comorbidities | AD Duration and Severity | Non-Atopic Comorbidities | AA Duration and Severity | Prior Treatment |
|---|---|---|---|---|---|---|
| 1 | 13/M | AD, allergic rhinoconjunctivitis | Early onset; moderate EASI: 10 IGA: 1 Itch NRS: 6 Sleep NRS: 4 DLQI: 10 | None | 1 year; AU (SALT score 100) | Dupilumab |
| 2 | 12/M | AD | Early onset; moderate EASI: 9 IGA: 1 Itch NRS: 6 Sleep NRS: 4 DLQI: 8 | None | 3 months; ophiasis (SALT score 51.5) | TSC, SCS, CsA |
| 3 | 14/M | AD | Early onset; moderate EASI: 8 IGA: 2 Itch NRS: 6 Sleep NRS: 5 DLQI: 10 | CD, EGE | 17 months; AU (SALT score 100) | SCS, dupilumab |
| Case Number | Daily Dosage | Treatment Duration, Months | SALT Scores at Follow-Up Visits | EBA (0–3) | ELA (03) | AD Scores at 1-Month Follow-Up Visit | Adverse Events |
|---|---|---|---|---|---|---|---|
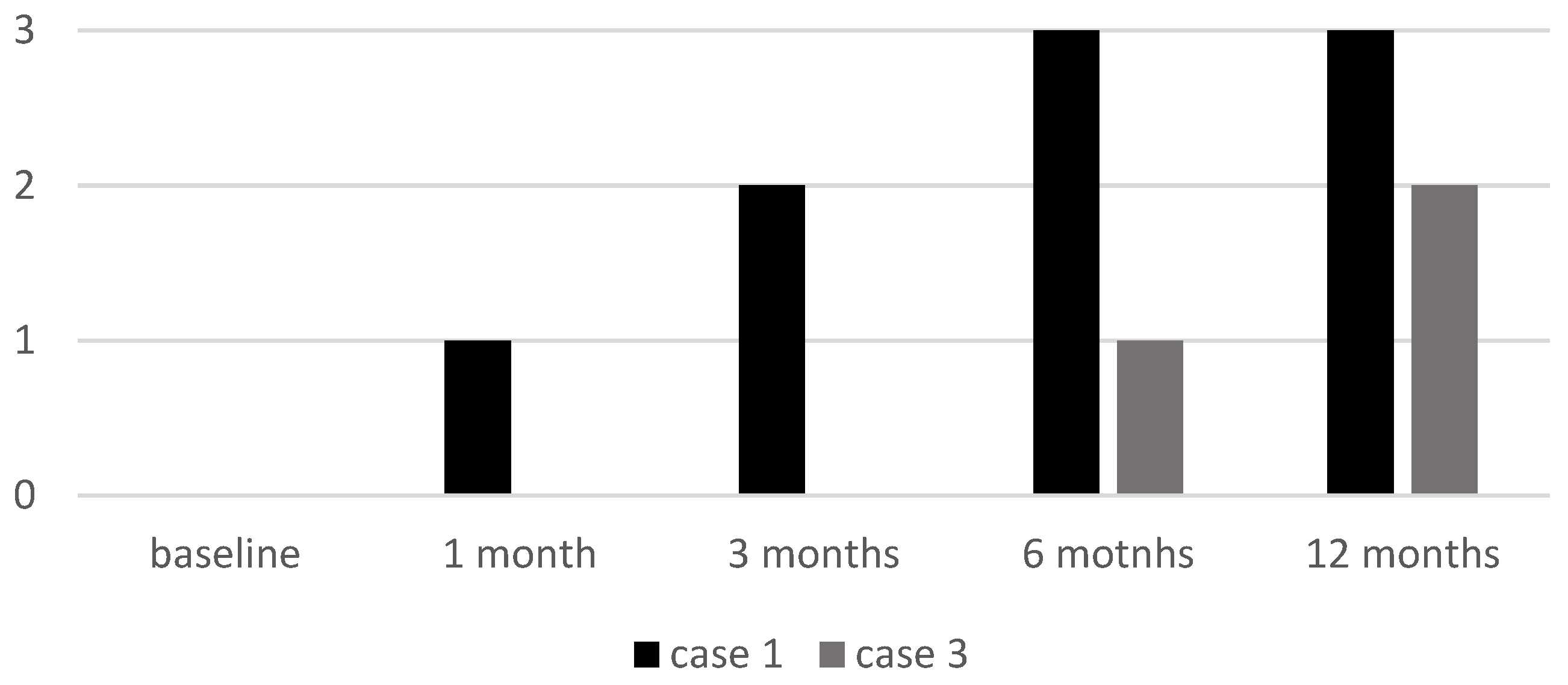
| 1 | 15 mg | 12 | 1 month: 96 3 months: 35.3 6 months: 21.8 12 months: 14 | Baseline: 0 1 month: 1 3 months: 2 6 months: 3 | Baseline: 0 1 month: 1 3 months: 2 6 months: 3 | EASI: 0 IGA: 0 Itch NRS: 0 Sleep NRS: 0 CDLQI: 0 | None |
| 2 | 15 mg | 12 | 1 month: 47.8 3 months: 35.3 6 months: 15 12 months: 1.8 | NI | NI | EASI: 0 IGA: 0 Itch NRS: 0 Sleep NRS: 0 CDLQI: 0 | Mild transient CPK elevation |
| 3 | 15 mg | 12 | 1 month: 92 3 months: 78 6 months: 45 12 months: 0 | Baseline: 0 1 month: 0 6 months: 1 12 months: 2 | Baseline: 0 1 month: 0 6 months: 1 12 months: 2 | EASI: 0 IGA: 0 Itch NRS: 0 Sleep NRS: 0 CDLQI: 0 | Mild transient CPK elevation |
| CASE 1 | T0 | T1 | T2 | T3 | T4 |
|---|---|---|---|---|---|
| Hb | 13.7 g/dL | 14.1 g/dL | 14.0 g/dL | 14.2 g/dL | 14.1 g/dL |
| Neutrophils | 2.8 × 103/uL | 3.4 × 103/uL | 3.2 × 103/uL | 3.0 × 103/uL | 3.2 × 103/uL |
| Eosinophils | 0.1 × 103/uL | 0.1 × 103/uL | 0.1 × 103/uL | 0.1 × 103/uL | 0.1 × 103/uL |
| IgE | 1264.0 IU/mL | 1150 IU/mL | 364 IU/mL | 252 IU/mL | 256 IU/mL |
| ESR | 4 mm/h | 4 mm/h | 3 mm/h | 3 mm/h | 3 mm/h |
| CRP | 0.6 mg/dL | 0.6 mg/dL | 0.5 mg/dL | 0.5 mg/dL | 0.4 mg/dL |
| Total Cholesterol | 145 mg/dL | 157 mg/dL | 150 mg/dL | 142 mg/dL | 155 mg/dL |
| Triglycerides | 46 mg/dL | 79 mg/dL | 62 mg/dL | 71 mg/dL | 57 mg/dL |
| AST | 22 U/L | 19 U/L | 20 U/L | 21 U/L | 19 U/L |
| ALT | 17 U/L | 19 U/L | 19 U/L | 18 U/L | 18 U/L |
| CPK | 190 U/L | 148 U/L | 142 U/L | 145 U/L | 153 U/L |
| CASE 2 | |||||
| Hb | 14.0 g/dL | 14.5 g/dL | 14.1 g/dL | 14.7 g/dL | 14.3 g/dL |
| Neutrophils | 1.96 × 103/uL | 4.18 × 103/uL | 1.98 × 103/uL | 2.23 × 103/uL | 2.15 × 103/uL |
| Eosinophils | 0.13 × 103/uL | 0.12 × 103/uL | 0.14 × 103/uL | 0.12 × 103/uL | 0.12 × 103/uL |
| IgE | 120 IU/mL | 110 IU/mL | 82 IU/mL | 63 IU/mL | 58 IU/mL |
| ESR | 21 mm/h | 19 mm/h | 15 mm/h | 11 mm/h | 10 mm/h |
| CRP | 3.22 mg/dL | 1.42 mg/dL | 0.8 mg/dL | 0.7 mg/dL | 0.3 mg/dL |
| Total Cholesterol | 155 mg/dL | 143 mg/dL | 155 mg/dL | 145 mg/dL | 148 mg/dL |
| Triglycerides | 105 mg/dL | 110 mg/dL | 66 mg/dL | 69 mg/dL | 72 mg/dL |
| AST | 27 U/L | 23 U/L | 38 U/L | 21 U/L | 22 U/L |
| ALT | 17 U/L | 13 U/L | 21 U/L | 13 U/L | 15 U/L |
| CPK | 83.80 U/L | 175 U/L | 278 U/L | 158 U/L | 143 U/L |
| CASE 3 | |||||
| Hb | 16.2 g/dL | 15.8 g/dL | 15.6 g/dL | 15.5 g/dL | 15.4 g/dL |
| Neutrophils | 4.14 × 103/uL | 3.88 × 103/uL | 3.37 × 103/uL | 3.57 × 103/uL | 3.83 × 103/uL |
| Eosinophils | 1.13 × 103/uL | 0.85 × 103/uL | 0.76 × 103/uL | 0.75 × 103/uL | 0.35 × 103/uL |
| IgE | 40.2 IU/mL | 39.9 IU/mL | 38.2 IU/mL | 38.1 IU/mL | 38.0 IU/mL |
| ESR | 9 mm/h | 8 mm/h | 5 mm/h | 5 mm/h | 4 IU/mL |
| CRP | 0.3 mg/dL | 0.3 mg/dL | 0.2 mg/dL | 0.2 mg/dL | 0.04 mg/dL |
| Total Cholesterol | 159.7 mg/dL | 148 mg/dL | 152.3 mg/dL | 154.2 mg/dL | 161 mg/dL |
| Triglycerides | 54.9 mg/dL | 24 mg/dL | 31 mg/dL | 36 mg/dL | 35 mg/dL |
| AST | 16 U/L | 21 U/L | 28 U/L | 23 U/L | 22 U/L |
| ALT | 12 U/L | 15 U/L | 23 U/L | 16 U/L | 23 U/L |
| CPK | 112 U/L | 382 U/L | 135 U/L | 143 U/L | 116 U/L |
| Study | Age (Years)/Sex (M/F) | Atopic Comorbidities | AD Duration and Severity | Non-Atopic Comorbidities | AA Duration and Severity | Prior Treatment | Daily Dosage | Treatment Duration, Months | Treatment Outcome AA; AD | Side Effects |
|---|---|---|---|---|---|---|---|---|---|---|
| Bourkas et al. [18] | 14/M | AD, allergic rhinitis, asthma, allergies to sesame seeds, grass, tree nuts, eggs | NA; severe | None | 13 years; almost the entire scalp | ICSI, TCS, TCI, CsA, LDOM, MTX | NA | 5 | Complete response; marked improvement | NA |
| Kołcz et al. [19] | 14/F | AD | Early childhood; mild | None | 16 months; AU (SALT score 100) | TSC, TCI, TMX, DPCP, NB UVB | 15 mg | 3 | Complete response (SALT score 0); complete response | Mild transient leukopenia |
| Yu et al. [20] | 9/F | AD | NA; mild (EASI score 2.5) | None | 7 years; AU (SALT score 98) | TCS, TMX, TCI, SCS, oral compound glycyrrhizin tablets | 15 mg | 5 | Complete response (SALT score 0); complete remission | None |
| Ha et al. [21] | 15/F | AD | NA; NA | None | 11 years; AT (SALT score 100) | CsA, SCS, ICSI, DPCP, EL, CT | 15 mg | 12 | SALT score 11.7; NA | None |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Battilotti, C.; Azzella, G.; Dattola, A.; Rossi, A.; Svara, F.; Chello, C.; Del Duca, E.; Paolino, G.; Nisticò, S.P.; Pellacani, G.; et al. Efficacy of Upadacitinib in Treating Alopecia Areata, Atopic Dermatitis, and Th1 Comorbidities in Pediatric Patients: A Comprehensive Case Series and Literature Review. J. Clin. Med. 2025, 14, 3881. https://doi.org/10.3390/jcm14113881
Battilotti C, Azzella G, Dattola A, Rossi A, Svara F, Chello C, Del Duca E, Paolino G, Nisticò SP, Pellacani G, et al. Efficacy of Upadacitinib in Treating Alopecia Areata, Atopic Dermatitis, and Th1 Comorbidities in Pediatric Patients: A Comprehensive Case Series and Literature Review. Journal of Clinical Medicine. 2025; 14(11):3881. https://doi.org/10.3390/jcm14113881
Chicago/Turabian StyleBattilotti, Chiara, Giulia Azzella, Annunziata Dattola, Alfredo Rossi, Francesca Svara, Camilla Chello, Ester Del Duca, Giovanni Paolino, Steven P. Nisticò, Giovanni Pellacani, and et al. 2025. "Efficacy of Upadacitinib in Treating Alopecia Areata, Atopic Dermatitis, and Th1 Comorbidities in Pediatric Patients: A Comprehensive Case Series and Literature Review" Journal of Clinical Medicine 14, no. 11: 3881. https://doi.org/10.3390/jcm14113881
APA StyleBattilotti, C., Azzella, G., Dattola, A., Rossi, A., Svara, F., Chello, C., Del Duca, E., Paolino, G., Nisticò, S. P., Pellacani, G., & Grieco, T. (2025). Efficacy of Upadacitinib in Treating Alopecia Areata, Atopic Dermatitis, and Th1 Comorbidities in Pediatric Patients: A Comprehensive Case Series and Literature Review. Journal of Clinical Medicine, 14(11), 3881. https://doi.org/10.3390/jcm14113881









