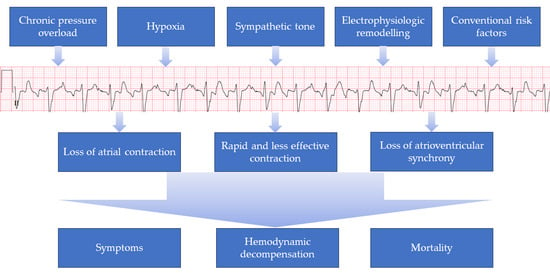
Arrhythmias in Patients with Pulmonary Hypertension and Right Ventricular Failure: Importance of Rhythm Control Strategies
Abstract
1. Introduction
2. Pathogenesis
3. Incidence
4. Significance of Atrial Arrhythmias in PH
5. Clinical Presentation
6. Management of Atrial Arrhythmias in PH
6.1. Emergency Management
6.2. Rate-Control Drugs
6.3. Rhythm-Control Drugs
6.4. Device Therapy
6.5. Catheter Ablation
Anticoagulation
7. Anesthetic Management of Patients with PH
7.1. Preoperative Risk Assessment in PH
7.2. Optimization of PH Prior to Procedure
7.3. Intraoperative Management of PH
7.4. Anesthetic Agents
8. Guidelines
9. Conclusions
Funding
Conflicts of Interest
References
- Wen, L.; Sun, M.-L.; An, P.; Jiang, X.; Sun, K.; Zheng, L.; Liu, Q.-Q.; Wang, L.; Zhao, Q.-H.; He, J.; et al. Frequency of supraventricular arrhythmias in patients with idiopathic pulmonary arterial hypertension. Am. J. Cardiol. 2014, 114, 1420–1425. [Google Scholar] [CrossRef]
- Humbert, M.; Kovacs, G.; Hoeper, M.M.; Badagliacca, R.; Berger, R.M.F.; Brida, M.; Carlsen, J.; Coats, A.J.S.; Escribano-Subias, P.; Ferrari, P.; et al. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur. Heart J. 2022, 43, 3618–3731. [Google Scholar] [CrossRef] [PubMed]
- Middleton, J.T.; Maulik, A.; Lewis, R.; Kiely, D.G.; Toshner, M.; Charalampopoulos, A.; Kyriacou, A.; Rothman, A. Arrhythmic Burden and Outcomes in Pulmonary Arterial Hypertension. Front. Med. 2019, 6, 169. [Google Scholar] [CrossRef] [PubMed]
- Cannillo, M.; Marra, W.G.; Gili, S.; D’Ascenzo, F.; Morello, M.; Mercante, L.; Mistretta, E.; Salera, D.; Zema, D.; Bissolino, A.; et al. Supraventricular Arrhythmias in Patients with Pulmonary Arterial Hypertension. Am. J. Cardiol. 2015, 116, 1883–1889. [Google Scholar] [CrossRef] [PubMed]
- Simonneau, G.; Gatzoulis, M.A.; Adatia, I.; Celermajer, D.; Denton, C.; Ghofrani, A.; Sanchez, M.A.G.; Kumar, R.K.; Landzberg, M.; Machado, R.F.; et al. Updated clinical classification of pulmonary hypertension. J. Am. Coll. Cardiol. 2013, 62, D34–D41. [Google Scholar] [CrossRef] [PubMed]
- Cirulis, M.M.; Ryan, J.J.; Archer, S.L. Pathophysiology, incidence, management, and consequences of cardiac arrhythmia in pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension. Pulm. Circ. 2019, 9, 2045894019834890. [Google Scholar] [CrossRef] [PubMed]
- Grapsa, J.; Gibbs, J.S.R.; Cabrita, I.Z.; Watson, G.F.; Pavlopoulos, H.; Dawson, D.; Gin-Sing, W.; Howard, L.S.G.E.; Nihoyannopoulos, P. The association of clinical outcome with right atrial and ventricular remodelling in patients with pulmonary arterial hypertension: Study with real-time three-dimensional echocardiography. Eur. Heart J. Cardiovasc. Imaging 2012, 13, 666–672. [Google Scholar] [CrossRef]
- Wanamaker, B.; Cascino, T.; McLaughlin, V.; Oral, H.; Latchamsetty, R.; Siontis, K.C. Atrial Arrhythmias in Pulmonary Hypertension: Pathogenesis, Prognosis and Management. Arrhythm. Electrophysiol. Rev. 2018, 7, 43–48. [Google Scholar] [CrossRef]
- Medi, C.; Kalman, J.M.; Ling, L.; Teh, A.W.; Lee, G.; Lee, G.; Spence, S.J.; Kaye, D.M.; Kistler, P.M. Atrial electrical and structural remodeling associated with longstanding pulmonary hypertension and right ventricular hypertrophy in humans. J. Cardiovasc. Electrophysiol. 2012, 23, 614–620. [Google Scholar] [CrossRef]
- Velez-Roa, S.; Ciarka, A.; Najem, B.; Vachiery, J.-L.; Naeije, R.; van de Borne, P. Increased sympathetic nerve activity in pulmonary artery hypertension. Circulation 2004, 110, 1308–1312. [Google Scholar] [CrossRef]
- Mercurio, V.; Peloquin, G.; Bourji, K.I.; Diab, N.; Sato, T.; Enobun, B.; Housten-Harris, T.; Damico, R.; Kolb, T.M.; Mathai, S.C.; et al. Pulmonary arterial hypertension and atrial arrhythmias: Incidence, risk factors, and clinical impact. Pulm. Circ. 2018, 8, 2045894018769874. [Google Scholar] [CrossRef] [PubMed]
- Olsson, K.M.; Nickel, N.P.; Tongers, J.; Hoeper, M.M. Atrial flutter and fibrillation in patients with pulmonary hypertension. Int. J. Cardiol. 2013, 167, 2300–2305. [Google Scholar] [CrossRef] [PubMed]
- Chu, J.W.; Kao, P.N.; Faul, J.L.; Doyle, R.L. High prevalence of autoimmune thyroid disease in pulmonary arterial hypertension. Chest 2002, 122, 1668–1673. [Google Scholar] [CrossRef] [PubMed]
- Tongers, J.; Schwerdtfeger, B.; Klein, G.; Kempf, T.; Schaefer, A.; Knapp, J.-M.; Niehaus, M.; Korte, T.; Hoeper, M.M. Incidence and clinical relevance of supraventricular tachyarrhythmias in pulmonary hypertension. Am. Heart J. 2007, 153, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Andersen, M.; Diederichsen, S.Z.; Svendsen, J.H.; Carlsen, J. Assessment of cardiac arrhythmias using long-term continuous monitoring in patients with pulmonary hypertension. Int. J. Cardiol. 2021, 334, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Fingrova, Z.; Ambroz, D.; Jansa, P.; Kuchar, J.; Lindner, J.; Kunstyr, J.; Aschermann, M.; Linhart, A.; Havranek, S. The prevalence and clinical outcome of supraventricular tachycardia in different etiologies of pulmonary hypertension. PLoS ONE 2021, 16, e0245752. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Zhu, Y.J.; Zhai, Z.Q.; Weng, S.X.; Ma, Y.Z.; Yu, F.Y.; Qi, Y.J.; Jiang, Y.Z.; Gao, X.; Xu, X.Q.; et al. Radiofrequency Catheter Ablation of Supraventricular Tachycardia in Patients With Pulmonary Hypertension: Feasibility and Long-Term Outcome. Front. Physiol. 2021, 12, 674909. [Google Scholar] [CrossRef]
- Hoeper, M.M.; Galiè, N.; Murali, S.; Olschewski, H.; Rubenfire, M.; Robbins, I.M.; Farber, H.W.; Mclaughlin, V.; Shapiro, S.; Pepke-Zaba, J.; et al. Outcome after cardiopulmonary resuscitation in patients with pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 2002, 165, 341–344. [Google Scholar] [CrossRef]
- Yang, J.Z.; Odish, M.F.; Mathers, H.; Pebley, N.; Wardi, G.; Papamatheakis, D.G.; Poch, D.S.; Kim, N.H.; Fernandes, T.M.; Sell, R.E. Outcomes of cardiopulmonary resuscitation in patients with pulmonary arterial hypertension. Pulm. Circ. 2022, 12, e12066. [Google Scholar] [CrossRef]
- Drakopoulou, M.; Nashat, H.; Kempny, A.; Alonso-Gonzalez, R.; Swan, L.; Wort, S.J.; Price, L.C.; McCabe, C.; Wong, T.; Gatzoulis, M.A.; et al. Arrhythmias in adult patients with congenital heart disease and pulmonary arterial hypertension. Heart 2018, 104, 1963–1969. [Google Scholar] [CrossRef]
- Sivak, J.A.; Raina, A.; Forfia, P.R. Assessment of the physiologic contribution of right atrial function to total right heart function in patients with and without pulmonary arterial hypertension. Pulm. Circ. 2016, 6, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Showkathali, R.; Tayebjee, M.H.; Grapsa, J.; Alzetani, M.; Nihoyannopoulos, P.; Howard, L.S.; Lefroy, D.C.; Gibbs, J.S.R. Right atrial flutter isthmus ablation is feasible and results in acute clinical improvement in patients with persistent atrial flutter and severe pulmonary arterial hypertension. Int. J. Cardiol. 2011, 149, 279–280. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Cano, M.J.; Gonzalez-Mansilla, A.; Escribano, P.; Delgado, J.; Arribas, F.; Torres, J.; Flox, A.; Riva, M.; Gomez, M.A.; Saenz, C. Clinical implications of supraventricular arrhythmias in patients with severe pulmonary arterial hypertension. Int. J. Cardiol. 2011, 146, 105–106. [Google Scholar] [CrossRef] [PubMed]
- Bradfield, J.; Shapiro, S.; Finch, W.; Tung, R.; Boyle, N.G.; Buch, E.; Mathuria, N.; Mandapati, R.; Shivkumar, K.; Bersohn, M. Catheter ablation of typical atrial flutter in severe pulmonary hypertension. J. Cardiovasc. Electrophysiol. 2012, 23, 1185–1190. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.; Genuardi, M.V.; Koczo, A.; Zou, R.H.; Thoma, F.W.; Handen, A.; Craig, E.; Hogan, C.M.; Girard, T.; Althouse, A.D.; et al. Atrial arrhythmias are associated with increased mortality in pulmonary arterial hypertension. Pulm. Circ. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Kamada, H.; Kaneyama, J.; Inoue, Y.Y.; Noda, T.; Ueda, N.; Nakajima, K.; Kamakura, T.; Wada, M.; Ishibashi, K.; Yamagata, K.; et al. Long term prognosis in patients with pulmonary hypertension undergoing catheter ablation for supraventricular tachycardia. Sci. Rep. 2021, 11, 16176. [Google Scholar] [CrossRef] [PubMed]
- Luesebrink, U.; Fischer, D.; Gezgin, F.; Duncker, D.; Koenig, T.; Oswald, H.; Klein, G.; Gardiwal, A. Ablation of typical right atrial flutter in patients with pulmonary hypertension. Heart Lung Circ. 2012, 21, 695–699. [Google Scholar] [CrossRef]
- Rich, S.; Seidlitz, M.; Dodin, E.; Osimani, D.; Judd, D.; Genthner, D.; McLaughlin, V.; Francis, G. The short-term effects of digoxin in patients with right ventricular dysfunction from pulmonary hypertension. Chest 1998, 114, 787–792. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Actelion. Tracleer (Bosentan) [Package Insert]. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/021290s044,209279s010lbl.pdf (accessed on 24 February 2024).
- Chung, M.K.; Patton, K.K.; Lau, C.-P.; Forno, A.R.D.; Al-Khatib, S.M.; Arora, V.; Birgersdotter-Green, U.M.; Cha, Y.-M.; Chung, E.H.; Cronin, E.M.; et al. 2023 HRS/APHRS/LAHRS guideline on cardiac physiologic pacing for the avoidance and mitigation of heart failure. Heart Rhythm. 2023, 20, e17–e91. [Google Scholar] [CrossRef]
- Meiburg, R.; Rijks, J.H.J.; Beela, A.S.; Bressi, E.; Grieco, D.; Delhaas, T.; Luermans, J.G.L.; Prinzen, F.W.; Vernooy, K.; Lumens, J. Comparison of novel ventricular pacing strategies using an electro-mechanical simulation platform. Europace 2023, 25, euad144. [Google Scholar] [CrossRef]
- Boyle, T.A.; Daimee, U.A.; Simpson, C.E.; Kolb, T.M.; Mathai, S.C.; Akhtar, T.; Nyhan, D.; Calkins, H.; Spragg, D. Left atrial ablation for the management of atrial tachyarrhythmias in patients with pulmonary hypertension: A case series. HeartRhythm Case Rep. 2022, 8, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Smilowitz, N.R.; Armanious, A.; Bangalore, S.; Ramakrishna, H.; Berger, J.S. Cardiovascular Outcomes of Patients with Pulmonary Hypertension Undergoing Noncardiac Surgery. Am. J. Cardiol. 2019, 123, 1532–1537. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, S.; Ruetzler, K.; Ghadimi, K.; Horn, E.M.; Kelava, M.; Kudelko, K.T.; Moreno-Duarte, I.; Preston, I.; Bovino, L.L.R.; Smilowitz, N.R.; et al. Evaluation and Management of Pulmonary Hypertension in Noncardiac Surgery: A Scientific Statement from the American Heart Association. Circulation 2023, 147, 1317–1343. [Google Scholar] [CrossRef] [PubMed]
- Benza, R.L.; Gomberg-Maitland, M.; Elliott, C.G.; Farber, H.W.; Foreman, A.J.; Frost, A.E.; McGoon, M.D.; Pasta, D.J.; Selej, M.; Burger, C.D.; et al. Predicting Survival in Patients with Pulmonary Arterial Hypertension: The REVEAL Risk Score Calculator 2.0 and Comparison with ESC/ERS-Based Risk Assessment Strategies. Chest 2019, 156, 323–337. [Google Scholar] [CrossRef]
- Price, L.C.; Wort, S.J.; Finney, S.J.; Marino, P.S.; Brett, S.J. Pulmonary vascular and right ventricular dysfunction in adult critical care: Current and emerging options for management: A systematic literature review. Crit. Care 2010, 14, R169. [Google Scholar] [CrossRef] [PubMed]
- Miotti, C.; Papa, S.; Manzi, G.; Scoccia, G.; Luongo, F.; Toto, F.; Malerba, C.; Cedrone, N.; Sciomer, S.; Ciciarello, F.; et al. The Growing Role of Echocardiography in Pulmonary Arterial Hypertension Risk Stratification: The Missing Piece. J. Clin. Med. 2021, 10, 619. [Google Scholar] [CrossRef] [PubMed]
- Subramani, S.; Sharma, A.; Arora, L.; Hanada, S.; Krishnan, S.; Ramakrishna, H. Perioperative Right Ventricular Dysfunction: Analysis of Outcomes. J. Cardiothorac. Vasc. Anesth. 2022, 36, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Augoustides, J.G.; Culp, K.; Smith, S. Rebound pulmonary hypertension and cardiogenic shock after withdrawal of inhaled prostacyclin. Anesthesiology 2004, 100, 1023–1025. [Google Scholar] [CrossRef]
- Ruetzler, K.; Smilowitz, N.R.; Berger, J.S.; Devereaux, P.J.; Maron, B.A.; Newby, L.K.; de Jesus Perez, V.; Sessler, D.I.; Wijeysundera, D.N. Diagnosis and Management of Patients with Myocardial Injury after Noncardiac Surgery: A Scientific Statement from the American Heart Association. Circulation 2021, 144, e287–e305. [Google Scholar] [CrossRef]
- Gooding, J.M.; Dimick, A.R.; Tavakoli, M.; Corssen, G. A physiologic analysis of cardiopulmonary responses to ketamine anesthesia in noncardiac patients. Anesth. Analg. 1977, 56, 813–816. [Google Scholar] [CrossRef]
- Martin, C.; Perrin, G.; Saux, P.; Papazian, L.; Albanese, J.; Gouin, F. Right ventricular end-systolic pressure-volume relation during propofol infusion. Acta Anaesthesiol. Scand. 1994, 38, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Joglar, J.A.; Chung, M.K.; Armbruster, A.L.; Benjamin, E.J.; Chyou, J.Y.; Cronin, E.M.; Deswal, A.; Eckhardt, L.L.; Goldberger, Z.D.; Gopinathannair, R.; et al. 2023 ACC/AHA/ACCP/HRS Guideline for the Diagnosis and Management of Atrial Fibrillation: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2024, 149, e1–e156. [Google Scholar] [CrossRef] [PubMed]

| Study | Study Design | Study Size | Patient Population | Primary Endpoint and Results | Type of Arrhythmia | Effect of Arrhythmia | Effect of Rhythm Control |
|---|---|---|---|---|---|---|---|
| Tongers et al., Am Heart J, 2007 [14] | Retrospective, observational, single-center | 231 | Consecutive patients followed for PAH or inoperable CTEPH | Incidence of SVA 31 episodes of SVA were observed in 27 of 231 patients (cumulative incidence, 11.7%; annual risk, 2.8% per patient) | AFL (n = 15), AF (n = 13), and AVNRT (n = 3) | SVA onset was associated with clinical deterioration and right ventricular failure (84% of SVA episodes); outcome was strongly associated with the type of SVA and restoration of sinus rhythm | Mortality was 6.3% (follow-up 26 ± 23 months) when sinus rhythm was restored (all cases of AVNRT and AFL), but was 82% with sustained AF (follow-up 11 ± 8 months) |
| Showkathali et al., Int J Cardiol, 2011 [22] | Retrospective, observational, single-center | 22 | Patients with AFL and PAH or CTEPH | Success of typical atrial flutter ablation AFL ablation was acutely successful and without complications. Three patients had recurrence and underwent successful redo procedures without further recurrence | Typical atrial flutter | NR | Functional class improved in 9 and remained the same in 11 patients; 6MWT was 275 ± 141 m before and increased to 293 ± 146 m following ablation (p = 0.301) |
| Luesebrink et al., Heart Lung Circ, 2012 [27] | Retrospective, observational, single-center | 38 with PAH; 196 controls | Patients undergoing ablation of cavo-tricuspid isthmus-dependent flutter with an 8 mm RF ablation catheter | Influence of PAH on typical atrial flutter ablation procedure Acutely successful ablation in all patients; patients with severe PAH had a significantly longer procedure time (78 ± 40 min vs. 62 ± 29 min; p = 0.033), total ablation time (20 ± 11 min vs. 15 ± 9 min; p = 0.02), and more ablation lesions (26 ± 16 vs. 19 ± 12; p = 0.018) compared to patients without PAH | Typical atrial flutter | NR | NR |
| Bradfield et al., JCE, 2012 [24] | Retrospective, observational, single-center | 12 | Consecutive patients with severe PAH (systolic pulmonary artery pressure > 60 mmHg) and AFL referred for ablation (4 congenital, 2 CTEPH, 6 PAH) | Describe flutter ablation in patients with severe PAH Acute success was obtained in 86% of procedures. Complications were seen in 14%. A total of 80% (8/10) of patients were free of AFL at 3 months; 75% (6/8) at 1 year | Typical atrial flutter | NR | SPAP decreased from 114 ± 44 mmHg to 82 ± 38 mmHg after ablation (p = 0.004); BNP levels were lower post ablation (787 ± 832 pg/mL vs. 522 ± 745 pg/mL, p = 0.02) |
| Kamada et al., Sci Rep, 2021 [26] | Retrospective, observational, single-center | 23 | 13 patients with congenital heart disease; 6 with idiopathic or other PAH; 3 with CTEPH; and 1 with hemodialysis-associated PH (group 5) | Procedural success rate; short- and long-term clinical outcomes Single-procedure success, 83%; 94% (17/18) in typical atrial flutter; 73% (8/11) in atrial tachycardia (AT); and 100% (1/1) in atrioventricular nodal reentrant tachycardia. | Typical atrial flutter, atrial tachycardia, and AVNRT | NR | Antiarrhythmic drugs, serum brain natriuretic peptide levels, and number of hospitalizations significantly decreased after RFCA SVT after the last RFCA was a significant risk factor of mortality (HR, 9.31; p = 0.016). |
| Zhou et al., Front Physiol, 2021 [17] | Retrospective, observational, single-center | 71 | Consecutive PH patients with SVA who were scheduled to undergo catheter ablation | Feasibility and long-term outcomes of catheter ablation in PH patients with SVA Acute success in 54, complications in 4 (6.7%); during median follow-up of 36 (range, 3–108) months, 7 patients with atrial flutter experienced recurrence (78.3% success rate) | Typical atrial flutter (n = 33, 43.5%) was the most common SVT type, followed by atrioventricular nodal reentrant tachycardia (n = 16, 21.1%) | NR | NR |
| Cannillo et al., Am J Cardiol, 2015 [4] | Retrospective, observational, single-center | 77 | Consecutive patients with PAH without history of SVA | All-cause mortality and re-hospitalization During a median follow-up of 35 months, 17 patients (22%) experienced SVA. The primary endpoint occurred in 13 patients (76%) in the SVA group and in 22 patients (37%) in the group without SVA (p = 0.004) | Persistent AF (8 patients, 47%); permanent AF (3, 17%); paroxysmal SVA (3, 17%: 2 with atrial ectopic tachycardia and 1 with atrioventricular nodal re-entry tachycardia); right atrial flutter (2, 12%); and paroxysmal AF (1, 6%) | SVA onset was associated with the worsening of functional class, NT-proBNP, 6 min walk distance, TAPSE, and DLCO; 9 patients (53%) among those with SVA died compared with 8 (13%) among those without (p = 0.001) | NR |
| Wen et al., Am J Card, 2014 [1] | Prospective, two-center cohort study | 280 | Consecutivepatients > 18 years of age with IPAH at 2 national referral centers in China | All-cause mortality Patients who developed SVAs had a significantly higher mortality than those who did not; estimated survivalat 1, 3, and 6 years was 85%, 64.2%, and 52.6% vs. 92%, 81.9%, and 74.5%,respectively; p = 0.008 | Atrial fibrillation (n = 16), atrial flutter (n = 13), and atrial tachycardia (n = 11) | In most patients (97.5%), the onset of SVA resulted in clinical deterioration or worsening right-sided cardiac failure | Patients who developed permanent SVA had a significantly lower survival rate than patients with transient SVA (p = 0.011) or without SVA (p < 0.001); survival was not statistically different between patients with transient SVA and those without SVA (p = 0.850) |
| Olsson et al., Int J Cardiol [12] | Prospective, single-center cohort study | 239 (PAH, n = 157; inoperable chronic thromboembolic pulmonary hypertension, n = 82) | Consecutive patients ≥ 18 years of age treated for PAH or inoperable CTEPH | Incidences of AF and AFL The cumulative 5-year incidence of new-onset atrial flutter and fibrillation was 25.1% (95% confidence interval, 13.8–35.4%) | AF 50% and AFL 50% | AF and AFL were frequently accompanied by clinical worsening (80%) and right heart failure (30%); new-onset atrial flutter and AF were independent risk factors for death | Stable sinus rhythm was successfully re-established in 21/24 (88%) with atrial flutter and in 16/24 (67%) with atrial fibrillation Higher mortality was observed in patients with persistent AF compared to patients in whom sinus rhythm was restored (estimated survival at 1, 2, and 3 years was 64%, 55%, and 27% versus 97%, 80%, and 57%, respectively) |
| Smith et al., Pulm Circ, 2018 [25] | Retrospective, observational, multi-center | 297 (group 1 PAH, n = 266; CTEPH, n = 31) | All patients in a healthcare system with PAH or CTEPH (excluding those who had undergone thromboembolectomy) | AF/AFL occurrence and survival 79 (26.5%) developed AF/AFL, either before or after a diagnosis of PH or CTEPH | AF in 46 (58.2%), atrial flutter in 25 (31.6%), and instances of both in 8 (10.1%) | AF/AFL was associated with a 3.81-fold increase in the hazard of death (95% CI, 2.64–5.52; p < 0.001) Mortality risk was present, whether paroxysmal or persistent AF/AFL | NR |
| Ruiz-Cano et al., Int J Cardiol, 2010 [23] | Retrospective, observational, single-center | 282 patients with PH; not reported but implied 28 with arrhythmias | Group 1 PAH: 6 patients (26.1%) had idiopathic PAH; 7 (30.4%), a connective tissue disease; 6 (26.1%), toxic oil syndrome; and 4 (17.4%), Eisenmenger syndrome | Safety and efficacy of EPS Efficacy 100% for AVNRT and 95% for typical flutter; safety not reported | AF (n = 12, 42.8%); atypical flutter (n = 7, 25%); typical flutter (n = 5, 17.8%); andAVNRT (n = 4, 14.2%) | Most episodes of SVA (82%) were symptomatic with clinicalworsening or RV failure Clinical deterioration was not observed in patients with AVNRT | Restoration of SR was associated with a clinical improvement in all the patients, with an average increase of 196 ± 163 m in 6MWT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anand, S.; Cronin, E.M. Arrhythmias in Patients with Pulmonary Hypertension and Right Ventricular Failure: Importance of Rhythm Control Strategies. J. Clin. Med. 2024, 13, 1866. https://doi.org/10.3390/jcm13071866
Anand S, Cronin EM. Arrhythmias in Patients with Pulmonary Hypertension and Right Ventricular Failure: Importance of Rhythm Control Strategies. Journal of Clinical Medicine. 2024; 13(7):1866. https://doi.org/10.3390/jcm13071866
Chicago/Turabian StyleAnand, Suneesh, and Edmond M. Cronin. 2024. "Arrhythmias in Patients with Pulmonary Hypertension and Right Ventricular Failure: Importance of Rhythm Control Strategies" Journal of Clinical Medicine 13, no. 7: 1866. https://doi.org/10.3390/jcm13071866
APA StyleAnand, S., & Cronin, E. M. (2024). Arrhythmias in Patients with Pulmonary Hypertension and Right Ventricular Failure: Importance of Rhythm Control Strategies. Journal of Clinical Medicine, 13(7), 1866. https://doi.org/10.3390/jcm13071866