Diagnostic Accuracy of Ultrasound Guided Percutaneous Pleural Needle Biopsy for Malignant Pleural Mesothelioma
Abstract
1. Introduction
2. Methods
2.1. Patients
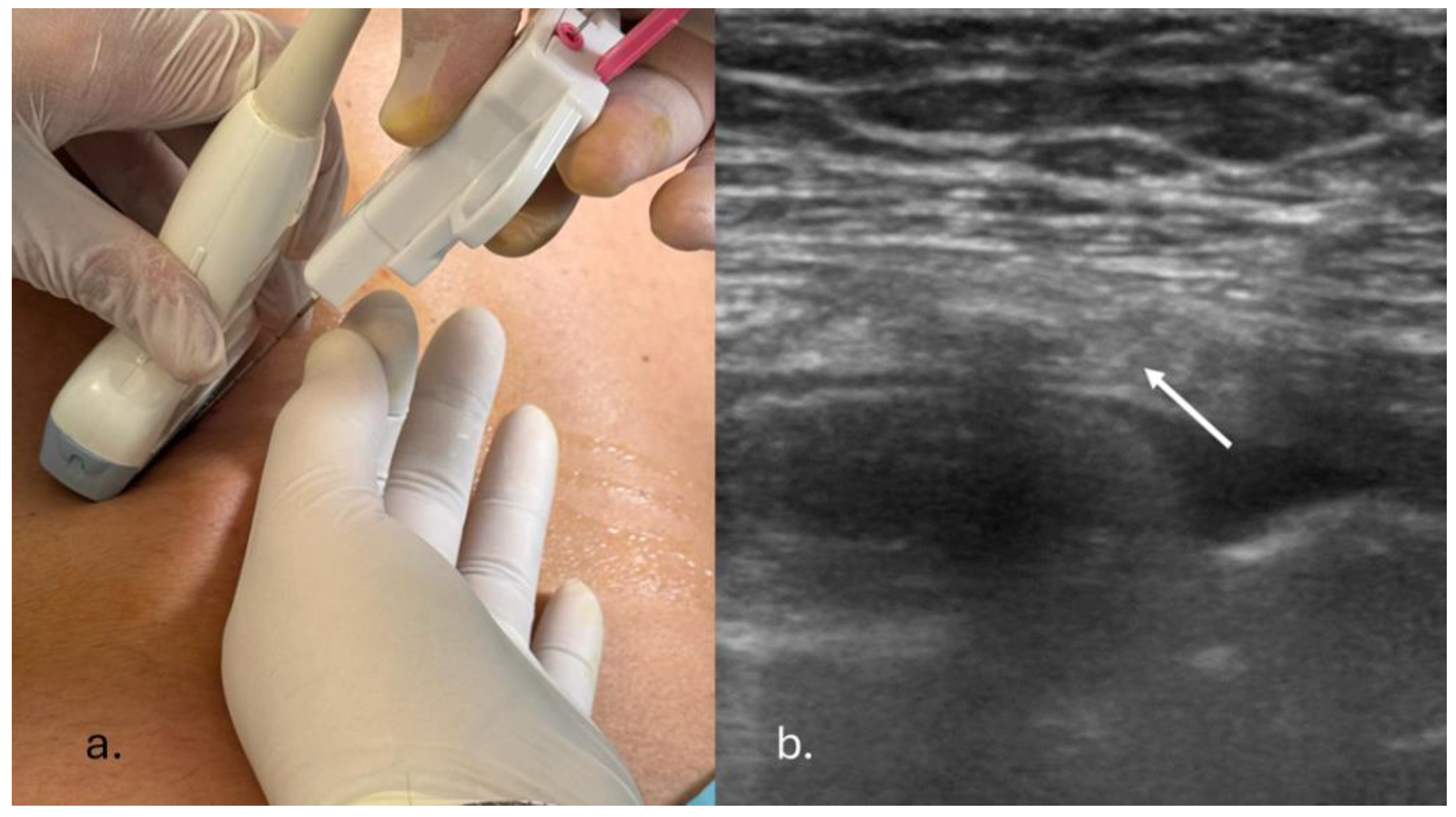
2.2. US-PPNB: Description of Procedure
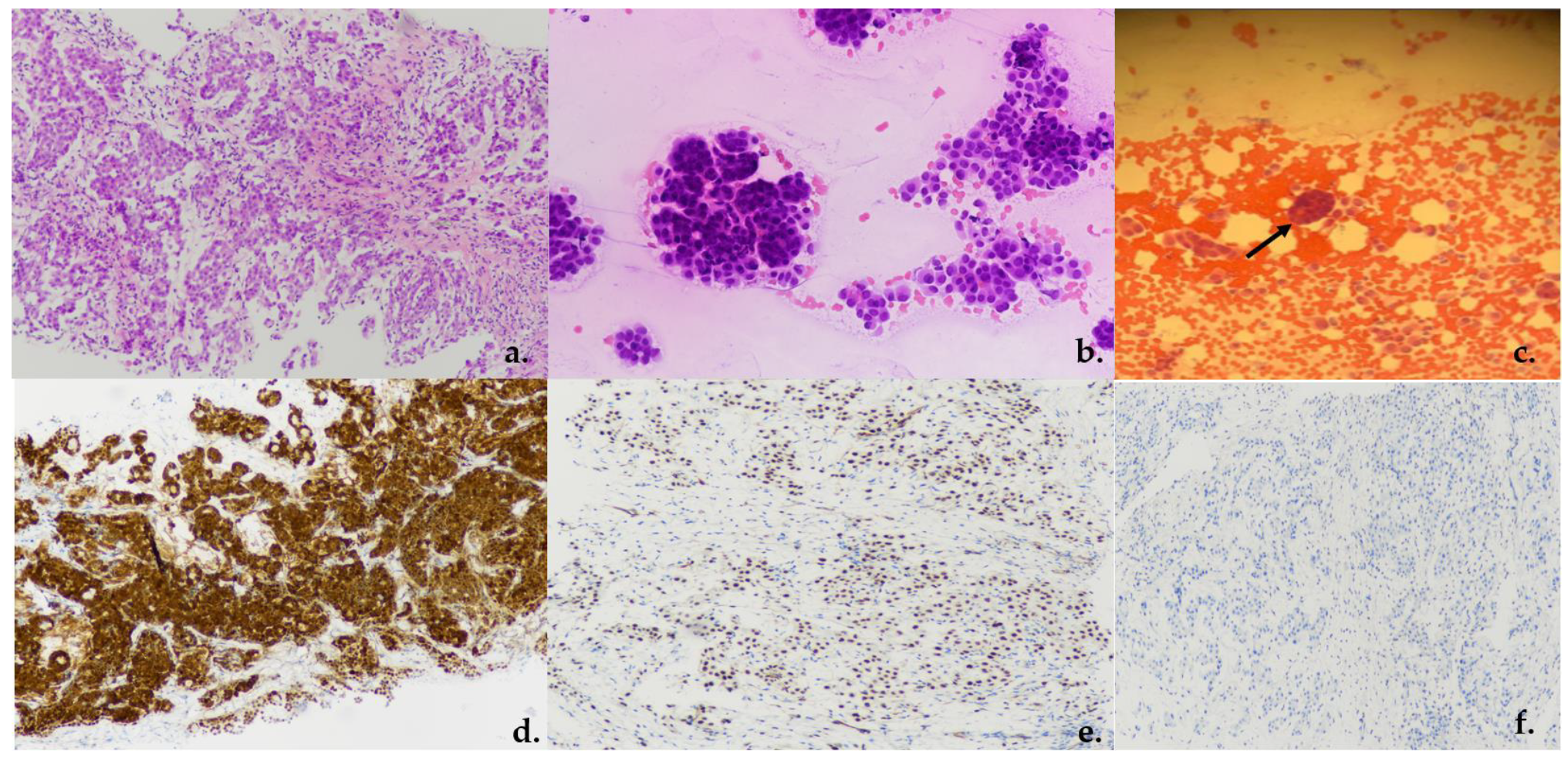
2.3. Assessment of Samples
2.4. Statistical Analysis
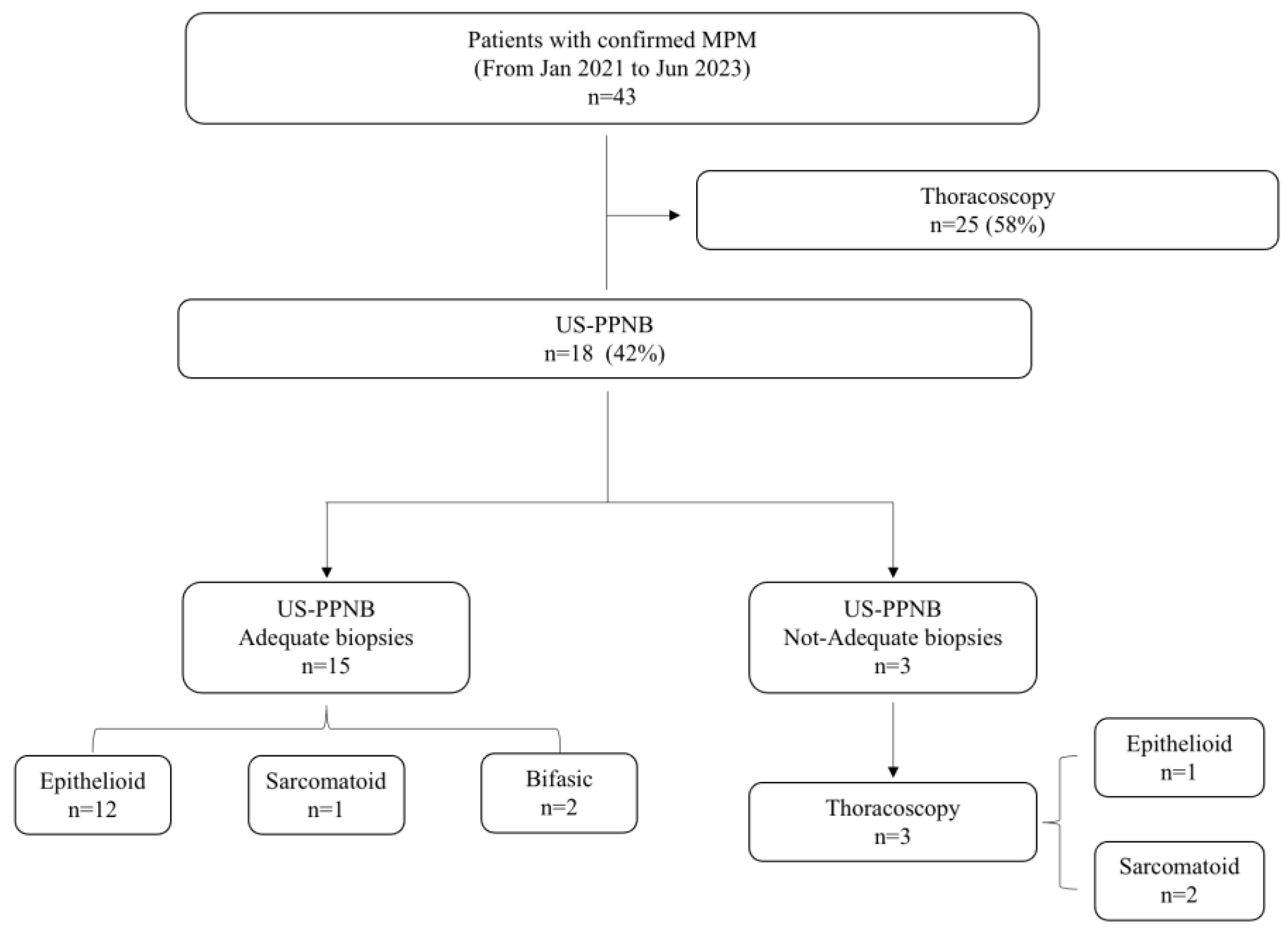
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bianco, A.; Valente, T.; De Rimini, M.L.; Sica, G.; Fiorelli, A. Clinical diagnosis of malignant pleural mesothelioma. J. Thorac. Dis. 2018, 10, S253–S261. [Google Scholar] [CrossRef] [PubMed]
- Sauter, J.L.; Dacic, S.; Galateau-Salle, F.; Attanoos, R.L.; Butnor, K.J.; Churg, A.; Husain, A.N.; Kadota, K.; Khoor, A.; Nicholson, A.G.; et al. The 2021 WHO Classification of Tumors of the Pleura: Advances Since the 2015 Classification. J. Thorac. Oncol. 2022, 17, 608–622. [Google Scholar] [CrossRef]
- Mollica, M.; Salvi, R.; Paoli, G.; Graziani, V.; Cerqua, F.S.; Iadevaia, C.; Lavoretano, S.; D’agnano, V.; Rocco, D.; Fiorelli, A.; et al. Lung cancer management: Challenges in elderly patients. J. Gerontol. Geriatr. 2019, 2019, 132–140. [Google Scholar]
- Mei, F.; Bonifazi, M.; Rota, M.; Cirilli, L.; Grilli, M.; Duranti, C.; Zuccatosta, L.; Bedawi, E.O.; McCracken, D.; Gasparini, S.; et al. Diagnostic Yield and Safety of Image-Guided Pleural Biopsy: A Systematic Review and Meta-Analysis. Respiration 2021, 100, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Dangi, A.; Roy, K.; Agrawal, S.; Chen, H.; Ashok, A.; Wible, C.; Osman, M.; Pratap, R.; Kothapalli, S.-R. A modular approach to neonatal whole-brain photoacoustic imaging. In Proceedings of the Photons Plus Ultrasound: Imaging and Sensing 2020, San Francisco, CA, USA, 2–5 February 2020; Volume 11240, p. 1124057. [Google Scholar]
- Rinaldi, L.; Milione, S.; Fascione, M.C.; Pafundi, P.C.; Altruda, C.; Di Caterino, M.; Monaco, L.; Reginelli, A.; Perrotta, F.; Porta, G.; et al. Relevance of lung ultrasound in the diagnostic algorithm of respiratory diseases in a real-life setting: A multicentre prospective study. Respirology 2019, 25, 535–542. [Google Scholar] [CrossRef]
- Stark, P. Imaging in Pulmonary Disease; Goldman, L., Schafer, Eds.; Goldman’s Cecil Medicine; Elsevier: Amsterdam, The Netherlands, 2012; Chapter 84; ISBN 978-1-4377-1604-7. [Google Scholar]
- Demi, L.; Wolfram, F.; Klersy, C.; De Silvestri, A.; Ferretti, V.V.; Muller, M.; Miller, D.; Feletti, F.; Wełnicki, M.; Buda, N.; et al. New International Guidelines and Consensus on the Use of Lung Ultrasound. J. Ultrasound Med. 2023, 42, 309–344. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, J.; Zhou, X.; Chen, W.; Zhang, S.; Li, Y.; Zhou, D.; He, L.; Tang, Q. Diagnostic ability and its influenced factors of ultrasound-guided percutaneous pleural needle biopsy diagnosis for malignant pleural mesothelioma. Front. Surg. 2022, 9, 1022505. [Google Scholar] [CrossRef]
- Bahar, E.; Yoon, H. Lidocaine: A Local Anesthetic, Its Adverse Effects and Management. Medicina 2021, 57, 782. [Google Scholar] [CrossRef] [PubMed]
- Conway, A.; Rolley, J.; Sutherland, J.R. Midazolam for sedation before procedures. Cochrane Database Syst. Rev. 2016, 2016, CD009491. [Google Scholar] [CrossRef]
- Messina, G.; Bove, M.; Natale, G.; Di Filippo, V.; Opromolla, G.; Rainone, A.; Leonardi, B.; Martone, M.; Fiorelli, A.; Vicidomini, G.; et al. Diagnosis of malignant pleural disease: Ultrasound as “a detective probe”. Thorac. Cancer 2023, 14, 223–230. [Google Scholar] [CrossRef]
- Wong, H.B.; Lim, G.H. Measures of diagnostic accuracy: Sensitivity, specificity, PPV and NPV. Proc. Singap. Healthc. 2011, 20, 316–318. [Google Scholar] [CrossRef]
- Tamburrini, M.; Desai, U.; Fanti, G.; Scarda, A.; Zampieri, F.; Zuccon, U.; Thakare, P.; Contoli, M.; Papi, A. Medical thoracoscopy without pleural fluid: How I do it. Monaldi Arch. Chest Dis. Arch. Monaldi Per Le Mal. Del Torace 2021, 92, 1. [Google Scholar] [CrossRef] [PubMed]
- Perrotta, F.; D’Agnano, V.; Scialò, F.; Komici, K.; Allocca, V.; Nucera, F.; Salvi, R.; Stella, G.M.; Bianco, A. Evolving concepts in COPD and lung cancer: A narrative review. Minerva Med. 2022, 113, 436–448. [Google Scholar] [CrossRef] [PubMed]
- Komici, K.; Bencivenga, L.; Navani, N.; D’Agnano, V.; Guerra, G.; Bianco, A.; Rengo, G.; Perrotta, F. Frailty in patients with lung cancer: A systematic review and meta-analysis. Chest 2022, 162, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Haaga, J.R.; LiPuma, J.P.; Bryan, P.J.; Balsara, V.J.; Cohen, A.M. Clinical comparison of small-and large-caliber cutting needles for biopsy. Radiology 1983, 146, 665–667. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, M.; Rinaldi, P.; Rella, R.; Fabrizi, G.; Petta, F.; Carlino, G.; Di Leone, A.; Mulè, A.; Bufi, E.; Romani, M.; et al. Effect of Needle Size in Ultrasound-guided Core Needle Breast Biopsy: Comparison of 14-, 16-, and 18-Gauge Needles. Clin. Breast Cancer 2017, 17, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Heilo, A.; Stenwig, A.E.; Solheim, O.P. Malignant pleural mesothelioma: US-guided histologic core-needle biopsy. Radiology 1999, 211, 657–659. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tang, J.; Zhou, X.; Zhou, D.; Wang, J.; Tang, Q. Ultrasound-guided pleural cutting needle biopsy: Accuracy and factors influencing diagnostic yield. J. Thorac. Dis. 2018, 10, 3244–3252. [Google Scholar] [CrossRef]
- Woolhouse, I.; Bishop, L.; Darlison, L.; Fonseka, D.D.; Edey, A.; Edwards, J.; Faivre-Finn, C.; Fennell, D.A.; Holmes, S.; Kerr, K.M.; et al. British Thoracic Society Guideline for the investigation and management of malignant pleural mesothelioma. Thorax 2018, 73, i1–i30. [Google Scholar] [CrossRef]
- Lichtenstein, D.; Goldstein, I.; Mourgeon, E.; Cluzel, P.; Grenier, P.; Rouby, J.-J. Comparative diagnostic performances of auscultation, chest radiography, and lung ultrasonography in acute respiratory distress syndrome. Anesthesiology 2004, 100, 9–15. [Google Scholar] [CrossRef]
- Stigt, J.A.; Boers, J.E.; Groen, H.J.M. Analysis of “dry” mesothelioma with ultrasound guided biopsies. Lung Cancer 2012, 78, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Zahid, I.; Sharif, S.; Routledge, T.; Scarci, M. What is the best way to diagnose and stage malignant pleural mesothelioma? Interact. Cardiovasc. Thorac. Surg. 2011, 12, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, H.; Terazawa, K.; Morita, N.; Otomi, Y.; Yamashita, K.; Nishitani, H. Is FDG-PET/CT useful for managing malignant pleural mesothelioma? J. Med. Investig. 2009, 56, 16–20. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Basu, S.; Saboury, B.; Torigian, D.A.; Alavi, A. Current evidence base of FDG-PET/CT imaging in the clinical management of malignant pleural mesothelioma: Emerging significance of image segmentation and global disease assessment. Mol. Imaging Biol. 2011, 13, 801–811. [Google Scholar] [CrossRef] [PubMed]
- Popat, S.; Baas, P.; Faivre-Finn, C.; Girard, N.; Nicholson, A.G.; Nowak, A.K.; Opitz, I.; Scherpereel, A.; Reck, M. Malignant pleural mesothelioma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up(☆). Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. 2022, 33, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Bianco, A.; Perrotta, F.; Barra, G.; Malapelle, U.; Rocco, D.; De Palma, R. Prognostic Factors and Biomarkers of Responses to Immune Checkpoint Inhibitors in Lung Cancer. Int. J. Mol. Sci. 2019, 20, 4931. [Google Scholar] [CrossRef] [PubMed]
- Sandach, P.; Seifert, R.; Fendler, W.P.; Hautzel, H.; Herrmann, K.; Maier, S.; Plönes, T.; Metzenmacher, M.; Ferdinandus, J. A Role for PET/CT in Response Assessment of Malignant Pleural Mesothelioma. Semin. Nucl. Med. 2022, 52, 816–823. [Google Scholar] [CrossRef]
- Bianco, A.; D’Agnano, V.; Matera, M.G.; Della Gravara, L.; Perrotta, F.; Rocco, D. Immune checkpoint inhibitors: A new landscape for extensive stage small cell lung cancer treatment. Expert Rev. Respir. Med. 2021, 15, 1415–1425. [Google Scholar] [CrossRef] [PubMed]
- Lococo, F.; Rena, O.; Torricelli, F.; Filice, A.; Rapicetta, C.; Boldorini, R.; Paci, M.; Versari, A. 18F-fluorodeoxyglucose positron emission tomography in malignant pleural mesothelioma: Diagnostic and prognostic performance and its correlation to pathological results. Interact. Cardiovasc. Thorac. Surg. 2020, 30, 593–596. [Google Scholar] [CrossRef]
- Xu, W.; Wen, Q.; Zhang, X.; Pan, M.; Liu, C.; Zha, L.; Huang, P. The Application of Contrast Enhanced Ultrasound for Core Needle Biopsy of Subpleural Pulmonary Lesions: Retrospective Analysis in 92 Patients. Ultrasound Med. Biol. 2021, 47, 1253–1260. [Google Scholar] [CrossRef]
- Yusuf, G.T.; Fang, C.; Tran, S.; Rao, D.; Bartlett-Pestell, S.; Stefanidis, K.; Huang, D.Y.; Sidhu, P.S. A pictorial review of the utility of CEUS in thoracic biopsies. Insights Imaging 2021, 12, 9. [Google Scholar] [CrossRef] [PubMed]
| Rib fractures |
| AMS |
| Uncontrolled cardiac arrythmia |
| PNT |
| Severe anemia |
| Chronic heart failure (FE < 40%) |
| Severe respiratory failure (P/F ratio < 250) |
| INR > 1.5 |
| Total number of patients | 18 |
| Males | 11 (61%) |
| Median age (sd) | 71.2 (±6.61) |
| Mean ECOG-PS | 1.55 |
| TNM | |
| 3 (16%) |
| 15 (84%) |
| Reported exposure to asbestos | 11 (61%) |
| Mean pleural lesion thickness (sd) | 13.5 mm (±9.45) |
| Mean SUVmax (sd) | 10.7 (±1.58) |
| Pleural effusion | 11 (61%) |
| Comorbidity | |
| Arterial hypertension | 16 (88.8%) |
| Chronic renal failure | 5 (27.8%) |
| COPD | 7 (38.9%) |
| Chronic respiratory failure | 7 (38.9%) |
| Pregress stroke | 3 (16.7%) |
| Chronic ischemic disease | 4 (22.2%) |
| Diabetes | 3 (16.7%) |
| Prostatic hypertofic disease | 6 (33.3%) |
| Mean number of samples (sd) | 2 (±0.9) |
| Accuracy | |
| Adequate biopsies | 15 (83%) |
| Not-adequate biopsies | 3 (17%) |
| Histology | |
| Epithelioid | 13 (72%) |
| Sarcomatoid | 3 (17%) |
| Bifasic | 2 (11%) |
| Adequate biopsies | |
| Epithelioid | 12 (72%) |
| Sarcomatoid | 1 (5%) |
| Bifasic | 2 (11%) |
| Not-adequate biopsies | |
| Epithelioid | 1 (5%) |
| Sarcomatoid | 2 (11%) |
| Bifasic | 0 (0%) |
| Pleural lesion thickness | |
| Adequate biopsies | 15.4 mm (±9.19) |
| Not-adequate biopsies | 3.77 mm (±0.60) |
| Pain assessment | |
| NRS < 5 | 14 (78%) |
| NRS > 5 | 4 (22%) |
| Complications | |
| PNT | 4 (22%) |
| Hemorrhagic complication | 0 (0%) |
| DOP | 21 min (±4.67) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iadevaia, C.; D’Agnano, V.; Pagliaro, R.; Nappi, F.; Lucci, R.; Massa, S.; Bianco, A.; Perrotta, F. Diagnostic Accuracy of Ultrasound Guided Percutaneous Pleural Needle Biopsy for Malignant Pleural Mesothelioma. J. Clin. Med. 2024, 13, 2600. https://doi.org/10.3390/jcm13092600
Iadevaia C, D’Agnano V, Pagliaro R, Nappi F, Lucci R, Massa S, Bianco A, Perrotta F. Diagnostic Accuracy of Ultrasound Guided Percutaneous Pleural Needle Biopsy for Malignant Pleural Mesothelioma. Journal of Clinical Medicine. 2024; 13(9):2600. https://doi.org/10.3390/jcm13092600
Chicago/Turabian StyleIadevaia, Carlo, Vito D’Agnano, Raffaella Pagliaro, Felice Nappi, Raffaella Lucci, Simona Massa, Andrea Bianco, and Fabio Perrotta. 2024. "Diagnostic Accuracy of Ultrasound Guided Percutaneous Pleural Needle Biopsy for Malignant Pleural Mesothelioma" Journal of Clinical Medicine 13, no. 9: 2600. https://doi.org/10.3390/jcm13092600
APA StyleIadevaia, C., D’Agnano, V., Pagliaro, R., Nappi, F., Lucci, R., Massa, S., Bianco, A., & Perrotta, F. (2024). Diagnostic Accuracy of Ultrasound Guided Percutaneous Pleural Needle Biopsy for Malignant Pleural Mesothelioma. Journal of Clinical Medicine, 13(9), 2600. https://doi.org/10.3390/jcm13092600






